-
Name
3-Nitrocinnamic acid
- EINECS 209-104-2
- CAS No. 555-68-0
- Article Data109
- CAS DataBase
- Density 1.411 g/cm3
- Solubility
- Melting Point 200-202 °C(lit.)
- Formula C9H7NO4
- Boiling Point 372.1 °C at 760 mmHg
- Molecular Weight 193.159
- Flash Point 166.5 °C
- Transport Information
- Appearance Light yellow fine crystalline powder
- Safety 22-24/25
- Risk Codes 36/37/38
-
Molecular Structure
-
Hazard Symbols
 Xi
Xi
- Synonyms Cinnamicacid, m-nitro- (6CI,7CI,8CI);3-(3-Nitrophenyl)-2-propenoic acid;3-(3-Nitrophenyl)acrylic acid;3-Nitrobenzenepropenoic acid;Ethyl 3-(3-nitrophenyl)-2-propenoate;NSC 5408;m-Nitrocinnamic acid;
- PSA 83.12000
- LogP 2.21580
Synthetic route

| Conditions | Yield |
|---|---|
| With 1,8-diazabicyclo[5.4.0]undec-7-ene; 3-amino propanoic acid In ethanol at 20℃; Knoevenagel-Doebner reaction; stereoselective reaction; | 98% |
| With piperidine; pyridine for 0.0333333h; microwave irradiation; | 97% |
| With ammonium acetate for 0.0666667h; Irradiation; | 95% |

| Conditions | Yield |
|---|---|
| With sodium carbonate In water at 120℃; for 12h; Green chemistry; | 96.5% |
| With potassium carbonate In water for 12h; Heck Reaction; Inert atmosphere; Reflux; | 95% |
| With tributyl-amine; silica-supported bidentate arsine palladium(0) In para-xylene at 100℃; for 6h; Heck arylation; | 87% |
-
-
141-82-2
malonic acid

-
-
68444-12-2
sodium α-hydroxy-m-nitrotoluene-α-sulphonate

-
-
555-68-0
(E)-3-Nitrocinnamic acid

| Conditions | Yield |
|---|---|
| With piperazine In ethylene glycol for 0.125h; Knoevenagel condensation; microwave irradiation; | 92% |

| Conditions | Yield |
|---|---|
| With potassium carbonate In water for 12h; Heck Reaction; Inert atmosphere; Reflux; | 92% |
-
-
111425-75-3, 36574-84-2
(E)-2'-hydroxy-3-nitrochalcone

-
-
555-68-0
(E)-3-Nitrocinnamic acid

| Conditions | Yield |
|---|---|
| With dihydrogen peroxide; potassium carbonate In acetonitrile at 20℃; for 5h; | 91% |

| Conditions | Yield |
|---|---|
| With pyridine; dmap; lithium chloride In 1-methyl-pyrrolidin-2-one at 185 - 190℃; for 8h; | 82% |
| With pyridine; dmap; sodium tetraborate decahydrate; N-benzyl-N,N,N-triethylammonium chloride In 1-methyl-pyrrolidin-2-one at 185 - 190℃; for 7h; | 82% |

| Conditions | Yield |
|---|---|
| With trichlorophosphate at 80 - 90℃; Condensation; Perkin reaction; | 80% |
| With tetraacetyl diborate | |
| With sodium acetate |
-
-
24163-36-8
3,3'-dinitrodiphenyliodonium bromide

-
-
79-10-7
acrylic acid

-
-
555-68-0
(E)-3-Nitrocinnamic acid

| Conditions | Yield |
|---|---|
| palladium dichloride In water for 0.0166667h; Heck reaction; microwave irradiation; | 80% |

-
A
-
40828-54-4
1H-benzimidazole-2-sulfonic acid

-
B
-
555-68-0
(E)-3-Nitrocinnamic acid

| Conditions | Yield |
|---|---|
| With dihydrogen peroxide; acetic acid for 24h; | A 59% B 56% |

| Conditions | Yield |
|---|---|
| With sodium hydroxide In ethanol at 0℃; for 2h; | 36% |
-
-
110-89-4
piperidine

