-
Name
Octanoic acid
- EINECS 204-677-5
- CAS No. 124-07-2
- Article Data515
- CAS DataBase
- Density 0.929 g/cm3
- Solubility 0.68 g/L (20 ºC)
- Melting Point 16 °C
- Formula C8H16O2
- Boiling Point 239.33 °C at 760 mmHg
- Molecular Weight 144.214
- Flash Point 107.379 °C
- Transport Information UN 3265 8/PG 3
- Appearance colourless oily liquid
- Safety 26-36/39-45-36/37/39-25-27
- Risk Codes 34
-
Molecular Structure
-
Hazard Symbols
 C,
C, Xi
Xi
- Synonyms 1-Heptanecarboxylic acid;n-Caprylic acid;n-Octanoic acid;n-Octylic acid;
- PSA 37.30000
- LogP 2.43150
Synthetic route

| Conditions | Yield |
|---|---|
| With nickel(II) iodide; manganese; C36H40N2 In N,N-dimethyl-formamide at 25℃; under 760.051 Torr; for 20h; regioselective reaction; | 92% |
| With rubidium carbonate; diethyl 2,6-dimethyl-1,4-dihydropyridine-3,5-dicarboxylate; (4s,6s)-2,4,5,6-tetra(9H-carbazol-9-yl)isophthalonitrile; (6,6’-dimethyl-2,2'-bipyridine)nickel(II) dibromide; tetra-(n-butyl)ammonium iodide In N,N-dimethyl-formamide at 10℃; under 750.075 Torr; for 20h; Schlenk technique; Sealed tube; Irradiation; | 48% |
| Stage #1: 2-bromoheptane; carbon dioxide With nickel(II) bromide dimethoxyethane; C32H32N2; C42H34F10IrN4(1+)*F6P(1-); N-ethyl-N,N-diisopropylamine; lithium tert-butoxide In N,N-dimethyl-formamide at 30℃; for 24h; Microwave irradiation; Schlenk technique; Stage #2: With hydrogenchloride In water; N,N-dimethyl-formamide Reagent/catalyst; | 25% |
| Stage #1: carbon dioxide With nickel(II) iodide; manganese; C36H40N2 In N,N-dimethyl-formamide at 25℃; under 760.051 Torr; Schlenk technique; Stage #2: 2-bromoheptane In N,N-dimethyl-formamide at 25℃; under 760.051 Torr; for 20h; Schlenk technique; Stage #3: With hydrogenchloride In water; N,N-dimethyl-formamide Reagent/catalyst; | 76 %Spectr. |

| Conditions | Yield |
|---|---|
| With nitric acid for 0.333333h; Ambient temperature; sonication; | 100% |
| With nitric acid for 0.333333h; Ambient temperature; sonication; | 100% |
| With ruthenium trichloride; iodobenzene; potassium peroxomonosulfate In water; acetonitrile at 20℃; for 16h; | 100% |

| Conditions | Yield |
|---|---|
| With tris[2-(4,6-difluorophenyl)pyridinato-C2,N]-iridium(III); oxygen In acetonitrile at 20℃; Irradiation; Sealed tube; Green chemistry; chemoselective reaction; | 99% |
| With diphenyl diselenide; dihydrogen peroxide In water at 20℃; for 3h; Green chemistry; | 99% |
| With copper acetylacetonate; oxygen; sodium hydroxide; 1,3-bis(2,4,6-trimethylphenyl)-2-imidazolidinylidene In water at 50℃; under 760.051 Torr; for 12h; Sealed tube; | 99% |

| Conditions | Yield |
|---|---|
| With nickel(II) iodide; manganese; C36H40N2 In N,N-dimethyl-formamide at 25℃; under 760.051 Torr; for 20h; regioselective reaction; | 81% |
-
-
1974-04-5
2-bromoheptane

