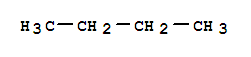Dayang Chem (Hangzhou) Co.,Ltd.
Dayangchem's R&D center can offer custom synthesis according to the contract research and development services for the fine chemicals, pharmaceutical, biotechnique and some of the other chemicals. DayangChem can provide different quantities
Henan Allgreen Chemical Co.,Ltd
he company has advanced technology, as well as a large number of excellent R & D team, to provide customers from the grams to one hundred kilograms and tons of high-quality products, competitive prices and quality se T rvice Appearance:
Cas:592-76-7
Min.Order:1 Kilogram
Negotiable
Type:Manufacturers
inquiryChemwill Asia Co., Ltd.
Our main production base is located in Xuzhou industry park. We are certified both to the ISO 9001 and ISO 14001 Standards, have a safety management system in place.Our R&D team masters core technology for process-design of target building block
Henan Tianfu Chemical Co., Ltd.
Now we would like to update our product list for you, kindly check the below information: 1. Catalyst series ( such as Noble Metal Catalyst, Phosphorous ligand, etc ) 2. Pharmaceutical Intermediate ( such as diabtes series, Anti
SHANGHAI T&W PHARMACEUTICAL CO., LTD.
A substitute for perfluorooctanoic acid, mainly used as a surfactant, dispersant, additive, etc Appearance:White solid or Colorless liquid Purity:99.3 % We will ship the goods in a timely manner as required We can provide relevant documents acc
Hangzhou J&H Chemical Co., Ltd.
J&H CHEM R&D center can offer custom synthesis according to the contract research and development services for the fine chemicals, pharmaceutical, biotechnique and some of the other chemicals. J&H CHEM has some Manufacturing base in Jia
Hangzhou Huarong Pharm Co., Ltd.
Hangzhou Huarong Pharm Co., Ltd.established since 2006 , has been actively developing specialty products for Finished Dosages, APIs, Intermediates, and Fine chemicals markets in North America, Europe, Korea, Japan, Mid-East and all over the World. Hu
Hunan chemfish Pharmaceutical co.,Ltd
Hunan chemfish Pharmaceutical co.,Ltd.located in Lugu High-tech industral park ,Hunan province . with its own R&D center and more than 10000㎡manufacture plant . Chemfish owns 40 reactors from 1000L to 8000L. With complete auxiliary equipment as
Zhuozhou Wenxi import and Export Co., Ltd
Product Description Description & Specification Category Pharmaceutical Raw Materials, Fine Chemicals, Bulk drug Standard Medical standard
Cas:592-76-7
Min.Order:1 Kilogram
FOB Price: $112.0
Type:Trading Company
inquiryAntimex Chemical Limied
Ansciep Chemical is a professional enterprise manufacturing and distributing fine chemicals and speciality chemicals. We have been dedicated to heterocycle compounds and phenyl rings for tens of years. This is our mature product for export. Our quali
Changchun Artel lmport and Export trade company
Supply top quality products with a reasonable price Application:api
Zhengzhou Kingorgchem Chemical Technology Co., Ltd.
Zhengzhou Kingorgchem Chemical Technology Co., Ltd. was founded on the basis of Organophosphorus Chemistry Lab of Institute of Chemistry Henan Academy of Sciences in 2015. The laboratory covers 600 m2 and the pilot plant covers 2000 m2. Kingorgchem i
Cas:592-76-7
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryXiamen BaiFuchem Co.,Ltd
BaiFuChem is a Professional chemical raw material supplier in China, our main products include Biochemical , Pharma Intermediate and Organic chemical etc. BaiFuChem have wealth of products,experience , expertise and state-of-the-art
Hangzhou Share Chemical Co., Ltd
At Share Chemical Company, we scrupulously abide by our policy of “Excellent Quality at a Reasonable Price”. We strive to satisfy all of our customers by providing the finest quality products supported by the finest in customer servi
Cerametek Materials(ShenZhen)Co., Ltd.
Cerametek Materials (CMT) is your industrial and R&D supplier: 1. Semiconductor materials: Compounds of II, III, IV, V, VI groups such as Metal Sulfides, Selenides, Tellurides, Arsenide, Antimonide, Phosphide... 2. Advanced ceramic and crys
Shanghai united Scientific Co.,Ltd.
United Scientific Company Located in Shanghai of China , is a competitive player in the global specialty and fine chemical market. Fenghua has both the expertise and flexibility to produce a wide range of chemicals. Focusing on developing the innovat
Nanjing Raymon Biotech Co., Ltd.
1-Heptene Storage:keep in dry and cool condition Package:25kg or according to cutomer's demand Application:Chemical research/pharma intermediate Transportation:By Sea,by Air,By courier like DHL or Fedx. Port:Shanghai/Shenzhen
Hangzhou Yierdechem Co. Ltd
With about ten years experiences in the field of pharmaceutical chemicals, Yierdechem has established solid business cooperation relationships with many large companys worldwide.As a leading supplier of API and pharmaceutical intermediates, holds its
Debye Scientific
Debyesci is here who supplied several kinds of chemical products to global pharmaceutical, drug discovery, agrochemical and biotechnology industries for four yearsOur key scientific leadership team has gained experience in top research and developmen
Changzhou Foreign Trade Corp
CFTC?PharmaChem?is?a?service?based?company?in?China,?a?pharmachem?division?of?Changzhou?Foreign?Trade?Corp.,supplying?raw?materials,?intermediates,?APIs?and?fine?chemicals?for?the?pharmaceutical?and?specialty?chemical?industries?worldwide. Applicatio
yuyongmei
YUYONGMEI was established in Aug,1999, located in the industrial park of Nanjing University of Technology, It is a private enterprises in Jiangsu Province with 1200 square meters’ R&D center. Our R&D center has a well-equipped synthetic laboratory a
Chemical Co.Ltd
Manufacturer,strong R&D,professional team Storage:Store in a cool, dry place. Store in a tightly closed container. Package:according to your requirement Application:ZhiShang Chemical is owned by ZhiShang Group, is a professional new-type chemicals en
Sigma-Aldrich Chemie GmbH
Package:5 g in ampule
Synthetic route

