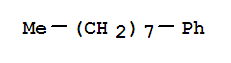Dayang Chem (Hangzhou) Co.,Ltd.
Dayangchem's R&D center can offer custom synthesis according to the contract research and development services for the fine chemicals, pharmaceutical, biotechnique and some of the other chemicals. DayangChem can provide different quantities
Cas:2189-60-8
Min.Order:1 Kilogram
FOB Price: $3.0
Type:Lab/Research institutions
inquiryHangzhou Think Chemical Co. Ltd
1-Phenyloctane[CAS:2189-60-8] for Fingolimod HANGZHOU THINK CHEMICAL CO., LTD. (THINKCHEM) is an integrative corporation of trade, research and contract manufacture. With about ten years of business experiences on the marketing &
Enke Pharma-tech Co.,Ltd. (Cangzhou, China )
Cangzhou Enke Pharma Tech Co.,ltd. is located in Cangzhou City, Hebei province ,where is a famous petroleum chemical industry city in China. Enke Pharma a high-tech enterprise ,and we are dedicated to developing and manufacturing new ap
Chemwill Asia Co., Ltd.
Cas:2189-60-8
Min.Order:1 Metric Ton
FOB Price: $1.0
Type:Manufacturers
inquiryHebei Nengqian Chemical Import and Export Co., LTD
Our Advantage Rich Experience Our products are sold all over Europe,North&South America, Sino-East, Asia and pacific area as well as Africa,we establish long term. Quality service Company cooperates with research institutes. We strictly con
Cas:2189-60-8
Min.Order:1 Kilogram
FOB Price: $1.0 / 10.0
Type:Trading Company
inquiryHenan Tianfu Chemical Co., Ltd.
Our company was built in 2009 with an ISO certificate.In the past 5 years, we have grown up as a famous fine chemicals supplier in China and we had established stable business relationships with Samsung,LG,Merck,Thermo Fisher Scientific and so on.O
Cas:2189-60-8
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryZhuozhou Wenxi import and Export Co., Ltd
WITH US,YOUR MONEY IN SAFE,YOUR BUSINESS IN SAFE 1)Quick Response Within 12 hours; 2)Quality Guarantee: All products are strictly tested by our QC, confirmed by QA and approved by third party lab in China, USA, Canada, Germany, UK, Italy, France et
Cas:2189-60-8
Min.Order:10 Gram
FOB Price: $146.0 / 176.0
Type:Trading Company
inquiryHenan Sinotech Import&Export Corporation
Product Name: CAS No.: 2189-60-8 Molecular formula: C14H22 Molecular weight: 190 Appearance: Colorless transparent liquid Appearance:Colorless transparent liquid Storage: Preserve i
Shanghai Upbio Tech Co.,Ltd
1.In No Less 10 years exporting experience. you can 100% received goods 2.Lower Price with higher quality 3,Free sample 4,We are sincerely responsible for the "product quality" and "After Service" Upbio is Specializ
Qingdao Beluga Import and Export Co., LTD
N-OCTYLBENZENE CAS:2189-60-8 Qingdao Belugas Import and Export Co., Ltd. is a scientific and technological company integrating research and development, production and trade of chemical intermediates, specializing in high quality organic intermediat
Cas:2189-60-8
Min.Order:1 Gram
Negotiable
Type:Lab/Research institutions
inquiryShandong Hanjiang Chemical Co., Ltd.
Hello, dear friend! I'm Hansen and Allen from China. Welcome to my lookchem mall! The following is a brief introduction of our company's products and services. If you are interested in our products, please contact us by emai
Henan Wentao Chemical Product Co., Ltd.
Henan Wentao Chemical Product Co.,Ltd is Located in Zhengzhou High-tech Development Zone with import and export license, We passed ISO 9001:2008 as well, Henan Wentao has developed more than 1000 compounds, which are widely used in the fields of prod
Hebei Quanhe Biotechnology Co. LTD
1. Timely and efficient service to ensure communication with customers 2. Produce products of different specifications and sizes according to your requirements. 3. Quality procedures and standards recognized by SGS. Advanced plant equipment ensures s
Cas:2189-60-8
Min.Order:1 Kilogram
FOB Price: $40.0 / 50.0
Type:Lab/Research institutions
inquiryJilin Artel New Materials Co., LTD
Price, service, company and transport advantage: 1.place of origin china with super quality by reasonable price. 2. it's customers' right to choose the package (ems, dhl, fedex, ups); 3. it's custo
Triumph International Development Limilted
Appearance:white or light yellow crystalline powder Storage:Store in a cool,dry place and keep away from direct strong light Package:As customer request Application:Used for research and industrial manufacture. Transportation:By
