-
Name
Hexamethyldisilane
- EINECS 215-911-0
- CAS No. 1450-14-2
- Article Data134
- CAS DataBase
- Density 0.735 g/cm3
- Solubility Insoluble in water, soluble in acetone, benzene, ethyl ether and heptane
- Melting Point 9-12 °C(lit.)
- Formula C6H18Si2
- Boiling Point 113.5 °C at 760 mmHg
- Molecular Weight 146.38
- Flash Point 5.8 °C
- Transport Information UN 1993 3/PG 2
- Appearance Colorless clear liquid
- Safety 16-23-45-9-33-26-7/9
- Risk Codes 11-36/37-43-36/37/38
-
Molecular Structure
-
Hazard Symbols
 F,
F, Xi
Xi
- Synonyms Disilane, hexamethyl-(6CI,7CI,8CI,9CI);NSC 294211;Permethyldisilane;Hexamethyldisilane (HMD);
- PSA 0.00000
- LogP 2.74120
Synthetic route

| Conditions | Yield |
|---|---|
| With sodium; lithium In tetrahydrofuran at 25 - 28℃; for 4h; Heating; Irradiation; ultrasound irrad. var. time, var. temperatures, var. metal purity; | 100% |
| With sodium; lithium In tetrahydrofuran at 25 - 28℃; for 4h; Irradiation; | 100% |
| With potassium fluoride; phosphorus; potassium In 5,5-dimethyl-1,3-cyclohexadiene at 90℃; for 12h; Reagent/catalyst; Temperature; | 97.3% |

| Conditions | Yield |
|---|---|
| In diethyl ether Heating; | A 68% B 100% |

-
-
119-61-9
benzophenone

-
-
56859-17-7
(trimethylsilyl)potassium

-
A
-
1450-14-2
1,1,1,2,2,2-hexamethyldisilane

-
B
-
464-72-2
tetraphenylethane-1,2-diol

| Conditions | Yield |
|---|---|
| In 1,2-dimethoxyethane for 12h; Ambient temperature; | A 85% B 80% |

-
-
75-77-4
chloro-trimethyl-silane

-
A
-
1450-14-2
1,1,1,2,2,2-hexamethyldisilane

-
B
-
107-46-0
Hexamethyldisiloxane

| Conditions | Yield |
|---|---|
| With sodium at 130 - 140℃; for 10h; | A 84% B n/a |
-
-
74768-58-4
1,1,3,3-tetramethylindan-2-thione

-
-
79184-72-8
2,2-dimesitylhexamethyltrisilane

-
A
-
1450-14-2
1,1,1,2,2,2-hexamethyldisilane

-
B
-
110354-69-3
C31H38SSi

| Conditions | Yield |
|---|---|
| In cyclohexane for 15h; Ambient temperature; Irradiation; low pressure mercury lamp; | A n/a B 82% |


-
-
2290-65-5
trimethylsilyl isothiocyanate

-
A
-
75-77-4
chloro-trimethyl-silane

-
B
-
1450-14-2
1,1,1,2,2,2-hexamethyldisilane

| Conditions | Yield |
|---|---|
| In toluene Heating at 120-150°C for 10 h.; | A 73% B 32% C 76% |
| In toluene molar ratio V-complex/Me3SiNCS=1:2, evacuated sealed ampul (120-150°C, 10 h); | A 73% B 32% C 76% |

-
-
2700-22-3
Benzylidenemalononitrile

-
A
-
1450-14-2
1,1,1,2,2,2-hexamethyldisilane

-
B
-
1867-37-4
Benzylmalononitrile

| Conditions | Yield |
|---|---|
| With phenanthrene In acetonitrile for 43h; Irradiation; 300 W high pressure mercury lamp, λ>280 nm; | A 75% B 10% |

| Conditions | Yield |
|---|---|
| In diethyl ether Si2Cl6 and 31% excess of Zn(CH3)2 in ether at 85°C in a closed tube;; | 74% |

-
-
59147-01-2
trimethylsilyl 2-iodobenzoate

-
A
-
1450-14-2
1,1,1,2,2,2-hexamethyldisilane

| Conditions | Yield |
|---|---|
| In toluene at 20℃; for 60h; Sealed tube; | A n/a B 69% |

