-
Name
Cyclohexanemethanol
- EINECS 202-857-8
- CAS No. 100-49-2
- Article Data408
- CAS DataBase
- Density 0.913 g/cm3
- Solubility
- Melting Point -43oC
- Formula C7H14O
- Boiling Point 181.1 °C at 760 mmHg
- Molecular Weight 114.188
- Flash Point 71.1 °C
- Transport Information
- Appearance Colorless liquid
- Safety 23-24/25
- Risk Codes
-
Molecular Structure
- Hazard Symbols
- Synonyms (Hydroxymethyl)cyclohexane;Cyclohexanecarbinol;Cyclohexylcarbinol;Cyclohexylmethylalcohol;NSC 5288;
- PSA 20.23000
- LogP 1.55900
Synthetic route

| Conditions | Yield |
|---|---|
| With hydrogen In water at 100℃; under 15001.5 Torr; for 1h; | 100% |
| With hydrogen In water at 100℃; under 22502.3 Torr; for 3h; | 99% |
| With hydrogen; tetra(n-butyl)ammonium hydrogensulfate; rhodium colloidal catalyst In water at 36℃; under 180018 Torr; for 62h; pH=7.5; Catalytic hydrogenation; | 73% |

| Conditions | Yield |
|---|---|
| With sodium tetrahydroborate In methanol at 0 - 25℃; for 2h; | 100% |
| With isopropyl alcohol; zirconium(IV) oxide for 6h; Heating; | 99% |
| With isopropyl alcohol; zirconium(IV) oxide for 6h; Rate constant; Heating; | 99% |

| Conditions | Yield |
|---|---|
| With hydrogen In water at 100℃; under 15001.5 Torr; for 1h; | 100% |
| With hydrogen In water at 75℃; under 22502.3 Torr; for 10h; Autoclave; Sealed tube; Ionic liquid; chemoselective reaction; | 100% |
| With hydrogen at 180℃; under 150015 Torr; for 10h; Conversion of starting material; | |
| With hydrogen In water at 30℃; under 22502.3 Torr; for 1h; Autoclave; chemoselective reaction; | |
| With hydrogen In water at 30℃; for 19h; Autoclave; |

| Conditions | Yield |
|---|---|
| With samarium diiodide; hexanal; samarium(III) trifluoromethanesulfonate In tetrahydrofuran; methanol; potassium hydroxide at 20℃; for 0.075h; Reduction; | 99% |
| With sodium tetrahydroborate; titanium tetrachloride In 1,2-dimethoxyethane for 14h; Ambient temperature; | 94% |
| With hydrogen; Rh/Al2O3; molybdenum hexacarbonyl In 1,2-dimethoxyethane at 150℃; under 76000 Torr; for 16h; | 93% |
-
-
88773-82-4
(tetrahydropyranoxymethyl)-cyclohexane

-
-
100-49-2
cyclohexylmethyl alcohol

| Conditions | Yield |
|---|---|
| Nafion-H In methanol for 4h; | 99% |

| Conditions | Yield |
|---|---|
| With hydrogen; Rh/Al2O3; molybdenum hexacarbonyl In 1,2-dimethoxyethane at 150℃; under 76000 Torr; for 16h; | 99% |
| With hydrogen In hexane at 130℃; under 15001.5 Torr; for 18h; Molecular sieve; chemoselective reaction; | 72 %Chromat. |
| With hydrogen In water at 220℃; under 37503.8 Torr; for 24h; Autoclave; | 94.6 %Chromat. |

