-
Name
L-Homoserine
- EINECS 211-590-6
- CAS No. 672-15-1
- Article Data45
- CAS DataBase
- Density 1.3126 (rough estimate)
- Solubility 1100 g/L (30 °C) in water
- Melting Point 203 °C
- Formula C4H9NO3
- Boiling Point 368.7 °C at 760 mmHg
- Molecular Weight 119.12
- Flash Point 176.8 °C
- Transport Information
- Appearance white to light yellow crystalline powder
- Safety 24/25
- Risk Codes 36/37/38
-
Molecular Structure
- Hazard Symbols
- Synonyms Butyric acid, 2-amino-4-hydroxy-, L- (8CI);Butanoic acid, 2-amino-4-hydroxy-, (S)-;2-amino-4-hydroxy-butanoic acid;2-Amino-4-hydroxybutyric acid;Butyric acid, 2-amino-4-hydroxy-, L-;(2S)-2-amino-4-hydroxy-butanoic acid;Homoserine (VAN);H-Homoserine;H-Hse-OH;L-Homo-serine;H-HoSer-OH;
- PSA 83.55000
- LogP -0.51900
Synthetic route
-
-
159487-41-9
Isopropyl (2S)-N-(trifluoroacetyl)homoserine

-
-
672-15-1
L-homoserine

| Conditions | Yield |
|---|---|
| With ethanol | 95% |
| With potassium hydroxide In ethanol for 2h; | 95% |

| Conditions | Yield |
|---|---|
| With water; sodium hydrogencarbonate pH=2 - 5; Reflux; | 92% |
| With sodium hydrogencarbonate In water pH=3 - 6; Inert atmosphere; Schlenk technique; Reflux; | 92% |
| With sodium hydrogencarbonate In water at 140℃; for 0.5h; | 69% |

| Conditions | Yield |
|---|---|
| With triethylamine In water for 2h; Ambient temperature; | 82% |
-
-
29625-73-8
2-acetamido-4-O-(2-acetamido-2-deoxy-β-D-glucopyranosyl)-1-N-(4-L-aspartoyl)-2-deoxy-β-D-glucopyranosylamine

-
A
-
672-15-1
L-homoserine

-
B
-
102039-77-0
N,N'-diacetylchitobiosamine

-
C
-
118943-00-3
1-acetamido-4-O-(2-acetamido-2-deoxy-β-D-glucopyranosyl)-2-amino-1,2-dideoxy-D-glucitol

-
D
-
119009-35-7
2-acetamido-4-O-(2-acetamido-2-deoxy-β-D-glucopyranosyl)-1-amino-1,2-dideoxy-D-glucitol

| Conditions | Yield |
|---|---|
| With lithium hydroxide; lithium borohydride; trilithium citrate In water; tert-butyl alcohol at 45℃; for 5h; Yield given; | A 80% B 72% C n/a D n/a |

-
-
29625-73-8
2-acetamido-4-O-(2-acetamido-2-deoxy-β-D-glucopyranosyl)-1-N-(4-L-aspartoyl)-2-deoxy-β-D-glucopyranosylamine

-
A
-
672-15-1
L-homoserine

-
B
-
102039-77-0
N,N'-diacetylchitobiosamine

-
C
-
118943-00-3
1-acetamido-4-O-(2-acetamido-2-deoxy-β-D-glucopyranosyl)-2-amino-1,2-dideoxy-D-glucitol

-
D
-
119009-35-7
2-acetamido-4-O-(2-acetamido-2-deoxy-β-D-glucopyranosyl)-1-amino-1,2-dideoxy-D-glucitol

-
E
-
29886-32-6
β-D-GlcpNAc(1->4)-D-GlcNAc-ol

| Conditions | Yield |
|---|---|
| With lithium hydroxide; lithium borohydride; trilithium citrate In water; tert-butyl alcohol at 45℃; for 5h; Product distribution; | A 80% B 72% C n/a D n/a E 5% |

