-
Name
Methyldiethoxyphosphine
- EINECS 239-805-9
- CAS No. 15715-41-0
- Article Data30
- CAS DataBase
- Density 0.9 g/cm3
- Solubility It slowly hydrolyses in water.
- Melting Point
- Formula C5H13O2P
- Boiling Point 124.497 °C at 760 mmHg
- Molecular Weight 136.131
- Flash Point 26.513 °C
- Transport Information UN 1993
- Appearance colorless liquid
- Safety 16-26-36/37/39
- Risk Codes 10-36/37/38
-
Molecular Structure
- Hazard Symbols Xn
- Synonyms Phosphonousacid, methyl-, diethyl ester (6CI,7CI,8CI,9CI);Diethoxymethylphosphine;Diethyl methanephosphonite;Diethyl methylphosphonite;Methyldiethoxyphosphine;O,O-Diethyl methylphosphonite;
- PSA 32.05000
- LogP 2.00110
Synthetic route

| Conditions | Yield |
|---|---|
| With triethylamine In Petroleum ether at 25 - 60℃; for 1.5h; Time; Flow reactor; Large scale; | 98% |
| Stage #1: ethanol With calcium oxide In 1,3,5-trimethyl-benzene for 2h; Reflux; Stage #2: methyldichlorophosphane In 1,3,5-trimethyl-benzene at 0 - 30℃; for 4h; Temperature; Solvent; Inert atmosphere; | 96.7% |
| at 10 - 120℃; for 0.0833333h; Temperature; Inert atmosphere; | 96% |
-
-
589-57-1
diethyl phosphorylchloridite

-
-
676-58-4
methylmagnesium chloride

-
-
15715-41-0
methyldiethoxyphosphine

| Conditions | Yield |
|---|---|
| Stage #1: diethyl phosphorylchloridite; methylmagnesium chloride In tetrahydrofuran at -10 - 0℃; for 2h; Inert atmosphere; Stage #2: With dibutyl ether; benzylamine In tetrahydrofuran at -5℃; for 10h; Reagent/catalyst; Temperature; | 89.3% |
| In tetrahydrofuran at 0℃; for 2.5h; Temperature; | |
| In tetrahydrofuran at -10 - -5℃; Inert atmosphere; | 10.15 g |
-
-
589-57-1
diethyl phosphorylchloridite

-
-
917-64-6
methyl magnesium iodide

-
-
15715-41-0
methyldiethoxyphosphine

| Conditions | Yield |
|---|---|
| In diethyl ether | 60% |
-
-
628-37-5
diethyl peroxide

-
-
14410-05-0
1,3,4-trimethyl-Δ3-phospholene

-
A
-
15715-41-0
methyldiethoxyphosphine

-
B
-
683-08-9
Diethyl methylphosphonate

-
C
-
34736-63-5
tetraethoxy-methyl-λ5-phosphane

-
D
-
15450-80-3
1,3,4-trimethyl-Δ3-phospholene oxide

-
E
-
15450-82-5
1,3,4-trimethyl-2,3-dihydro-1H-phosphole 1-oxide

| Conditions | Yield |
|---|---|
| In [D3]acetonitrile for 1.91667h; Rate constant; Product distribution; further reaction times; | A 10 % Spectr. B 6 % Spectr. C 15 % Spectr. D 7 % Spectr. E n/a |

-
-
589-57-1
diethyl phosphorylchloridite

-
-
75-16-1
methylmagnesium bromide

-
-
15715-41-0
methyldiethoxyphosphine

| Conditions | Yield |
|---|---|
| In diethyl ether at -12 - 0℃; for 16h; | 30.4 g |
| In diethyl ether -20 deg C then r.t., 1 h; |

| Conditions | Yield |
|---|---|
| Stage #1: methylene chloride With magnesium In tetrahydrofuran; toluene for 2h; Stage #2: diethyl phosphorylchloridite In tetrahydrofuran; toluene at 15 - 30℃; for 6h; Solvent; Temperature; |
-
-
64-17-5
ethanol