| Conditions | Yield |
|---|---|
| With quinoline; oct-1-ene; hydrogen; Lindlar's catalyst | A 98.3% B 1.7% |
| With hydrogen; silica gel; rhodium(1+) In ethanol; toluene at 40℃; under 1320.1 Torr; Product distribution; | A 32% B 1% |
| With hydrogen; copper at 200℃; |
-
-
13389-36-1
6-iodohept-1-ene

-
A
-
592-76-7
1-Heptene

-
B
-
73783-40-1
1-(iodomethyl)-2-methylcyclopentane

-
C
-
1192-18-3
cis-1,2-dimethylcyclopentane

-
-
822-50-4
trans-1,2-dimethylcyclopentane

| Conditions | Yield |
|---|---|
| With lithium triethylborohydride In tetrahydrofuran for 2h; Product distribution; Mechanism; or with deuterated dicyclohexylphosphine (DCPD) or other reagents; | A 98% B 9% C n/a D n/a |


| Conditions | Yield |
|---|---|
| TR97305 at 300℃; Dehydrochlorination; | 97% |

| Conditions | Yield |
|---|---|
| With triphenylphosphine; copper dichloride In tetrahydrofuran at 60℃; | 96% |

-
-
592-76-7
1-Heptene

| Conditions | Yield |
|---|---|
| Cu(acac)2-PPh3 In tetrahydrofuran | 95% |

| Conditions | Yield |
|---|---|
| With hydrogen at 40℃; under 3000.3 Torr; for 0.333333h; Kinetics; Reagent/catalyst; Temperature; | 93% |
| With piperazine; hydrogen In ethanol at 80℃; under 4500.45 Torr; for 24h; | 90% |
| With methanol; hydrogen; nickel Hydrogenation; |

| Conditions | Yield |
|---|---|
| With bis(cyclopentadienyl)titanium dichloride; magnesium In tetrahydrofuran for 12h; Ambient temperature; | 92% |

| Conditions | Yield |
|---|---|
| With Grubbs catalyst first generation In toluene at 40℃; under 3750.38 Torr; for 0.5h; Temperature; Reagent/catalyst; Pressure; Inert atmosphere; Glovebox; Sealed tube; | 91.3% |
| With 1-Propyl acetate; ethylaluminum dichloride; tungsten(VI) oxychloride diethyl etherate In toluene at 85℃; under 2850.29 - 3000.3 Torr; for 2h; |
-
-
628-71-7
1-Heptyne

