-
Name
Allantoin
- EINECS 202-592-8
- CAS No. 97-59-6
- Article Data114
- CAS DataBase
- Density 1.652 g/cm3
- Solubility Slightly soluble in water. Freely soluble in alkalis
- Melting Point 230 °C (dec.)(lit.)
- Formula C4H6N4O3
- Boiling Point 478oC
- Molecular Weight 158.117
- Flash Point 230-234 °C
- Transport Information
- Appearance White crystalline powder
- Safety 22-24/25-36-26
- Risk Codes 22-36/37/38
-
Molecular Structure
-
Hazard Symbols
 Xi,
Xi, Xn
Xn
- Synonyms (2,5-dioxoimidazolidin-4-yl)urea;Glyoxylic(acid) diureide;urea, N-(2,5-dioxo-4-imidazolidinyl)-;Cutemol emollient;[(4S)-2,5-dioxoimidazolidin-4-yl]urea;5-Ureido-2,4-imidazolidindion;[(4R)-2,5-dioxoimidazolidin-4-yl]urea;Urea, (2,5-dioxo-4-imidazolidinyl)-;Urea, (2,5-dioxo-4-imidazolidinyl)- (9CI);Glyoxyldiureide;Urea, (2, 5-dioxo-4-imidazolidinyl)-;Glyoxylic diureide;(2,5-Dioxo-4-imidazolidinyl)urea;5-Ureidohydantoin;Allantoin (5-Ureidohydantoin);Urea,(2,5-dioxo-4-imidazolidinyl)-;
- PSA 113.32000
- LogP -0.43100
Synthetic route

| Conditions | Yield |
|---|---|
| With Co[PyPS]2Mo11VO40 at 90℃; for 10h; Temperature; Reagent/catalyst; Microwave irradiation; | 89.3% |
| With titanium dioxide-based composite solid catalyst A1 at 75℃; for 4h; | 85% |
| at 60℃; for 1.5h; Concentration; | 82.4% |
-
-
69-93-2
uric Acid

-
A
-
120-89-8
parabanic acid

-
B
-
5676-27-7
Oxalyldiurea

-
C
-
105245-87-2
dehydro-allantoin

-
D
-
97-59-6
Allantoin

| Conditions | Yield |
|---|---|
| With lithium hydroxide; iodine In water for 0.0833333h; excess of I2; | A n/a B n/a C 75% D n/a |
| With lithium hydroxide; iodine In water for 0.0833333h; Mechanism; also with substituted uric acid; effect of amount of I2; 2H and 13C labelling experiment; |


| Conditions | Yield |
|---|---|
| With lithium hydroxide; iodine In water at 4℃; equimolar amount of I2; | 70% |
| With sodium hydroxide; oxygen; pyrographite | |
| With water; ozone |

| Conditions | Yield |
|---|---|
| With potassium hydroxide; potassium permanganate In water for 4h; Product distribution; Mechanism; Ambient temperature; labelled 14C in position of 5; | A 26% B 14% |

| Conditions | Yield |
|---|---|
| In nitromethane for 1h; Reflux; | 15% |

| Conditions | Yield |
|---|---|
| With potassium hydroxide; potassium permanganate In water for 4h; Ambient temperature; Yield given; | A n/a B 14% |

| Conditions | Yield |
|---|---|
| With bromine; acetic acid |

| Conditions | Yield |
|---|---|
| With dihydrogen peroxide; copper dichloride |

| Conditions | Yield |
|---|---|
| With potassium nitrite; acetic acid |

| Conditions | Yield |
|---|---|
| With water |
-
-
874531-63-2
4,5-dihydroxy-2-oxo-5-ureido-imidazolidine-4-carboxylic acid

-
-
97-59-6
Allantoin

| Conditions | Yield |
|---|---|
| With water at 60 - 80℃; | |
| With nitric acid |

| Conditions | Yield |
|---|---|
| With water at 110 - 115℃; | |
| at 110℃; | |
| at 110℃; |
-
-
69-93-2
uric Acid

-
A
-
120-89-8
parabanic acid

-
B
-
61066-33-9, 61066-34-0, 61066-35-1, 61127-23-9
pyrimidine-2,4,5,6(1H,3H)-tetraone

-
C
-
36597-25-8
4-hydroxy-2,5-dioxoimidazolidine-4-carboxamide

-
D
-
3237-50-1
5,5-dihydroxy-pyrimidine-2,4,6-trione

-
E
-
57-13-6
urea

-
F
-
97-59-6
Allantoin

| Conditions | Yield |
|---|---|
| In methanol Mechanism; electrochemical oxidation; |

-
-
69-93-2
uric Acid

-
A
-
61066-33-9, 61066-34-0, 61066-35-1, 61127-23-9
pyrimidine-2,4,5,6(1H,3H)-tetraone

-
B
-
57-13-6
urea

-
C
-
97-59-6
Allantoin

| Conditions | Yield |
|---|---|
| at 25℃; Product distribution; Mechanism; Rate constant; electrochemical oxidation; buffer (pH=2-10); various times, potentials and electrodes; | |
| In phosphate buffer at 24℃; pH=2.3 - 11.2; Product distribution; Further Variations:; effect of surfactants; differential pulse voltammetric oxidation; Electrochemical reaction; |