-
-
111655-78-8
bis(trimethylsilyl)diphenylgermane

-
-
513-81-5
2,3-dimethyl-buta-1,3-diene

-
A
-
1450-14-2
1,1,1,2,2,2-hexamethyldisilane

-
B
-
768-32-1
trimethylphenylsilane

-
C
-
5764-69-2
1,1-diphenyl-3,4-dimethyl-1-germacyclopent-3-ene

| Conditions | Yield |
|---|---|
| In pentane Irradiation (UV/VIS); (N2), 1 h irradiated; gas chromatography; elem. anal., NMR, mass. spectra; | A 68% B 27% C 61% |

-
-
75-77-4
chloro-trimethyl-silane

-
-
7677-24-9
trimethylsilyl cyanide

-
A
-
1450-14-2
1,1,1,2,2,2-hexamethyldisilane

-
B
-
72223-62-2
2-(Bis-trimethylsilanyl-methyl)-1,1,1,3,3,3-hexamethyl-disilazane

-
C
-
138482-32-3
2-(Bis-trimethylsilanyl-methyl)-1,1,3,3,3-pentamethyl-1-trimethylsilanylmethyl-disilazane

| Conditions | Yield |
|---|---|
| With lithium In tetrahydrofuran at 0℃; Yields of byproduct given; | A 65% B n/a C n/a |

-
-
134847-65-7
methyl(phenyl)bis(trimethylsilyl)germane

-
-
513-81-5
2,3-dimethyl-buta-1,3-diene

-
A
-
1450-14-2
1,1,1,2,2,2-hexamethyldisilane

-
B
-
134847-66-8
1,3,4-trimethyl-1-(trimethylsilyl)-1-germacyclopent-3-ene

-
C
-
134847-67-9
(2,3-dimethyl-3-butenyl)methyl(trimethylsilyl){o-(trimethylsilyl)phenyl}germane

-
D
-
768-32-1
trimethylphenylsilane

-
E
-
51533-82-5
1,3,4-trimethyl-1-phenyl-1-germacyclopent-3-ene

| Conditions | Yield |
|---|---|
| In pentane Irradiation (UV/VIS); (N2), 1.5 h irradiated; column chromatography; elem. anal., NMR, mass. spectra; | A 60% B 28% C 5% D 35% E 55% |


-
-
59147-01-2
trimethylsilyl 2-iodobenzoate

-
A
-
1450-14-2
1,1,1,2,2,2-hexamethyldisilane

-
B
-
1428262-40-1
η5-cyclopentadienevanadium bis-o-iodobenzoate

-
C
-
542-92-7
cyclopenta-1,3-diene

| Conditions | Yield |
|---|---|
| In toluene at 100℃; for 100h; Time; Sealed tube; | A n/a B 59% C n/a |

-
-
768-32-1
trimethylphenylsilane

-
-
17946-71-3
lithium trimethylstannyl

-
A
-
1450-14-2
1,1,1,2,2,2-hexamethyldisilane

-
B
-
934-56-5
trimethyl(phenyl)stannane

-
C
-
661-69-8
hexamethyldistannane

-
D
-
16393-88-7
trimethyl(trimethylstannyl)silane

-
E
-
71-43-2
benzene

| Conditions | Yield |
|---|---|
| In tetrahydrofuran LiSnMe3 added to PhSiMe3 in THF, stirred at 24°C for 1 h or 50°C for 30 min, hydrolysis with water; org. layer extd. with ether, dried over Na2SO4; products determined by GLC; | A 0% B 0% C 50% D 0% E 0% |

-
-
20932-80-3
1,1,1,4,4,4-hexakis(trimethylsilyl)butin-2

-
A
-
1450-14-2
1,1,1,2,2,2-hexamethyldisilane

-
B
-
6231-76-1
1,2-bis(trimethylsilyl)ethane

-
C
-
70641-35-9
1,1,4,4-tetrakis(trimethylsilyl) 1,2,3-butatriene

-
D
-
14630-40-1
Bis(trimethylsilyl)ethyne

| Conditions | Yield |
|---|---|
| at 650℃; under 0.001 Torr; Further byproducts given; | A 7% B 5% C 41% D 16% |