-
-
15715-41-0
methyldiethoxyphosphine

| Conditions | Yield |
|---|---|
| With aluminium at -10 - 150℃; for 2h; Temperature; | 71.62 g |
-
-
15715-41-0
methyldiethoxyphosphine

-
-
16391-07-4
ethyl methylphosphinate

| Conditions | Yield |
|---|---|
| With water at 22℃; for 17h; | 100% |
| With water at 5 - 20℃; for 18h; | |
| In water at 0 - 22℃; for 17h; | |
| With water; hexadecyltributylammonium chloride; sodium chloride at -5 - 0℃; Inert atmosphere; | 8.8 g |
| With water at 20℃; for 18h; Inert atmosphere; |

| Conditions | Yield |
|---|---|
| for 2.5h; Heating; | 100% |

| Conditions | Yield |
|---|---|
| Stage #1: methyldiethoxyphosphine; butan-1-ol In water; toluene at 45 - 50℃; for 5h; Inert atmosphere; Stage #2: With tetrabutoxytitanium In water; toluene Reflux; | 98.26% |
| With Amberlyst 15 In water; toluene at 50 - 140℃; Inert atmosphere; | 97.9% |
-
-
1071-71-2
ethyl 6-chloro-6-oxohexanoate

-
-
15715-41-0
methyldiethoxyphosphine

-
-
115693-04-4
5-Ethoxy-adipyl-P-methyl-phosphinsaeure-O-ethylester

| Conditions | Yield |
|---|---|
| at 30℃; | 98% |
-
-
15715-41-0
methyldiethoxyphosphine

-
-
380153-45-7
[(E)-(R)-4-[(1R,2R,3S,5R)-2-((Z)-6-Bromo-hex-2-enyl)-3,5-bis-(tert-butyl-dimethyl-silanyloxy)-cyclopentyl]-2-(tert-butyl-dimethyl-silanyloxy)-but-3-enyloxy]-benzene

-
-
380153-32-2
(6-{3,5-bis(tert-butyldimethylsilanoylxy)-2-[3-(tert-cutyldimethylsilanoylxy)-4-phenoxybut-1-enyl]cyclopentyl}-hex-4-enyl)methylphosphinic acid ethyl ester

| Conditions | Yield |
|---|---|
| In toluene at 145℃; for 22h; Arbuzov reaction; | 98% |
-
-
15715-41-0
methyldiethoxyphosphine

-
-
138500-85-3
4-bromomethylphenylboronic acid pinacol ester

-
-
1273492-75-3
ethyl methyl(4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)benzyl)phosphinate

| Conditions | Yield |
|---|---|
| In 1,4-dioxane at 100℃; for 1h; Inert atmosphere; | 98% |
-
-
15715-41-0
methyldiethoxyphosphine


| Conditions | Yield |
|---|---|
| With dichloro(1,1'-bis(diphenylphosphanyl)ferrocene)palladium(II)*CH2Cl2; triethylamine In N,N-dimethyl-formamide at 130℃; for 0.0833333h; Sealed tube; Microwave irradiation; | 98% |

| Conditions | Yield |
|---|---|
| With acetic acid at 70 - 75℃; for 4h; Inert atmosphere; | 97.6% |
-
-
15715-41-0
methyldiethoxyphosphine

-
-
292638-85-8
acrylic acid methyl ester

| Conditions | Yield |
|---|---|
| With acetic acid at 5 - 20℃; for 2h; | 96.3% |
-
-
15715-41-0
methyldiethoxyphosphine

-
-
23145-65-5
1-bromomethyl-3-methoxy-4-nitrobenzene

-
-
1257996-50-1
(3-methoxy-4-nitrobenzyl)-methyl phosphonic acid ethyl ester

| Conditions | Yield |
|---|---|
| In toluene for 16h; Reflux; | 96% |
-
-
15715-41-0
methyldiethoxyphosphine