| Conditions | Yield |
|---|---|
| With methanol; potassium permanganate; trimethylsulphonium iodide at 25℃; under 760.051 Torr; chemoselective reaction; | 99% |
| With ((N-((6-((di-tert-butylphosphino)methyl)pyridin-2-yl)methyl)-2-methylpropan-2-amine))CoCl2; potassium tert-butylate; hydrogen; sodium triethylborohydride In tetrahydrofuran at 130℃; under 37503.8 Torr; for 48h; Inert atmosphere; Autoclave; High pressure; | 51 %Chromat. |
| With C21H35BrMnN2O2P; hydrogen; potassium hydride; 1,3,5-trimethyl-benzene In toluene at 100℃; under 15001.5 Torr; for 43h; Autoclave; Inert atmosphere; | 99 %Spectr. |

| Conditions | Yield |
|---|---|
| With C66H102N4OP2Ru; hydrogen In toluene at 105℃; under 22502.3 Torr; for 20h; Inert atmosphere; Glovebox; | A n/a B 99% |
| With C30H37ClN4ORu; hydrogen; sodium t-butanolate In toluene at 105℃; under 4500.45 Torr; for 20h; Glovebox; Sealed tube; Overall yield = 99 %; |

| Conditions | Yield |
|---|---|
| With C66H102N4OP2Ru; hydrogen In toluene at 105℃; under 22502.3 Torr; for 20h; Inert atmosphere; Glovebox; | A n/a B 99% |
| With C30H37ClN4ORu; hydrogen; sodium t-butanolate In toluene at 105℃; under 4500.45 Torr; for 20h; Glovebox; Sealed tube; Overall yield = >99 %; |
-
-
18448-47-0
methyl cyclohex-1-ene-1-carboxylate

-
-
100-49-2
cyclohexylmethyl alcohol

| Conditions | Yield |
|---|---|
| With C13H34BFeNOP2; hydrogen In tetrahydrofuran at 100℃; under 22502.3 Torr; for 18h; Autoclave; Inert atmosphere; | 98% |
| With [bis({2‐[bis(propan‐2‐yl)phosphanyl]ethyl})amine](borohydride)(carbonyl)(hydride)iron(II); hydrogen In tetrahydrofuran at 120℃; under 22502.3 Torr; for 19h; Autoclave; | 85 %Chromat. |
-
-
103668-33-3, 3309-97-5
3-hydroxymethylcyclohexene

-
-
100-49-2
cyclohexylmethyl alcohol

| Conditions | Yield |
|---|---|
| With hydrogen; nickel In ethanol at 20℃; under 18240 Torr; for 1.5h; | 97% |
| With formic acid In water at 140℃; under 3750.38 Torr; for 4h; Inert atmosphere; |
-
-
62-53-3
aniline

-
-
2043-61-0
cyclohexanecarbaldehyde

-
A
-
79952-92-4
N-(cyclohexylmethyl)aniline

-
B
-
100-49-2
cyclohexylmethyl alcohol

| Conditions | Yield |
|---|---|
| Stage #1: aniline; cyclohexanecarbaldehyde at 25℃; for 0.166667h; Stage #2: With sodium tetrahydroborate; benzoic acid at 25℃; for 0.333333h; | A 97% B n/a |
| Stage #1: cyclohexanecarbaldehyde at 40℃; for 0.166667h; Stage #2: aniline With sodium tetrahydroborate at 40℃; for 3h; Stage #3: With methanol at 0℃; for 0.5h; chemoselective reaction; |

-
-
58587-03-4
S-phenyl cyclohexanecarbothioate

-
-
100-49-2
cyclohexylmethyl alcohol

| Conditions | Yield |
|---|---|
| With sodium tetrahydroborate In ethanol for 19h; Ambient temperature; | 96% |

| Conditions | Yield |
|---|---|
| With butyltriphenylphosphonium tetrahydroborate at 20℃; for 0.0333333h; | 96% |
| With zinc(II) tetrahydroborate; N,N,N,N,-tetramethylethylenediamine In tetrahydrofuran; diethyl ether at 0℃; for 5h; | 93% |
| With zinc(II) tetrahydroborate; N,N,N,N,-tetramethylethylenediamine In diethyl ether at 0℃; for 6h; | 93% |
| Multi-step reaction with 2 steps 1: triethylamine / dichloromethane / 20 °C / Inert atmosphere 2: sodium 2-methyl-2-adamantoxide; sodium hydride; dichlorobis(dicyclohexylphosphinomethylpyridine)-ruthenium (II); hydrogen / toluene; mineral oil / 48 h / 160 °C / 60006 Torr / Inert atmosphere; Autoclave View Scheme |