-
-
29625-73-8
2-acetamido-4-O-(2-acetamido-2-deoxy-β-D-glucopyranosyl)-1-N-(4-L-aspartoyl)-2-deoxy-β-D-glucopyranosylamine

-
A
-
672-15-1
L-homoserine

-
B
-
102039-77-0
N,N'-diacetylchitobiosamine

-
C
-
119009-35-7
2-acetamido-4-O-(2-acetamido-2-deoxy-β-D-glucopyranosyl)-1-amino-1,2-dideoxy-D-glucitol

-
D
-
29886-32-6
β-D-GlcpNAc(1->4)-D-GlcNAc-ol

| Conditions | Yield |
|---|---|
| With lithium hydroxide; lithium borohydride; trilithium citrate In water; tert-butyl alcohol at 45℃; for 5h; | A 80% B 72% C n/a D 5% |


| Conditions | Yield |
|---|---|
| With water; methyl iodide at 20℃; for 24h; Inert atmosphere; Reflux; | 74% |
| Stage #1: L-methionine With methyl iodide In water for 24h; Inert atmosphere; Stage #2: With potassium hydrogencarbonate for 10h; pH=3 - 6; Reflux; Stage #3: With hydrogenchloride In water pH=5 - 6; | 74% |
| Stage #1: L-methionine With methyl iodide In methanol; water for 48h; Stage #2: With sodium hydrogencarbonate In methanol; water for 15h; Reflux; | 62% |
| With sodium hydrogencarbonate; methyl iodide In methanol; water for 12h; Reflux; |
-
-
1149755-80-5
(S)-tert-butyl 1-(furan-2-yl)-3-hydroxypropylcarbamate

-
-
672-15-1
L-homoserine

| Conditions | Yield |
|---|---|
| Stage #1: (S)-tert-butyl 1-(furan-2-yl)-3-hydroxypropylcarbamate With ozone In methanol at -78℃; for 0.166667h; Stage #2: With dimethylsulfide In methanol at 20℃; for 12h; Stage #3: With hydrogenchloride In 1,4-dioxane at 0 - 20℃; | 62% |
-
-
63-68-3
L-methionine

-
-
87625-62-5
ptaquiloside

-
A
-
672-15-1
L-homoserine

-
B
-
34175-96-7
2(R)-pterosin B

-
C
-
114751-11-0
(R)-2,5,7-Trimethyl-6-(2-methylsulfanyl-ethyl)-indan-1-one

-
D
-
114751-12-1
(S)-2-Amino-4-methylsulfanyl-butyric acid 2-((R)-2,4,6-trimethyl-3-oxo-indan-5-yl)-ethyl ester

| Conditions | Yield |
|---|---|
| With sodium hydroxide; Amberlite CG 50 (H form) In water; acetone at 37℃; for 12h; 1.) pH: 9.2; | A 10% B 26% C 12% D 4% |

-
-
29625-73-8
2-acetamido-4-O-(2-acetamido-2-deoxy-β-D-glucopyranosyl)-1-N-(4-L-aspartoyl)-2-deoxy-β-D-glucopyranosylamine

-
A
-
672-15-1
L-homoserine

-
B
-
56-84-8
L-Aspartic acid

-
C
-
29886-32-6
β-D-GlcpNAc(1->4)-D-GlcNAc-ol

| Conditions | Yield |
|---|---|
| With sodium hydroxide; sodium tetrahydroborate | A 15% B 3% C 20% |


| Conditions | Yield |
|---|---|
| In alkalischen und in verduennten neutralen wss. Loesungen; | |
| With water; sodium hydrogencarbonate at 100℃; for 24h; | 8.8 g |
-
-
879008-07-8
N-chloroacetyl of racemic homoserine lacton

-
-
672-15-1
L-homoserine

| Conditions | Yield |
|---|---|
| Enzymatische Herstellung; |
-
-
16856-13-6
(+)-β-methyl-L-aspartate hydrochloride