-
-
124-38-9
carbon dioxide

-
A
-
116454-37-6, 128441-06-5, 1188-02-9
2-methylheptanoic acid

-
B
-
124-07-2
Octanoic acid

| Conditions | Yield |
|---|---|
| Stage #1: 2-bromoheptane; carbon dioxide With nickel(II) iodide; manganese; 2,9-diethyl-4,7-diphenyl-1,10-phenanthroline In N,N-dimethyl-formamide at 30℃; under 760.051 Torr; for 17h; Schlenk technique; Stage #2: With hydrogenchloride In water; N,N-dimethyl-formamide Reagent/catalyst; Temperature; Solvent; | A n/a B 72% |
| With rubidium carbonate; diethyl 2,6-dimethyl-1,4-dihydropyridine-3,5-dicarboxylate; (4s,6s)-2,4,5,6-tetra(9H-carbazol-9-yl)isophthalonitrile; (6,6’-dimethyl-2,2'-bipyridine)nickel(II) dibromide In N,N-dimethyl-formamide at 10℃; under 750.075 Torr; for 20h; Reagent/catalyst; Schlenk technique; Sealed tube; Irradiation; Overall yield = 48 %; Overall yield = 17.3 mg; |


| Conditions | Yield |
|---|---|
| With nickel(II) iodide; manganese; C36H40N2 In N,N-dimethyl-formamide at 25℃; under 760.051 Torr; for 20h; regioselective reaction; | 72% |

| Conditions | Yield |
|---|---|
| With palladium on activated carbon; W(OTf)6; hydrogen In neat (no solvent) at 135℃; under 760.051 Torr; for 12h; | 98% |
| With palladium 10% on activated carbon; W(OTf)6; hydrogen at 135℃; under 760.051 Torr; for 12h; | 92% |
-
-
116435-51-9, 1974-05-6, 116724-27-7
3-bromoheptane

-
-
124-38-9
carbon dioxide

-
A
-
116454-37-6, 128441-06-5, 1188-02-9
2-methylheptanoic acid

-
B
-
124-07-2
Octanoic acid

| Conditions | Yield |
|---|---|
| Stage #1: 3-bromoheptane; carbon dioxide With nickel(II) iodide; manganese; C36H40N2 In N,N-dimethyl-formamide at 25℃; under 760.051 Torr; for 17h; Schlenk technique; Stage #2: With hydrogenchloride In water; N,N-dimethyl-formamide | A n/a B 81% |


| Conditions | Yield |
|---|---|
| With nickel(II) iodide; manganese; C36H40N2 In N,N-dimethyl-formamide at 25℃; under 760.051 Torr; for 20h; regioselective reaction; | 56% |

-
-
124-07-2
Octanoic acid

| Conditions | Yield |
|---|---|
| With palladium 10% on activated carbon; W(OTf)6; hydrogen; acetic acid at 180℃; under 22502.3 Torr; for 10h; Autoclave; | 80% |
| With palladium on activated charcoal; W(OTf)6; hydrogen; acetic acid at 180℃; under 22502.3 Torr; for 10h; Autoclave; | 80% |
-
-
998-93-6
4-bromoheptane

-
-
124-38-9
carbon dioxide

-
A
-
116454-37-6, 128441-06-5, 1188-02-9
2-methylheptanoic acid

-
B
-
124-07-2
Octanoic acid

| Conditions | Yield |
|---|---|
| Stage #1: 4-bromoheptane; carbon dioxide With nickel(II) iodide; manganese; C36H40N2 In N,N-dimethyl-formamide at 25℃; under 760.051 Torr; for 17h; Schlenk technique; Stage #2: With hydrogenchloride In water; N,N-dimethyl-formamide | A n/a B 72% |


| Conditions | Yield |
|---|---|
| With hydrogenchloride; sodium sulfate; OsO4 In ethyl acetate; N,N-dimethyl-formamide; tert-butyl alcohol | 90% |


| Conditions | Yield |
|---|---|
| With carbon monoxide; hydrogen; triethylamine; acetylacetonatodicarbonylrhodium(l); triphenylphosphine In dichloromethane at 25℃; under 7500.75 Torr; for 24h; | 42% |

| Conditions | Yield |
|---|---|
| With nickel(II) iodide; manganese; C36H40N2 In N,N-dimethyl-formamide at 25℃; under 760.051 Torr; for 20h; regioselective reaction; | 50% |
-
-
14779-25-0
3-(5-methylfuran-2-yl) acrylic acid