| Conditions | Yield |
|---|---|
| With dimethylsulfide borane complex In 2-methyltetrahydrofuran at 90℃; under 7500.75 Torr; for 0.333333h; Inert atmosphere; Flow reactor; | 95% |
| Stage #1: methyl cyclohexylcarboxylate With 3-phenyl-propionaldehyde; diethylaluminum benzenethiolate In hexane; toluene at -78℃; for 1.08333h; Inert atmosphere; Stage #2: With diisobutylaluminium hydride In hexane; toluene at -78 - 0℃; Inert atmosphere; Stage #3: With hydrogenchloride In hexane; water; toluene at 0℃; chemoselective reaction; | 93% |
| With C18H28Br2N4Ru; potassium tert-butylate; hydrogen In 1,4-dioxane at 105℃; under 22502.3 Torr; for 8h; | 93% |
-
-
164071-20-9
cyclohexylmethyl trifluoroacetate

-
A
-
75-89-8
2,2,2-trifluoroethanol

-
B
-
100-49-2
cyclohexylmethyl alcohol

| Conditions | Yield |
|---|---|
| With trans-[(2,6-bis(di-tert-butylphosphinomethyl)pyridine)Fe(H)2(CO)]; hydrogen; sodium methylate In 1,4-dioxane at 40℃; under 18751.9 Torr; for 48h; Glovebox; Inert atmosphere; | A 95% B n/a |
| With trimethylamine-N-oxide; tricarbonyl(η4-1,3-bis(trimethylsilyl)-4,5,6,7-tetrahydro-2H-inden-2-one)iron; hydrogen In toluene at 110℃; under 52505.3 Torr; for 17h; Catalytic behavior; Solvent; Temperature; Inert atmosphere; Glovebox; |
-
-
104-94-9
4-methoxy-aniline

-
-
2043-61-0
cyclohexanecarbaldehyde

-
A
-
6709-45-1
N-cyclohexylmethyl-p-anisidine

-
B
-
100-49-2
cyclohexylmethyl alcohol

| Conditions | Yield |
|---|---|
| Stage #1: 4-methoxy-aniline; cyclohexanecarbaldehyde at 25℃; for 0.166667h; Stage #2: With sodium tetrahydroborate; boric acid at 25℃; for 0.333333h; | A 94% B n/a |


-
-
100-49-2
cyclohexylmethyl alcohol

| Conditions | Yield |
|---|---|
| With sodium hydroxide In water Inert atmosphere; | 94% |

| Conditions | Yield |
|---|---|
| With ethylmagnesium bromide; poly(methylhydrosiloxane); bis(cyclopentadienyl)titanium dichloride In tetrahydrofuran; diethyl ether for 5h; Ambient temperature; | 93% |
| With lithium borohydride In diethyl ether; toluene at 100℃; for 1h; | 91% |
| With potassium borohydride; hafnium tetrachloride In tetrahydrofuran at 40℃; for 10.5h; Inert atmosphere; Cooling with ice; | 86% |
-
-
72581-32-9, 1679-51-2
cyclohex-3-enylmethanol