-
A
-
592-76-7
1-Heptene

-
B
-
592-78-9
hept-3-ene

-
C
-
592-77-8
hept-2-ene

-
D
-
142-82-5
n-heptane

| Conditions | Yield |
|---|---|
| With hydrogen; palladium dichloride In N,N-dimethyl-formamide under 18751.5 Torr; for 0.416667h; Product distribution; Ambient temperature; various time; | A 89% B 1.8% C 2.9% D 6.3% |

-
-
83367-40-2
1-chloromethanesulfonyl-hexane

-
-
592-76-7
1-Heptene

| Conditions | Yield |
|---|---|
| With sodium hydroxide; Aliquat 336 In dichloromethane for 30h; Heating; | 85% |
-
-
38334-98-4
6-bromo-1-heptene

-
-
16643-09-7
trimethylstannyl sodium

-
A
-
592-76-7
1-Heptene

-
B
-
76879-52-2
1-hepten-6-yltrimethyltin

-
D
-
822-50-4, 1192-18-3, 13012-46-9, 134932-08-4, 2452-99-5
1,2-dimethylcyclopentane

| Conditions | Yield |
|---|---|
| In tetrahydrofuran stannane added to bromide, stirred under nitrogen at 0°C 30 min; quenched with aq. ammonium chloride; | A 2.4% B 6.1% C 80.7% D 2.3% |
| With tert-butylamine In tetrahydrofuran stannane added to bromide, additive 10 molar equiv., stirred under nitrogen at 0°C 30 min; quenched with aq. ammonium chloride; | A 3% B 3.6% C 74.5% D 3% |
| In tetrahydrofuran bromide added to stannane, stirred under nitrogen at 0°C 30 min; quenched with aq. ammonium chloride; | A 2% B 13% C 69% D 1% |

-
-
15661-92-4
1-methyl-5-hexenyl chloride

-
-
16643-09-7
trimethylstannyl sodium

-
A
-
592-76-7
1-Heptene

-
B
-
76879-52-2
1-hepten-6-yltrimethyltin

-
D
-
822-50-4, 1192-18-3, 13012-46-9, 134932-08-4, 2452-99-5
1,2-dimethylcyclopentane

| Conditions | Yield |
|---|---|
| With tert-butylamine In tetrahydrofuran stannane added to chloride, 10 molar equiv. of additive, stirred under nitrogen at 0°C 3 h; quenched with aq. ammonium chloride; | A 0.4% B 79.9% C 8.7% D <1 |
| With dicyclohexylphosphane In tetrahydrofuran stannane added to chloride, 10 molar equiv. of additive, stirred under nitrogen at 0°C 3 h; quenched with aq. ammonium chloride; | A 10% B 78.7% C 2.2% D 1.6% |
| In tetrahydrofuran stannane added to chloride, stirred under nitrogen at 0°C 3 h; quenched with aq. ammonium chloride; | A 0.5% B 78.2% C 12.2% D <1 |


| Conditions | Yield |
|---|---|
| With palladium on silica gel; hydrogen at 245℃; under 760.051 Torr; for 4h; Temperature; Flow reactor; | A 12% B 70% C 18% |

-
-
13389-36-1
6-iodohept-1-ene

-
-
16643-09-7
trimethylstannyl sodium

-
A
-
592-76-7
1-Heptene

-
B
-
76879-52-2
1-hepten-6-yltrimethyltin

-
D
-
822-50-4, 1192-18-3, 13012-46-9, 134932-08-4, 2452-99-5
1,2-dimethylcyclopentane