-
-
672-15-1
L-homoserine

| Conditions | Yield |
|---|---|
| With lithium borohydride In tetrahydrofuran |

-
-
672-15-1
L-homoserine

| Conditions | Yield |
|---|---|
| hydrolysis; |
-
-
33515-32-1
S-methyl-L-methionine hydrobromide

-
-
672-15-1
L-homoserine

| Conditions | Yield |
|---|---|
| With sodium hydrogencarbonate In methanol; water for 20h; Heating; Yield given; |
-
-
123052-97-1
tert-butyl (2S,5S)-2-(tert-butyl)-5-(2-hydroxyethyl)-3-methyl-4-oxo-1-imidazolidinecarboxylate

-
-
672-15-1
L-homoserine

| Conditions | Yield |
|---|---|
| With hydrogenchloride; Dowex 50-WX8; acetic acid; trifluoroacetic acid 1.) CH2Cl2, 30 min, 0 deg C; 12 h, room temperature; 2.) 100-105 deg C, toluene, water, 36 h; Yield given. Multistep reaction; |
-
-
50-00-0
formaldehyd

-
-
96293-17-3, 105024-93-9, 105112-33-2
(S)-N-(2-benzoylphenyl)-1-benzylpyrrolidine-2-carboxamide

-
-
56-40-6
glycine

-
A
-
312-84-5
D-Serine

-
B
-
56-45-1
L-serin

-
C
-
672-15-1
L-homoserine

| Conditions | Yield |
|---|---|
| With nickel nitrate; water; sodium methylate; triethylamine 1.) MeOH, 2 h, 50 deg C; 2.) MeOH, 50 deg C; Yield given. Multistep reaction. Yields of byproduct given; |


| Conditions | Yield |
|---|---|
| With hydrogen; hydrogen In methanol for 20h; Ambient temperature; | A 60 % Spectr. B 40 % Spectr. |
-
-
64-18-6
formic acid

-
-
63-68-3
L-methionine

-
A
-
672-15-1
L-homoserine

-
B
-
56-84-8
L-Aspartic acid

-
C
-
3226-65-1
L-methionine sulfoxide

-
D
-
56-86-0
L-glutamic acid

-
E
-
7314-32-1
L-methionine sulfone

-
F
-
14857-77-3
L-homocysteic acid

| Conditions | Yield |
|---|---|
| With hydrogen; oxygen In water at 40 - 50℃; Product distribution; Mechanism; A flame from a burner fed with H2-O2 mixtures of different compositions were blown onto the surface of an aq. solution of pH=3; |

-
-
63-68-3
L-methionine

-
A
-
1492-24-6
L-2-aminobutyric acid

-
B
-
672-15-1
L-homoserine

-
C
-
56-84-8
L-Aspartic acid

-
D
-
3226-65-1
L-methionine sulfoxide

-
E
-
7314-32-1
L-methionine sulfone

-
F
-
14857-77-3
L-homocysteic acid

| Conditions | Yield |
|---|---|
| With hydrogen; oxygen In water at 40 - 50℃; Product distribution; Mechanism; A flame from a burner fed with H2-O2 mixtures of different compositions were blown onto the surface of an aq. solution of pH=3; |

-
-
63-68-3
L-methionine

-
A
-
672-15-1
L-homoserine

-
B
-
56-84-8
L-Aspartic acid

-
C
-
7314-32-1
L-methionine sulfone

-
D
-
14857-77-3
L-homocysteic acid

| Conditions | Yield |
|---|---|
| With formic acid; hydrogen; oxygen In water at 40 - 50℃; a flame from a burner fed with H2+O2 was blown to the surface of an aq. solution; H2:O2=50:50; Further byproducts given; |