-
-
100-49-2
cyclohexylmethyl alcohol

| Conditions | Yield |
|---|---|
| Stage #1: cyclohex-3-enylmethanol With pyridinium p-toluenesulfonate In 1,2-dichloro-ethane at 80℃; for 20h; Stage #2: With triethylamine In dichloromethane at 20℃; for 6h; Stage #3: With trifluoroacetic acid In methanol; dichloromethane at 140℃; for 0.0666667h; microwave irradiation; Further stages.; | 93% |
| Multi-step reaction with 2 steps 1: 66 percent / 4-dimethylaminopyridine; Et3N / CH2Cl2 / 24 h / 20 °C 2: 74 percent / H2 / Pd/C / methanol / 24 h / 20 °C View Scheme |

| Conditions | Yield |
|---|---|
| With (carbonyl)(chloro)(hydrido)tris(triphenylphosphine)ruthenium(II); water; hydrogen In 1,4-dioxane at 140℃; under 7500.75 Torr; for 18h; Autoclave; | 93% |
| With formaldehyd; [ruthenium(II)(η6-1-methyl-4-isopropyl-benzene)(chloride)(μ-chloride)]2 In water; toluene at 90℃; | 87% |

| Conditions | Yield |
|---|---|
| With mer-[(1,4-bis(diphenylphosphino)butane)aquatrichlororuthenium(III)]; hydrogen In tetrahydrofuran at 160℃; under 67506.8 Torr; for 24h; Catalytic behavior; | 92% |
| With dodecacarbonyl-triangulo-triruthenium; 2-(dicyclohexylphosphino)-1-methyl-1H-imidazole; water; hydrogen; lithium chloride In 1-methyl-pyrrolidin-2-one at 130℃; under 60 Torr; for 20h; Autoclave; regioselective reaction; | 76% |
| With [bis(2-methylallyl)cycloocta-1,5-diene]ruthenium(II); 2-(dicyclohexylphosphanyl)-1-(2-methoxyphenyl)-1H-imidazole; hydrogen In toluene at 160℃; under 37503.8 Torr; for 24h; Autoclave; Inert atmosphere; regioselective reaction; | 51% |
| With hydrogen; cobalt(II) acetylacetonate dihydrate at 99.9℃; under 84756.7 Torr; for 7h; |
-
-
1122-56-1
cyclohexylcarboxamide

-
A
-
3218-02-8
cyclohexylmethylamine

-
B
-
100-49-2
cyclohexylmethyl alcohol

| Conditions | Yield |
|---|---|
| With hydrogen at 160℃; under 75007.5 Torr; | A 91% B 8% |
| With dodecacarbonyl-triangulo-triruthenium; hydrogen; molybdenum hexacarbonyl In 1,2-dimethoxyethane at 160℃; under 75007.5 Torr; for 16h; Inert atmosphere; | |
| With hydrogen In 1,2-dimethoxyethane at 160℃; under 75007.5 Torr; for 16h; | |
| With 5 wt% ruthenium/carbon; water; hydrogen at 59.84℃; under 60006 Torr; for 4h; Reagent/catalyst; Autoclave; Sealed tube; |

| Conditions | Yield |
|---|---|
| With C24H20ClN2OPRu; potassium tert-butylate; hydrogen In tetrahydrofuran at 110℃; under 10640.7 Torr; for 36h; Inert atmosphere; Schlenk technique; | 91% |
| With hydrogen In toluene at 160℃; under 45004.5 Torr; for 15h; Catalytic behavior; Autoclave; | 76% |
| With water; hydrogen at 59.84℃; under 60006 Torr; for 48h; Reagent/catalyst; Temperature; Concentration; Pressure; Autoclave; Sealed tube; | |
| Multi-step reaction with 2 steps 1: hydrogen; water / 4 h / 59.84 °C / 60006 Torr / Autoclave; Sealed tube 2: hydrogen / water / 24 h / 59.84 °C / 60006 Torr View Scheme |
-
-
54829-38-8
S-benzyl cyclohexanecarbothioate

-
-
100-49-2
cyclohexylmethyl alcohol

| Conditions | Yield |
|---|---|
| With sodium tetrahydroborate In ethanol for 24h; Ambient temperature; | 90% |
-
-
88773-80-2
cyclohexylmethyl trimethylsilyl ether

