Dayang Chem (Hangzhou) Co.,Ltd.
Dayangchem’s R&D center can offer custom synthesis according to the contract research and development services for the fine chemicals, pharmaceutical, biotechnique and some of the other chemicals. DayangChem can provide different quantiti
Cas:1663-39-4
Min.Order:1 Kilogram
FOB Price: $3.0
Type:Lab/Research institutions
inquiryHangzhou Dingyan Chem Co., Ltd
Items Standard Result Appearance Colorless liquid accord Amount% ≥99.5
Cas:1663-39-4
Min.Order:1 Kilogram
FOB Price: $3.0 / 10.0
Type:Manufacturers
inquirySimagchem Corporation
Welcome to Simagchem, your partner in China as a premier supply of bulk specialty chemicals for industry and life science. We introduce experienced quality product and exceptional JIT service with instant market intelligence in China to benefit our
Cas:1663-39-4
Min.Order:0 Metric Ton
Negotiable
Type:Manufacturers
inquiryHefei TNJ chemical industry co.,ltd
Classification: Alcohol CAS No.: 75-65-0 Other Names: TBA MF: C4H10O EINECS No.: 200-889-7 Place of
Cas:1663-39-4
Min.Order:1 Metric Ton
Negotiable
Type:Trading Company
inquiryAlity Chemical Corporation
The above product is Ality Chemical's strong item with best price, good quality and fast supply. Ality Chemical has been focusing on the research and production of this field for over 14 years. At the same time, we are always committed to providi
Chemwill Asia Co., Ltd.
Our main production base is located in Xuzhou industry park. We are certified both to the ISO 9001 and ISO 14001 Standards, have a safety management system in place.Our R&D team masters core technology for process-design of target building block
Cas:1663-39-4
Min.Order:5 Kiloliter
FOB Price: $1.2 / 5.0
Type:Manufacturers
inquiryJinan Finer Chemical Co., Ltd
Product Description Product website: http://www.finerchem.com Product Name tert-Butyl acrylate CAS No. 1663-39-4
Cas:1663-39-4
Min.Order:1 Metric Ton
FOB Price: $12.0
Type:Lab/Research institutions
inquiryHenan Tianfu Chemical Co., Ltd.
Product Name: tert-Butyl acrylate Synonyms: tert-butylpropenoate;ACRYLIC ACID TERT-BUTYL ESTER;TERTIARY-BUTYL ACRYLATE;TERT-BUTYL ACRYLATE;TBA;T-BUTYL ACRYLATE;2-Propenoic acid, 1,1-dimethylethyl ester;2-Propenoicacid,1,1-dimethylethyl
Cas:1663-39-4
Min.Order:1 Metric Ton
FOB Price: $8900.0
Type:Lab/Research institutions
inquiryHangzhou Keyingchem Co.,Ltd
Hangzhou KeyingChem Co., Ltd. exported this product to many countries and regions at best price. If you are looking for the material’s manufacturer or supplier in China, KeyingChem is your best choice. Pls contact with us freely for getting det
Cas:1663-39-4
Min.Order:0 Metric Ton
Negotiable
Type:Lab/Research institutions
inquiryHangzhou J&H Chemical Co., Ltd.
J&H CHEM R&D center can offer custom synthesis according to the contract research and development services for the fine chemicals, pharmaceutical, biotechnique and some of the other chemicals. J&H CHEM has some Manufacturing base in Jia
HANGZHOU YUNUO CHEMICAL CO.,LTD
supplier in China Appearance:colorless liquid Storage:Stored in cool, dry and ventilation place; Away from fire and heat Package:100g/bottle,1kg/bottle,25kg/drum or as per your request Application:organic synthesis Transportation:BY AIR Port:S
Siwei Development Group Ltd.
Product name: Tert-Butyl Acrylate CAS No.:1663-39-4 Molecule Formula:C7H12O2 Molecule Weight:128.17 Purity: 99.0% Package: 200kg/drum Description:Colorless liquid Manufacture Standards:Enterprise Standard TESTING I
Cas:1663-39-4
Min.Order:1 Kilogram
Negotiable
Type:Trading Company
inquiryHenan Wentao Chemical Product Co., Ltd.
We are leading fine chemicals supplier in China with ISO certificate, Our main business covers the fields below: 1.Noble Metal Catalysts (Pt.Pd...) 2.Organic Phosphine Ligands (Tert-butyl-phosphine.Cyclohexyl-phosphine...) 3.OLED
Cas:1663-39-4