Ality Chemical Corporation
The above product is Ality Chemical's strong item with best price, good quality and fast supply. Ality Chemical has been focusing on the research and production of this field for over 14 years. At the same time, we are always committed to providi
Hangzhou J&H Chemical Co., Ltd.
J&H CHEM R&D center can offer custom synthesis according to the contract research and development services for the fine chemicals, pharmaceutical, biotechnique and some of the other chemicals. J&H CHEM has some Manufacturing base in Jia
Zibo Hangyu Biotechnology Development Co., Ltd
Zibo Hangyu Biotechnology Development Co., Ltd is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality pharmaceuticals, intermediates, special chemicals and OLED intermediates and other fine chemi
Cas:2189-60-8
Min.Order:10 Gram
FOB Price: $100.0
Type:Lab/Research institutions
inquiryHANGZHOU YUNUO CHEMICAL CO.,LTD
Superior quality, moderate price & quick delivery. Appearance:colourless liquid Storage:Stored in cool, dry and ventilation place; Away from fire and heat Package:25kg/drum, or as per your request. Application:Used as Pharmaceutical Intermediat
Zibo Dorne chemical technology co. LTD
Product Details Grade: pharmaceutical grade Purity:99%+ ProductionCapacity: 1000 Kilogram/Month Scope of use: For scientific research only(The product must be used legally) Our Advantage 1. Best quality with competitive price. 2. Quick shipping,
Siwei Development Group Ltd.
Product name: 1-Phenyloctane CAS No.:2189-60-8 Molecule Formula:C14H22 Molecule Weight:190.32 Purity: 98.0% Package: 25kg/drum Description:Colorless transparent liquid Manufacture Standards:Enterprise Standard TESTING ITEMS
Cas:2189-60-8
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryXiamen Jenny Chemical Technology Co., Ltd.
GMP standard, high purity, competitive price, in stock 1. Quick Response: within 6 hours after receiving your email. 2. Quality Guarantee: All products are strictly tested by our QC, confirmed by QA, and approved by a third-party lab in China, USA,
Shanghai Minstar Chemical Co., Ltd
Product Name: N-OCTYLBENZENE MF: C14H22 MW: 190.32 EINECS: 218-582-1 Mol File: 2189-60-8.mol N-OCTYLBENZENE Structure N-OCTYLBENZENE Chemical Properties Melting point -36 °C (lit.) Boiling point 261-263 °C (lit.) dens
Hangzhou Huarong Pharm Co., Ltd.
Hangzhou Huarong Pharm Co., Ltd.established since 2006 , has been actively developing specialty products for Finished Dosages, APIs, Intermediates, and Fine chemicals markets in North America, Europe, Korea, Japan, Mid-East and all over the World. Hu
KAISA GROUP INC
1.Applied in food field.it can improve the immune system and prolong life. 2.Appliedin cosmetic field.it can improve the skin care. 3.Applied in pharmaceutical field.it can treat various dieases. 4.Our product quality assurance will make our customer
Cas:2189-60-8
Min.Order:1 Metric Ton
FOB Price: $7.0 / 8.0
Type:Trading Company
inquiryWuhu Nuowei Chemistry Technologies Co., Ltd.
Wuhu Nuovo Chemical Technology Co., Ltd. was established in August 2014, mainly engaged in the development, production and sales of ionic liquids, ribose, nucleosides, nucleotides and related chemicals; Products are mainly used in new energy, new ma
Beijing Greenchem Technology Co.,Ltd. ( Panjin Greenchem Technology Co.Ltd .)
Capability on chemical synthesis 1. Beijing High-Tech Enterprises 2. Strong R&D Team 3. 8 years of experiences in R & D of high-tech Catalyst; 4. 5000 production techniques, 69 items of national patents, and 360 kinds of products on sal
Kono Chem Co.,Ltd
Product Name: N-OCTYLBENZENE Synonyms: 1-PHENYLOCTANE;OCTYLBENZENE;N-OCTYLBENZENE;PHENYLOCTANE;Benzene, octyl-;Octane, 1-phenyl-;octane,1-phenyl-;oct…Appearance:Solid powder Storage:Sealed,light and oxygen resistant Package:aluminum foil bag,carton
Win-Win chemical Co.Ltd
Stock products, own laboratoryAppearance:Colorless liquid Package:Grams, Kilograms Application:For R&D Transportation:According to customer request Port:Shanghai
Cas:2189-60-8
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryGIHI CHEMICALS CO.,LIMITED
Lower price, sample is available,SDS test documents are available,large stock in warehouseAppearance:White powder Storage:Sealed and preserved Package:200/Kilograms Application:Fine chemical intermediates, used as the main raw material for the synthe
Synthetic route
-
-
28665-60-3
(E)-1-phenyl-1-octene