-
-
110-86-1
pyridine

-
-
141-82-2
malonic acid

-
-
99-61-6
3-nitro-benzaldehyde

-
-
555-68-0
(E)-3-Nitrocinnamic acid

-
-
91-22-5
quinoline

-
-
141-82-2
malonic acid

-
-
99-61-6
3-nitro-benzaldehyde

-
-
555-68-0
(E)-3-Nitrocinnamic acid

-
-
141-82-2
malonic acid

-
-
99-61-6
3-nitro-benzaldehyde

-
A
-
555-68-0
(E)-3-Nitrocinnamic acid

-
B
-
5678-47-7
3-amino-3-(3-nitro-phenyl)-propionic acid

| Conditions | Yield |
|---|---|
| With ammonium acetate In ethanol for 24h; Heating; | |
| With ammonia |


| Conditions | Yield |
|---|---|
| With hydrogenchloride Diazotization.Eintragen in ein Gemisch von Maleinsaeure (oder Acrylsaeure), Kupfer(II)-chlorid, Natriumacetat und Wasser; |

-
-
555-68-0
(E)-3-Nitrocinnamic acid

| Conditions | Yield |
|---|---|
| With sodium iodide In N,N-dimethyl-formamide at 29.9℃; Kinetics; Thermodynamic data; Mechanism; effect of temperature; ΔH(excit.), ΔS(excit), ΔG(excit.); |

| Conditions | Yield |
|---|---|
| With sodium acetate at 180 - 185℃; |

| Conditions | Yield |
|---|---|
| With sodium hydroxide; water; sodium hydride modified Wittig's reaction; 1.)THF, R.T., 3 h; 2.)CH3OH, reflux, 8 h; Yield given. Multistep reaction; |

| Conditions | Yield |
|---|---|
| Stage #1: m-iodonitrobenzene; poly(ethylene glycol) diacrylate With palladium diacetate; tetrabutylammomium bromide; potassium carbonate at 80℃; for 8h; Heck reaction; Stage #2: With sodium hydroxide at 20℃; |

| Conditions | Yield |
|---|---|
| With pyridine; malonic acid In water |
-
-
350490-16-3
butyl (3-nitro)cinnamate

-
-
555-68-0
(E)-3-Nitrocinnamic acid

| Conditions | Yield |
|---|---|
| With trifluoroacetic acid In dichloromethane at 0℃; for 4h; |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: triethylamine; tris-(dibenzylideneacetone)dipalladium(0); tris-(o-tolyl)phosphine / N,N-dimethyl-formamide / 12 h / 90 °C / Inert atmosphere 2: lithium hydroxide monohydrate; water / tetrahydrofuran / 12 h / 25 °C View Scheme |

| Conditions | Yield |
|---|---|
| With lithium hydroxide monohydrate; water In tetrahydrofuran at 25℃; for 12h; |
-
-
99-61-6
3-nitro-benzaldehyde

-
A
-
555-68-0
(E)-3-Nitrocinnamic acid

-
B
-
19883-74-0
(2S)-2-amino-3-(3-nitrophenyl)propanoic acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: ammonium formate / ethanol / 4 h / Reflux 2: phenylalanine ammonia lyase from Streptomyces maritimus; phenylalanine ammonia α-lyase from Anabaena variabilis / aq. buffer / 24 h / 30 °C / pH 8 / Resolution of racemate; Enzymatic reaction View Scheme |
-
-
5678-47-7
3-amino-3-(3-nitro-phenyl)-propionic acid