-
-
63-68-3
L-methionine

-
A
-
672-15-1
L-homoserine

-
B
-
56-84-8
L-Aspartic acid

-
C
-
3226-65-1
L-methionine sulfoxide

-
D
-
7314-32-1
L-methionine sulfone

-
E
-
498-20-4, 2338-03-6, 15106-57-7, 23632-91-9
(S)-aspartate β-semialdehyde

-
F
-
14857-77-3
L-homocysteic acid

| Conditions | Yield |
|---|---|
| With hydrogen; oxygen In water Product distribution; various sulfur-containing amino acids under different reaction conditions; |


| Conditions | Yield |
|---|---|
| Equilibrium constant; |

| Conditions | Yield |
|---|---|
| In konzentrierten und in schwach sauren wss. Loesungen; |

-
-
672-15-1
L-homoserine

| Conditions | Yield |
|---|---|
| With water; silver(l) oxide at 20℃; |

-
-
672-15-1
L-homoserine

| Conditions | Yield |
|---|---|
| With methanol; platinum durch Hydrogenolyse; |
-
-
1115-84-0, 3493-12-7, 41844-44-4, 71267-08-8
L-S-methylmethionine sulfonium chloride

-
A
-
75-18-3
dimethylsulfide

-
B
-
672-15-1
L-homoserine

| Conditions | Yield |
|---|---|
| With sodium hydroxide at 100℃; Kinetics; |

-
-
672-15-1
L-homoserine

| Conditions | Yield |
|---|---|
| With hydrogenchloride; water at 100℃; for 1h; |

| Conditions | Yield |
|---|---|
| With AHSL acylase Enzymatic reaction; |

| Conditions | Yield |
|---|---|
| With thionyl chloride at 0 - 20℃; for 12.5h; | 100% |

| Conditions | Yield |
|---|---|
| With sodium hydroxide at 2 - 10℃; | 100% |

| Conditions | Yield |
|---|---|
| With hydrogenchloride In water for 0.25h; | 99% |
| With hydrogenchloride for 5h; Reflux; | 99% |
| With hydrogenchloride a) reflux, 3 h, b) RT, 16 h; | 94% |

| Conditions | Yield |
|---|---|
| With thionyl chloride | 98% |
| With thionyl chloride at 0 - 20℃; for 80h; Heating / reflux; | 92% |
-
-
672-15-1
L-homoserine

-
-
38622-91-2, 36635-61-7
[(p-methylphenyl)sulfonylmethyl]isonitrile

-
-
78-84-2
isobutyraldehyde

| Conditions | Yield |
|---|---|
| With 2,2,2-trifluoroethanol at 30 - 40℃; for 94h; | 97% |


| Conditions | Yield |
|---|---|
| Stage #1: L-homoserine; urea In water at 100℃; for 8h; Stage #2: With hydrogenchloride In water at 90℃; for 6h; | 96% |
-
-
672-15-1
L-homoserine

-
-
24424-99-5
di-tert-butyl dicarbonate

-
-
41088-86-2
L-2-(tert-butyloxycarbonylamino)-4-hydroxybutyric acid

| Conditions | Yield |
|---|---|
| With sodium carbonate In acetone at 19℃; for 8.3h; Temperature; Cooling with ice; | 95% |
| With sodium hydroxide In 1,4-dioxane; water at 0 - 25℃; for 24.5h; | 93% |
| With sodium hydroxide In water; acetone at 0 - 20℃; for 4h; Reagent/catalyst; | 93% |
-
-
672-15-1
L-homoserine

| Conditions | Yield |
|---|---|
| With hydrogen bromide; acetic acid at 80℃; for 6h; Sealed tube; | 95% |
| With hydrogen bromide; acetic acid at 80℃; for 5h; |

| Conditions | Yield |
|---|---|
| With sodium carbonate In acetone at 18℃; for 14.6h; Temperature; Cooling with ice; | 94% |
| With sodium hydrogencarbonate In water for 3h; | 92% |
| With sodium hydrogencarbonate at 20℃; for 24h; | 89% |

| Conditions | Yield |
|---|---|
| With 2,2,2-trifluoroethanol at 30 - 40℃; for 18h; | 93% |


| Conditions | Yield |
|---|---|
| With potassium carbonate In ethanol at 60 - 70℃; diastereoselective reaction; | 93% |