-
-
2189-60-8
Octylbenzene

| Conditions | Yield |
|---|---|
| Stage #1: (E)-1-phenyl-1-octene With Dimethylphenylsilane; copper(l) chloride In various solvent(s) at 20℃; for 24h; Reduction; Stage #2: With water; toluene-4-sulfonic acid In various solvent(s) at 20℃; Hydrolysis; | 100% |

| Conditions | Yield |
|---|---|
| With sodium hydroxide; (1,1'-bis(diphenylphosphino)ferrocene)palladium(II) dichloride In tetrahydrofuran at 65℃; for 16h; Product distribution; other: B-alkylboron compounds, catalysts, bases, solvents, temperatures; | 99% |
| With sodium hydroxide; cis-dichloro-1,1'-bis(diphenylphosphino)ferrocene palladium(II) In tetrahydrofuran; water Reaction mixt. is stirred for 16 h at temp. 65°C, cooled to room temp., oxidized with soln. of NaOH in THF-H2O.; Extracted with hexane, washed with water, dried (MgSO4), identified by GLC.; | 99% |
| With potassium carbonate; cis-dichloro-1,1'-bis(diphenylphosphino)ferrocene palladium(II) In N,N-dimethyl-formamide Reaction mixt. is stirred for 16 h at temp. 50°C, cooled to room temp., oxidized with soln. of K2CO3 in DMF.; Extracted with hexane, washed with water, dried (MgSO4), identified by GLC.; | 98% |

| Conditions | Yield |
|---|---|
| With (1R,2R)-bis(dimethylamino)cyclohexane In tetrahydrofuran at 25℃; for 0.25h; | 99% |
| With 1,3-bis-(diphenylphosphino)propane; cobalt(II) chloride In tetrahydrofuran at -15℃; for 0.5h; | 33% |
| With N,N,N,N,-tetramethylethylenediamine; iron(III) chloride In tetrahydrofuran at 0℃; for 0.5h; | 97 % Spectr. |

| Conditions | Yield |
|---|---|
| With 1,3-bis(mesityl)imidazolium chloride; palladium diacetate In tetrahydrofuran; 1-methyl-pyrrolidin-2-one at 20℃; for 1h; Kumada reaction; | 97% |
| With (1R,2R)-bis(dimethylamino)cyclohexane In tetrahydrofuran at 25℃; for 0.25h; | 10% |
| With N,N,N,N,-tetramethylethylenediamine; iron(III) chloride In tetrahydrofuran at 40℃; for 0.5h; | 45 % Spectr. |

| Conditions | Yield |
|---|---|
| With [polymer-incarcerated nickel nanocatalyst] PICB-NHC-Ni (0.25 mol % as Ni) In tetrahydrofuran at 20℃; for 12h; | 97% |
| With N,N,N,N,-tetramethylethylenediamine; [((Me)NN2)NiCl] In tetrahydrofuran at 20℃; for 2h; Kumada-Corriu-Tamao coupling reaction; Inert atmosphere; | 92% |
| With [Fe(bis(oxazolinylphenyl)amido-Ph)Cl2] In tetrahydrofuran at -40℃; for 1h; Inert atmosphere; | 92% |
-
-
16967-02-5
1-phenyl-1-octyne

