-
Name
Tiagabine
- EINECS
- CAS No. 115103-54-3
- Article Data5
- CAS DataBase
- Density 1.208 g/cm3
- Solubility
- Melting Point 192 °C
- Formula C20H25NO2S2
- Boiling Point 568 °C at 760 mmHg
- Molecular Weight 375.556
- Flash Point 297.3 °C
- Transport Information
- Appearance white to off-white crystalline solid
- Safety 26-37/39
- Risk Codes 36/37/38
-
Molecular Structure
-
Hazard Symbols
 Xi
Xi
- Synonyms 3-Piperidinecarboxylicacid, 1-[4,4-bis(3-methyl-2-thienyl)-3-butenyl]-, (3R)- (9CI);3-Piperidinecarboxylic acid, 1-[4,4-bis(3-methyl-2-thienyl)-3-butenyl]-, (R)-;(R)-Tiagabine; Gabatril; NO 050328; NO 328; TGB; Tiagabine
- PSA 97.02000
- LogP 5.78470
Synthetic route
-
-
145821-58-5
ethyl (R)-1-<4,4-bis(3-methyl-2-thienyl)-3-butenyl>-3-piperidinecarboxylate

-
-
115103-54-3
tiagabine

| Conditions | Yield |
|---|---|
| With hydrogenchloride In water | 90% |
| With hydrogenchloride In water | 90% |
| With hydrogenchloride In water | 86% |
| With hydrogenchloride In water | 86% |
| With sodium hydroxide In ethanol for 4h; Ambient temperature; |

-
-
115103-54-3
tiagabine

| Conditions | Yield |
|---|---|
| With sodium hydrogencarbonate In water at 20℃; for 3h; Product distribution / selectivity; | |
| With sodium hydroxide In water pH=6; Product distribution / selectivity; Aqueous phosphate buffer; Sonographic reaction; No light exposure; | |
| Stage #1: tiagabine hydrochloride With sodium hydroxide In dichloromethane; water at 20℃; for 1 - 2.5h; Stage #2: With sodium hydrogencarbonate In dichloromethane; water at 20℃; for 1 - 2h; Product distribution / selectivity; |
-
-
14282-76-9
2-bromo-3-methylthiophene

-
-
115103-54-3
tiagabine

| Conditions | Yield |
|---|---|
| Multi-step reaction with 6 steps 1: 1.) Mg / 1.) Et2O, 1 h, 2.) Et2O, 1 h 2: 76 percent / MnO2 / CH2Cl2 / 16 h / Ambient temperature 3: 1.) Mg / 1.) THF, reflux, 0.5 h, 2.) THF, reflux, 1.5 h 4: 46 percent / 48percent aq. HBr / acetic acid / 1.5 h / 10 °C 5: 50 percent / KI, K2CO3 / acetone / 44 h / Ambient temperature 6: 12 M aq. NaOH / ethanol / 4 h / Ambient temperature View Scheme | |
| Multi-step reaction with 4 steps 1: 1.) n-BuLi / 1.) Et2O, 10 deg C, 15 min, 2.) Et2O, -70 deg C, 2.5 h 2: 20percent aq. H2SO4 / propan-2-ol / 3 h / Ambient temperature 3: 50 percent / KI, K2CO3 / acetone / 44 h / Ambient temperature 4: 12 M aq. NaOH / ethanol / 4 h / Ambient temperature View Scheme | |
| Multi-step reaction with 6 steps 1.1: magnesium / tetrahydrofuran / 1 h / 35 °C 1.2: 2 h / 0 °C / Reflux 2.1: hydrogenchloride / water; methanol / 1 °C / Reflux 3.1: triethylamine / diethyl ether / 1 h 4.1: potassium iodide; potassium carbonate / acetone / 120 h / 20 °C 5.1: sodium hydroxide / water; methanol / 2 h / 20 - 50 °C 6.1: hydrogen; C63H78IrNOP(2+)*C32H12BF24(1-) / methanol / 12 h / 60 °C / 4560.31 Torr / Autoclave View Scheme |
-
-
5834-16-2
3-methyl-2-thiophenecarboxaldehdye