-
-
672-15-1
L-homoserine

-
-
28814-72-4, 46318-21-2, 50975-79-6
(S)-cis-3,6-bis-(2-hydroxy-ethyl)-piperazine-2,5-dione

| Conditions | Yield |
|---|---|
| In ethylene glycol at 150℃; for 36h; | 93% |
-
-
672-15-1
L-homoserine

-
-
38622-91-2, 36635-61-7
[(p-methylphenyl)sulfonylmethyl]isonitrile

-
-
630-19-3
pivalaldehyde

| Conditions | Yield |
|---|---|
| With 2,2,2-trifluoroethanol at 30 - 40℃; for 94h; | 92% |

-
-
672-15-1
L-homoserine

-
-
18162-48-6
tert-butyldimethylsilyl chloride

-
-
474023-97-7
(2S)-amino-4-(tert-butyldimethylsilanyloxy)butanoic acid

| Conditions | Yield |
|---|---|
| With 1,8-diazabicyclo[5.4.0]undec-7-ene In acetonitrile at 0 - 20℃; for 16.0833h; | 92% |
| With 1,8-diazabicyclo[5.4.0]undec-7-ene In acetonitrile at 0 - 20℃; for 16.0833h; | 92% |
| With 1,8-diazabicyclo[5.4.0]undec-7-ene In acetonitrile at 0 - 20℃; for 16.0833h; | 92% |

| Conditions | Yield |
|---|---|
| With sodium hydroxide In 1,4-dioxane; water for 0.75h; Ambient temperature; | 91% |
-
-
672-15-1
L-homoserine

-
-
52773-66-7
benzyloxycarbonyl-O-benzyl-L-tyrosine 1-succinimidyl ester

-
-
1161754-51-3
C28H30N2O7

| Conditions | Yield |
|---|---|
| With potassium hydrogencarbonate In tetrahydrofuran at 20℃; | 90% |
-
-
52462-29-0
[ruthenium(II)(η6-1-methyl-4-isopropyl-benzene)(chloride)(μ-chloride)]2

-
-
672-15-1
L-homoserine

| Conditions | Yield |
|---|---|
| With sodium hydroxide In methanol at 20℃; for 3.5h; diastereoselective reaction; | 90% |

| Conditions | Yield |
|---|---|
| With sodium hydroxide at 20℃; for 7h; | 89% |
-
-
672-15-1
L-homoserine

-
-
19794-26-4
L-homoserine O-sulfate

| Conditions | Yield |
|---|---|
| With sulfuric acid at 20℃; for 3.5h; | 88% |

| Conditions | Yield |
|---|---|
| With sodium hydrogencarbonate In water; acetone Cooling; | 88% |
-
-
672-15-1
L-homoserine

-
-
89985-91-1
tert-butyl pyridin-2-yl carbonate

-
-
41088-86-2
L-2-(tert-butyloxycarbonylamino)-4-hydroxybutyric acid

| Conditions | Yield |
|---|---|
| With sodium hydroxide In 1,4-dioxane; water at 0℃; for 24h; | 88% |

| Conditions | Yield |
|---|---|
| With 2,2,2-trifluoroethanol at 30 - 40℃; for 48h; | 87% |