-
-
2189-60-8
Octylbenzene

| Conditions | Yield |
|---|---|
| Stage #1: 1-phenyl-1-octyne With tris-(dibenzylideneacetone)dipalladium(0); tricyclohexylphosphine In 1,4-dioxane at 20℃; for 0.25h; Inert atmosphere; Stage #2: With formic acid In 1,4-dioxane at 80℃; Inert atmosphere; chemoselective reaction; | 96% |

| Conditions | Yield |
|---|---|
| With copper(l) iodide; lithium chloride In tetrahydrofuran at 20℃; for 1h; | 96% |

| Conditions | Yield |
|---|---|
| Stage #1: phenyllithium With ZnI2(tmeda) In tetrahydrofuran; dibutyl ether at 0℃; for 0.166667h; Stage #2: 1-octyl p-toluenesulfonate With iron(III) chloride; magnesium bromide In tetrahydrofuran; dibutyl ether at 0 - 25℃; | 95% |
| (i) CuI, (ii) /BRN= 2418747/; Multistep reaction; |

| Conditions | Yield |
|---|---|
| Stage #1: oct-1-ene With 9-borabicyclo[3.3.1]nonane dimer In tetrahydrofuran at 20℃; for 17h; Stage #2: iodobenzene With potassium phosphate; 2H(1+)*Cl4Pd(2-)*2H3N; poly[N-isopropylacrylamide-co-diphenyl(4'-styryl)phosphine] In tetrahydrofuran; 1,4-dioxane at 100℃; for 1.5h; Suzuki-Miyaura reaction; | 95% |

| Conditions | Yield |
|---|---|
| With N,N,N,N,-tetramethylethylenediamine; cobalt(III) acetylacetonate In tetrahydrofuran at 0℃; for 0.666667h; Inert atmosphere; chemoselective reaction; | 95% |
| With triphenyl phosphite; silver(I) bromide In diethyl ether; hexane for 5h; Inert atmosphere; Reflux; | 88% |
| With C20H26Cl2FeN4 In tetrahydrofuran at 45℃; for 0.666667h; Inert atmosphere; | 72 %Spectr. |
| With N,N,N,N,-tetramethylethylenediamine; C34H46Cl4N10Ni2O2 In tetrahydrofuran at 20℃; for 1.33333h; Reagent/catalyst; Temperature; Solvent; Inert atmosphere; Schlenk technique; | 83 %Spectr. |

| Conditions | Yield |
|---|---|
| With TlOH; (1,2-bis(diphenylphosphanyl)ethane)dichloridopalladium(II) In water; benzene at 50℃; for 16h; | 93% |
| With TlOH; (1,2-bis(diphenylphosphanyl)ethane)dichloridopalladium(II) In water; benzene at 50°C, for 16 h; | 93% |
| With TlOH In tetrahydrofuran; water at 50°C, for 16 h; | 75% |
| With Ti2CO3 In tetrahydrofuran at 50°C, for 16 h; | 60% |
| With sodium hydroxide; cis-dichloro-1,1'-bis(diphenylphosphino)ferrocene palladium(II) In tetrahydrofuran Reaction mixt. is stirred for 16 h at 65°C, cooled to room temp., oxidized with aq. soln of NaOH.; Extracted with hexane, washed with water, dried (MgSO4), identified by GLC.; | 1% |