Min.Order:1 Kilogram
FOB Price: $3.0
Type:Lab/Research institutions
inquiryHangzhou Huarong Pharm Co., Ltd.
Hangzhou Huarong Pharm Co., Ltd.established since 2006 , has been actively developing specialty products for Finished Dosages, APIs, Intermediates, and Fine chemicals markets in North America, Europe, Korea, Japan, Mid-East and all over the World. Hu
Hunan chemfish Pharmaceutical co.,Ltd
Appearance:95%+ Package:R&D,Pilot run Transportation:per client require Port:Express ,Air, Sea
Triumph International Development Limilted
Triumph has the complete production of G- KG - MT service chain,we can make the new technology into productivity quickly in the research and development of new products. Main Business Custom Synthesis:
Kono Chem Co.,Ltd
high purity lowest priceAppearance:solid or liquid Storage:in sealed air resistant place Package:drum and bag Application:for pharma use Transportation:by sea or air Port:Beijing or Guangzhou
JINHUA HUAYI CHEMICAL CO., LTD.
Jinhua huayi chemical co., ltd. is dedicated to the development, production and marketing of chemicals. On the basis of equality and mutual benefit, and under the principle of customer first, credit first, quality first, we are ready to join hands
Cas:1663-39-4
Min.Order:100 Gram
Negotiable
Type:Lab/Research institutions
inquiryHenan Allgreen Chemical Co.,Ltd
high quality Storage:Sealed, dry, microtherm , avoid light and smell. Package:According to the demand of customer Application:Organic synthesis Transportation:by air or by sea
Aecochem Corp.
Our clients, like BASF,CHEMO,Brenntag,ASR,Evonik,Merck and etc.Appearance:COA Storage:in stock Application:MSDS/TDS
Zhuozhou Wenxi import and Export Co., Ltd
Product Description Description & Specification Category Pharmaceutical Raw Materials, Fine Chemicals, Bulk drug Standard Medical standard
Cas:1663-39-4
Min.Order:1 Kilogram
FOB Price: $112.0
Type:Trading Company
inquiryAntimex Chemical Limied
Ansciep Chemical is a professional enterprise manufacturing and distributing fine chemicals and speciality chemicals. We have been dedicated to heterocycle compounds and phenyl rings for tens of years. This is our mature product for export. Our quali
Cas:1663-39-4
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryXiamen AmoyChem Co.,Ltd
Amoychem is committed to providing the top-quality chemical products and services Internationally. We offer our customers with friendly, professional service and reliable, high performance products that have been manufactured according to the accredi
Luyunjia Chemistry Xiamen Limited
Xiamen Luyunjia Trading Co.,Ltd Package:1kg/bag; 5002kg/drum, or as customer's request. Application:Xiamen Luyunjia Trading Co.,Ltd Transportation:DHL, EMS, FedEx, TNT, AIR, SEA Port:Beijing,Shanghai,Guangzhou ,China main port
Changchun Artel lmport and Export trade company
Supply top quality products with a reasonable price Application:api
Hangzhou ZeErRui Chemical Co., Ltd.
Hangzhou ZeErRui Chemical Co., Ltd. located in Lingang industrial areas, our plant covers an area of 6000 square meters.ZeErRui dedicated to the development, production and marketing of chemicals. We have earned ourselves a good reputation at home an
Cas:1663-39-4
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryHunan Longxianng Runhui Trading Co.,Ltd
tert-butyl acrylateAppearance:colorless liquid Package:barrel or tank Application:4. Applications tert-Butyl acrylate forms homopolymers and copolymers. Copolymers of tert-butyl acrylate can be prepared with acrylic acid and its salts, amides and es
Hunan Russell Chemicals Technology Co.,Ltd
high purity lowest priceAppearance:solid or liquid Storage:in sealed air resistant place Package:Foil bag; Drum; Plastic bottle Application:Pharma;Industry;Agricultural Transportation:by sea or air Port:any port in China
Synthetic route