-
-
100-49-2
cyclohexylmethyl alcohol

| Conditions | Yield |
|---|---|
| With Nafion-H(R); silica gel In hexane at 20℃; for 0.333333h; | 90% |

| Conditions | Yield |
|---|---|
| With C30H26Cl2N3PRu; hydrogen; sodium ethanolate In toluene at 80℃; under 38002.6 Torr; for 16h; Catalytic behavior; Autoclave; Inert atmosphere; Schlenk technique; | 90% |
-
-
93-58-3
benzoic acid methyl ester

-
A
-
4630-82-4
methyl cyclohexylcarboxylate

-
B
-
100-49-2
cyclohexylmethyl alcohol

-
C
-
100-51-6
benzyl alcohol

| Conditions | Yield |
|---|---|
| With hydrogen; Aliquat 336; rhodium(III) chloride In water; 1,2-dichloro-ethane at 30℃; under 760 Torr; for 5h; | A 89% B 10 % Chromat. C 1% |
| With hydrogen; Aliquat 336; rhodium(III) chloride In water; 1,2-dichloro-ethane at 30℃; under 760 Torr; for 5h; | A 89 % Chromat. B 10% C 1% |


| Conditions | Yield |
|---|---|
| Stage #1: carbon monoxide; Cyclohexyl iodide With diethoxymethylane; [CuCl(IPrMe)]; lithium methanolate In tetrahydrofuran at 60℃; under 2280.15 Torr; for 16h; Sealed tube; Stage #2: With tetrabutyl ammonium fluoride In tetrahydrofuran; diethyl ether at 20℃; for 2h; | 89% |
| With 2,2'-azobis(isobutyronitrile); triphenylgermane; sodium cyanoborohydride In tetrahydrofuran; benzene at 105℃; under 72400.7 Torr; | 62% |
| With tetrabutylammonium borohydride In acetonitrile at 25℃; under 760.051 Torr; for 3h; UV-irradiation; | 36% |
| With tetrabutylammonium tricarbonylnitrosyl ferrate; sodium trimethoxyborohydride In tetrahydrofuran at 50℃; under 3040 Torr; Product distribution; |

| Conditions | Yield |
|---|---|
| With 2,2,6,6-tetramethyl-piperidine-N-oxyl; 1-(diacetoxyiodo)-4-methylbenzene In chloroform at 20℃; for 24h; | 100% |
| With 2,2,6,6-tetramethyl-piperidine-N-oxyl; oxygen; copper(I) bromide dimethylsulfide complex In chlorobenzene at 80℃; for 8h; | 99% |
| With sodium hydrogencarbonate; sodium bromide In dichloromethane at 20℃; Electrochemical reaction; | 99% |

| Conditions | Yield |
|---|---|
| With peracetic acid; C24H29INO5 In acetic acid at 30℃; for 24h; | 100% |
| With 4-acetylamino-2,2,6,6-tetramethylpiperidine-1-oxoammonium tetrafluoroborate In water; acetonitrile at 20℃; | 96% |
| With sodium bromate; 4 In water at 60℃; for 15h; | 95% |

| Conditions | Yield |
|---|---|
| With trimethylsilyl bromide; 4 A molecular sieve; tetrabutylammomium bromide; cobalt(II) bromide In dichloromethane at 22 - 28℃; for 16h; Product distribution; other alcohols; var. time and reaction conditions; | 100% |
| With trimethylsilyl bromide; 4 A molecular sieve; tetrabutylammomium bromide; cobalt(II) bromide In dichloromethane at 22 - 28℃; for 16h; | 100% |

| Conditions | Yield |
|---|---|
| With oxygen at 60℃; under 760.051 Torr; for 24h; Irradiation; | 100% |
| With iodine; potassium carbonate In tert-butyl alcohol at 20℃; for 27h; | 93% |
| With pyridinium hydrobromide perbromide In water at 20℃; for 14h; | 92% |
-
-
124072-61-3
N-t-butoxycarbonyl-N-methyl-β-alanine