| Conditions | Yield |
|---|---|
| With Ti2CO3; (1,1'-bis(diphenylphosphino)ferrocene)palladium(II) dichloride In tetrahydrofuran at 50℃; for 16h; | 93% |
| With Ti2CO3 In tetrahydrofuran at 50°C, for 16 h; | 93% |
| With TlOH In tetrahydrofuran; water at 50°C, for 16 h; | 41% |
| With potassium hydroxide In tetrahydrofuran; water at 50°C, for 16 h; | <1 |

| Conditions | Yield |
|---|---|
| With aluminium trichloride at 60℃; for 0.166667h; | 93% |

| Conditions | Yield |
|---|---|
| With sodium hydroxide; cis-dichloro-1,1'-bis(diphenylphosphino)ferrocene palladium(II) In tetrahydrofuran A mixt. of iodobenzene, catalyst and octyldisiamylborane is stirred for 16 h at 65°C, cooled to room temp., oxidized with aq. soln of NaOH.; Extracted with hexane, washed with water, dried (MgSO4), identified by GLC.; | 93% |

| Conditions | Yield |
|---|---|
| With potassium fluoride; palladium diacetate; chlorobenzene In tetrahydrofuran; water at 20℃; for 1h; Inert atmosphere; chemoselective reaction; | 92% |
| With 5%-palladium/activated carbon; hydrogen In methanol under 2585.81 - 3102.97 Torr; | 91.5% |
| With triethylsilane; titanium tetrachloride In dichloromethane at 0 - 5℃; for 3 - 5h; Reagent/catalyst; Solvent; Temperature; Large scale; | 90% |
-
-
3742-75-4
3-phenylpropyl 4-methylbenzenesulfonate

-
-
6393-56-2
pentylmagnesium chloride

-
-
2189-60-8
Octylbenzene

| Conditions | Yield |
|---|---|
| With copper(l) iodide; lithium chloride In tetrahydrofuran at 20℃; for 1h; | 92% |

| Conditions | Yield |
|---|---|
| With copper(l) iodide; o-(di(tert-butyl)phosphino)-N,N-dimethylaniline In N,N-dimethyl-formamide at 100℃; for 24h; Inert atmosphere; Glovebox; | 91% |
-
-
591-50-4
iodobenzene

-
-
629-27-6
1-Iodooctane

-
-
201230-82-2
carbon monoxide

-
A
-
2189-60-8
Octylbenzene

-
B
-
6008-36-2
1-phenylnonan-1-one

| Conditions | Yield |
|---|---|
| With copper; zinc; tetrakis(triphenylphosphine) palladium(0) In tetrahydrofuran at 50℃; under 760 Torr; for 23h; | A 3% B 90% |


| Conditions | Yield |
|---|---|
| With palladium diacetate; di-tert-butyl(methyl)phosphonium tetrafluoroborate salt; potassium tert-butylate In tert-Amyl alcohol at 20℃; Product distribution; Further Variations:; Reagents; Solvents; Suzuki cross-coupling; | 90% |
| With potassium carbonate In toluene at 90℃; for 9h; Reagent/catalyst; Suzuki-Miyaura Coupling; Green chemistry; | 88% |
| With potassium phosphate; bis(1,5-cyclooctadiene)nickel (0); bis(N-methylimidazol-2-yl)methane In N,N-dimethyl acetamide at 80℃; for 2h; Product distribution / selectivity; Suzuki Coupling; | 79% |

| Conditions | Yield |
|---|---|
| With copper(l) iodide; lithium chloride In tetrahydrofuran at 20℃; for 1h; | 90% |
-
-
111-83-1
1-bromo-octane

-
-
603-35-0
triphenylphosphine

-
A
-
2189-60-8
Octylbenzene

-
B
-
6737-43-5
n-octyldiphenylphosphine

| Conditions | Yield |
|---|---|
| Stage #1: triphenylphosphine With lithium In tetrahydrofuran at 20℃; for 3h; Inert atmosphere; Stage #2: 1-bromo-octane In tetrahydrofuran at 5 - 70℃; for 6.25h; | A 89.4% B 86.3% |
| Stage #1: triphenylphosphine With lithium In tetrahydrofuran at 20℃; for 3h; Inert atmosphere; Stage #2: 1-bromo-octane In tetrahydrofuran at 5 - 70℃; for 3.25h; Inert atmosphere; |


| Conditions | Yield |
|---|---|
| With potassium phosphate monohydrate; palladium diacetate; tricyclohexylphosphine for 0.0833333h; Suzuki-Miyaura coupling; Inert atmosphere; Microwave irradiation; Neat (no solvent); | 88% |