| Conditions | Yield |
|---|---|
| With strong acid cation exchange resin; polymerization inhibitor A; polymerization inhibitor B at 10℃; for 13h; Temperature; | 99.7% |
| With tempol; 10H-phenothiazine; sulfuric acid | |
| With mesoporous Fe-SBA-15-SO3H at 60℃; for 12h; Autoclave; |
-
-
13831-03-3
tert-butyl prop-2-ynoate

-
-
1663-39-4
tert-Butyl acrylate

| Conditions | Yield |
|---|---|
| With hydrogen; [Cp2Mo(μ-SH)2Rh(PPh3)2][PF6] In acetone at 20℃; under 760.051 Torr; for 5h; Product distribution; Further Variations:; Catalysts; Reaction partners; | 99% |

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at 20℃; for 0.25h; | 96% |

| Conditions | Yield |
|---|---|
| With N,N,N,N,-tetramethylethylenediamine; acetic anhydride at 0 - 60℃; for 5h; | 66% |
| With N,N,N,N,N,N-hexamethylphosphoric triamide; Bu3PI2 In diethyl ether for 24h; | 51% |
| With nickel(II) polyacrylate; water |
-
-
292638-85-8
acrylic acid methyl ester

-
-
75-65-0
tert-butyl alcohol

-
A
-
1663-39-4
tert-Butyl acrylate

-
B
-
3852-09-3
methyl 3-methoxypropionate

-
C
-
81048-08-0
3-tert.-Butoxypropionsaeuremethylester

-
D
-
112032-54-9
3-Methoxy-propionic acid tert-butyl ester

-
E
-
21150-74-3
tert-butyl β-tert-butoxypropionate

| Conditions | Yield |
|---|---|
| With n-butyllithium; bis(acetylacetonate)nickel(II) In benzene at 90℃; for 5h; Product distribution; other time, other temp.; | A 33% B 26% C 9% D 32% E 3% |


| Conditions | Yield |
|---|---|
| With calcium carbonate at 70℃; | |
| With pyridine | |
| With triethylamine In chloroform at 10℃; for 12h; |

| Conditions | Yield |
|---|---|
| With 2,3-Dimethylaniline at 150℃; |
-
-
57-57-8
β-Propiolactone

-
-
74-88-4
methyl iodide

-
A
-
1663-39-4
tert-Butyl acrylate

-
B
-
6149-41-3
methyl ester (3-hydroxy) propionic acid

-
C
-
292638-85-8
acrylic acid methyl ester

| Conditions | Yield |
|---|---|
| With 18-crown-6 ether; potassium tert-butylate 1) THF, 20 deg C, 15 min; Yield given. Multistep reaction. Yields of byproduct given; |


| Conditions | Yield |
|---|---|
| With hydrogenchloride; tetracarbonyl nickel |


| Conditions | Yield |
|---|---|
| With sulfuric acid |

-
-
548756-56-5
tert-butyl 3-hydroxy-3-(2-hydroxy-3-methoxyphenyl)-2-methylenenpropanoate

-
A
-
1663-39-4
tert-Butyl acrylate

-
B
-
148-53-8
3-methoxy-2-hydroxybenzaldehyde

| Conditions | Yield |
|---|---|
| With 1,4-diaza-bicyclo[2.2.2]octane In chloroform-d1 retro-Baylis-Hillman reaction; |
-
-
548756-58-7
tert-butyl 3-(5-bromo-2-hydroxyphenyl)-3-hydroxy-2-methylenenpropanoate

-
A
-
1663-39-4
tert-Butyl acrylate

-
B
-
1761-61-1
5-bromosalicyclaldehyde

| Conditions | Yield |
|---|---|
| With 1,4-diaza-bicyclo[2.2.2]octane In chloroform-d1 retro-Baylis-Hillman reaction; |
-
-
548756-57-6
tert-butyl 3-(3-ethoxy-2-hydroxyphenyl)-3-hydroxy-2-methylenenpropanoate

-
A
-
1663-39-4
tert-Butyl acrylate

-
B
-
492-88-6
3-ethoxysalicylaldehyde

| Conditions | Yield |
|---|---|
| With 1,4-diaza-bicyclo[2.2.2]octane In chloroform-d1 retro-Baylis-Hillman reaction; |
-
-
548756-59-8
tert-butyl 3-(3,5-dibromo-2-hydroxyphenyl)-3-hydroxy-2-methylenenpropanoate