-
-
100-49-2
cyclohexylmethyl alcohol

-
-
100-51-6
benzyl alcohol

-
-
654651-68-0
Boc-MeβAla-OCH2cHex

| Conditions | Yield |
|---|---|
| With dmap; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride In dichloromethane at 0 - 20℃; | 100% |

-
-
74124-79-1
di(succinimido) carbonate

-
-
100-49-2
cyclohexylmethyl alcohol

-
-
922723-33-9
N-succinimidyl carbonic acid cyclohexylmethyl ester

| Conditions | Yield |
|---|---|
| With triethylamine In dichloromethane at 20℃; | 100% |

| Conditions | Yield |
|---|---|
| With dmap; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride; N,N-dimethyl-formamide at 20 - 50℃; Inert atmosphere; | 100% |
-
-
1224680-14-1
(S)-4-(3-(tert-butyldimethylsilyloxy)phenyl)-6-ethyl-2-oxo-1,2,3,4-tetrahydropyrimidin-5-carboxylic acid

-
-
100-49-2
cyclohexylmethyl alcohol

-
-
1224680-16-3
C26H40N2O4Si

| Conditions | Yield |
|---|---|
| With dmap; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride In N,N-dimethyl-formamide at 50℃; Inert atmosphere; | 100% |

| Conditions | Yield |
|---|---|
| With dmap; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride In N,N-dimethyl-formamide at 50℃; Inert atmosphere; | 100% |
-
-
146176-60-5
2,2,4,4-tetramethyl-1,3-oxazolidine-3-carbonyl chloride

-
-
100-49-2
cyclohexylmethyl alcohol

| Conditions | Yield |
|---|---|
| Stage #1: cyclohexylmethyl alcohol With sodium hydride In tetrahydrofuran; mineral oil at 20℃; for 0.5h; Inert atmosphere; Stage #2: 2,2,4,4-tetramethyl-1,3-oxazolidine-3-carbonyl chloride In tetrahydrofuran; mineral oil at 20℃; for 12h; Inert atmosphere; | 100% |
-
-
116922-60-2
3-bromo-4-fluoropyridine

-
-
100-49-2
cyclohexylmethyl alcohol

| Conditions | Yield |
|---|---|
| Stage #1: cyclohexylmethyl alcohol With potassium tert-butylate In dimethyl sulfoxide at 20℃; for 0.75h; Stage #2: 3-bromo-4-fluoropyridine In dimethyl sulfoxide for 3h; | 100% |
-
-
124-63-0
methanesulfonyl chloride

-
-
100-49-2
cyclohexylmethyl alcohol

-
-
14100-97-1
cyclohexylmethyl methanesulfonate

| Conditions | Yield |
|---|---|
| With triethylamine In dichloromethane at 0 - 20℃; for 6h; Inert atmosphere; | 99% |
| With triethylamine In dichloromethane at 0℃; for 16h; | 96% |
| With triethylamine In dichloromethane at 0℃; for 0.5h; Inert atmosphere; | 96% |
-
-
130427-27-9
(1S,3S,4S,7R,8R)-7,8-Bis-benzyloxy-3-phenylsulfanyl-2-oxa-5-thia-bicyclo[2.2.2]octane

-
-
100-49-2
cyclohexylmethyl alcohol

-
-
130427-34-8
(1S,3S,4S,7R,8R)-7,8-Bis-benzyloxy-3-cyclohexylmethoxy-2-oxa-5-thia-bicyclo[2.2.2]octane

| Conditions | Yield |
|---|---|
| With N-Bromosuccinimide In dichloromethane at -40℃; for 0.166667h; MS 4A; | 99% |
-
-
139190-72-0, 139190-74-2, 139190-75-3
Phenyl 2-deoxy-3,4-O-isopropylidene-1-thio-D-fucopyranoside