-
-
115103-54-3
tiagabine

| Conditions | Yield |
|---|---|
| Multi-step reaction with 6 steps 1: 1.) Mg / 1.) Et2O, 1 h, 2.) Et2O, 1 h 2: 76 percent / MnO2 / CH2Cl2 / 16 h / Ambient temperature 3: 1.) Mg / 1.) THF, reflux, 0.5 h, 2.) THF, reflux, 1.5 h 4: 46 percent / 48percent aq. HBr / acetic acid / 1.5 h / 10 °C 5: 50 percent / KI, K2CO3 / acetone / 44 h / Ambient temperature 6: 12 M aq. NaOH / ethanol / 4 h / Ambient temperature View Scheme |
-
-
30717-55-6
bis(3-methyl-2-thienyl)methanone

-
-
115103-54-3
tiagabine

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: 1.) Mg / 1.) THF, reflux, 0.5 h, 2.) THF, reflux, 1.5 h 2: 46 percent / 48percent aq. HBr / acetic acid / 1.5 h / 10 °C 3: 50 percent / KI, K2CO3 / acetone / 44 h / Ambient temperature 4: 12 M aq. NaOH / ethanol / 4 h / Ambient temperature View Scheme |
-
-
148319-25-9
bis(3-methyl-2-thienyl)methanol

-
-
115103-54-3
tiagabine

| Conditions | Yield |
|---|---|
| Multi-step reaction with 5 steps 1: 76 percent / MnO2 / CH2Cl2 / 16 h / Ambient temperature 2: 1.) Mg / 1.) THF, reflux, 0.5 h, 2.) THF, reflux, 1.5 h 3: 46 percent / 48percent aq. HBr / acetic acid / 1.5 h / 10 °C 4: 50 percent / KI, K2CO3 / acetone / 44 h / Ambient temperature 5: 12 M aq. NaOH / ethanol / 4 h / Ambient temperature View Scheme |
-
-
109857-81-0
2-[4-bromo-1-(3-methylthien-2-yl)but-1-enyl]-3-methylthiophene

-
-
115103-54-3
tiagabine

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 50 percent / KI, K2CO3 / acetone / 44 h / Ambient temperature 2: 12 M aq. NaOH / ethanol / 4 h / Ambient temperature View Scheme |
-
-
148319-26-0
cyclopropylbis(3-methyl-2-thienyl)methanol

-
-
115103-54-3
tiagabine

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: 46 percent / 48percent aq. HBr / acetic acid / 1.5 h / 10 °C 2: 50 percent / KI, K2CO3 / acetone / 44 h / Ambient temperature 3: 12 M aq. NaOH / ethanol / 4 h / Ambient temperature View Scheme |
-
-
922184-80-3
4-Bromo-1,1-bis-(3-methyl-thiophen-2-yl)-butan-1-ol

-
-
115103-54-3
tiagabine

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: 20percent aq. H2SO4 / propan-2-ol / 3 h / Ambient temperature 2: 50 percent / KI, K2CO3 / acetone / 44 h / Ambient temperature 3: 12 M aq. NaOH / ethanol / 4 h / Ambient temperature View Scheme |
-
-
847233-27-6
4,4-bis(3-methyl-2-thienyl)-3-buten-1-ol

-
-
115103-54-3
tiagabine

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: triethylamine / diethyl ether / 1 h 2: potassium iodide; potassium carbonate / acetone / 120 h / 20 °C 3: sodium hydroxide / water; methanol / 2 h / 20 - 50 °C 4: hydrogen; C63H78IrNOP(2+)*C32H12BF24(1-) / methanol / 12 h / 60 °C / 4560.31 Torr / Autoclave View Scheme |

-
-
115103-54-3
tiagabine

| Conditions | Yield |
|---|---|
| With C63H78IrNOP(2+)*C32H12BF24(1-); hydrogen In methanol at 60℃; under 4560.31 Torr; for 12h; Autoclave; enantioselective reaction; | n/a |
-
-
300-08-3
arecoline hydrobromide