-
-
81129-52-4
1-carboxy-2,4,6,8-tetraazabicyclo<3.3.0>octa-4-ene-3,7-dione

-
-
97-59-6
Allantoin

| Conditions | Yield |
|---|---|
| With hydroxide at 25℃; Rate constant; |

| Conditions | Yield |
|---|---|
| at 25℃; Product distribution; Mechanism; Rate constant; electrochemical oxidation; buffer (pH=6-10); various times, potentials and electrodes; |
-
-
6960-30-1, 151359-24-9
5-hydroxyisourate

-
-
97-59-6
Allantoin

| Conditions | Yield |
|---|---|
| With phosphate buffer In water at 22℃; Rate constant; var. ionic strength of buffer; |

-
A
-
36597-25-8
4-hydroxy-2,5-dioxoimidazolidine-4-carboxamide

-
B
-
36468-53-8
β-D-ribofuranose


-
D
-
97-59-6
Allantoin

| Conditions | Yield |
|---|---|
| In water at 22℃; Rate constant; pH 7.0; decomposition rate constants; |

-
-
590-28-3
potassium cyanate

-
-
24764-63-4
5-amino-imidazolidine-2,4-dione

-
-
7732-18-5
water

-
-
97-59-6
Allantoin


| Conditions | Yield |
|---|---|
| at 38℃; |

-
-
461-72-3
2,4-imidazolidinedione

-
-
7726-95-6
bromine

-
-
64-19-7
acetic acid

-
-
57-13-6
urea

-
-
97-59-6
Allantoin


| Conditions | Yield |
|---|---|
| saeuert man die alkal.Loesung mit Essigsaeure, und nach weiterem Stehenlassen oder Eindampfen der Loesung; |


-
-
97-59-6
Allantoin

| Conditions | Yield |
|---|---|
| With hydrogenchloride | |
| With water |

-
-
97-59-6
Allantoin

| Conditions | Yield |
|---|---|
| With ammonia | |
| With potassium hydroxide | |
| With potassium hydroxide |

| Conditions | Yield |
|---|---|
| With acetic anhydride |

| Conditions | Yield |
|---|---|
| With N-Bromosuccinimide; potassium iodide; potassium hydroxide In water at 0 - 25℃; for 3h; Temperature; Reagent/catalyst; Darkness; | 91.32% |
-
-
127099-85-8, 780722-26-1
N-Cyanoguanidine

-
-
97-59-6
Allantoin

| Conditions | Yield |
|---|---|
| With sulfuric acid In water for 20h; Heating; Green chemistry; | 81% |
-
-
1590389-14-2
potassium (4-fluorobenzoyl)trifluoroborate

-
-
97-59-6
Allantoin

| Conditions | Yield |
|---|---|
| With 1,3-dichloro-5,5-dimethylhydantoin In tetrahydrofuran at 40℃; for 4h; pH=3; chemoselective reaction; | 80% |
-
-
64-19-7
acetic acid

-
-
97-59-6
Allantoin

-
-
2207-75-2
monopotassium 1,2,3,4-tetrahydro-2,4-dioxo-1,3,5-triazine-6-carboxylate

| Conditions | Yield |
|---|---|
| Stage #1: Allantoin With bromine; potassium iodide; potassium hydroxide In water at 0 - 25℃; for 30h; Stage #2: acetic acid In water at 0 - 5℃; pH=5; Time; | 76.1% |

| Conditions | Yield |
|---|---|
| With potassium carbonate In ethanol for 3h; Heating; | 52% |
-
-
60971-83-7
1-(3-iodopropyl)-3,7-dimethyl-2,3,6,7-tetrahydro-1H-2,6-purinedione

-
-
97-59-6
Allantoin

| Conditions | Yield |
|---|---|
| Stage #1: Allantoin With sodium methylate In methanol for 1.5h; Heating; Stage #2: 1-(3-iodopropyl)-3,7-dimethyl-2,3,6,7-tetrahydro-1H-2,6-purinedione In methanol for 36h; Heating; | 40% |
-
-
97-59-6
Allantoin

-
-
108-95-2
phenol

-
A
-
2420-17-9
5-(4-hydroxy-phenyl)-imidazolidine-2,4-dione

-
B
-
77972-18-0
D,L-5-(2'-hydroxyphenyl)hydantoin

| Conditions | Yield |
|---|---|
| hydrogenchloride at 65℃; for 24h; | A 19.2% B 8% |
| hydrogenchloride at 65℃; for 24h; | A 19.2% B 8% |


| Conditions | Yield |
|---|---|
| With potassium hydroxide | |
| Darstellung; |

| Conditions | Yield |
|---|---|
| With hydrogenchloride nachfolgende Einw. von salpetriger Saeure; |

| Conditions | Yield |
|---|---|
| With hydrogen iodide |

| Conditions | Yield |
|---|---|
| With sodium amalgam; sulfuric acid |

| Conditions | Yield |
|---|---|
| With permanganate(VII) ion; acetic acid |

| Conditions | Yield |
|---|---|
| With ammonium persulfate; sodium acetate | |
| With potassium permanganate; bi carbonate .alkali | |
| With potassium permanganate; acetic acid |

| Conditions | Yield |
|---|---|
| With dihydrogen peroxide |

| Conditions | Yield |
|---|---|
| With potassium hydroxide | |
| With potassium hydroxide; water | |
| enzymatische Spaltung durch Brei oder Presssaft von Leguminosensamen; | |
| With water Alkaline conditions; |

| Conditions | Yield |
|---|---|
| With barium dihydroxide |
-
-
97-59-6
Allantoin