-
-
100-49-2
cyclohexylmethyl alcohol

-
-
139190-77-5
(3aS,4R,6S,7aR)-6-Cyclohexylmethoxy-2,2,4-trimethyl-tetrahydro-[1,3]dioxolo[4,5-c]pyran

| Conditions | Yield |
|---|---|
| With N-Bromosuccinimide In dichloromethane a.) 0 deg C, 1 h, b.) 25 deg C, 16 h; | 99% |
| With N-Bromosuccinimide In dichloromethane a.) 0 deg C, 1 h, b.) 25 deg C, 16 h; effect of conformational assistance of the glycosyl donor on stereoselective glycosylation, other glycosyl donors, other alcohols, other solvents; | 99% |

| Conditions | Yield |
|---|---|
| With Cp*2Sm(THF)2 In toluene for 0.000833333h; Ambient temperature; | 99% |
-
-
100-49-2
cyclohexylmethyl alcohol

-
-
357940-81-9
1-deoxy-2,3,4.6-tetrakis-O-phenylmethyl-1-phenylsulfinyl-mannopyranose

| Conditions | Yield |
|---|---|
| With 5 Angstroem MS; Sulfate; zirconium(IV) oxide In diethyl ether at 25℃; for 3h; | 99% |

| Conditions | Yield |
|---|---|
| In toluene for 3h; Heating; | 99% |

-
-
108-77-0
1,3,5-trichloro-2,4,6-triazine

-
-
100-49-2
cyclohexylmethyl alcohol

-
-
502767-34-2
2,4-dichloro-6-cyclohexylmethoxy-[1,3,5]triazine

| Conditions | Yield |
|---|---|
| With potassium hydrogencarbonate In toluene | 99% |
| With potassium hydrogencarbonate; 18-crown-6 ether In toluene | 99% |
| With potassium carbonate; 18-crown-6 ether In toluene for 18h; Heating / reflux; | 99% |
| With potassium hydrogencarbonate; 18-crown-6 ether In toluene for 18h; Heating / reflux; | |
| With potassium hydrogencarbonate; 18-crown-6 ether In toluene for 18h; Heating / reflux; |
-
-
70-55-3
toluene-4-sulfonamide

-
-
100-49-2
cyclohexylmethyl alcohol

-
-
86328-85-0
N-(cyclohexylmethyl)-4-methylbenzenesulfonamide

| Conditions | Yield |
|---|---|
| With potassium hydroxide In toluene at 130℃; for 96h; Inert atmosphere; | 99% |
| With bis[dichloro(pentamethylcyclopentadienyl)iridium(III)]; potassium tert-butylate In toluene for 17h; Inert atmosphere; Reflux; | 97% |
| With [(η5-C5Me5)Ir(6,6'-dihydroxy-2,2'-bipyridine)(H2O)]OTf2; caesium carbonate In water at 120℃; for 15h; Inert atmosphere; Schlenk technique; | 85% |

| Conditions | Yield |
|---|---|
| titanium (IV) oxide bis(2,4-pentanedionate) In xylene Reflux; | 99% |

| Conditions | Yield |
|---|---|
| With 2,4,6-tripropyl-1,3,5,2,4,6-trioxatriphosphinane-2,4,6-trioxide; N-ethyl-N,N-diisopropylamine In tetrahydrofuran; ethyl acetate; toluene at 20℃; for 4h; Inert atmosphere; | 99% |