-
-
2117-28-4
bis(trimethylsilyl)methane

-
-
2154-71-4
bis-trifluoromethyl-aminooxyl

-
A
-
1450-14-2
1,1,1,2,2,2-hexamethyldisilane

-
B
-
420-56-4
trimethylsilyl fluoride

-
C
-
359-63-7
N,N-bis(trifluoromethyl)hydroxylamine

-
D
-
371-71-1
Perfluoro-2-azapropen

| Conditions | Yield |
|---|---|
| With bis(trimethylsilyl)methane for 4h; Ambient temperature; Further byproducts given; | A 15% B 40% C 33% D 31% |

-
-
2117-28-4
bis(trimethylsilyl)methane

-
-
2154-71-4
bis-trifluoromethyl-aminooxyl

-
A
-
1450-14-2
1,1,1,2,2,2-hexamethyldisilane

-
B
-
420-56-4
trimethylsilyl fluoride

-
C
-
359-63-7
N,N-bis(trifluoromethyl)hydroxylamine

-
D
-
20485-84-1
O-Trimethylsilyl-bis-(trifluormethyl)-hydroxylamin

| Conditions | Yield |
|---|---|
| for 4h; Ambient temperature; Further byproducts given; | A 15% B 40% C 33% D 21% |

-
-
37899-63-1
(dimethylphenylgermyl)trimethylsilane

-
A
-
1450-14-2
1,1,1,2,2,2-hexamethyldisilane

-
B
-
993-07-7
trimethylsilan

-
C
-
768-32-1
trimethylphenylsilane

-
D
-
7366-21-4
dimethylphenylgermane

-
E
-
22702-72-3
1,1,2,2-tetramethyl-1,2-diphenyldigermane

| Conditions | Yield |
|---|---|
| With methanol In benzene Irradiation (UV/VIS); irradiation of catenate in benzene containing an excess of methanol with a 110 W low pressure Hg arc lamp at room temp. under Ar for 1-4 h; identification by GC and GC-MS; | A <1 B n/a C 37% D <1 E <1 |
| In cyclohexane Irradiation (UV/VIS); irradiation of catenate in cyclohexane with a 110 W low pressure Hg arc lamp at room temp. under Ar for 1-4 h; identification by GC and GC-MS; | A <1 B 4% C 24% D <1 E <1 |

-
-
4110-02-5
tris(trimethylsilyl)silyllithium

-
A
-
1450-14-2
1,1,1,2,2,2-hexamethyldisilane

-
B
-
4098-98-0
tetrakis(trimethylsilyl)silane

-
C
-
5181-43-1
2,2,3,3-tetrakis(trimethylsilyl)hexamethyltetrasilane

-
D
-
130377-71-8
C6H18Li2Si3

| Conditions | Yield |
|---|---|
| at 140 - 150℃; | A n/a B 10% C 27% D 35% |

-
-
617-86-7
triethylsilane

-
-
155570-15-3
1-<(N,N-dimethylamino)methyl>-2-benzene

-
A
-
1450-14-2
1,1,1,2,2,2-hexamethyldisilane

| Conditions | Yield |
|---|---|
| In cyclohexane for 1.66667h; Irradiation; | A n/a B 20% C 33% D 24% |

-
-
513-81-5
2,3-dimethyl-buta-1,3-diene

-
-
155570-15-3
1-<(N,N-dimethylamino)methyl>-2-benzene

-
A
-
1450-14-2
1,1,1,2,2,2-hexamethyldisilane

| Conditions | Yield |
|---|---|
| for 4.5h; Irradiation; | A 33% B 9% C 16% |

-
-
155570-15-3
1-<(N,N-dimethylamino)methyl>-2-benzene

-
A
-
1450-14-2
1,1,1,2,2,2-hexamethyldisilane

| Conditions | Yield |
|---|---|
| With triethylsilane In cyclohexane for 1.66667h; Irradiation; | A n/a B 20% C 33% D 24% |

-
-
64-17-5
ethanol

-
-
394739-08-3
bis[2,4,6-tris((dimethylamino)methyl)phenoxy]bis(trimethylsilyl)silane