-
-
115103-54-3
tiagabine

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: potassium carbonate; water / 0.5 h / 20 °C 2: potassium iodide; potassium carbonate / acetone / 120 h / 20 °C 3: sodium hydroxide / water; methanol / 2 h / 20 - 50 °C 4: hydrogen; C63H78IrNOP(2+)*C32H12BF24(1-) / methanol / 12 h / 60 °C / 4560.31 Torr / Autoclave View Scheme |
-
-
92600-27-6
1-(1-chloroethyl) 3-methyl 5,6-dihydropyridine-1,3(2H)-dicarboxylate

-
-
115103-54-3
tiagabine

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1.1: methanol / 2 h / Reflux 1.2: 0.5 h 2.1: potassium iodide; potassium carbonate / acetone / 120 h / 20 °C 3.1: sodium hydroxide / water; methanol / 2 h / 20 - 50 °C 4.1: hydrogen; C63H78IrNOP(2+)*C32H12BF24(1-) / methanol / 12 h / 60 °C / 4560.31 Torr / Autoclave View Scheme |
-
-
495-19-2
norarecoline

-
-
115103-54-3
tiagabine

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: potassium iodide; potassium carbonate / acetone / 120 h / 20 °C 2: sodium hydroxide / water; methanol / 2 h / 20 - 50 °C 3: hydrogen; C63H78IrNOP(2+)*C32H12BF24(1-) / methanol / 12 h / 60 °C / 4560.31 Torr / Autoclave View Scheme |
-
-
847233-26-5
1,1-bis(3-methylthiophen-2-yl)butane-1,4-diol

-
-
115103-54-3
tiagabine

| Conditions | Yield |
|---|---|
| Multi-step reaction with 5 steps 1: hydrogenchloride / water; methanol / 1 °C / Reflux 2: triethylamine / diethyl ether / 1 h 3: potassium iodide; potassium carbonate / acetone / 120 h / 20 °C 4: sodium hydroxide / water; methanol / 2 h / 20 - 50 °C 5: hydrogen; C63H78IrNOP(2+)*C32H12BF24(1-) / methanol / 12 h / 60 °C / 4560.31 Torr / Autoclave View Scheme |
-
-
847233-13-0
4,4-bis(3-methylthiophen-2-yl)but-3-en-1-yl methanesulfonate

-
-
115103-54-3
tiagabine

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: potassium iodide; potassium carbonate / acetone / 120 h / 20 °C 2: sodium hydroxide / water; methanol / 2 h / 20 - 50 °C 3: hydrogen; C63H78IrNOP(2+)*C32H12BF24(1-) / methanol / 12 h / 60 °C / 4560.31 Torr / Autoclave View Scheme |

-
-
115103-54-3
tiagabine

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: sodium hydroxide / water; methanol / 2 h / 20 - 50 °C 2: hydrogen; C63H78IrNOP(2+)*C32H12BF24(1-) / methanol / 12 h / 60 °C / 4560.31 Torr / Autoclave View Scheme |
-
-
115103-54-3
tiagabine

-
-
1006863-69-9
tiagabine hydrobromide

| Conditions | Yield |
|---|---|
| With hydrogen bromide In di-isopropyl ether; ethyl acetate; acetonitrile at 20℃; Product distribution / selectivity; | 94% |

| Conditions | Yield |
|---|---|
| In methanol; acetonitrile Product distribution / selectivity; | 91% |
| In tetrahydrofuran; dichloromethane; isopropyl alcohol at 20℃; for 48h; Product distribution / selectivity; |
-
-
115103-54-3
tiagabine

| Conditions | Yield |
|---|---|
| With hydrogenchloride In dichloromethane; water at 20℃; for 1h; | 90% |
| With hydrogenchloride In methanol; dichloromethane; chloroform at 3℃; for 24h; |

| Conditions | Yield |
|---|---|
| In methanol; ethyl acetate; acetonitrile at 20℃; for 1h; Product distribution / selectivity; | 77% |
| In tetrahydrofuran; dichloromethane; isopropyl alcohol at 20℃; for 48h; Product distribution / selectivity; |

| Conditions | Yield |
|---|---|
| In methanol; acetonitrile Product distribution / selectivity; | 68% |
| In methanol; dichloromethane at 20℃; for 48h; Product distribution / selectivity; |

| Conditions | Yield |
|---|---|
| In methanol; ethyl acetate; acetonitrile at 20℃; for 1h; | 32% |

