-
Name
Tetrazole
- EINECS 206-023-4
- CAS No. 288-94-8
- Article Data144
- CAS DataBase
- Density 1.477 g/cm3
- Solubility soluble in water
- Melting Point 156-158 °C
- Formula CH2N4
- Boiling Point 220.233 °C at 760 mmHg
- Molecular Weight 70.0537
- Flash Point 114.573 °C
- Transport Information UN 1993 3/PG 2
- Appearance white crystals or crystalline powder
- Safety 16-26-36
- Risk Codes 11-20/21/22-36-5-4
-
Molecular Structure
-
Hazard Symbols
 F,
F, Xn
Xn
- Synonyms 1H-Tetrazole(8CI,9CI);1H-TZ;NSC 36712;Tetraazacyclopentadiene;1H-Tetrazole;
- PSA 54.46000
- LogP -0.80030
Synthetic route

| Conditions | Yield |
|---|---|
| With sodium azide; ammonium chloride; acetic acid at 90℃; for 10h; | 91.55% |
| Stage #1: orthoformic acid triethyl ester With hydrogenchloride; sodium azide; ammonia; pyridine hydrochloride; acetic acid at 90℃; for 12h; Stage #2: With water; sodium hydroxide at 20℃; | 71% |
| With sodium azide; ammonium chloride; acetic acid at 95℃; for 18h; | 60.5% |

| Conditions | Yield |
|---|---|
| With sodium azide; triethylamine hydrochloride In N,N-dimethyl-formamide at 130℃; for 4h; Microwave irradiation; Inert atmosphere; | 85% |

| Conditions | Yield |
|---|---|
| With hypophosphorous acid; sodium nitrite |
-
-
14213-13-9
4-tetrazol-1-yl-phenylamine

-
-
288-94-8
1H-tetrazole

| Conditions | Yield |
|---|---|
| With sulfuric acid; permanganate(VII) ion |

| Conditions | Yield |
|---|---|
| With hydrogen azide; ethanol at 100℃; | |
| With hydrogen azide; water at 70 - 80℃; | |
| With tris-(2-chloro-ethyl)-amine; benzene at 110℃; |

| Conditions | Yield |
|---|---|
| With hydrogenchloride; sodium azide at 110℃; |

-
-
288-94-8
1H-tetrazole

| Conditions | Yield |
|---|---|
| With water at 85℃; Rate constant; pH 2.07 - 12.47; |

-
-
288-94-8
1H-tetrazole

| Conditions | Yield |
|---|---|
| With permanganate(VII) ion; acetic acid Man kocht die nach Entfernung des Braunsteins eingeengte Loesung mit verd. Salpetersaeure; |

-
-
288-94-8
1H-tetrazole

| Conditions | Yield |
|---|---|
| With sodium hydroxide; permanganate(VII) ion |

| Conditions | Yield |
|---|---|
| beim Erwaermen; |

-
-
288-94-8
1H-tetrazole

| Conditions | Yield |
|---|---|
| With hydrogenchloride; tin(ll) chloride | |
| With copper(I) sulfate; water; hypophosphorous acid; copper(II) sulfate at 60℃; | |
| With copper(I) sulfate; alkali arsenite; water; copper(II) sulfate at 60℃; |
-
-
288-94-8
1H-tetrazole

-
-
4105-38-8
Tri-O-acetyluridine

-
-
76991-96-3
4-(tetrazol-1-yl)-1-(2',3',5'-tri-O-acetyl-β-D-ribofuranosyl)-pyrimedin-2(1H)-one

| Conditions | Yield |
|---|---|
| With diphenyl hydrogen phosphate; p-toluenesulfonyl chloride In pyridine at 20℃; for 36h; Substitution; | 100% |
| With pyridine; diphenyl hydrogen phosphate; p-toluenesulfonyl chloride at 20℃; for 36h; | 95% |
| With diphenyl hydrogen phosphate; p-toluenesulfonyl chloride In pyridine for 36h; Ambient temperature; | 88% |
| With diphenyl hydrogen phosphate; p-toluenesulfonyl chloride In pyridine for 36h; Ambient temperature; | 88% |
-
-
288-94-8
1H-tetrazole

-
-
273214-60-1
[3-15N]-4-(tetrazol-1-yl)-1-(2',3',5'-tri-O-acetyl-β-D-ribofuranosyl)pyrimidin-2(1H)-one

| Conditions | Yield |
|---|---|
| With diphenyl hydrogen phosphate; p-toluenesulfonyl chloride In pyridine at 20℃; for 36h; Substitution; | 99% |
-
-
288-94-8
1H-tetrazole

-
-
64890-08-0
tetrazolium perchlorate

| Conditions | Yield |
|---|---|
| With perchloric acid In water at 80℃; for 0.166667h; | 98% |
-
-
288-94-8
1H-tetrazole