-
A
-
1450-14-2
1,1,1,2,2,2-hexamethyldisilane

-
B
-
161787-77-5
[2,4,6-tris((dimethylamino)methyl)phenoxy]trimethylsilane

| Conditions | Yield |
|---|---|
| at 120℃; for 48h; | A 2% B 33% C 32% D 31% |
| In cyclohexane for 4h; Product distribution; Irradiation; | A 7% B 23% C 20% D 6% |

-
-
75-77-4
chloro-trimethyl-silane

-
-
114953-59-2
phenyldi-n-butylchlorosilane

-
A
-
1450-14-2
1,1,1,2,2,2-hexamethyldisilane

-
B
-
114953-60-5
1-phenyl-1,1-di-n-butyltrimethyldisilane

| Conditions | Yield |
|---|---|
| With potassium alloy; sodium In benzene for 144h; Heating; | A n/a B 32% |

-
-
513-81-5
2,3-dimethyl-buta-1,3-diene

-
-
394739-08-3
bis[2,4,6-tris((dimethylamino)methyl)phenoxy]bis(trimethylsilyl)silane

-
A
-
1450-14-2
1,1,1,2,2,2-hexamethyldisilane

-
B
-
161787-77-5
[2,4,6-tris((dimethylamino)methyl)phenoxy]trimethylsilane


| Conditions | Yield |
|---|---|
| at 120℃; for 168h; Product distribution; | A 3% B 32% C 29% D 2% |

-
-
75-77-4
chloro-trimethyl-silane

-
A
-
1450-14-2
1,1,1,2,2,2-hexamethyldisilane

-
B
-
1586-73-8
tris(trimethylsilyl)amine

| Conditions | Yield |
|---|---|
| With potassium graphite; Co2(2,2,2-tri(diisopropylphosphinomethylamino)triphenylamine(-3H)); nitrogen In tetrahydrofuran at 25.84℃; under 760.051 Torr; for 12h; Catalytic behavior; Kinetics; Reagent/catalyst; Inert atmosphere; | A n/a B 30% |
| With trans-[Mo[N2]2(depf)2]; nitrogen; lithium In tetrahydrofuran at 22 - 24℃; under 760.051 Torr; for 100h; | A 16.53 mmol B 0.55 mmol |
| With trans-[Mo[N2]2(depf)2]; nitrogen; sodium In TETRAHYDROPYRANE at 22 - 24℃; under 760.051 Torr; for 100h; | A 0.29 mmol B 1.08 mmol |
-
-
23695-65-0
adamantane-2-thione

-
-
79184-72-8
2,2-dimesitylhexamethyltrisilane

-
A
-
1450-14-2
1,1,1,2,2,2-hexamethyldisilane

| Conditions | Yield |
|---|---|
| In cyclohexane for 15h; Ambient temperature; Irradiation; low pressure mercury lamp; | A n/a B 29% |


-
A
-
1450-14-2
1,1,1,2,2,2-hexamethyldisilane

-
B
-
106420-40-0
{(C5H5)2Nb(CH2Si(CH3)3)Cl}(1+)*PF6(1-)={(C5H5)2Nb(CH2Si(CH3)3)Cl}PF6

| Conditions | Yield |
|---|---|
| With CH2Cl2 In dichloromethane addn. of cold (-78°C) CH2Cl2 to mixt. of Cp2Nb(SiMe3)Cl and (Cp2Fe)PF6 in 1:1 molar ratio cooled to -78°C under inert atm., allowed to warm to room temp.; further products; evapd., extd. (Et2O), evapn. gave Cp2Fe (NMR, IR); residue extd. (CH2Cl2), soln. filtered, concd., cooling to -45°C gave cryst. target product (elem.anal.); crystn. (MeCN) of residue gave (Cp2NbCl2)PF6 (IR); others products analyzed by GC; | A <1 B 28% |

-
-
37899-63-1
(dimethylphenylgermyl)trimethylsilane

-
-
513-81-5
2,3-dimethyl-buta-1,3-diene

-
A
-
1450-14-2
1,1,1,2,2,2-hexamethyldisilane

-
B
-
768-32-1
trimethylphenylsilane

| Conditions | Yield |
|---|---|
| In cyclohexane Irradiation (UV/VIS); irradiation of catenate in cyclohexane containing an excess of 2,3-dimethylbutadiene with a 110 W low pressure Hg arc lamp at room temp. under Ar for 1-4 h; identification by GC and GC-MS; | A <1 B 23% |

