-
Name
Calcium lactate
- EINECS 212-406-7
- CAS No. 814-80-2
- Article Data18
- CAS DataBase
- Density
- Solubility
- Melting Point
- Formula C6H10CaO6
- Boiling Point 227.6 °C at 760 mmHg
- Molecular Weight 218.22
- Flash Point 109.9 °C
- Transport Information
- Appearance White powder.
- Safety 26-36/37/39
- Risk Codes 36/37/38
-
Molecular Structure
- Hazard Symbols
- Synonyms Lacticacid, calcium salt (2:1) (8CI);Calcium DL-lactate;Propanoicacid, 2-hydroxy-, calcium salt (2:1);Calphosan;Conclyte Ca;E 327;L-Cal;Pationic 1230;Pationic 1240;Prequist Powder;Puracal PP/FCC;Puracal PP/USP;Tokal;Calcium Lactate FCC/USP;
- PSA 120.72000
- LogP -3.76580
Synthetic route

| Conditions | Yield |
|---|---|
| In not given heating 3 h 25% saccharose soln. with CaO in excess at 197°C;; | 65% |
| In not given |

| Conditions | Yield |
|---|---|
| In not given at 215°C and 30 at;; addn. of CaO in autoclave and saturation in heat at pH = 8.2; treatment with active carbon; concg.;; | 45% |
| In not given at 215°C and 30 at;; addn. of CaO in autoclave and saturation in heat at pH = 8.2; treatment with active carbon; concg.;; | 45% |


| Conditions | Yield |
|---|---|
| With water; C25H37IrN6(2+)*2F6P(1-); calcium hydroxide at 180℃; for 20h; Catalytic behavior; Reagent/catalyst; Autoclave; | 18% |
-
-
10326-41-7
D-Lactic acid

-
-
814-80-2
calcium lactate

| Conditions | Yield |
|---|---|
| With calcium hydroxide In water |
-
-
74-90-8
hydrogen cyanide

-
-
7664-93-9
sulfuric acid

-
-
75-07-0
acetaldehyde

-
A
-
7664-41-7
ammonia

-
B
-
814-80-2
calcium lactate

| Conditions | Yield |
|---|---|
| In sodium hydroxide reaction of 98% acetaldehyde in NaOH with HCN; heating soln with 50% H2SO4 and excess of Ca(OH)2; filtration in heat; cooling down;; |


| Conditions | Yield |
|---|---|
| In not given heating equimolar mixture at 70°C; reaction takes place in autoclave;; ultrapure;; | |
| In not given heating equimolar mixture at 70°C; reaction takes place in autoclave;; ultrapure;; |

| Conditions | Yield |
|---|---|
| In not given fermenting 13% glucose soln. in presence of CaCO3;; | |
| In not given fermenting 13% glucose soln. in presence of CaCO3;; |
-
-
849585-22-4
LACTIC ACID

-
-
814-80-2
calcium lactate

| Conditions | Yield |
|---|---|
| With water; calcium oxide In glycerol Product distribution / selectivity; | |
| With calcium carbonate In ethanol; water for 0.166667h; Sonication; |

| Conditions | Yield |
|---|---|
| With copper(II) oxide In water at 230℃; for 0.5h; Sealed tube; Inert atmosphere; Green chemistry; |

| Conditions | Yield |
|---|---|
| Stage #1: D,L-lactide In water at 130 - 170℃; for 2.83333h; Sealed tube; Stage #2: With calcium carbonate In water for 0.166667h; Reagent/catalyst; Temperature; Heating; |

| Conditions | Yield |
|---|---|
| With ethanol; sulfuric acid at 75℃; for 1h; | A 86.6% B n/a |

| Conditions | Yield |
|---|---|
| With carbon dioxide at 180℃; for 7.37h; Temperature; Sealed tube; Green chemistry; | 58.22% |
| With sulfuric acid at 60 - 80℃; bei pH 1-1.4; |