| Conditions | Yield |
|---|---|
| With toluene-4-sulfonic acid In benzene for 24h; Reflux; | 99% |
Cyclohexanemethanol Consensus Reports
Reported in EPA TSCA Inventory.
Cyclohexanemethanol Specification
The Hexahydrobenzyl alcohol is an organic compound with the formula C7H14O. The IUPAC name of this chemical is cyclohexylmethanol. With the CAS registry number 100-49-2, it is also named as 1,2,3, 6-Tetrahydrobenzaldehyde. The product's category is Alcohol Aldehyde & Acid Series. Besides, it is a colorless liquid, which should be stored in a cool and ventilated place. It is an organic synthesis intermediate.
Physical properties about Hexahydrobenzyl alcohol are: (1)ACD/LogP: 1.87; (2)ACD/LogD (pH 5.5): 1.87; (3)ACD/LogD (pH 7.4): 1.87; (4)ACD/BCF (pH 5.5): 15.59; (5)ACD/BCF (pH 7.4): 15.59; (6)ACD/KOC (pH 5.5): 248.55; (7)ACD/KOC (pH 7.4): 248.55; (8)#H bond acceptors: 1; (9)#H bond donors: 1; (10)#Freely Rotating Bonds: 2; (11)Polar Surface Area: 9.23 Å2; (12)Index of Refraction: 1.454; (13)Molar Refractivity: 33.89 cm3; (14)Molar Volume: 125 cm3; (15)Polarizability: 13.43×10-24cm3; (16)Surface Tension: 33.7 dyne/cm; (17)Density: 0.913 g/cm3; (18)Flash Point: 71.1 °C; (19)Enthalpy of Vaporization: 48.58 kJ/mol; (20)Boiling Point: 181.1 °C at 760 mmHg; (21)Vapour Pressure: 0.254 mmHg at 25°C.
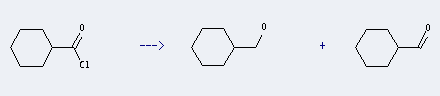
Preparation: this chemical can be prepared by cyclohexanecarbonyl chloride. This reaction is a kind of Reduction. It will need reagent Bu3SnH, Ph3P, catalyst InCl3 and solvent tetrahydrofuran. The reaction time is 2 hours with reaction temperature of 20 °C. The yield is about 62%.
Uses of Hexahydrobenzyl alcohol: it can be used to produce acetic acid cyclohexylmethyl ester at temperature of 25 - 30 °C. It will need reagent Woelm-200-N alumina with reaction time of 1 hour. The yield is about 85%.

You can still convert the following datas into molecular structure:
(1)SMILES: OCC1CCCCC1
(2)InChI: InChI=1/C7H14O/c8-6-7-4-2-1-3-5-7/h7-8H,1-6H2
(3)InChIKey: VSSAZBXXNIABDN-UHFFFAOYAW
(4)Std. InChI: InChI=1S/C7H14O/c8-6-7-4-2-1-3-5-7/h7-8H,1-6H2
(5)Std. InChIKey: VSSAZBXXNIABDN-UHFFFAOYSA-N
The toxicity data is as follows:
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| mouse | LD50 | intraperitoneal | 250mg/kg (250mg/kg) | National Technical Information Service. Vol. AD277-689, |
Related Products
- Cyclohexanemethanol
- Cyclohexanemethanol, 4-amino-, trans-
- Cyclohexanemethanol, a,a-dicyclohexyl-
- Cyclohexanemethanol, a-ethynyl-, 1-propanoate
- Cyclohexanemethanol, alpha,alpha,4-trimethyl-, 1-acetate
- Cyclohexanemethanol, α-butyl-
- Cyclohexanemethanol,1,2,2,3,3,4,4,5,5,6,6-undecafluoro-
- Cyclohexanemethanol,1-fluoro-
- Cyclohexanemethanol,2-[(phenylmethyl)amino]-, (1S,2R)-
- Cyclohexanemethanol,4,4-difluoro-
- 100492-04-4
- 10049-21-5
- 10049-24-8
- 10049-83-9
- 1004990-28-6
- 1005009-98-2
- 100501-57-3
- 100501-59-5
- 100501-62-0
- 1005030-66-9
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View