-
-
1450-14-2
1,1,1,2,2,2-hexamethyldisilane

-
-
591-87-7
Allyl acetate

-
A
-
187737-37-7
propene

-
B
-
2754-27-0
trimethylsilyl acetate

-
C
-
762-72-1
allyl-trimethyl-silane

-
D
-
4964-03-8
(E)-trimethyl(prop-1-en-1-yl)silane

| Conditions | Yield |
|---|---|
| tetrakis(triphenylphosphine) palladium(0) at 180℃; for 2h; high pressure, under nitrogen; | A 2 % Chromat. B 100% C 48% D 48% |

-
-
1450-14-2
1,1,1,2,2,2-hexamethyldisilane

-
-
25115-43-9
(α,α-dichlorobenzyl)trimethylsilane

-
-
24894-87-9
(E)-α,β-bis(trimethylsilyl)stilbene

| Conditions | Yield |
|---|---|
| With tetrakis(triphenylphosphine) palladium(0) In 1,3,5-trimethyl-benzene at 130℃; for 18h; | 100% |
-
-
1450-14-2
1,1,1,2,2,2-hexamethyldisilane

-
-
53119-90-7
α,β,4-trichlorobenzyltrimethylsilane

| Conditions | Yield |
|---|---|
| With tetrakis(triphenylphosphine) palladium(0) In 1,3,5-trimethyl-benzene at 130℃; for 24h; | 100% |

| Conditions | Yield |
|---|---|
| With hydrogenchloride; diethyl ether at 120℃; for 70h; Sealed tube; | 100% |
| With carbon monoxide; hydrogen; nickel at 200℃; under 750.06 Torr; for 2h; | 59% |
| With bromobenzene; calcium In water for 1h; Ambient temperature; | 5% |
-
-
1450-14-2
1,1,1,2,2,2-hexamethyldisilane

-
-
614-47-1
1,3-diphenyl-propen-3-one

-
-
30262-98-7
(E)-1,3-diphenyl-3-trimethylsilyl-2-propene

| Conditions | Yield |
|---|---|
| With copper(I) trifluoromethanesulfonate benzene In various solvent(s) at 100℃; for 19h; | 100% |
-
-
1450-14-2
1,1,1,2,2,2-hexamethyldisilane

-
-
17190-05-5
3',4'-dimethoxy-1,1'-biphenyl-4-ol

| Conditions | Yield |
|---|---|
| In pyridine at 115℃; for 3h; | 100% |
-
-
1450-14-2
1,1,1,2,2,2-hexamethyldisilane

-
-
53059-30-6
3'-nitro-[1,1'-biphenyl]-4-ol

-
-
923294-79-5
4-(3'-nitrophenyl)phenol trimethylsilyl ether

| Conditions | Yield |
|---|---|
| In pyridine at 115℃; for 3h; | 100% |

| Conditions | Yield |
|---|---|
| In pyridine at 115℃; for 3h; | 100% |
-
-
106-44-5
p-cresol

-
-
1450-14-2
1,1,1,2,2,2-hexamethyldisilane

-
-
17902-32-8
trimethyl(4-methylphenoxy)silane

| Conditions | Yield |
|---|---|
| In pyridine at 115℃; for 3h; | 100% |
| With lanthanum(III) nitrate hexahydrate In acetonitrile at 20℃; | 90% |
-
-
1450-14-2
1,1,1,2,2,2-hexamethyldisilane

-
-
92-69-3
4-Phenylphenol

-
-
1023-13-8
4-phenylphenol trimethylsilyl ether

| Conditions | Yield |
|---|---|
| In pyridine at 115℃; for 3h; | 100% |
-
-
1450-14-2
1,1,1,2,2,2-hexamethyldisilane

-
-
321-62-0
2'-fluoro-[1,1'-biphenyl]-4-ol

-
-
923294-77-3
4-(2'-fluorophenyl)phenol trimethylsilyl ether

| Conditions | Yield |
|---|---|
| In pyridine at 115℃; for 3h; | 100% |
-
-
1450-14-2
1,1,1,2,2,2-hexamethyldisilane