-
A
-
787544-61-0
(R)-3-amino-3-(3-nitrophenyl)propanoic acid

-
B
-
555-68-0
(E)-3-Nitrocinnamic acid

-
C
-
19883-74-0
(2S)-2-amino-3-(3-nitrophenyl)propanoic acid

| Conditions | Yield |
|---|---|
| With phenylalanine ammonia α-lyase from Anabaena variabilis; phenylalanine ammonia lyase from Streptomyces maritimus In aq. buffer at 30℃; for 24h; pH=8; Reagent/catalyst; Resolution of racemate; Enzymatic reaction; enantioselective reaction; |


| Conditions | Yield |
|---|---|
| Stage #1: 3-nitrocinnamaldehyde With titanium tetrachloride; ethyl bromoacetate; triphenylphosphine In dichloromethane at 0 - 20℃; for 6h; Stage #2: With potassium hydroxide In methanol at 20℃; for 2h; |

| Conditions | Yield |
|---|---|
| With phosphate buffer; Camellia sinensis for 240h; pH=6.4; | 100% |
| With quinoline; copper at 185 - 195℃; | |
| With Camellia sinensis at 25℃; for 120h; pH=6.4; Decarboxylation; |
-
-
555-68-0
(E)-3-Nitrocinnamic acid

-
-
136265-09-3
(Z)-ethyl 3-(3-nitrophenyl)acrylate

| Conditions | Yield |
|---|---|
| With sulfuric acid In ethanol | 99% |
-
-
4112-65-6
5-hydroxy-2-methylisoindoline-1,3-dione

-
-
555-68-0
(E)-3-Nitrocinnamic acid

-
-
1242154-02-4
2-methylisoindole-1,3-dion-5-yl (E)-3-(3-nitrophenyl) acrylate

| Conditions | Yield |
|---|---|
| With dmap; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride In dichloromethane at 20℃; for 4h; | 98% |

| Conditions | Yield |
|---|---|
| With sulfuric acid for 0.0833333h; Esterification; Irradiation; | 97% |
| With sulfuric acid Reflux; | 97.3% |
| With sulfuric acid |

| Conditions | Yield |
|---|---|
| Multistep reaction; | 97% |

-
-
555-68-0
(E)-3-Nitrocinnamic acid

-
-
74710-09-1
o-phenylenediamine sulphate

-
-
55969-91-0
2-(3-nitro-styryl)-1H-benzoimidazole

| Conditions | Yield |
|---|---|
| In ethylene glycol for 3h; Heating; | 97% |

| Conditions | Yield |
|---|---|
| Stage #1: (E)-3-Nitrocinnamic acid With sodium tetrahydroborate In tetrahydrofuran for 0.166667h; Stage #2: With iodine In tetrahydrofuran for 1h; | 97% |
-
-
555-68-0
(E)-3-Nitrocinnamic acid

-
-
100-46-9
benzylamine

-
-
1085449-49-5
(E)-N-benzyl-3-(3-nitrophenyl)acrylamide

| Conditions | Yield |
|---|---|
| With tris(2,2,2-trifluoroethyl) borate In acetonitrile at 80℃; for 15h; | 95% |
-
-
555-68-0
(E)-3-Nitrocinnamic acid

-
-
333-20-0
potassium thioacyanate

-
-
22715-07-7
(E)-3-(3-nitrophenyl)acryloyl isothiocyanate

| Conditions | Yield |
|---|---|
| Stage #1: (E)-3-Nitrocinnamic acid With trichloroisocyanuric acid; triphenylphosphine In toluene at 0℃; for 0.25h; Stage #2: potassium thioacyanate In toluene at 0 - 20℃; for 1.5h; | 95% |
-
-
555-68-0
(E)-3-Nitrocinnamic acid

-
-
100-51-6
benzyl alcohol

-
-
518045-06-2
(E)-benzyl 3-(3-nitrophenyl)acrylate

| Conditions | Yield |
|---|---|
| Stage #1: (E)-3-Nitrocinnamic acid With N-chlorobenzotriazole; triphenylphosphine In dichloromethane for 0.25h; Cooling; Stage #2: benzyl alcohol With triethylamine In dichloromethane at 20℃; for 1.33333h; | 92% |
-
-
555-68-0
(E)-3-Nitrocinnamic acid