| Conditions | Yield |
|---|---|
| With carbon dioxide at 180℃; Temperature; Sealed tube; Green chemistry; | 46.5% |
| With sulfuric acid at 100℃; bei pH 1 bis 1.4; |

| Conditions | Yield |
|---|---|
| With bis(acetylacetonato)dioxidomolybdenum(VI) In water at 200 - 270℃; Inert atmosphere; | 20% |

| Conditions | Yield |
|---|---|
| With sulfuric acid |

| Conditions | Yield |
|---|---|
| With sulfuric acid; benzene unter azeotropem Abdestillieren des Reaktionswassers; | |
| With sulfuric acid; toluene unter azeotropem Abdestillieren des Reaktionswassers; |

| Conditions | Yield |
|---|---|
| With sulfuric acid; benzene unter azeotropem Abdestillieren des Reaktionswassers; | |
| With sulfuric acid; toluene unter azeotropem Abdestillieren des Reaktionswassers; |

| Conditions | Yield |
|---|---|
| With sulfuric acid; benzene unter azeotropem Abdestillieren des Reaktionswassers; | |
| With sulfuric acid; toluene unter azeotropem Abdestillieren des Reaktionswassers; |

| Conditions | Yield |
|---|---|
| With sulfuric acid; toluene unter azeotropem Abdestillieren des Reaktionswassers; | |
| With sulfuric acid; benzene unter azeotropem Abdestillieren des Reaktionswassers; |

| Conditions | Yield |
|---|---|
| With water at 160℃; |

| Conditions | Yield |
|---|---|
| With sulfuric acid; benzene unter azeotropem Abdestillieren des Reaktionswassers; | |
| With sulfuric acid; toluene unter azeotropem Abdestillieren des Reaktionswassers; |
-
-
563-17-7
potassium ethyl sulfate

-
-
814-80-2
calcium lactate

-
-
97-64-3, 2676-33-7
ethyl 2-hydroxypropionate

| Conditions | Yield |
|---|---|
| bei der Einw. verschiedener Hefen; |
-
-
814-80-2
calcium lactate

-
-
849585-22-4
LACTIC ACID

| Conditions | Yield |
|---|---|
| With sulfuric acid In water at 72 - 75℃; for 0.75h; pH=2.0; Purification / work up; | |
| With sulfuric acid In water at 30℃; for 2h; | |
| With sulfuric acid; ammonium chloride In water | |
| With sulfuric acid |
-
-
814-80-2
calcium lactate

-
-
19201-34-4
1,1'-bis(1,1'-carboxyethyl) ether

| Conditions | Yield |
|---|---|
| at 280℃; |
-
-
814-80-2
calcium lactate

-
-
513-86-0, 52217-02-4
3-hydroxy-2-butanon

| Conditions | Yield |
|---|---|
| Einw. von Bacillus subtilis; |

| Conditions | Yield |
|---|---|
| With calcium hydroxide |

| Conditions | Yield |
|---|---|
| bei der Vergaerung durch eine Mischkultur von zwei nicht bezeichneten Bakterien; |

| Conditions | Yield |
|---|---|
| bei der Vergaerung durch Bakterien der Gruppe Bacillus subtilis; substances of butanediol-(2.3), whose stereochemical homogeneity and configuration is unknown; |

| Conditions | Yield |
|---|---|
| at 60℃; |

| Conditions | Yield |
|---|---|
| With sulfuric acid |

| Conditions | Yield |
|---|---|
| With sulfuric acid; benzene unter azeotropem Abdestillieren des Reaktionswassers; | |
| With sulfuric acid; toluene unter azeotropem Abdestillieren des Reaktionswassers; |

| Conditions | Yield |
|---|---|
| With sulfuric acid; benzene unter azeotropem Abdestillieren des Reaktionswassers; | |
| With sulfuric acid; toluene unter azeotropem Abdestillieren des Reaktionswassers; |

| Conditions | Yield |
|---|---|
| In water at 83 - 95℃; for 1.03333 - 1.25h; |
-
-
366491-15-8
1-butyl-3-methyl-1H-imidazol-3-iumhydrogencarbonate