-
-
62668-02-4
(E)-1,3-diphenyl-2-propen-1-ol

-
-
106137-98-8
(R)-(E)-1,3-diphenyl-3-trimethylsilylpropene

| Conditions | Yield |
|---|---|
| With tetrakis(acetonitrile)palladium(II) bis(tetrafluoroborate); allyl alcohol In methanol; dimethyl sulfoxide at 60℃; Inert atmosphere; | 100% |
| With Pd(BF4)2(MeCN)4 In methanol; dimethyl sulfoxide at 50℃; for 15h; Air; regioselective reaction; | 69% |

| Conditions | Yield |
|---|---|
| With sodium methylate In N,N,N,N,N,N-hexamethylphosphoric triamide | 99% |
| With sodium hydroxide; (o-hydroxyphenyl)diphenylphosphine; PdCl(ϖ-C3H5)2 In tetrahydrofuran at 100℃; for 10h; Substitution; Silylation; | 83% |
| With sodium acetate In N,N-dimethyl acetamide at 120℃; for 24h; Catalytic behavior; Solvent; Reagent/catalyst; Inert atmosphere; | 70% |
-
-
1450-14-2
1,1,1,2,2,2-hexamethyldisilane

-
-
114716-44-8
(1R*,2R*)-(Z)-4-chloro-2-methyl-1-phenyl-3-buten-1-ol

| Conditions | Yield |
|---|---|
| for 96h; Ambient temperature; | 99% |
-
-
1450-14-2
1,1,1,2,2,2-hexamethyldisilane

-
-
57-14-7
1,1-dimethylhydrazine

-
-
684-26-4
N,N-dimethyl-N',N'-bis(trimethylsilyl)hydrazine

| Conditions | Yield |
|---|---|
| With potassium hydride In tetrahydrofuran; N,N,N,N,N,N-hexamethylphosphoric triamide for 24h; Ambient temperature; | 99% |
-
-
1450-14-2
1,1,1,2,2,2-hexamethyldisilane

-
-
187532-11-2
(2R,3S)-3-Isopropyl-N-(2,4,6-trimethylphenylsulfonyl)-2-vinylaziridine

| Conditions | Yield |
|---|---|
| Stage #1: 1,1,1,2,2,2-hexamethyldisilane With n-butyllithium In tetrahydrofuran; N,N,N,N,N,N-hexamethylphosphoric triamide; hexane at -78℃; for 1h; Metallation; Stage #2: In tetrahydrofuran; N,N,N,N,N,N-hexamethylphosphoric triamide; hexane at -78 - -30℃; transmetallation; Stage #3: (2R,3S)-3-Isopropyl-N-(2,4,6-trimethylphenylsulfonyl)-2-vinylaziridine In tetrahydrofuran; N,N,N,N,N,N-hexamethylphosphoric triamide; hexane at -78℃; Ring cleavage; | 99% |

| Conditions | Yield |
|---|---|
| Stage #1: pyridine; 1,1,1,2,2,2-hexamethyldisilane With sodium methylate In N,N,N,N,N,N-hexamethylphosphoric triamide for 2h; Stage #2: With water Further stages.; | 99% |

| Conditions | Yield |
|---|---|
| Stage #1: pyridine; 1,1,1,2,2,2-hexamethyldisilane With sodium methylate In N,N,N,N,N,N-hexamethylphosphoric triamide for 0.25h; Stage #2: With water Further stages.; | 99% |

| Conditions | Yield |
|---|---|
| With allyl(cyclopentadiene)palladium(II); trimethylsilyl trifluoromethanesulfonate In benzene at 20℃; for 1h; | 99% |

| Conditions | Yield |
|---|---|
| With palladium diacetate; caesium carbonate; triphenylphosphine In N,N-dimethyl-formamide at 60℃; for 12h; Schlenk technique; Inert atmosphere; | 99% |