-
-
140-88-5
ethyl acrylate

-
-
15090-27-4
ethyl 3-(ethoxymethylphosphinyl)propionate

| Conditions | Yield |
|---|---|
| In ethanol at 5℃; for 2h; | 95.8% |
| With acetic acid at 5 - 25℃; for 2.5h; Inert atmosphere; | 95% |

| Conditions | Yield |
|---|---|
| With sulfuric acid In water at 50℃; Reflux; | 95.8% |
-
-
15715-41-0
methyldiethoxyphosphine

-
-
1829-28-3
ethyl 2-iodobenzoate

-
-
57020-81-2
o-ethoxycarbonylphenyl-methylphosphinic acid ethyl ester

| Conditions | Yield |
|---|---|
| With nickel dichloride at 170℃; | 95% |

| Conditions | Yield |
|---|---|
| at 110℃; for 17h; Michaelis-Arbuzov reaction; | 95% |

| Conditions | Yield |
|---|---|
| at 25 - 30℃; for 2.33333h; Inert atmosphere; | 95% |

-
-
15715-41-0
methyldiethoxyphosphine

-
-
2032-35-1
Bromoacetaldehyde diethyl acetal

| Conditions | Yield |
|---|---|
| In ethanol at 80 - 100℃; for 24h; Solvent; | 95% |
| In toluene at 110℃; for 4h; Arbuzov Reaction; | 91% |
-
-
15715-41-0
methyldiethoxyphosphine

| Conditions | Yield |
|---|---|
| 94% |
-
-
15715-41-0
methyldiethoxyphosphine


| Conditions | Yield |
|---|---|
| at 125℃; for 3h; | 94% |
-
-
4360-63-8
2-bromomethyl-1,3-dioxolane

-
-
15715-41-0
methyldiethoxyphosphine

| Conditions | Yield |
|---|---|
| In toluene at 110℃; for 5h; Inert atmosphere; | 93.8% |
| at 120℃; for 2h; Arbuzov Reaction; | 82.6% |
-
-
28562-53-0
4-acetoxy azetidinone

-
-
15715-41-0
methyldiethoxyphosphine

-
-
84673-24-5
O-ethyl(4-oxoazetidin-2-yl)methylphosphinate

| Conditions | Yield |
|---|---|
| at 60℃; for 1h; | 93% |
| at 60℃; for 1h; | 89% |

| Conditions | Yield |
|---|---|
| With lanthanum(III) chloride at 140℃; for 10h; Reagent/catalyst; Temperature; Inert atmosphere; | 91.3% |
-
-
1001419-35-7
1,1-dimethylethyl [4-(bromomethyl)-1,3-thiazol-2-yl]carbamate

-
-
15715-41-0
methyldiethoxyphosphine

-
-
1001419-75-5
C12H21N2O4PS

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at 75℃; for 16h; | 91% |

| Conditions | Yield |
|---|---|
| With tetrabutylammomium bromide; magnesium bromide In water at 140℃; for 15h; Temperature; Solvent; Reagent/catalyst; | 90.2% |
-
-
15715-41-0
methyldiethoxyphosphine

-
-
122690-13-5
4-bromo-2-phthalimidobutyric acid methyl ester

-
-
109541-73-3
METHYL DL-2-(1,3-DIHYDRO-1,3-DIOXO-2H-ISOINDOL-2-YL)-4-(ETHOXYMETHYLPHOSPHINYL)BUTANOATE

| Conditions | Yield |
|---|---|
| In toluene at 100℃; | 90% |
-
-
15715-41-0
methyldiethoxyphosphine

-
-
7661-21-4
6-Bromohexanoic Acid Phenylamide

-
-
606092-26-6
methyl-(5-phenylcarbamoyl-pentyl)-phosphinic acid ethyl ester

| Conditions | Yield |
|---|---|
| at 120℃; for 10h; | 89% |
| at 120℃; for 10h; Arbuzov reaction; | 200 mg |
-
-
15715-41-0
methyldiethoxyphosphine

-
-
134745-23-6
ethyl 2-oxo-3-butenoate

| Conditions | Yield |
|---|---|
| at 5℃; for 8h; Inert atmosphere; | 87.8% |
-
-
15715-41-0
methyldiethoxyphosphine