-
-
13030-62-1
3',5'-di-O-acetyl-2'-deoxyuridine

-
-
175601-12-4
Acetic acid (2R,3S,5R)-2-acetoxymethyl-5-(2-oxo-4-tetrazol-1-yl-2H-pyrimidin-1-yl)-tetrahydro-furan-3-yl ester

| Conditions | Yield |
|---|---|
| With diphenyl hydrogen phosphate; p-toluenesulfonyl chloride In pyridine for 12h; Ambient temperature; | 97% |

| Conditions | Yield |
|---|---|
| With sodium hydroxide | 97% |
-
-
288-94-8
1H-tetrazole

-
-
144-55-8
sodium hydrogencarbonate

-
-
40615-39-2
5'-O-(4-4'-dimethoxytrityl)thymidine

-
-
327596-87-2
N,N,N',N'-tetraisopropyl-O-[2-[N-isopropyl-N-(4-methoxybenzoyl)amino]ethyl] phosphordiamidite

| Conditions | Yield |
|---|---|
| In hexane; dichloromethane; ethyl acetate; toluene; acetonitrile | 97% |

-
-
288-94-8
1H-tetrazole

-
-
17455-13-9
18-crown-6 ether

-
-
1233953-72-4
K(μ2-(B(hydride)2(tetrazolate)2)-κ2N,H)(18-crown-6-κ6)

| Conditions | Yield |
|---|---|
| With H2 In neat (no solvent) mixing KBH4, N compd. (2, 3 or 4 equiv.), heating at 100-130°C until gas evolution cessation, treatment with soln. of 1 equiv. of 18-crown-6 in C4H8O; isolation of solid; | 97% |


| Conditions | Yield |
|---|---|
| Stage #1: 1H-tetrazole With potassium borohydride at 100 - 130℃; Stage #2: 18-crown-6 ether In tetrahydrofuran | 97% |

| Conditions | Yield |
|---|---|
| With triethylamine In N,N-dimethyl-formamide at 60℃; for 0.166667h; Microwave irradiation; | 97% |
| With triethylamine In N,N-dimethyl-formamide Microwave irradiation; |

| Conditions | Yield |
|---|---|
| With phosphoric acid In dichloromethane at 30 - 35℃; for 7h; | 96.6% |

| Conditions | Yield |
|---|---|
| In ethanol for 0.05h; Solvent; Sonication; | 96% |


| Conditions | Yield |
|---|---|
| With aq. NaOH In water mixed, aq. 1.0M NaOH added, stirred (1 h); filtered, washed (H2O, C2H5OH, acetone), dried (in vac., 2 h); elem. anal.; | 95.9% |

| Conditions | Yield |
|---|---|
| In acetonitrile for 0.333333h; Ambient temperature; | 95% |
| In acetonitrile | |
| In dichloromethane; acetonitrile at 20℃; for 0.5h; Inert atmosphere; | |
| In acetonitrile |

| Conditions | Yield |
|---|---|
| With 4-chlorophenylphosphorodichloridate In pyridine for 5h; Ambient temperature; | 95% |

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetone at 20 - 25℃; for 3h; | 95% |
-
-
288-94-8
1H-tetrazole

-
-
61671-79-2
3',5'-di-O-acetyl-2'-O-methyluridine

-
-
1085342-81-9
4-(tetrazol-1-yl)-1-(3',5'-di-O-acetyl-2'-O-methyl-β-D-ribofuranosyl)pyrimidin-2-(1H)-one

| Conditions | Yield |
|---|---|
| With pyridine; diphenyl hydrogen phosphate; p-toluenesulfonyl chloride at 20℃; for 36h; | 95% |

| Conditions | Yield |
|---|---|
| With sulfuric acid for 0.833333h; | 94% |
-
-
288-94-8
1H-tetrazole

-
-
85510-77-6
1-chloro-3-(5-phenyl-1H-tetrazol-1-yl)propan-2-ol

| Conditions | Yield |
|---|---|
| With sodium hydroxide In ethanol at 50℃; for 3h; | 93% |
-
-
288-94-8
1H-tetrazole

-
-
13440-29-4
1H-tetrazole sodium salt

| Conditions | Yield |
|---|---|
| With sodium hydroxide In water | 93% |
-
-
288-94-8
1H-tetrazole

-
-
96107-81-2
sodium 5-chlorotetrazolate

| Conditions | Yield |
|---|---|
| With sodium hypochlorite; acetic acid In water at 55℃; for 6h; Temperature; Time; | 92.76% |
-
-
288-94-8
1H-tetrazole

-
-
404839-22-1
[3',4'-13C2]3',5'-di-O-acetyl-2'-deoxyuridine

-
-
404839-23-2
4-(tetrazol-1-yl)-1-([3',4'-13C2]3',5'-di-O-acetyl-2'-deoxy-β-D-ribofuranosyl)pyrimidin-2-one

| Conditions | Yield |
|---|---|
| With pyridine; diphenyl hydrogen phosphate; p-toluenesulfonyl chloride at 20℃; for 1.5h; | 92% |
-
-
288-94-8
1H-tetrazole

-
-
619-73-8
4-nitrobenzyl chloride

-
-
144035-43-8
1-(tetrazol-2-ylmethyl)-4-nitrobenzene

| Conditions | Yield |
|---|---|
| With trifluorormethanesulfonic acid at 60℃; for 5h; | 92% |
-
-
288-94-8
1H-tetrazole