-
-
505-23-7
1,3 dithiane

-
-
74-96-4
ethyl bromide

-
-
1450-14-2
1,1,1,2,2,2-hexamethyldisilane

- 2-ethyl-2-(trimethylsilyl)-1,3-dithiane
-
72658-48-1
2-ethyl-2-(trimethylsilyl)-1,3-dithiane

| Conditions | Yield |
|---|---|
| Mechanism; multistep reaction; other electrophiles, influence of temperature and reaction time; | 98% |
| Yield given. Multistep reaction; |

-
-
55-22-1
pyridine-4-carboxylic acid

-
-
1450-14-2
1,1,1,2,2,2-hexamethyldisilane

-
-
25436-39-9
trimethylsilyl 4-pyridinecarboxylate

| Conditions | Yield |
|---|---|
| RT, then warmed slowly until violent gas evolution occured; | 98% |
-
-
51-21-8
5-fluorouracil

-
-
1450-14-2
1,1,1,2,2,2-hexamethyldisilane

-
-
17242-85-2
5-fluoro-2,4-bis(trimethylsilyloxy)pyrimidine

| Conditions | Yield |
|---|---|
| With ammonium sulfate for 14h; Heating; | 98% |
-
-
1450-14-2
1,1,1,2,2,2-hexamethyldisilane

-
-
100-51-6
benzyl alcohol

-
-
14642-79-6
benzyloxy-trimethylsilane

| Conditions | Yield |
|---|---|
| With lanthanum(III) nitrate hexahydrate In acetonitrile at 20℃; | 98% |
| With 1% Au/TiO2 In ethyl acetate at 25℃; for 1h; Inert atmosphere; | 96% |
| With natural kaolinitic clay In tetrachloromethane for 2h; Ambient temperature; | 90% |
-
-
1450-14-2
1,1,1,2,2,2-hexamethyldisilane

-
-
187531-92-6
(2R,3S)-3-methyl-N-(4-methylbenzenesulfonyl)-2-vinylaziridine

| Conditions | Yield |
|---|---|
| Stage #1: 1,1,1,2,2,2-hexamethyldisilane With n-butyllithium In tetrahydrofuran; N,N,N,N,N,N-hexamethylphosphoric triamide; hexane at -78℃; for 1h; Metallation; Stage #2: In tetrahydrofuran; N,N,N,N,N,N-hexamethylphosphoric triamide; hexane at -78 - -30℃; transmetallation; Stage #3: (2R,3S)-3-methyl-N-(4-methylbenzenesulfonyl)-2-vinylaziridine In tetrahydrofuran; N,N,N,N,N,N-hexamethylphosphoric triamide; hexane at -78℃; for 0.5h; Ring cleavage; | 98% |
-
-
619-73-8
4-nitrobenzyl chloride

-
-
1450-14-2
1,1,1,2,2,2-hexamethyldisilane

-
-
14856-73-6
4-nitrobenzyl trimethylsilyl ether

| Conditions | Yield |
|---|---|
| With lanthanum(III) nitrate hexahydrate In acetonitrile at 20℃; | 98% |

| Conditions | Yield |
|---|---|
| With lanthanum(III) nitrate hexahydrate In acetonitrile at 20℃; | 98% |
| With 1% Au/TiO2 In ethyl acetate at 25℃; for 1.5h; Inert atmosphere; | 97% |
-
-
1450-14-2
1,1,1,2,2,2-hexamethyldisilane