-
-
124-07-2
Octanoic acid

| Conditions | Yield |
|---|---|
| With palladium on activated charcoal; W(OTf)6; hydrogen; acetic acid at 180℃; under 22502.3 Torr; for 10h; Autoclave; | 86% |

| Conditions | Yield |
|---|---|
| With 1,10-Phenanthroline; oxygen; cobalt(II) nitrate In dimethyl sulfoxide at 100℃; under 9000.9 Torr; for 12h; | A 36% B 53% |

| Conditions | Yield |
|---|---|
| Stage #1: non-1-ene With oxygen; ozone In tetrahydrofuran at 0℃; Stage #2: With semicarbazide hydrochloride In tetrahydrofuran at 0 - 20℃; Inert atmosphere; | 95% |
| Stage #1: non-1-ene With ozone In dichloromethane; acetic acid at 0℃; Stage #2: With semicarbazide hydrochloride In dichloromethane; acetic acid at 0 - 20℃; | 95% |
| With Oxone; osmium(VIII) oxide In N,N-dimethyl-formamide; tert-butyl alcohol at 20℃; for 3h; | 90% |
-
-
592-76-7
1-Heptene

-
-
124-38-9
carbon dioxide

-
A
-
116454-37-6, 128441-06-5, 1188-02-9
2-methylheptanoic acid

-
B
-
124-07-2
Octanoic acid

| Conditions | Yield |
|---|---|
| With nickel(II) iodide; manganese; Bathocuproine; water In N,N-dimethyl-formamide at 50℃; under 760.051 Torr; for 40h; | A n/a B 49% |


| Conditions | Yield |
|---|---|
| With ethanol; lithium; nickel dichloride; 4,4'-di-tert-butylbiphenyl In tetrahydrofuran at 20℃; for 12h; | 93% |
| With sodium hydroxide; Triethoxysilane; water; palladium diacetate for 4h; Ambient temperature; | 92% |
| With sodium tetrahydroborate; sodium hydroxide In water at 20 - 60℃; | 92% |
| With ethanol; sodium |

| Conditions | Yield |
|---|---|
| With hydroxylamine nitrate; sodium hydroxide In methanol at 0 - 50℃; for 3h; | A 84% B 3 g |

| Conditions | Yield |
|---|---|
| Stage #1: non-1-ene With oxygen; ozone In tetrahydrofuran at 0℃; Stage #2: decanoic acid hydrazide In tetrahydrofuran at 0 - 20℃; for 72h; | A 12% B 78% |


| Conditions | Yield |
|---|---|
| With hydrogenchloride; 2,2,6,6-Tetramethyl-1-piperidinyloxy free radical; sodium nitrite In dichloromethane; water at 20℃; under 760.051 Torr; for 12h; in air; | A 92% B 5.2% |
| With Succinimide; sodium hypochlorite solution; 2,2,6,6-Tetramethyl-1-piperidinyloxy free radical; potassium carbonate In ethyl acetate at 0 - 10℃; for 2h; | A 86% B 7% |
| With potassium chromate; copolyesteramide (from N,N'-bis(4-methoxycarbonylbenzoyl)hexamethylenediamine, 1,6-hexanediol, poly(ethylene glycol)); sulfuric acid In dichloromethane at -5℃; for 0.25h; | A 83% B 2.1% |
-
-
1871-67-6
(E)-oct-2-enoic acid

-
-
201230-82-2
carbon monoxide

-
A
-
124-13-0
Octanal

-
B
-
124-07-2
Octanoic acid

-
C
-
1039763-53-5
C9H16O3

| Conditions | Yield |
|---|---|
| With tris(2,4-di-tert-butylphenyl)phosphite; hydrogen; acetylacetonatodicarbonylrhodium(l) In dichloromethane at 25℃; under 7500.75 Torr; for 24h; | A 23% B 33% C 12% |

-
-
592-77-8
hept-2-ene

-
-
124-38-9
carbon dioxide

-
A
-
116454-37-6, 128441-06-5, 1188-02-9
2-methylheptanoic acid

-
B
-
124-07-2
Octanoic acid

| Conditions | Yield |
|---|---|
| With nickel(II) iodide; manganese; Bathocuproine; water In N,N-dimethyl-formamide at 50℃; under 760.051 Torr; for 40h; | A n/a B 15% |


| Conditions | Yield |
|---|---|
| With hydrogen; trifluoroacetic acid In n-heptane at 190℃; under 22502.3 Torr; for 10h; Reagent/catalyst; Temperature; Pressure; Autoclave; High pressure; | 91 %Chromat. |
-
-
1871-67-6
(E)-oct-2-enoic acid