-
-
35418-05-4, 141236-48-8
(E)-3-nitrocinnamic acid chloride

| Conditions | Yield |
|---|---|
| With thionyl chloride In benzene | 91% |
| With thionyl chloride In benzene Heating; | 90% |
| With phosphorus pentachloride; trichlorophosphate |
-
-
555-68-0
(E)-3-Nitrocinnamic acid

-
-
16024-82-1
methyl 2-isothiocyanatobenzoate

| Conditions | Yield |
|---|---|
| Multistep reaction; | 90% |

| Conditions | Yield |
|---|---|
| With sulfonated nanohydroxyapatite functionalized with 2-aminoethyl dihydrogen phosphate In dichloromethane at 41℃; for 3h; | 90% |
-
-
555-68-0
(E)-3-Nitrocinnamic acid

-
-
108-24-7
acetic anhydride

-
-
32862-98-9
(E)-3-(acetylamino)cinnamic acid

| Conditions | Yield |
|---|---|
| Stage #1: (E)-3-Nitrocinnamic acid With hydrogenchloride; tin(ll) chloride In water at 45 - 75℃; for 1h; Stage #2: acetic anhydride With acetic acid at 110℃; for 2h; | 89% |
-
-
555-68-0
(E)-3-Nitrocinnamic acid

-
-
569348-67-0
3-nitro-cinnamoyl azide

| Conditions | Yield |
|---|---|
| With sodium azide; N,N-dimethyl-formamide; trichlorophosphate at 10 - 15℃; for 2h; | 88% |

| Conditions | Yield |
|---|---|
| With phosphoric acid; trifluoroacetic acid at 20℃; for 0.5h; | 88% |
-
-
1041756-57-3
(4-(4-aminobutyl)piperidin-1-yl)(phenyl)methanone

-
-
555-68-0
(E)-3-Nitrocinnamic acid

-
-
1296137-02-4
(E)-N-(4-(1-benzoylpiperidin-4-yl)butyl)-3-(3-nitrophenyl)acrylamide

| Conditions | Yield |
|---|---|
| With benzotriazol-1-ol; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride; triethylamine In dichloromethane at 20℃; | 87% |

| Conditions | Yield |
|---|---|
| With hydroxylamine sulfate; hydroxylamine-O-sulfonic acid In water at 10℃; for 6h; pH=7.6; | 86% |
-
-
555-68-0
(E)-3-Nitrocinnamic acid

-
-
82594-66-9, 115665-66-2, 115665-74-2
(E)-1-(2-bromo-vinyl)-3-nitro-benzene

| Conditions | Yield |
|---|---|
| With dibromamine-T; potassium carbonate In acetonitrile at 20℃; for 0.166667h; stereoselective reaction; | 85% |
| With diphosphorus tetraiodide; tetraethylammonium bromide In carbon disulfide at 20℃; for 14h; | 80% |
| With tetraethylammonium bromide; Dess-Martin periodane In dichloromethane at 20℃; for 16h; | 65% |
| Multi-step reaction with 2 steps 1: 84 percent / Br2 / acetic acid / Heating 2: Et3N / dimethylformamide / Heating View Scheme | |
| With [bis(acetoxy)iodo]benzene; tetraethylammonium bromide In dichloromethane at 25℃; for 16h; | 2.6 g |
-
-
555-68-0
(E)-3-Nitrocinnamic acid

-
-
100-66-3
methoxybenzene

-
-
396725-25-0
(2E)-1-(4-methoxyphenyl)-3-(3-nitrophenyl)prop-2-en-1-one

| Conditions | Yield |
|---|---|
| With phosphoric acid; trifluoroacetic acid at 20℃; for 0.5h; | 85% |

| Conditions | Yield |
|---|---|
| With methanol; hydrogen; acetic acid; palladium on activated charcoal | 84% |
| With ethanol; platinum Hydrogenation; | |
| Multi-step reaction with 2 steps 1: sodium sulfide; aq. NaOH solution 2: mercury-cathode; sodium sulfate; water View Scheme | |
| palladium on charcoal In ethanol; water |
-
-
555-68-0
(E)-3-Nitrocinnamic acid