-
-
4128-31-8
rac-octan-2-ol

-
-
18023-52-4
trimethyl-(1-methyl-heptyloxy)-silane

| Conditions | Yield |
|---|---|
| With lanthanum(III) nitrate hexahydrate In acetonitrile at 20℃; | 98% |
Hexamethyldisilane Specification
The IUPAC name of Hexamethyldisilane is trimethyl(trimethylsilyl)silane. With the CAS registry number 1450-14-2 and EINECS 215-911-0, it is also named as Disilane, 1,1,1,2,2,2-hexamethyl-. The product's categories are Standards for NMR; Analytical Chemistry; NMR Spectrometry; Si (Classes of Silicon Compounds); Silicon Compounds (for Synthesis); Si-Si Compounds; Synthetic Organic Chemistry; Blocking Agents; Protective Agents; Silylating Agents; Special Silanes; Disilanes; DisilanesVapor Deposition Precursors; Organometallic Reagents; Organosilicon; Precursors by Metal. It is colorless clear liquid which is insoluble in water, soluble in acetone, benzene, ethyl ether and heptane. What's more, Hexamethyldisilane is a dimer of the trimethylsilyl (TMS) group, linked through a silicon-silicon bond. And it can be cleaved by iodine to give trimethylsilyl iodide. Additionally, this chemical should be sealed in the container and stored in the cool and dry place. People should ensure that the workplace has well-ventilated equipment.
The other characteristics of this product can be summarized as: (1)ACD/LogP: 2.50; (2)# of Rule of 5 Violations: 0; (3)ACD/LogD (pH 5.5): 2.5; (4)ACD/LogD (pH 7.4): 2.5; (5)ACD/BCF (pH 5.5): 46.85; (6)ACD/BCF (pH 7.4): 46.85; (7)ACD/KOC (pH 5.5): 546.38; (8)ACD/KOC (pH 7.4): 546.38; (9)#H bond acceptors: 0; (10)#H bond donors: 0; (11)#Freely Rotating Bonds: 1; (12)Polar Surface Area: 0 Å2; (13)Index of Refraction: 1.39; (14)Molar Refractivity: 47.28 cm3; (15)Molar Volume: 199 cm3; (16)Polarizability: 18.74×10-24 cm3; (17)Surface Tension: 14.6 dyne/cm; (18)Density: 0.735 g/cm3; (19)Flash Point: 5.8 °C; (20)Enthalpy of Vaporization: 33.74 kJ/mol; (21)Boiling Point: 113.5 °C at 760 mmHg; (22)Vapour Pressure: 24.5 mmHg at 25°C.
Preparation of Hexamethyldisilane: It can be obtained by chloro-trimethyl-silane. This reaction needs reagent Li/2 percent Na and solvent tetrahydrofuran at temperature of 25-28 °C. The reaction time is 4 hours. The yield is 100%.

Uses of Hexamethyldisilane: It can be used as raw material in the preparation of trimethylsilyl lithium, sodium trimethylsilyl and trimethylsilyl potassium. It is also used in pharmaceutical synthesis and can be used as terminator of poly carbosilane chain. In addition, it can react with furan-2-carbonyl chloride to get furan-2-yl-trimethylsilanyl-methanone. This reaction needs reagents (p-allyl-PdCl)2, P(OEt)3 and solvent toluene by heating. The reaction time is 6 hours. The yield is 60%.
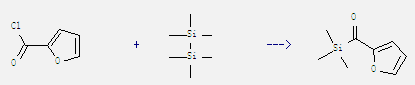
When you are using this chemical, please be cautious about it as the following:
It is highly flammable, so people should keep it away from sources of ignition. It is also irritating to eyes, respiratory system and skin. People must not breathe vapour. In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. When using this chemical, take precautionary measures against static discharges. In case of accident or if you feel unwell, seek medical advice immediately (show the label whenever possible.)
People can use the following data to convert to the molecule structure.
1. SMILES:C[Si](C)(C)[Si](C)(C)C
2. InChI:InChI=1/C6H18Si2/c1-7(2,3)8(4,5)6/h1-6H3
3. InChIKey:NEXSMEBSBIABKL-UHFFFAOYAV
The following are the toxicity data which has been tested.
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| guinea pig | LD50 | oral | > 20gm/kg (20000mg/kg) | Gigiena i Sanitariya. For English translation, see HYSAAV. Vol. 59(4), Pg. 14, 1994. | |
| mouse | LD50 | oral | > 20gm/kg (20000mg/kg) | Gigiena i Sanitariya. For English translation, see HYSAAV. Vol. 59(4), Pg. 14, 1994. | |
| rat | LD50 | oral | > 20gm/kg (20000mg/kg) | Gigiena i Sanitariya. For English translation, see HYSAAV. Vol. 59(4), Pg. 14, 1994. |
Related Products
- Hexamethyldisilane
- 14501-64-5
- 145017-98-7
- 145018-73-1
- 145022-44-2
- 145022-83-9
- 1450-23-3
- 145027-96-9
- 14503-45-8
- 145037-81-6
- 145038-50-2
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View