-
A
-
90-59-5
3,5-Dibromosalicylaldehyde

-
B
-
1663-39-4
tert-Butyl acrylate

| Conditions | Yield |
|---|---|
| With 1,4-diaza-bicyclo[2.2.2]octane In chloroform-d1 retro-Baylis-Hillman reaction; |

| Conditions | Yield |
|---|---|
| Stage #1: acrolein With tert-butylhypochlorite In tetrachloromethane at 30℃; for 3.33333h; Stage #2: tert-butyl alcohol In tetrachloromethane at 50 - 75℃; for 22h; |
-
-
15026-17-2
succinic acid mono-tert-butyl ester

-
-
1663-39-4
tert-Butyl acrylate

| Conditions | Yield |
|---|---|
| With 2,2-dimethylpropanoic anhydride; triphenylphosphine; palladium dichloride at 190℃; for 2h; Inert atmosphere; | 31 %Chromat. |
| Multi-step reaction with 2 steps 1.1: triethylamine / dichloromethane / 0.17 h / Inert atmosphere 1.2: 20 °C / Inert atmosphere 2.1: palladium dichloride; lithium chloride / 3 h / 155 - 160 °C / Inert atmosphere; Schlenk technique; Sealed tube View Scheme | |
| Multi-step reaction with 2 steps 1: HATU; N-ethyl-N,N-diisopropylamine / dichloromethane / 4 h / 20 °C 2: triphenylphosphine / tetrahydrofuran; water / 20 °C View Scheme |

-
-
1663-39-4
tert-Butyl acrylate

| Conditions | Yield |
|---|---|
| With lithium chloride; palladium dichloride at 155 - 160℃; for 3h; Inert atmosphere; Schlenk technique; Sealed tube; | 46 %Chromat. |

-
-
1663-39-4
tert-Butyl acrylate

| Conditions | Yield |
|---|---|
| With triphenylphosphine In tetrahydrofuran; water at 20℃; |

-
-
1663-39-4
tert-Butyl acrylate

| Conditions | Yield |
|---|---|
| With Dess-Martin periodane In dichloromethane at 20℃; |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: tetrahydrofuran / 0 - 20 °C 2: Dess-Martin periodane / dichloromethane / 20 °C View Scheme |
-
-
548756-55-4
tert-butyl 3-hydroxy-3-(2-hydroxyphenyl)-2-methylenenpropanoate

-
A
-
1663-39-4
tert-Butyl acrylate

-
B
-
90-02-8
salicylaldehyde

| Conditions | Yield |
|---|---|
| With 1,4-diaza-bicyclo[2.2.2]octane In [D3]acetonitrile at 20℃; Kinetics; Inert atmosphere; |

| Conditions | Yield |
|---|---|
| With 4-methoxy-phenol at 60℃; for 10h; |

| Conditions | Yield |
|---|---|
| Stage #1: acrylic acid With potassium carbonate In N,N-dimethyl-formamide at 0℃; for 0.75h; Inert atmosphere; Stage #2: t-butyl bromide In N,N-dimethyl-formamide at 100℃; for 24h; Inert atmosphere; |
-
-
1663-39-4
tert-Butyl acrylate

-
-
138034-79-4, 49855-41-6
tert-butyl 2,3-dibromopropanoate

| Conditions | Yield |
|---|---|
| With bromine In dichloromethane at 0 - 20℃; for 18h; | 100% |
| With bromine In dichloromethane at 5℃; for 2h; | 90% |
| With bromine In chloroform for 15h; Ambient temperature; | 81% |
-
-
1663-39-4
tert-Butyl acrylate

-
-
1122-91-4
4-bromo-benzaldehyde

-
-
208036-26-4
(E)-t-butyl 4-formylcinnamate

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide at 100℃; for 6h; Heck-Mizoroki reaction; | 100% |
| With palladium diacetate; triethylamine; tris-(o-tolyl)phosphine Heating; | 91% |
| With palladium on silica; triethylamine In N,N-dimethyl-formamide for 0.2h; Heck Reaction; Microwave irradiation; Green chemistry; | 89% |
-
-
1663-39-4
tert-Butyl acrylate

-
-
2357-39-3
2-trifluoromethyl-4-isopropyl-Δ3-oxazolin-5-one

-
-
87341-14-8
tert-butyl 3-[5-oxo-4-(propan-2-yl)-2-(trifluoromethyl)-2,5-dihydro-1,3-oxazol-2-yl]propanoate

| Conditions | Yield |
|---|---|
| With triethylamine In dichloromethane at 0 - 20℃; | 100% |
| With triethylamine In dichloromethane 1.) -10 deg C, 2.) 25 deg C, 12 h; | 80% |
-
-
1663-39-4
tert-Butyl acrylate