-
-
108736-44-3
Methyl-3-oxo-pentenoat

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at 5℃; for 6h; Inert atmosphere; | 87.1% |
-
-
15715-41-0
methyldiethoxyphosphine

-
-
252858-13-2
2-decarboxy-2-iodoprostaglandin F2α triacetate

-
-
380153-41-3
2-decarboxy-2-(O-ethyl-P-methylphosphinico)prostaglandin F2α triacetate

| Conditions | Yield |
|---|---|
| In toluene at 100℃; for 6h; Arbuzov reaction; | 86% |
Methyldiethoxyphosphine Specification
The IUPAC name of Methyldiethoxyphosphine is diethoxy(methyl)phosphane. With the CAS registry number 15715-41-0, it is also named as O,O-Diethyl methylphosphonite. The product's category is Phosphorus Compounds, and it is air & moisture sensitive. Besides, it is colorless liquid, which should be stored in dry inert gas in a dark, closed and ventilated place at room temperature.
The other characteristics of this product can be summarized as: (1)EINECS: 239-805-9; (2)ACD/LogP: 2.78; (3)# of Rule of 5 Violations: 0; (4)ACD/LogD (pH 5.5): 2.78; (5)ACD/LogD (pH 7.4): 2.78; (6)ACD/BCF (pH 5.5): 76.87; (7)ACD/BCF (pH 7.4): 76.87; (8)ACD/KOC (pH 5.5): 778.82; (9)ACD/KOC (pH 7.4): 778.82; (10)H bond acceptors: 2; (11)H bond donors: 0; (12)Freely Rotating Bonds: 4; (13)Density: 0.9 g/cm3; (14)Flash Point: 26.5 °C; (15)Enthalpy of Vaporization: 34.74 kJ/mol; (16)Boiling Point: 124.5 °C at 760 mmHg; (17)Vapour Pressure: 15.4 mmHg at 25 °C.
Preparation of Methyldiethoxyphosphine: this chemical can be prepared by Ethanol and Methylphosphinous acid dichloride.
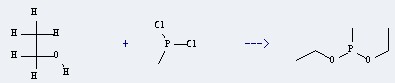
This reaction needs N,N-Diethylaniline and Pentane at temperature of 0 °C. The reaction time is 30 min. The yield is 90 %.
Uses of Methyldiethoxyphosphine: it can react with Adipic acid ethyl ester chloride to get 5-Ethoxy-adipyl-P-methyl-phosphinsaeure-O-ethylester.
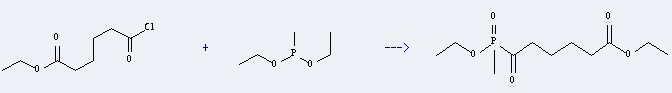
This reaction will occur at temperature of 30 °C. The yield is 98 %.
When you are using this chemical, please be cautious about it as the following: it is flammable, so please keep away from sources of ignition. And it is also irritating to eyes, respiratory system and skin. You should wear suitable protective clothing, gloves and eye/face protection when use it. Moreover, in case of contact with eyes, please rinse immediately with plenty of water and seek medical advice.
People can use the following data to convert to the molecule structure.
(1)SMILES:O(P(OCC)C)CC
(2)InChI:InChI=1/C5H13O2P/c1-4-6-8(3)7-5-2/h4-5H2,1-3H3
(3)InChIKey:NSSMTQDEWVTEKN-UHFFFAOYAD
(4)Std. InChI:InChI=1S/C5H13O2P/c1-4-6-8(3)7-5-2/h4-5H2,1-3H3
(5)Std. InChIKey:NSSMTQDEWVTEKN-UHFFFAOYSA-N
Related Products
- Methyldiethoxyphosphine
- 15715-42-1
- 15716-30-0
- 1571-65-9
- 1571-69-3
- 157169-68-1
- 157169-71-6
- 15717-17-6
- 15717-25-6
- 1571-72-8
- 1571-75-1
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View