-
-
2189-60-8
Octylbenzene

| Conditions | Yield |
|---|---|
| With cyclohexa-1,4-diene; 9-ethyl-N3,N3,N6,N6,-tetramethyl-9H-carbazole-3,6-diamine; N-ethyl-N,N-diisopropylamine In N,N-dimethyl acetamide at 23℃; for 48h; Reagent/catalyst; Solvent; Inert atmosphere; UV-irradiation; Schlenk technique; | 86% |

| Conditions | Yield |
|---|---|
| With palladium diacetate; bis(1-adamantyl)phosphine oxide In tetrahydrofuran; 1-methyl-pyrrolidin-2-one at 25℃; for 20h; Kumada-Corriu coupling; Inert atmosphere; | 85% |
| dilithium tetrachlorocuprate In tetrahydrofuran at 20℃; Alkylation; | 70% |
| With [CoI(1,3-bis(2,6-diisopropylphenyl)imidazol-2-ylidene)]2(μ-I)2 In tetrahydrofuran at 20℃; for 18h; Kumada-Tamao-Corriu cross-coupling; Inert atmosphere; | 16% |
| With N,N,N,N,-tetramethylethylenediamine; [((Me)NN2)NiCl] In tetrahydrofuran at 20℃; Kumada-Corriu-Tamao coupling reaction; Inert atmosphere; | 90 %Chromat. |
| With Fe2Cl4(C3H2N2(C6H3(CH(CH3)2)2)2)2 In tetrahydrofuran at -30 - 23℃; for 1h; Kumada Cross-Coupling; Inert atmosphere; Glovebox; | 71 %Spectr. |

| Conditions | Yield |
|---|---|
| With potassium phosphate; bis(1,5-cyclooctadiene)nickel (0); bis(N-methylimidazol-2-yl)methane In N,N-dimethyl acetamide at 80℃; for 9h; Suzuki Coupling; | 85% |

| Conditions | Yield |
|---|---|
| With 1,8-diazabicyclo[2.2.2.]octane; (1,2,3,4,5-pentamethyl-cyclopenta-2,4-dienylmethyl)PPh2*BH3; nickel dichloride In tetrahydrofuran; diethyl ether; toluene at 25℃; for 3h; | 83% |
| With (1R,2R)-bis(dimethylamino)cyclohexane In tetrahydrofuran at 25℃; for 0.25h; | 80% |
| dilithium tetrachlorocuprate In tetrahydrofuran at 20℃; for 16h; | 76% |

| Conditions | Yield |
|---|---|
| With {1,2-bis(diphenylphosphino)ethane}dichloroiron; magnesium bromide ethyl etherate In tetrahydrofuran at 40℃; for 3h; | 83% |

| Conditions | Yield |
|---|---|
| With sodium hydroxide; cis-dichloro-1,1'-bis(diphenylphosphino)ferrocene palladium(II) In tetrahydrofuran A mixt. of iodobenzene, catalyst and octyldisiamylborane is stirred for 16 h at 65°C, cooled to room temp., oxidized with aq. soln of NaOH.; Extracted with hexane, washed with water, dried (MgSO4), identified by GLC.; | 82% |

| Conditions | Yield |
|---|---|
| With nickel(II) bromide dimethoxyethane; pyridine-2,6-bis(N-cyanocarboxamidine); lithium chloride; zinc In 1-methyl-pyrrolidin-2-one at 80℃; for 24h; Catalytic behavior; Reagent/catalyst; Inert atmosphere; | 82% |
-
-
111-83-1
1-bromo-octane