-
-
18369-83-0
methyl thiochloroformate

-
-
82995-84-4
5-<(methylthio)carbonyl>tetrazole

| Conditions | Yield |
|---|---|
| With triethylamine In tetrahydrofuran Ambient temperature; | 91% |
-
-
288-94-8
1H-tetrazole

-
-
13464-19-2
phenyl chlorothioformate

-
-
82995-86-6
Tetrazole-1-carbothioic acid S-phenyl ester

| Conditions | Yield |
|---|---|
| With triethylamine In tetrahydrofuran Ambient temperature; | 90% |
-
-
288-94-8
1H-tetrazole

-
-
114006-32-5
C13H16BrN3O2

-
-
114006-67-6
N-[1-Cyclohexyl-1-tetrazol-1-yl-meth-(Z)-ylidene]-N'-(4-nitro-phenyl)-hydrazine

| Conditions | Yield |
|---|---|
| With triethylamine In ethanol for 1h; Ambient temperature; | 90% |
-
-
288-94-8
1H-tetrazole

-
-
114006-31-4
C17H18BrN3O2

-
-
114006-66-5
N-[2,2-Dimethyl-3-phenyl-1-tetrazol-1-yl-prop-(Z)-ylidene]-N'-(4-nitro-phenyl)-hydrazine

| Conditions | Yield |
|---|---|
| With triethylamine In ethanol for 1h; Ambient temperature; | 90% |

| Conditions | Yield |
|---|---|
| In ethanol; water stirring (room temp., 2 h); filtration, washing (EtOH), drying in air; elem. anal.; | 90% |
| In water crystn. from weak HNO3-soln.; | |
| In water crystn. from weak HNO3-soln.; |
Tetrazole Specification
The Tetrazole, with the CAS registry number 288-94-8, is also known as Tetraazacyclopentadiene. It belongs to the product categories of Tetrazoles; Other Reagents; Chemistry; Peptide Synthesis; Solvents and Mixtures for Peptide Synthesis; Specialty Synthesis; Biotech Solvents Solvents; Solvent Bottles; Solvents. Its EINECS number is 206-023-4. This chemical's molecular formula is CH2N4 and molecular weight is 70.05. What's more, its systematic name is 2H-Tetrazole. This chemical is used as a peptide coupling agent. Tetrazole was first prepared by the reaction of anhydrous hydrazoic acid and hydrogen cyanide under pressure.
Physical properties of Tetrazole are: (1)ACD/LogP: -0.6; (2)# of Rule of 5 Violations: 0; (3)ACD/LogD (pH 5.5): -1.41; (4)ACD/LogD (pH 7.4): -2.52; (5)ACD/BCF (pH 5.5): 1.00; (6)ACD/BCF (pH 7.4): 1.00; (7)ACD/KOC (pH 5.5): 1.73; (8)ACD/KOC (pH 7.4): 1.00; (9)#H bond acceptors: 4; (10)#H bond donors: 1; (11)#Freely Rotating Bonds: 0; (12)Polar Surface Area: 54.46 Å2; (13)Index of Refraction: 1.543; (14)Molar Refractivity: 14.96 cm3; (15)Molar Volume: 47.427 cm3; (16)Polarizability: 5.931×10-24cm3; (17)Surface Tension: 98.3 dyne/cm; (18)Density: 1.477 g/cm3; (19)Flash Point: 114.573 °C; (20)Enthalpy of Vaporization: 45.665 kJ/mol; (21)Boiling Point: 220.233 °C at 760 mmHg; (22)Vapour Pressure: 0.1 mmHg at 25°C.
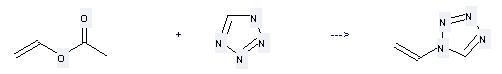
Uses of Tetrazole: it can be used to produce 1-vinyl-1H-tetrazole at the temperature of 50 °C. It will need reagents mercurous acetate, H2SO4 with the reaction time of 20 hours. The yield is about 60%.
When you are using this chemical, please be cautious about it as the following:
This chemical is highly flammable, so you should keep it away from sources of ignition - No smoking. It is harmful by inhalation, in contact with skin and if swallowed. This substance is irritating to eyes. When heating, it may cause an explosion. It forms very sensitive explosive metallic compounds. In case of contact with eyes, you should rinse immediately with plenty of water and seek medical advice. When using it, you need wear suitable protective clothing.
You can still convert the following datas into molecular structure:
(1)SMILES: n1nnnc1
(2)Std. InChI: InChI=1S/CH2N4/c1-2-4-5-3-1/h1H,(H,2,3,4,5)
(3)Std. InChIKey: KJUGUADJHNHALS-UHFFFAOYSA-N
Related Products
- Tetrazole
- TETRAZOLE-5-DIAZONIUM CHLORIDE
- 2889-61-4
- 28896-49-3
- 28898-02-4
- 28900-10-9
- 28900-64-3
- 28901-96-4
- 289031-45-4
- 28903-24-4
- 28903-71-1
- 289038-17-1
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View