| Conditions | Yield |
|---|---|
| In tetrahydrofuran stannane added to iodide, stirred under nitrogen at 0°C 30 min; quenched with aq. ammonium chloride; | A 8.2% B 9.5% C 69.2% D 8.6% |
| With tert-butylamine In tetrahydrofuran stannane added to iodide, additive 10 molar equiv., stirred under nitrogen at 0°C 30 min; quenched with aq. ammonium chloride; | A 6.7% B 7.2% C 62% D 10% |
| With cyclohexa-1,4-diene In tetrahydrofuran stannane added to iodide, additive 10 molar equiv., stirred under nitrogen at 0°C 30 min; quenched with aq. ammonium chloride; | A 6.2% B 18% C 52.2% D 6.4% |
| With dicyclohexylphosphane In tetrahydrofuran stannane added to iodide, additive 10 molar equiv., stirred under nitrogen at 0°C 30 min; quenched with aq. ammonium chloride; elem. anal.; | A 44.9% B 3.8% C 16% D 21.3% |

-
-
110-43-0
n-pentyl methyl ketone

-
A
-
592-76-7
1-Heptene

-
B
-
52390-72-4, 543-49-7
2-Heptanol

-
C
-
592-77-8
hept-2-ene

| Conditions | Yield |
|---|---|
| With isopropyl alcohol; magnesium oxide at 149.9℃; | A n/a B 66.3% C n/a |

-
-
22104-77-4
hept-2-en-1-ol

-
-
592-76-7
1-Heptene

| Conditions | Yield |
|---|---|
| With lithium aluminium tetrahydride In 1,2-dimethoxyethane for 120h; Heating; | 60% |

| Conditions | Yield |
|---|---|
| In diethyl ether for 20h; from -15 degC to 20 degC; | 60% |
| In diethyl ether for 20h; Mechanism; var. aldehydes: several Ge- and Cr- reagents; |
-
-
75-09-2
dichloromethane

-
-
44767-62-6
n-hexylmagnesium chloride

-
A
-
592-76-7
1-Heptene

-
B
-
111-65-9
octane

-
C
-
629-50-5
Tridecane

| Conditions | Yield |
|---|---|
| With C31H37ClN3NiO2(1-)*Li(1+) In tetrahydrofuran at 25℃; for 0.333333h; Inert atmosphere; Overall yield = 97.5 %; | A 59% B 14.3% C 23.6% |
| With C31H37ClFeN3O2 In tetrahydrofuran at 25℃; for 0.0833333h; Inert atmosphere; |


| Conditions | Yield |
|---|---|
| With aluminum oxide at 500℃; for 0.5h; | 51% |
| With aluminum oxide at 380 - 400℃; | |
| With aluminum oxide at 430 - 450℃; | |
| With colophonium at 350℃; | |
| With aluminum oxide at 350 - 380℃; |

| Conditions | Yield |
|---|---|
| In tetrahydrofuran; diethyl ether 1) -70 deg C, 1-2 h, 2) -70 - 20 deg C, 15 h; | 44% |

| Conditions | Yield |
|---|---|
| With Hoveyda-Grubbs catalyst second generation; ethene In toluene at 75℃; for 24h; Reagent/catalyst; Inert atmosphere; | 41% |
-
-
111-70-6
n-heptan1ol

-
A
-
592-76-7
1-Heptene

-
B
-
629-64-1
diheptyl ether

-
C
-
51197-57-0
2-heptyloxy-heptane

| Conditions | Yield |
|---|---|
| Al3+-montmorillonite at 200℃; for 4h; Product distribution; Mechanism; | A 9% B 39% C 1.5% D n/a E n/a |


| Conditions | Yield |
|---|---|
| With zirconium(IV) oxide at 450℃; under 760.051 Torr; for 2h; Inert atmosphere; | A 36% B 23% |

| Conditions | Yield |
|---|---|
| With P, S at 180 - 190℃; for 3.5h; Product distribution; | A 8.6% B 33.3% C 14.6% |


| Conditions | Yield |
|---|---|
| 33% | |
| With dibutyl ether | |
| Herstellung; | |
| With diethyl ether |
-
-
629-04-9
1-Bromoheptane

-
-
143-08-8
nonyl alcohol

-
A
-
592-76-7
1-Heptene

-
B
-
629-64-1
diheptyl ether

-
C
-
76043-65-7
heptyl-nonyl ether

| Conditions | Yield |
|---|---|
| With sodium hydroxide; tetrabutylammomium bromide at 80℃; for 6h; | A 4.5% B 12% C 86 % Chromat. |