-
-
18193-72-1
2,3-dibromo-3-(3-nitro-phenyl)-propionic acid

| Conditions | Yield |
|---|---|
| With bromine In acetic acid Heating; | 84% |
-
-
555-68-0
(E)-3-Nitrocinnamic acid

-
-
615-28-1
o-phenylenediamine dihydrochloride

-
-
55969-91-0
2-(3-nitro-styryl)-1H-benzoimidazole

| Conditions | Yield |
|---|---|
| In ethylene glycol for 5h; Condensation; Cycloaddition; Heating; | 84% |

| Conditions | Yield |
|---|---|
| With Aliquat 336; potassium carbonate microwave irradiation; | 84% |
-
-
461-82-5
4-(trifluoromethoxy)aniline

-
-
555-68-0
(E)-3-Nitrocinnamic acid

-
-
1615697-50-1
(E)-N-(4-trifluoromethoxyphenyl)-3-(4-nitrophenyl)acrylamide

| Conditions | Yield |
|---|---|
| Stage #1: 4-(trifluoromethoxy)aniline; (E)-3-Nitrocinnamic acid With triethylamine; trichlorophosphate In chloroform at 0℃; for 0.166667h; Inert atmosphere; Stage #2: With triethylamine In chloroform at 0℃; for 0.5h; Inert atmosphere; | 83% |
3-nitrocinnamic acid Chemical Properties
3-nitrocinnamic acid(555-68-0) is also named as AKOS B004090;3-(3-NITROPHENYL)ACRYLIC ACID;LABOTEST-BB LT00005570;RARECHEM BK HC P253;OTAVA-BB BB7020401717;M-NITROCINNAMIC ACID;m-Nitrocinnamic acid;Cinnamic acid, m-nitro;trans-3-Nitrocinnamic acid;3-nitro-cinnamic acid;Cinnamic acid, m-nitro-,and so on.3-nitrocinnamic acid(555-68-0) is light yellow fine crystalline powder.
CAS: 555-68-0
Molecular Formula: C9H7NO4
Molecular Weight: 193.16
Molecular structure: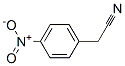
EINECS: 209-104-2
Melting point: 200-202 °C(lit.)
Merck: 14,6595
BRN: 1872713
ACD/LogD (pH 5.5): 0.34
ACD/LogD (pH 7.4): -1.19
ACD/BCF (pH 5.5): 1
ACD/BCF (pH 7.4): 1
ACD/KOC (pH 5.5): 4.6
ACD/KOC (pH 7.4): 1
H bond acceptors: 5
H bond donors: 1
Freely Rotating Bonds: 3
Index of Refraction: 1.655
Molar Refractivity: 50.25 cm3
Molar Volume: 136.8 cm3
Polarizability: 19.92 10-24cm3
Surface Tension: 64.2 dyne/cm
Density: 1.411 g/cm3
Flash Point: 166.5 °C
Enthalpy of Vaporization: 65.32 kJ/mol
Boiling Point: 372.1 °C at 760 mmHg
Vapour Pressure: 3.39E-06 mmHg at 25°C
3-nitrocinnamic acid Uses
3-nitrocinnamic acid Toxicity Data With Reference
3-nitrocinnamic acid Safety Profile

Risk Statements: 36/37/38 (36/37/38: Irritating to eyes, respiratory system and skin)
Safety Statements: 22-24/25 (22: Do not breathe dust 24/25: Avoid contact with skin and eyes)
WGK Germany: 3
Hazard Note: Irritant
HS Code: 29163900
Related Products
- 3-nitrocinnamic acid
- 55570-14-4
- 55570-91-7
- 5557-49-3
- 555-76-0
- 55577-25-8
- 55577-88-3
- 5557-83-5
- 55578-79-5
- 5557-88-0
- 55579-01-6
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View