-
-
24388-23-6
2-phenyl-4,4,5,5-tetramethyl-1,3,2-dioxoborole

-
-
2189-60-8
Octylbenzene

| Conditions | Yield |
|---|---|
| With C25H33Cl2FeLiN2*0.75C4H8O; lithium ethylmethyl amide In benzene at 20℃; for 24h; Suzuki-Miyaura Coupling; Glovebox; Inert atmosphere; | 80% |
| With 2,2-bis((S)-4-phenyl-4,5-dihydrooxazol-2-yl)acetonitrile; (2,2-bis((S)-4-phenyl-4,5-dihydrooxazol-2-yl)acetonitrile)FeCl; lithium ethylmethyl amide In benzene at 50℃; for 48.17h; Suzuki-Miyaura Coupling; Inert atmosphere; Sealed tube; | 77% |
| With nickel(II) bromide dimethoxyethane; potassium tert-butylate; trans-N,N'-dimethylcyclohexane-1,2-diamine; iso-butanol; sodium iodide at 60℃; Inert atmosphere; | 69% |

| Conditions | Yield |
|---|---|
| With aluminium trichloride In dichloromethane | 100% |
-
-
2189-60-8
Octylbenzene

-
-
54997-91-0
4-octylbenzene-1-sulfonyl chloride

| Conditions | Yield |
|---|---|
| With chlorosulfonic acid In chloroform at 0 - 20℃; for 0.25h; | 100% |
| With chlorosulfonic acid In chloroform at 20℃; for 20h; | 80% |
| With chlorosulfonic acid In dichloromethane at 20℃; for 6h; | 74% |
| With chlorosulfonic acid In chloroform at 20℃; for 20h; |

| Conditions | Yield |
|---|---|
| With hydrogen In neat (no solvent) at 50℃; under 760.051 Torr; for 64h; Temperature; | 99% |
| With nickel-aluminium oxide at 160 - 170℃; under 36775.4 - 51485.6 Torr; Hydrogenation; | |
| With nickel kieselguhr at 200 - 255℃; under 110326 Torr; Hydrogenation; | |
| (hydrogenation); |
-
-
2189-60-8
Octylbenzene

-
-
598-21-0
2-Bromoacetyl bromide

-
-
64068-76-4
α-bromomethyl p(n-octyl)phenyl cetone

| Conditions | Yield |
|---|---|
| With aluminum (III) chloride In dichloromethane at -10℃; Friedel-Crafts Acylation; | 98% |
| With aluminum (III) chloride In dichloromethane at -10 - 20℃; Friedel-Crafts Acylation; | 62% |
| With aluminum (III) chloride In 1,2-dichloro-ethane at 0 - 20℃; Friedel-Crafts acylation; | 60% |

| Conditions | Yield |
|---|---|
| aluminium chloride In dichloromethane | 96% |
| aluminium chloride In dichloromethane | 96% |
| With aluminum (III) chloride In dichloromethane at -5 - 30℃; for 2h; | 92.2% |

-
-
2189-60-8
Octylbenzene

| Conditions | Yield |
|---|---|
| In acetonitrile at 90℃; for 19h; Inert atmosphere; | 92% |

| Conditions | Yield |
|---|---|
| With hydrogenchloride; bromine; sodium methylate; aluminium trichloride In carbon disulfide; ethanol; 1,1,2-Trichloro-1,2,2-trifluoroethane; water | 91% |


| Conditions | Yield |
|---|---|
| With N-hydroxyphthalimide; 6-((cobalt(II) 4,9,16,23-tetraaminephthalocyanin-4-yl))cellulose; oxygen; potassium hydroxide In o-xylene for 14h; Reflux; Green chemistry; | 91% |
| With Oxone; potassium bromide In nitromethane at 20℃; for 24h; | 87% |
| With tert.-butylhydroperoxide; 4C12H25O4S(1-)*Fe2O(4+)*10H2O In water at 30℃; for 50h; | 72% |

| Conditions | Yield |
|---|---|
| With aluminium trichloride In carbon disulfide for 12h; Heating; | 89% |
| With carbon disulfide; aluminium trichloride at 50℃; | |
| With aluminium trichloride | |
| With aluminium trichloride In carbon disulfide |
Related products
Raw Materials
Downstream Products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View