-
-
693-03-8
n-butyl magnesium bromide

-
-
60-29-7
diethyl ether

-
-
557-40-4
Allyl ether

-
A
-
592-76-7
1-Heptene

-
B
-
107-18-6
allyl alcohol

-
C
-
107-02-8
acrolein

-
-
693-03-8
n-butyl magnesium bromide

-
-
60-29-7
diethyl ether

-
-
1746-13-0
allyl phenyl ether

-
A
-
592-76-7
1-Heptene

-
B
-
108-95-2
phenol

-
-
693-03-8
n-butyl magnesium bromide

-
-
60-29-7
diethyl ether

-
-
4125-43-3
O-allyl guaiacol

-
A
-
592-76-7
1-Heptene

-
B
-
90-05-1
2-methoxy-phenol

| Conditions | Yield |
|---|---|
| at 60℃; |


| Conditions | Yield |
|---|---|
| With hydrogen; molybdenum(VI) oxide at 400℃; | |
| With methyllithium; calcium carbonate | |
| Multi-step reaction with 2 steps 1: PCl5 2: sodium View Scheme | |
| Multi-step reaction with 2 steps 1: toluene; phosphorus pentachloride 2: sodium; cumene View Scheme |

| Conditions | Yield |
|---|---|
| Pt-thiourea Product distribution; Ambient temperature; various catalysts; | 100% |
| Pt-thiourea | 100% |
| Stage #1: 1-Heptene With diethylenetriaminepentaacetic acid (DTPA)-functionalized silica supported Pt at 60℃; for 0.5h; Stage #2: Dichloromethylsilane at 60℃; for 4h; Reagent/catalyst; | 98.9% |

| Conditions | Yield |
|---|---|
| at 90℃; for 5h; Inert atmosphere; | 100% |
| at 90℃; for 5h; Inert atmosphere; | 100% |
| With dihydrogen hexachloroplatinate; 7,8-dicarba-nido-undecaborate(1-) at 20℃; for 24h; Product distribution; other reaction time, various catalyst composition; | 78% |

| Conditions | Yield |
|---|---|
| With water; 4-methylmorpholine N-oxide; osmium; Cu-Al-hydrotalcite In toluene at 60℃; for 6h; | 100% |
| With 4-methylmorpholine N-oxide; polyaniline-supported Os In water; acetone; acetonitrile at 20℃; | 94% |
| With CF3O3S(1-)*C20H18F6N4O6OsS2(1+); dihydrogen peroxide In water; tert-butyl alcohol at 70℃; for 4h; pH=4; Inert atmosphere; | 89% |

| Conditions | Yield |
|---|---|
| Irradiation (UV/VIS); UV-irradiation at 25-50 °C for 18 h;; | 100% |
| Irradiation (UV/VIS); UV-irradiation at 25-50 °C for 18 h;; | 100% |
| With 2,2'-azobis(isobutyronitrile); diphenyl diselenide In neat (no solvent) heating a mixt. of tin compd., alkene, Se compd. and AIBN at 80°Cfor 2.5 h with stirring; | 96% |
-
-
592-76-7
1-Heptene

| Conditions | Yield |
|---|---|
| With tellurium tetrachloride In chloroform at 20℃; for 6h; regioselective reaction; | 100% |
| With tellurium tetrachloride In tetrachloromethane at 20℃; regioselective reaction; | 100% |

| Conditions | Yield |
|---|---|
| With tellurium tetrachloride In dichloromethane at 20℃; regioselective reaction; | 100% |
| With tellurium tetrachloride In chloroform at 20℃; for 20h; regioselective reaction; | 96% |
-
-
592-76-7
1-Heptene

| Conditions | Yield |
|---|---|
| With tellurium tetrachloride In benzene for 10h; Reflux; regioselective reaction; | 100% |

| Conditions | Yield |
|---|---|
| With tellurium tetrachloride In dichloromethane at 40℃; for 7h; Solvent; regioselective reaction; | 100% |
| Stage #1: 1-Heptene With tellurium tetrachloride In chloroform at 0 - 20℃; for 14h; Stage #2: ethanol In chloroform for 5h; Reflux; | 100% |
-
-
592-76-7
1-Heptene