-
-
814-80-2
calcium lactate

-
-
797050-71-6
1-butyl-3-methyl-3H-imidazol-1-ium lactate

| Conditions | Yield |
|---|---|
| Heating / reflux; |

| Conditions | Yield |
|---|---|
| Stage #1: tributyl-amine; calcium lactate With acetic acid In water Stage #2: carbon dioxide In water under 760.051 Torr; for 0.5h; Product distribution / selectivity; |

Calcium lactate Chemical Properties
Structure of Calcium lactate (CAS NO.814-80-2):
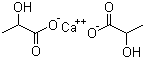
IUPAC Name: calcium 2-hydroxypropanoate
Empirical Formula: C6H10CaO6
Molecular Weight: 218.218
EINECS: 212-406-7
Flash Point: 109.9 °C
Enthalpy of Vaporization: 53.96 kJ/mol
Boiling Point: 227.6 °C at 760 mmHg
Vapour Pressure: 0.015 mmHg at 25°C
Product Categories: Nutritional fortification substances
Synonyms of Calcium lactate (CAS NO.814-80-2): 2-Hydroxypropanoic acid calcium salt ; Calcium 2-hydroxypropanoate (1:2) ; Calphosan ; Conclyte calcium ; Hemicalcium L-lactate ; Lactic acid, calcium salt (2:1) ; Propanoic acid, 2-hydroxy-, calcium salt (2:1)
Calcium lactate Uses
Calcium lactate (CAS NO.814-80-2) is used in foods (as a baking powder) and given medicinally. the E number is E327.
1.When Calcium lactate (CAS NO.814-80-2) is added to chewing gum containing xylitol, it will increases the remineralization of tooth enamel.
Calcium lactate (CAS NO.814-80-2) is also added to fresh-cut fruits to keep them firm and extend their shelf life, such as cantaloupes.
2.In medicine, Calcium lactate (CAS NO.814-80-2) is most commonly used as an antacid,also used to treat Calcium deficiencies. Calcium lactate (CAS NO.814-80-2) can be absorbed at various PHs ,so it does not need to be taken with food for absorption.
Calcium lactate Production
Calcium lactate (CAS NO.814-80-2) is manufactured by the reaction of calcium carbonate (CaCO3) or hydroxide [Ca(OH)2] with lactic acid.
Ca(OH)2 + HOCH2CH2CO2H → Ca(OCOCH2CH2OH)2
Calcium lactate Toxicity Data With Reference
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| dog | LDLo | intravenous | 80mg/kg (80mg/kg) | International Journal of Toxicology. Vol. 17(Suppl, | |
| mouse | LDLo | intravenous | 140mg/kg (140mg/kg) | Journal of the American Pharmaceutical Association. Vol. 27, Pg. 484, 1938. | |
| rabbit | LDLo | intravenous | 180mg/kg (180mg/kg) | International Journal of Toxicology. Vol. 17(Suppl, |
Calcium lactate Safety Profile
Risk Statements: 36/37/38
R36/37/38:Irritating to eyes, respiratory system and skin.
Safety Statements: 26-36/37/39
S26: In case of contact with eyes, rinse immediately with plenty of water and seek medical advice.
S36/37/39:Wear suitable protective clothing, gloves and eye/face protection.
TSCA Yes
Related Products
- Calcium
- Calcium (1)-bis(2-hydroxy-4-methylvalerate)
- Calcium (2S)-2-[(4-chlorobenzoyl)amino]-3-(1H-indol-3-yl)propanoate
- Calcium 2,4-dichlorophenolate
- Calcium 2-ethylhexanoate
- Calcium 2-hydroxypropanoate
- Calcium 2-methoxyethoxide
- Calcium 2-oxoglutarate
- Calcium 3-methyl-2-oxovalerate
- Calcium 4-aminosalicylate
- 81-48-1
- 81486-17-1
- 81486-22-8
- 81486-91-1
- 81487-29-8
- 814-88-0
- 814-89-1
- 81490-05-3
- 814-90-4
- 814-91-5
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View