-
-
672-15-1
L-homoserine

-
-
39537-41-2
γ-Cl-Abu

| Conditions | Yield |
|---|---|
| With hydrogenchloride; tetrabutylammomium bromide In water at 80℃; for 9h; Sealed tube; | 87% |

| Conditions | Yield |
|---|---|
| Stage #1: L-homoserine; Allyl chloroformate With sodium carbonate In water; acetonitrile at 20℃; Stage #2: allyl bromide With sodium hydrogencarbonate In N,N-dimethyl-formamide | 86% |


| Conditions | Yield |
|---|---|
| With potassium hydrogencarbonate In diethyl ether; water for 20h; Ambient temperature; | 85% |

| Conditions | Yield |
|---|---|
| With potassium hydrogencarbonate In methanol for 48h; Ambient temperature; | 85% |
-
-
672-15-1
L-homoserine

-
-
82911-69-1
N-(9H-fluoren-2-ylmethoxycarbonyloxy)succinimide

-
-
172525-85-8
Nα-(Fluoren-9-ylmethoxycarbonyl)-L-homoserine

| Conditions | Yield |
|---|---|
| With sodium hydrogencarbonate In acetonitrile | 85% |
| With sodium hydrogencarbonate | |
| With sodium carbonate In 1,4-dioxane; water |
-
-
672-15-1
L-homoserine

-
-
15159-65-6
(2S)-2-amino-4-bromobutanoic acid hydrobromide

| Conditions | Yield |
|---|---|
| With hydrogen bromide at 75 - 80℃; for 5h; | 85% |
| With hydrogen bromide; acetic acid at 75 - 80℃; for 5h; Autoclave; | 85% |
| With hydrogen bromide; acetic acid at 50℃; for 12h; Sealed tube; | 78% |
L-Homoserine Chemical Properties
IUPAC Name: 2-Amino-4-hydroxybutanoic acid
Synonyms of L-Homoserine (CAS NO.672-15-1): Butanoic acid, 2-amino-4-hydroxy-, (S)- ; Homoserine (VAN) ; Butyric acid, 2-amino-4-hydroxy-, L- (8CI)
CAS NO: 672-15-1
Molecular Formula: C4H9NO3
Molecular Weight: 119.12
Molecular Structure: 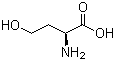
EINECS: 211-590-6
H bond acceptors: 4
H bond donors: 4
Freely Rotating Bonds: 5
Polar Surface Area: 38.77 Å2
Index of Refraction: 1.51
Molar Refractivity: 27.17 cm3
Molar Volume: 90.7 cm3
Surface Tension: 63.5 dyne/cm
Density: 1.312 g/cm3
Flash Point: 176.8 °C
Enthalpy of Vaporization: 71.22 kJ/mol
Boiling Point: 368.7 °C at 760 mmHg
Vapour Pressure: 6.16E-07 mmHg at 25°C
Melting Point: 203 °C (dec.)(lit.)
Alpha: -8.5 º (c=2, H2O 22 ºC)
Storage temp: Store at RT.
Water Solubility: 1100 g/L (30 °C)
Appearance: white to light yellow crystalline powder
Product Categories of L-Homoserine (CAS NO.672-15-1): Serine [Ser, S];Unusual Amino Acids;Amino Acids;Amino Acids & Derivatives
L-Homoserine Uses
L-Homoserine (CAS NO.672-15-1) is an intermediate in the biosynthesis of three essential amino acids: methionine, threonine (an isomer of homoserine), and isoleucine.
L-Homoserine Safety Profile
Safety Information about L-Homoserine (CAS NO.672-15-1):
Safety Statements: 24/25
S24/25: Avoid contact with skin and eyes.
WGK Germany: 3
F: 10
HS Code: 29225000
L-Homoserine Specification
General Information: As in any fire, wear a self-contained breathing apparatus in pressure-demand, MSHA/NIOSH (approved or equivalent), and full protective gear.
Extinguishing Media: Use water spray, dry chemical, carbon dioxide, or chemical foam.
Handling: Avoid breathing dust, vapor, mist, or gas. Avoid contact with skin and eyes.
Storage: Store in a cool, dry place. Store in a tightly closed container. Keep refrigerated. (Store below 4°C/39°F.)
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View