-
-
201230-82-2
carbon monoxide

-
A
-
124-13-0
Octanal

-
B
-
124-07-2
Octanoic acid

| Conditions | Yield |
|---|---|
| With hydrogen; acetylacetonatodicarbonylrhodium(l) In dichloromethane at 25℃; under 7500.75 Torr; for 24h; | |
| With hydrogen; acetylacetonatodicarbonylrhodium(l) In dichloromethane at 25℃; under 7500.75 Torr; for 20.5h; | A 91 % Chromat. B 4.5 % Spectr. |


| Conditions | Yield |
|---|---|
| With sodium bromate; sodium hydrogensulfite for 2h; Ambient temperature; | A 3% B 94% |
| Stage #1: octanol With gold on titanium oxide In water at 90℃; for 0.166667h; Inert atmosphere; Stage #2: With dihydrogen peroxide In water at 90℃; for 1.08333h; Inert atmosphere; chemoselective reaction; | A 90% B n/a |
| With sodium tungstate; dihydrogen peroxide In water at 90℃; for 4h; | A 87% B 2% |

| Conditions | Yield |
|---|---|
| Stage #1: non-1-ene With oxygen; ozone In dichloromethane; acetic acid at 0℃; Stage #2: toluene-4-sulfonic acid hydrazide In dichloromethane; acetic acid at 20℃; Inert atmosphere; | A 19% B 42% |


| Conditions | Yield |
|---|---|
| With oxygen at 100℃; under 3750.38 Torr; for 5h; Autoclave; | 11 %Chromat. |
-
-
64-17-5
ethanol

-
-
124-13-0
Octanal

-
A
-
54889-48-4
1,1-diethoxy-octane

-
B
-
124-07-2
Octanoic acid

-
C
-
106-32-1
octanoic acid ethyl ester

| Conditions | Yield |
|---|---|
| With oxygen at 100℃; under 3750.38 Torr; for 5h; Autoclave; | A 27 %Chromat. B 8 %Chromat. C 7 %Chromat. |
| With oxygen at 100℃; under 3750.38 Torr; for 5h; Reagent/catalyst; Autoclave; | A 8 %Chromat. B 13 %Chromat. C 58 %Chromat. |
| With oxygen at 100℃; under 3750.38 Torr; for 5h; Reagent/catalyst; Autoclave; | A 63 %Chromat. B 6 %Chromat. C 12 %Chromat. |

-
-
124-07-2
Octanoic acid

-
-
54107-24-3
3-(2-vinyloxyethoxy)-1,2-propylene carbonate

-
-
127827-87-6
Octanoic acid 1-[2-(2-oxo-[1,3]dioxolan-4-ylmethoxy)-ethoxy]-ethyl ester

| Conditions | Yield |
|---|---|
| With heptafluorobutyric Acid at 75℃; for 3h; | 100% |
-
-
124-07-2
Octanoic acid

-
-
59565-09-2
2-(vinyloxy)ethyl isothiocyanate

| Conditions | Yield |
|---|---|
| With triethylamine at 45 - 50℃; for 1h; | 100% |
-
-
124-07-2
Octanoic acid

-
-
125034-39-1
2-(Trimethylsilyl)ethyl 6-O-tert-butyldimethylsilyl-2-deoxy-4-O-diphenoxyphosphinyl-3-O-<(3R)-3-hydroxytetradecanoyl>-2-<(3R)-3-<(2-trimethylsilylethoxy)methoxy>tetradecanamido>-β-D-glucopyranoside

-
-
125056-34-0
2-(Trimethylsilyl)ethyl 6-O-tert-butyldimethylsilyl-2-deoxy-4-O-diphenoxyphosphinyl-3-O-<(3R)-3-octanoyloxytetradecanoyl>-2-<(3R)-3-<(2-trimethylsilylethoxy)methoxy>tetradecanamido>-β-D-glucopyranoside

| Conditions | Yield |
|---|---|
| With dmap; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride In dichloromethane Ambient temperature; | 100% |

| Conditions | Yield |
|---|---|
| With palladium on silica gel; hydrogen at 300℃; under 760.051 Torr; for 4h; Temperature; Flow reactor; | 100% |
| With hydrogen; silica gel; palladium at 330℃; Ni/Al2O3, 180 deg C; | 97% |
| With Au0012O19676(00)Pd042(98)Si038; hydrogen at 260℃; under 760.051 Torr; Catalytic behavior; Reagent/catalyst; |

| Conditions | Yield |
|---|---|
| In 1,2-dimethoxyethane at 0℃; for 3h; Condensation; | 100% |

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at 40 - 50℃; for 5h; | 100% |