-
-
99-90-1
para-bromoacetophenone

-
-
389091-50-3
(E)-tert-butyl 3-(4-acetylphenyl)prop-2-enoate

| Conditions | Yield |
|---|---|
| With 1,4-diaza-bicyclo[2.2.2]octane; potassium carbonate; palladium diacetate In N,N-dimethyl-formamide at 120℃; for 15h; Heck cross-coupling reaction; | 100% |
| With 1,3-disubstituted imidazolium bromide; potassium carbonate; 4-methylmorpholine N-oxide; palladium diacetate In 1-methyl-pyrrolidin-2-one at 120℃; for 2h; Heck reaction; | 99% |
| With potassium carbonate In N,N-dimethyl-formamide at 120℃; for 1h; Mizoroki-Heck reaction; | 99% |
-
-
1663-39-4
tert-Butyl acrylate

-
-
829-85-6
diphenylphosphane

-
-
175407-89-3
3-Diphenylphosphanyl-propionic acid tert-butyl ester

| Conditions | Yield |
|---|---|
| With tetramethyl ammoniumhydroxide In tetrahydrofuran | 100% |
| In 2-methyltetrahydrofuran at 90℃; for 4h; Inert atmosphere; Sealed tube; Green chemistry; regioselective reaction; | 81% |
| In 2-methyltetrahydrofuran at 90℃; for 4h; Inert atmosphere; | 81% |

| Conditions | Yield |
|---|---|
| With triethylamine; palladium In N,N-dimethyl acetamide at 100℃; for 24h; Heck reaction; | 100% |
| With C36H36Cl2N6Pd; triethylamine In methanol at 70℃; for 24h; Reagent/catalyst; Concentration; Heck Reaction; | 100% |
| With PdCl2(1-[2-(diphenylphosphanyl)ethyl]-3,5-dimethylpyrazole); tetrabutylammomium bromide; triethylamine In N,N-dimethyl-formamide at 140℃; for 0.16h; Catalytic behavior; Time; Heck Reaction; Inert atmosphere; Schlenk technique; | 100% |
-
-
1663-39-4
tert-Butyl acrylate

-
-
624-31-7
4-tolyl iodide

-
-
136053-53-7, 125951-00-0
tert-butyl 4-methylcinnamate

| Conditions | Yield |
|---|---|
| With tetrabutyl ammonium fluoride; palladium dichloride at 40℃; for 3h; Heck reaction; Inert atmosphere; neat (no solvent); | 100% |
| With triethylamine; palladium In N,N-dimethyl acetamide at 80℃; for 36h; Heck reaction; | 99% |
| With poly(N-isopropylacerylamide)-SCS-PdCl; TEA In n-heptane; N,N-dimethyl acetamide; water at 95℃; for 10h; Heck coupling; | 99% |
-
-
1663-39-4
tert-Butyl acrylate

-
-
586-78-7
para-nitrophenyl bromide

-
-
370839-59-1
(E)-tert-butyl 3-(4-nitrophenyl)prop-2-enoate

| Conditions | Yield |
|---|---|
| With tributyl-amine; [1,2-bis(4-Me-2-pyridylethynyl)benzene]dichloropalladium(II) at 100℃; for 7h; Heck reaction; | 100% |
| With {1,3-bis[2,6-bis(propan-2-yl)phenyl]-1,3-dihydro-2H-imidazol-2-ylidene}dichloro(pyridine)palladium; caesium carbonate In neat (no solvent) at 100℃; for 12h; Heck Reaction; | 99% |
| With C22H24Cl2N4Pd; tetrabutylammomium bromide; triethylamine In methanol at 70℃; for 24h; Reagent/catalyst; Concentration; Heck Reaction; | 97% |
-
-
1663-39-4
tert-Butyl acrylate

-
-
696-62-8
para-iodoanisole

-
-
53484-52-9
tert-butyl (E)-3-(4-methoxyphenyl)prop-2-enoate

| Conditions | Yield |
|---|---|
| With 1,4-diaza-bicyclo[2.2.2]octane; potassium carbonate; palladium diacetate In N,N-dimethyl-formamide at 120℃; for 6h; Heck cross-coupling reaction; | 100% |
| With tetrabutyl ammonium fluoride; palladium dichloride at 60℃; for 12h; Heck reaction; Inert atmosphere; neat (no solvent); | 100% |
| With water; palladium diacetate; caesium carbonate at 140℃; for 4h; Heck reaction; Ionic liquid; Inert atmosphere; | 100% |
-
-
1663-39-4
tert-Butyl acrylate