| Conditions | Yield |
|---|---|
| With selenium dibromide In tetrachloromethane at -20 - 20℃; for 18h; regioselective reaction; | 100% |
| Stage #1: 1-Heptene With selenium(IV) oxide; hydrogen bromide In diethyl ether; water at 0 - 25℃; for 0.5h; Stage #2: With sodium metabisulfite In benzene at 5 - 25℃; for 2h; | 85% |
| With selenium dibromide In chloroform at 20 - 25℃; |
-
-
592-76-7
1-Heptene

| Conditions | Yield |
|---|---|
| With selenium(II) chloride In chloroform at -60 - 20℃; for 18h; regioselective reaction; | 100% |
| Stage #1: 1-Heptene With hydrogenchloride; selenium(IV) oxide In diethyl ether; water at 0 - 25℃; for 7h; Stage #2: With sodium metabisulfite In benzene at 5 - 25℃; for 2h; | 82% |

| Conditions | Yield |
|---|---|
| With rhodium(III) chloride trihydrate at 80℃; for 8h; | 99.6% |

| Conditions | Yield |
|---|---|
| for 2h; UV-irradiation; | 99.1% |

| Conditions | Yield |
|---|---|
| With Pd(II)(15-crown-5-phen)Cl2; dinitrogen monoxide In N,N-dimethyl acetamide; water at 150℃; under 2250.23 Torr; for 18h; | 99% |
| With 2(p-CH2-C6H4CN)2[(CH2)3SO3Na]2-calix[4]arene*PdCl2; oxygen; copper dichloride under 3750.3 Torr; for 2h; Wacker oxidation; | 89% |
| With dihydrogen peroxide In water; acetonitrile at 55℃; for 12h; Wacker Oxidation; | 77% |
-
-
592-76-7
1-Heptene

-
-
109761-18-4, 121406-00-6
3β-nitromethyl-2α-(2-isopentenyl)cyclopentan-1-one

-
-
121406-04-0, 121469-57-6, 132072-80-1
(2R,3S)-2-(3-Methyl-but-2-enyl)-3-(5-pentyl-4,5-dihydro-isoxazol-3-yl)-cyclopentanone

| Conditions | Yield |
|---|---|
| With phenyl isocyanate; triethylamine In benzene for 48h; | 99% |
-
-
592-76-7
1-Heptene

| Conditions | Yield |
|---|---|
| With platinum In benzene at 20℃; | 99% |

| Conditions | Yield |
|---|---|
| zirconocene dichloride In dichloromethane 20°C; | 99% |
| zirconium (IV) butoxide In neat (no solvent) 20°C; | 51% |
| zirconium (IV) butoxide In dichloromethane 20°C; | 51% |
-
-
592-76-7
1-Heptene

-
-
42474-21-5
1,2-dibromoheptane

| Conditions | Yield |
|---|---|
| With bromine In tetrachloromethane at -10 - 20℃; for 16h; | 98% |
| With [bis(acetoxy)iodo]benzene; potassium bromide In dichloromethane; water at 20℃; for 0.25h; | 94% |
| With bromine In chloroform at 0 - 5℃; | 88% |
-
-
592-76-7
1-Heptene

-
-
1873-88-7
1,1,1,3,5,5,5-heptamethyltrisiloxan

-
-
14579-46-5
3-heptyl-1,1,1,3,5,5,5-heptamethyltrisiloxane

| Conditions | Yield |
|---|---|
| With 1,2,3-trimethylimidazolium methylsulphate; bis(cyclooctadiene (μ-silyloxytrimethyl) rhodium(I)) at 90℃; for 2h; Product distribution; Further Variations:; Catalysts; Reagents; | 98% |
| With C77H70N2OPtSi2 In toluene at 80℃; for 24h; Catalytic behavior; Reagent/catalyst; Inert atmosphere; Schlenk technique; regioselective reaction; | 95% |
| dihydrogen hexachloroplatinate In isopropyl alcohol Heating; | |
| (1,5-cyclooctadiene)rhodium/Aerosil 200 at 100℃; for 1h; | 98 % Chromat. |