-
-
861794-23-2
diureido-acetic acid hydrazide

| Conditions | Yield |
|---|---|
| With hydrazine hydrate |

| Conditions | Yield |
|---|---|
| With lithium carbonate Bei der elektrochemischen Oxydation an einer Bleidioxydanode; |
-
-
90-46-0
9-hydroxyxanthene

-
-
97-59-6
Allantoin

-
-
87980-02-7
1-(2,5-dioxo-imidazolidin-4-yl)-3-xanthen-9-yl-urea

| Conditions | Yield |
|---|---|
| With acetic acid |
Allantoin Specification
The Allantoin, with the CAS registry number 97-59-6, is also known as 1-(2,5-Dioxo-4-imidazolidinyl)urea. It belongs to the product categories of Substrates; Intermediates & Fine Chemicals; Metabolites & Impurities; Pharmaceuticals. Its EINECS registry number is 202-592-8. This chemical's molecular formula is C4H6N4O3 and molecular weight is 158.12. What's more, its IUPAC name is called (2,5-Dioxoimidazolidin-4-yl)urea. It should be stored in a cool, dry and well-ventilated place. In fish, Allantoin is broken down further (into ammonia) before excretion. It is a major metabolic intermediate in many other organisms including plants and bacteria. It is frequently present in toothpaste, mouthwash, and other oral hygiene products, in shampoos, lipsticks, anti-acne products, sun care products, and clarifying lotions, various cosmetic lotions and creams, and other cosmetic and pharmaceutical products.
Physical properties about Allantoin are: (1)ACD/LogP: -1.523; (2)# of Rule of 5 Violations: 1; (3)ACD/LogD (pH 5.5): -1.52; (4)ACD/LogD (pH 7.4): -1.62; (5)ACD/BCF (pH 5.5): 1.00; (6)ACD/BCF (pH 7.4): 1.00; (7)ACD/KOC (pH 5.5): 3.53; (8)ACD/KOC (pH 7.4): 2.81; (9)#H bond acceptors: 7; (10)#H bond donors: 5; (11)#Freely Rotating Bonds: 1; (12)Polar Surface Area: 113.32 Å2; (13)Index of Refraction: 1.616; (14)Molar Refractivity: 33.422 cm3; (15)Molar Volume: 95.701 cm3; (16)Polarizability: 13.25×10-24cm3; (17)Surface Tension: 82.625 dyne/cm; (18)Density: 1.652 g/cm3.
Preparation of Allantoin: this chemical can be prepared by 7,9-dihydro-3H-purine-2,6,8-trione. This reaction needs reagents I2, LiOH and solvent H2O at temperature of 4 °C. The yield is 70 %.
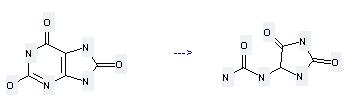
Uses of Allantoin: it is used to produce other chemicals. For example, it can react with 1-(3-iodo-propyl)-3,7-dimethyl-3,7-dihydro-purine-2,6-dione to get {1-[3-(3,7-dimethyl-2,6-dioxo-2,3,6,7-tetrahydro-purin-1-yl)-propyl]-2,5-dioxo-imidazolidin-4-yl}-urea. The reaction occurs with reagent NaOMe and other condition of heating for 1.5 hours. The yield is 40 %.
![Allantoin can react with 1-(3-iodo-propyl)-3,7-dimethyl-3,7-dihydro-purine-2,6-dione to get {1-[3-(3,7-dimethyl-2,6-dioxo-2,3,6,7-tetrahydro-purin-1-yl)-propyl]-2,5-dioxo-imidazolidin-4-yl}-urea.](/UserFilesUpload/Uses of Allantoin.jpg)
When you are dealing with this chemical, you should be very careful. This chemical is inflammation to the skin, eyes and respiratory system or other mucous membranes. It has serious damage to eyes. If swallowed, it's harmful to health. Therefore, you should wear suitable protective clothing. The gas can not be breathed and you should avoid contacting with skin and eyes. In case of contacting with eyes, you should rinse immediately with plenty of water and seek medical advice.
You can still convert the following datas into molecular structure:
(1) SMILES: C1(C(=O)NC(=O)N1)NC(=O)N
(2) InChI: InChI=1S/C4H6N4O3/c5-3(10)6-1-2(9)8-4(11)7-1/h1H,(H3,5,6,10)(H2,7,8,9,11)
(3) InChIKey: POJWUDADGALRAB-UHFFFAOYSA-N
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View