-
-
456-27-9
4-nitrobenzenediazonium tetrafluoroborate

-
-
370839-59-1
(E)-tert-butyl 3-(4-nitrophenyl)prop-2-enoate

| Conditions | Yield |
|---|---|
| With 1,6,11-triazacyclopentadeca-3,8,12-triene Pd In ethanol at 20℃; for 2.75h; Heck reaction; | 100% |
| With 3-benzyl-1-(2-hydroxy-2-phenylethyl)imidazolium chloride; palladium diacetate In ethanol at 36℃; for 3h; Heck-Matsuda reaction; | 96% |
| With dichloro bis(acetonitrile) palladium(II) In water at 0 - 25℃; for 1.5h; | 96% |
-
-
1663-39-4
tert-Butyl acrylate

-
-
52436-75-6
(4-tert-butylphenyl)diazonium tetrafluoroborate

| Conditions | Yield |
|---|---|
| With 1,6,11-triazacyclopentadeca-3,8,12-triene Pd In ethanol at 20℃; for 4h; Heck reaction; | 100% |
-
-
104-92-7
1-bromo-4-methoxy-benzene

-
-
1663-39-4
tert-Butyl acrylate

-
-
53484-52-9
tert-butyl (E)-3-(4-methoxyphenyl)prop-2-enoate

| Conditions | Yield |
|---|---|
| With C31H38ClN3Pd; potassium carbonate In 1-methyl-pyrrolidin-2-one at 140℃; for 18h; Heck-Mizoroki coupling; Inert atmosphere; | 100% |
| With IMes-Pd(dmba)Cl; potassium carbonate In 1-methyl-pyrrolidin-2-one at 140℃; for 18h; Heck-Mizoroki reaction; Inert atmosphere; | 100% |
| With 1,3-disubstituted imidazolium bromide; potassium carbonate; palladium diacetate In 1,4-dioxane at 105℃; for 20h; Heck reaction; | 99% |
-
-
1663-39-4
tert-Butyl acrylate

-
-
156-87-6
propan-1-ol-3-amine

-
-
644968-23-0
tert-butyl 3-[(2-tert-butoxycarbonyl-ethyl)-(3-hydroxy-propyl)-amino]-propionate

| Conditions | Yield |
|---|---|
| In methanol at 20℃; for 24h; Darkness; Inert atmosphere; | 100% |
| In methanol at 20 - 30℃; for 8h; | 99% |
| In methanol at 20℃; for 6h; | 98% |
-
-
1663-39-4
tert-Butyl acrylate

- poly-tert-butyl acrylate, obtained by anionic polymerization, initiated with the tBuOK/iBu3Al system, Mn = 10500, Mw/Mn = 1.18; monomer(s): tert-butyl acrylate
-
poly-tert-butyl acrylate, obtained by anionic polymerization, initiated with the tBuOK/iBu3Al system, Mn = 10500, Mw/Mn = 1.18; monomer(s): tert-butyl acrylate

| Conditions | Yield |
|---|---|
| With potassium tert-butylate; triisobutylaluminum In hexane; toluene at 0℃; for 1h; | 100% |
-
-
1663-39-4
tert-Butyl acrylate

- poly-tert-butyl acrylate, obtained by anionic polymerization, initiated with the tBuOK/iBu3Al system, Mn = 20000, Mw/Mn = 1.12; monomer(s): tert-butyl acrylate
-
poly-tert-butyl acrylate, obtained by anionic polymerization, initiated with the tBuOK/iBu3Al system, Mn = 20000, Mw/Mn = 1.12; monomer(s): tert-butyl acrylate

| Conditions | Yield |
|---|---|
| With potassium tert-butylate; triisobutylaluminum In hexane; toluene at 0℃; for 1h; | 100% |
-
-
1663-39-4
tert-Butyl acrylate