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at 40 - 50℃; for 5h; | 100% |

| Conditions | Yield |
|---|---|
| In cyclohexane at 40℃; Solvent; | 100% |
-
-
143-07-7
lauric acid

-
-
75-94-5
trichlorovinylsilane

-
-
124-07-2
Octanoic acid

-
-
544-63-8
n-tetradecanoic acid

-
-
1330066-17-5
C36H68O6Si

| Conditions | Yield |
|---|---|
| In toluene at 60 - 150℃; for 4h; | 99.82% |


| Conditions | Yield |
|---|---|
| With choline chloride; zinc(II) chloride at 110℃; for 6h; | 99% |
| With immobilized lipase Novozym 435 from Candida antarctica B supported on a macroporous acrylic resin In carbon dioxide at 63.7℃; under 76657.7 Torr; for 0.333333h; Supercritical conditions; Enzymatic reaction; liquid CO2; | 99.5% |

| Conditions | Yield |
|---|---|
| at 105℃; for 6h; Temperature; Concentration; | 99.24% |

| Conditions | Yield |
|---|---|
| With acetic acid at 275℃; for 0.5h; | 99.2% |
| With tungsten(VI) oxide at 175℃; under 1 Torr; for 22h; Reagent/catalyst; | 93% |
| at 100℃; Beim Erhitzen in Gegenwart von aus Naphthalin,Oelsaeure und konz.Schwefelsaeure in Petrolaether dargestelltem Twitchells Reagens; |

| Conditions | Yield |
|---|---|
| polyaniline sulfate at 70℃; for 24h; | 99% |
| With N-Bromosuccinimide at 70℃; for 2h; Time; | 97% |
| With 1,3-dibromo-5,5-dimethylimidazolidine-2,4-dione at 70℃; for 2h; | 97% |

| Conditions | Yield |
|---|---|
| With samarium diiodide; heptanal; samarium(III) trifluoromethanesulfonate In tetrahydrofuran; methanol; potassium hydroxide at 20℃; for 0.075h; Reduction; | 99% |
| With 1,1,3,3-Tetramethyldisiloxane; copper(II) bis(trifluoromethanesulfonate) In toluene at 80℃; for 16h; sealed tube; | 91% |
| With hydrogen In neat (no solvent) at 180℃; under 37503.8 Torr; for 12h; | 91% |

| Conditions | Yield |
|---|---|
| With Novozym 435 (Candida antarctica lipase B on Lewatit E); hydroxylamine In water at 40℃; for 20h; Condensation; Enzymatic reaction; | 99% |
| Stage #1: Octanoic acid With acetic anhydride for 0.166667h; Stage #2: With hydroxylamine hydrochloride for 0.666667h; | 95.81% |
| With liverextract; hydroxylamine | |
| With hydroxylamine; 1,1'-carbonyldiimidazole | |
| With hydroxylamine; adenosine monophosphate ligase SfaB from Streptomyces thioluteus; ATP; magnesium chloride; Cleland's reagent In aq. buffer at 30℃; for 6h; pH=8; Enzymatic reaction; |

| Conditions | Yield |
|---|---|
| With [Cl(C6F13C2H4)2SnOSn(C2H4C6F13)2Cl]2 In various solvent(s) at 150℃; for 10h; | 99% |
| With pyrographite; toluene-4-sulfonic acid for 0.00777778h; Esterification; Microwave irradiation (675 W); | 90% |
| With tris(2-methoxyphenyl)bismuthine In benzene for 12h; Heating; | 86% |

| Conditions | Yield |
|---|---|
| With pyridine; <(chlorosulfinyloxy)methylene>dimethylammonium chloride In dichloromethane at 20℃; for 6h; | 99% |