| Conditions | Yield |
|---|---|
| With [(C18H37)2(CH3)2N]3[SiO4H(WO5)3]; dihydrogen peroxide In ethyl acetate at 59.84℃; for 3h; | 98% |
| With 3-chloro-benzenecarboperoxoic acid In dichloromethane at 0 - 20℃; for 12h; Inert atmosphere; | 98% |
| With 3-chloro-benzenecarboperoxoic acid In dichloromethane for 3h; | 95% |
-
-
592-76-7
1-Heptene

-
-
80605-28-3
trans-1,2-dibutylcyclopentane

| Conditions | Yield |
|---|---|
| With (2,6-((2,4,6-tricyclopentyl-C6H2)N-CMe)2C5H3N)Fe(N2) In neat (no solvent) at 23℃; for 24h; Sealed tube; Inert atmosphere; diastereoselective reaction; | 98% |
-
-
592-76-7
1-Heptene

-
-
19736-58-4
2,3,5,6-tetrafluoro-4-mercaptopyridine

| Conditions | Yield |
|---|---|
| With benzo[de]benzo[4,5]imidazo[2,1-a]isoquinolin-7-one In cyclohexane for 18h; Inert atmosphere; Schlenk technique; Irradiation; | 98% |
-
-
592-76-7
1-Heptene

-
-
4141-91-7
1,1,1,3,3,3-hexafluoroisopropyl iodide

-
-
141810-09-5
1,1,1-Trifluoro-4-iodo-2-(trifluoromethyl)nonane

| Conditions | Yield |
|---|---|
| With 2,2'-azobis(isobutyronitrile) at 65℃; | 97% |
-
-
592-76-7
1-Heptene

-
-
16486-97-8
1-Chloro-1,1,2,2,3,3,4,4,5,5,6,6-dodecafluoro-6-iodo-hexane

-
-
103190-37-0
1-Chloro-1,1,2,2,3,3,4,4,5,5,6,6-dodecafluoro-8-iodo-tridecane

| Conditions | Yield |
|---|---|
| tetrakis(triphenylphosphine) palladium(0) at 20℃; for 0.333333h; | 97% |
| nickel In ethanol at 80℃; for 8h; | 96% |
| With magnesium In N,N-dimethyl-formamide at 80℃; for 10h; | 89.5% |
| With ammonium peroxydisulfate; sodium formate In N,N-dimethyl-formamide at 40℃; for 4h; | 86% |
| With iron In N,N-dimethyl-formamide at 80℃; for 2h; | 79% |
-
-
592-76-7
1-Heptene

-
-
3619-91-8
1,3-bisphosphinopropane

-
-
288145-95-9
1,3-bis[di(n-heptyl)phosphino]propane

| Conditions | Yield |
|---|---|
| at 20℃; for 10h; Addition; Irradiation; | 97% |

| Conditions | Yield |
|---|---|
| With tributyl-amine; palladium diacetate In N,N-dimethyl-formamide at 100℃; for 0.333333h; Heck reaction; Microwave irradiation; | 97% |
| With tributyl-amine; palladium diacetate In 1,4-dioxane; N,N-dimethyl-formamide at 100℃; for 0.333333h; Microwave irradiation; | 97% |
-
-
592-76-7
1-Heptene

-
-
124-38-9
carbon dioxide

-
-
2386-64-3
ethylmagnesium chloride

-
-
45019-28-1
4-Methylnonanoic acid

| Conditions | Yield |
|---|---|
| Stage #1: 1-Heptene; ethylmagnesium chloride With triisopropyl phosphite; tantalum pentachloride In tetrahydrofuran at 20℃; for 3h; Stage #2: carbon dioxide In tetrahydrofuran at 0℃; for 3h; Stage #3: With hydrogenchloride In tetrahydrofuran; water | 97% |


| Conditions | Yield |
|---|---|
| With C36H62Cl4Rh2; cesium acetate In methanol at 20℃; for 24h; Darkness; regioselective reaction; | 97% |
Related products
Raw Materials
Downstream Products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View