- poly-tert-butyl acrylate, obtained by anionic polymerization, initiated with the tBuOK/iBu3Al system, Mn = 29700, Mw/Mn = 1.09; monomer(s); tert-butyl acrylate
-
poly-tert-butyl acrylate, obtained by anionic polymerization, initiated with the tBuOK/iBu3Al system, Mn = 29700, Mw/Mn = 1.09; monomer(s); tert-butyl acrylate

| Conditions | Yield |
|---|---|
| With potassium tert-butylate; triisobutylaluminum In hexane; toluene at 0℃; for 2h; | 100% |
-
-
1663-39-4
tert-Butyl acrylate

- poly-tert-butyl acrylate, obtained by anionic polymerization, initiated with the tBuOK/iBu3Al system, Mn = 47700, Mw/Mn = 1.06; monomer(s): tert-butyl acrylate
-
poly-tert-butyl acrylate, obtained by anionic polymerization, initiated with the tBuOK/iBu3Al system, Mn = 47700, Mw/Mn = 1.06; monomer(s): tert-butyl acrylate

| Conditions | Yield |
|---|---|
| With potassium tert-butylate; triisobutylaluminum In hexane; toluene at 0℃; for 2h; | 100% |
-
-
1663-39-4
tert-Butyl acrylate

- poly-tert-butyl acrylate, obtained by anionic polymerization, initiated with the tBuOK/iBu3Al system, Mn = 58900, Mw/Mn = 1.05; monomer(s): tert-butyl acetate
-
poly-tert-butyl acrylate, obtained by anionic polymerization, initiated with the tBuOK/iBu3Al system, Mn = 58900, Mw/Mn = 1.05; monomer(s): tert-butyl acetate

| Conditions | Yield |
|---|---|
| With potassium tert-butylate; triisobutylaluminum In hexane; toluene at 0℃; for 2h; | 100% |
-
-
1663-39-4
tert-Butyl acrylate

- poly-tert-butyl acrylate, obtained by anionic polymerization, initiated with the tBuOK/iBu3Al system, Mn = 83400, Mw/Mn = 1.05; monomer(s): tert-butyl acrylate
-
poly-tert-butyl acrylate, obtained by anionic polymerization, initiated with the tBuOK/iBu3Al system, Mn = 83400, Mw/Mn = 1.05; monomer(s): tert-butyl acrylate

| Conditions | Yield |
|---|---|
| With potassium tert-butylate; triisobutylaluminum In hexane; toluene at 0℃; for 2h; | 100% |
-
-
1663-39-4
tert-Butyl acrylate

- poly-tert-butyl acrylate, obtained by anionic polymerization, initiated with the tBuOK/iBu3Al system, Mn = 250600, Mw/Mn = 1.17; monomer(s): tert-butyl acrylate
-
poly-tert-butyl acrylate, obtained by anionic polymerization, initiated with the tBuOK/iBu3Al system, Mn = 250600, Mw/Mn = 1.17; monomer(s): tert-butyl acrylate

| Conditions | Yield |
|---|---|
| With potassium tert-butylate; triisobutylaluminum In hexane; toluene at 0℃; for 3h; | 100% |
-
-
1663-39-4
tert-Butyl acrylate

- poly-tert-butyl acrylate, obtained by anionic polymerization, initiated with the tBuOK/Et3Al system, Mn = 170900, Mw/Mn = 1.06; monomer(s): tert-butyl acrylate
-
poly-tert-butyl acrylate, obtained by anionic polymerization, initiated with the tBuOK/Et3Al system, Mn = 170900, Mw/Mn = 1.06; monomer(s): tert-butyl acrylate

| Conditions | Yield |
|---|---|
| With potassium tert-butylate; triethylaluminum In toluene at 0℃; for 1h; | 100% |
-
-
1663-39-4
tert-Butyl acrylate

- poly-tert-butyl acrylate, obtained by anionic polymerization, initiated with the tBuOK/Et3Al system, Mn = 305900, Mw/Mn = 1.25; monomer(s): tert-butyl acrylate
-
poly-tert-butyl acrylate, obtained by anionic polymerization, initiated with the tBuOK/Et3Al system, Mn = 305900, Mw/Mn = 1.25; monomer(s): tert-butyl acrylate

| Conditions | Yield |
|---|---|
| With potassium tert-butylate; triethylaluminum In toluene at 0℃; for 2h; | 100% |
-
-
1663-39-4
tert-Butyl acrylate