| Conditions | Yield |
|---|---|
| In methanol; acetonitrile at 20℃; Product distribution / selectivity; | |
| In dichloromethane; ethyl acetate; acetone at 20℃; for 48h; Product distribution / selectivity; |
Tiagabine Chemical Properties
Molecule structure of 3-Piperidinecarboxylic acid, 1-(4,4-bis(3-methyl-2-thienyl)-3-butenyl)-, (R)- (CAS NO.115103-54-3):
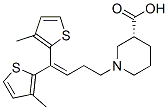
IUPAC Name: (3R)-1-[4,4-Bis(3-methylthiophen-2-yl)but-3-enyl]piperidine-3-carboxylic acid
Molecular Weight: 375.548 g/mol
Molecular Formula: C20H25NO2S2
Density: 1.208 g/cm3
Melting Point: 192 °C
Boiling Point: 568 °C at 760 mmHg
Flash Point: 297.3 °C
Index of Refraction: 1.604
Molar Refractivity: 106.97 cm3
Molar Volume: 310.6 cm3
Polarizability: 42.4×10-24 cm3
Surface Tension: 49.8 dyne/cm
Enthalpy of Vaporization: 89.69 kJ/mol
Vapour Pressure: 9.71E-14 mmHg at 25 °C
XLogP3-AA: 2.7
H-Bond Donor: 1
H-Bond Acceptor: 3
Rotatable Bond Count: 6
Exact Mass: 375.13267
MonoIsotopic Mass: 375.13267
Topological Polar Surface Area: 40.5
Heavy Atom Count: 25
Complexity: 474
Defined Atom StereoCenter Count: 1
Canonical SMILES: CC1=C(SC=C1)C(=CCCN2CCCC(C2)C(=O)O)C3=C(C=CS3)C
Isomeric SMILES: CC1=C(SC=C1)C(=CCCN2CCC[C@H](C2)C(=O)O)C3=C(C=CS3)C
InChI: InChI=1S/C20H25NO2S2/c1-14-7-11-24-18(14)17(19-15(2)8-12-25-19)6-4-10-21-9-3-5-16(13-21)20(22)23/h6-8,11-12,16H,3-5,9-10,13H2,1-2H3,(H,22,23)/t16-/m1/s1
InChIKey: PBJUNZJWGZTSKL-MRXNPFEDSA-N
Product Categories: Intermediates & Fine Chemicals; Neurochemicals; Pharmaceuticals
Tiagabine Uses
3-Piperidinecarboxylic acid, 1-(4,4-bis(3-methyl-2-thienyl)-3-butenyl)-, (R)- (CAS NO.115103-54-3) is used as a GABA uptake inhibitor.
Tiagabine Safety Profile
Hazard Codes:  Xi
Xi
Risk Statements: 36/37/38
R36/37/38:Irritating to eyes, respiratory system and skin.
Safety Statements: 26-37/39
S26: In case of contact with eyes, rinse immediately with plenty of water and seek medical advice.
S37/39:Wear suitable gloves and eye/face protection.
Hazardous Substances Data: 115103-54-3(Hazardous Substances Data)
Tiagabine Specification
3-Piperidinecarboxylic acid, 1-(4,4-bis(3-methyl-2-thienyl)-3-butenyl)-, (R)- (CAS NO.115103-54-3) is also named as HSDB 7527 ; Tiagabina ; Tiagabina [INN-Spanish] ; Tiagabine ; Tiagabinum ; Tiagabinum [INN-Latin] ; UNII-Z80I64HMNP ; (-)-(R)-1-(4,4-Bis(3-methyl-2-thienyl)-3-butenyl)nipecotic acid . 3-Piperidinecarboxylic acid, 1-(4,4-bis(3-methyl-2-thienyl)-3-butenyl)-, (R)- (CAS NO.115103-54-3) is white to off-white crystalline solid.
Related Products
- Tiagabine
- Tiagabine hydrochloride
- 115104-25-1
- 115104-40-0
- 115-10-6
- 1151-14-0
- 115114-32-4
- 1151-15-1
- 115-11-7
- 115118-93-9
- 115122-63-9
- 1151397-80-6
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View