| Conditions | Yield |
|---|---|
| With pyridine; <(chlorosulfinyloxy)methylene>dimethylammonium chloride In dichloromethane at 20℃; for 6h; | 99% |
Octanoic acid Consensus Reports
Octanoic acid Specification
The IUPAC name of this chemical is Octanoic acid. With the CAS registry number 124-07-2 and EINECS registry number 204-677-5, it is also named as 1-Heptanecarboxylicacid. In addition, the molecular formula is C8H16O2 and the molecular weight is 144.21. It is a kind of colourless oily liquid and belongs to the classes of Miscellaneous Natural Products; Alkylcarboxylic Acids; Monofunctional & alpha,omega-Bifunctional Alkanes; Monofunctional Alkanes. And it is minimally soluble in water with a slightly unpleasant rancid-like smell and taste.
Physical properties about this chemical are: (1)ACD/LogP: 2.74; (2)ACD/LogD (pH 5.5): 1.94; (3)ACD/LogD (pH 7.4): 0.146; (4)ACD/BCF (pH 5.5): 11.324; (5)ACD/BCF (pH 7.4): 1; (6)ACD/KOC (pH 5.5): 117.554; (7)ACD/KOC (pH 7.4): 1.887; (8)#H bond acceptors: 2; (9)#H bond donors: 1; (10)#Freely Rotating Bonds: 6; (11)Polar Surface Area: 37.3 Å2; (12)Index of Refraction: 1.437; (13)Molar Refractivity: 40.676 cm3; (14)Molar Volume: 155.215 cm3; (15)Polarizability: 16.125 ×10-24cm3; (16)Surface Tension: 33.075 dyne/cm; (17)Density: 0.929 g/cm3; (18)Flash Point: 107.379 °C; (19)Enthalpy of Vaporization: 50.325 kJ/mol; (20)Boiling Point: 239.33 °C at 760 mmHg; (21)Vapour Pressure: 0.022 mmHg at 25°C.
Preparation of Octanoic acid: it can be prepared by octanoyl chloride. The other product is octanoic acid octyl ester. This reaction will need reagent LiInH4 and solvent diethyl ether. The reaction should react in temperature of 0 °C for 1 hour and in the room temperature for 12 hours. The yield is about 22%.
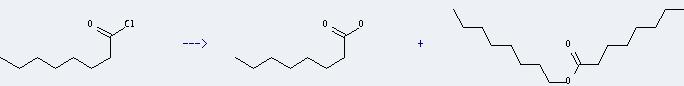
Uses of Octanoic acid: it is used commercially in the production of esters used in perfumery and also in the manufacture of dyes. And it is also used in the treatment of some bacterial infections and used as an algaecide, bactericide, and fungicide in nurseries, greenhouses, garden centers, and interiorscapes on ornamentals. Moreover, it is also used as disinfectant in health care facilities, schools/colleges, animal care/veterinary facilities, industrial facilities, office buildings, recreational facilities. In addition, it can be used to get octan-1-ol. This reaction will need reagents Sm and 10percent HCl and solvent methanol. The reaction time is 30 minutes with ambient temperature. The yield is about 39%.

When you are using this chemical, please be cautious about it as the following:
This chemical can cause burns. During using it, wear suitable protective clothing, gloves and eye/face protection and avoid contact with eyes. In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. And in case of accident or if you feel unwell, seek medical advice immediately (show the label whenever possible.).
You can still convert the following datas into molecular structure:
(1)SMILES: CCCCCCCC(=O)O
(2)InChI: InChI=1/C8H16O2/c1-2-3-4-5-6-7-8(9)10/h2-7H2,1H3,(H,9,10)
(3)InChIKey: WWZKQHOCKIZLMA-UHFFFAOYAH
The toxicity data is as follows:
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| mouse | LD50 | intravenous | 600mg/kg (600mg/kg) | BEHAVIORAL: CONVULSIONS OR EFFECT ON SEIZURE THRESHOLD | Acta Pharmacologica et Toxicologica. Vol. 18, Pg. 141, 1961. |
| rabbit | LD50 | skin | > 5gm/kg (5000mg/kg) | Food and Cosmetics Toxicology. Vol. 19, Pg. 237, 1981. | |
| rat | LD50 | oral | 10080mg/kg (10080mg/kg) | BEHAVIORAL: SOMNOLENCE (GENERAL DEPRESSED ACTIVITY) GASTROINTESTINAL: "HYPERMOTILITY, DIARRHEA" | Food and Cosmetics Toxicology. Vol. 2, Pg. 327, 1964. |
Related Products
- Octanoic acid
- Octanoic acid zirconium salt
- Octanoic acid, 2,2-dimethyl-, ethenyl ester
- Octanoic acid, 2-bromo-
- Octanoic acid, 2-butyl-
- Octanoic acid, 2-ethyl-
- Octanoic acid, 4-oxo-
- Octanoic acid, 5-oxo-
- Octanoic acid, 6,8-dimercapto-
- Octanoic acid, lead salt
- 1240725-47-6
- 124072-61-3
- 124072-89-5
- 12408-47-8
- 124-09-4
- 12410-14-9
- 124-10-7
- 124111-47-3
- 12411-64-2
- 124-11-8
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View