- poly-tert-butyl acrylate, obtained by anionic polymerization, initiated with the tBuOK/Me3Al system, Mn = 437900, Mw/Mn = 1.61; monomer(s): tert-butyl acrylate
-
poly-tert-butyl acrylate, obtained by anionic polymerization, initiated with the tBuOK/Me3Al system, Mn = 437900, Mw/Mn = 1.61; monomer(s): tert-butyl acrylate

| Conditions | Yield |
|---|---|
| With potassium tert-butylate; trimethylaluminum In toluene at 0℃; for 2h; | 100% |
-
-
1663-39-4
tert-Butyl acrylate

- poly-tert-butyl acrylate, obtained by anionic polymerization, initiated with the tBuOK/iBu3Al system, Mn = 5300, Mw/Mn = 1.26; monomer(s): tert-butyl acrylate
-
poly-tert-butyl acrylate, obtained by anionic polymerization, initiated with the tBuOK/iBu3Al system, Mn = 5300, Mw/Mn = 1.26; monomer(s): tert-butyl acrylate

| Conditions | Yield |
|---|---|
| With potassium tert-butylate; triisobutylaluminum In toluene at 0℃; for 1h; | 100% |
-
-
1663-39-4
tert-Butyl acrylate

-
-
146743-72-8
3-[Bis-(2-tert-butoxycarbonyl-ethyl)-amino]-propionic acid tert-butyl ester

| Conditions | Yield |
|---|---|
| With ammonium hydroxide In methanol at 120℃; under 7500.75 Torr; for 3h; Microwave irradiation; | 100% |
| With ammonia In methanol at 20℃; for 6h; | 98% |
-
-
636-98-6
p-nitrobenzene iodide

-
-
1663-39-4
tert-Butyl acrylate

-
-
370839-59-1
(E)-tert-butyl 3-(4-nitrophenyl)prop-2-enoate

| Conditions | Yield |
|---|---|
| With 1,4-diaza-bicyclo[2.2.2]octane; potassium carbonate; palladium diacetate In N,N-dimethyl-formamide at 80℃; for 4h; Heck cross-coupling reaction; | 100% |
| With tetrabutyl ammonium fluoride; palladium dichloride at 40℃; for 3h; Heck reaction; Inert atmosphere; neat (no solvent); | 100% |
| With 1,4-diaza-bicyclo[2.2.2]octane; poly(ethylene glycol)-400; potassium carbonate; palladium diacetate In water at 80℃; for 4h; Heck cross-coupling; | 98% |
-
-
876061-13-1
2-(benzylsulfanyl)-N-(2-bromo-4,6-difluorophenyl)-N-methylacetamide

-
-
1663-39-4
tert-Butyl acrylate

-
-
876061-15-3
(E)-tert-butyl 3-(2-(2-(benzylsulfanyl)-N-methylacetamido)-3,5-difluorophenyl)acrylate

| Conditions | Yield |
|---|---|
| With triethylamine; tris-(o-tolyl)phosphine; palladium diacetate In o-xylene at 100℃; for 7h; Heck reaction; microwave irradiation; | 100% |
| With triethylamine; tris-(o-tolyl)phosphine; palladium diacetate In o-xylene at 100℃; for 7h; Heck coupling; microwave irradiation; | 87% |

| Conditions | Yield |
|---|---|
| With C33H33N2(1+)*Cl(1-); palladium diacetate; potassium carbonate In water; N,N-dimethyl-formamide for 2h; Reagent/catalyst; Heck Reaction; Inert atmosphere; Sealed tube; Heating; | 100% |
| With C30H42Cl2N2Pd; potassium carbonate In N,N-dimethyl-formamide at 80℃; for 8h; Catalytic behavior; Reagent/catalyst; Heck Reaction; Inert atmosphere; Schlenk technique; | 99% |
| With potassium carbonate In N,N-dimethyl-formamide at 80℃; for 4h; Heck Reaction; | 98% |
-
-
1663-39-4
tert-Butyl acrylate

| Conditions | Yield |
|---|---|
| With potassium tert-butylate; triisobutylaluminum In hexane; toluene at 0℃; for 1h; | 100% |
-
-
1663-39-4
tert-Butyl acrylate

| Conditions | Yield |
|---|---|
| With potassium tert-butylate; triisobutylaluminum In hexane; toluene at 0℃; for 2h; | 100% |
Related products
Downstream Products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View

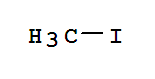




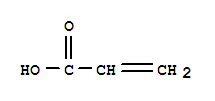








 F,
F, Xn
Xn