-
Name
TRI-N-OCTYLALUMINIUM
- EINECS 213-964-4
- CAS No. 1070-00-4
- Article Data10
- CAS DataBase
- Density 0.701 g/mL at 25 °C
- Solubility
- Melting Point <-40 °C
- Formula C24H51Al
- Boiling Point 126.4oC at 760mmHg
- Molecular Weight 366.65
- Flash Point -6 °F
- Transport Information UN 2924 3/PG 2
- Appearance clear colorless solution
- Safety 9-16-23-26-36/37/39-45-61
- Risk Codes 11-14-20/21/22-35-48-67-65-62-51-48/20
-
Molecular Structure
-
Hazard Symbols
 F;
F;  C;
C;  N
N
- Synonyms Trioctylaluminum;Aluminum, trioctyl-;Trioctylalane;
- PSA 0.00000
- LogP 9.94350
Synthetic route

| Conditions | Yield |
|---|---|
| at 110℃; Inert atmosphere; Schlenk technique; | 100% |
-
-
16853-85-3
lithium aluminium tetrahydride

-
-
111-66-0
oct-1-ene

-
-
7325-26-0
di(n-octyl)aluminum chloride

-
-
1070-00-4
trioctylaluminum

| Conditions | Yield |
|---|---|
| In further solvent(s) byproducts: LiCl; LiAlH4, oct-1-ene loaded in a vertical-type ball mill under Ar or N2; mixt. heated with agitation for 20 min at 70°C; (C8H17)2AlCl added for 0.5 h (LiAlH4:(C8H17)2AlCl:oct-1-ene=1:1:4.28); mixt. agitated for 1h at 95-100°C; cooled; ppt. filtered off; olefin removed (vac., 1 Torr) at 80-90°C; elem. anal.; | 98% |


| Conditions | Yield |
|---|---|
| In octane under N2 or Ar; suspn. of (AlH3)(n) in octane heated to 120°C; olefin (1 equiv.) added dropwise; after 30 min temp. raised to 125°C (reflux); cooled to 100-110°C; olefin (2.1 equiv.) added over 3-4 h; kept (90-100°C, 1 h); cooled; filtered through glass filter; solvent amd excess olefin distd. off in vac. below 80°C (7 Torr); elem. anal.; | 91% |
| With triisobutylaluminum In octane under N2 or Ar; vertical ball mill loaded with (AlH3)(n), i-Bu3Al, olefin (molar ratio 1:0.14:4.8) and octane; heated gradually to 100°C with stirring; cooled; stored at 90-100°C for 2 h; balls and blend sepd.; residue washed with octane; filtrate evapd. at 80-90°C (7 Torr); elem. anal.; | 88.6% |
| With Na(i-Bu)3AlH In octane under N2 or Ar; mixt. of (AlH3)(n), Na(i-Bu)3AlH (molar ratio 1:0.24) and octane heated to 80°C for 1 h; olefin added; temp. spontaneously raised to 100°C for 20 min; cooled; olefin additionally added dropwise at 90°C for 0.5-1 h; filtered; solvent and excess olefin distd. off in vac. below 80°C(7 Torr); mixt. of (C8H17)3Al and NaAl(C8H17)4 obtained; elem. anal.; |

| Conditions | Yield |
|---|---|
| With lithium aluminium tetrahydride; aluminium trichloride In octane byproducts: LiCl; under N2 or Ar; mixt. of γ-AlH3, excess LiAlH4, octane and olefin heated to 100-110°C for 1 h; additional olefin added at 90-100°C for 2 h; filtered; solvent and excess olefin removed in vac. at 60-70°C (1Torr); AlCl3 added; heated at 80°C; filtered; elem. anal.; | 89% |

| Conditions | Yield |
|---|---|
| bis(1,5-cyclooctadiene)nickel (0) 0°C; | |
| tris(ethene)nickel(0) 0°C; |

| Conditions | Yield |
|---|---|
| In (2)H8-toluene at 75℃; for 2h; Inert atmosphere; Schlenk technique; | |
| In toluene at 75℃; for 0.5h; Solvent; Inert atmosphere; Schlenk technique; |

| Conditions | Yield |
|---|---|
| Stage #1: 1-Iodooctane With n-butyllithium In tetrahydrofuran Inert atmosphere; Glovebox; Stage #2: aluminum (III) chloride In tetrahydrofuran at 20℃; for 0.75h; Inert atmosphere; Glovebox; |
-
-
1070-00-4
trioctylaluminum

-
A
-
3091-25-6
n-octyltin trichloride

-
B
-
3542-36-7
di-n-octyltin dichloride

-
C
-
2587-76-0
trioctyltin chloride

| Conditions | Yield |
|---|---|
| With tert-butyl methyl ether; tin(IV) chloride at 50℃; for 0.75h; Product distribution / selectivity; | A 98% B 98% C 98% |
| With 1,3-dioxane; tin(IV) chloride at 50℃; for 0.75h; Product distribution / selectivity; | A 97.3% B 97.3% C 97.3% |
| With diethyl ether; tin(IV) chloride at 50℃; for 0.75h; Product distribution / selectivity; | A 96.8% B 96.8% C 96.8% |
| With tetrahydrofuran; tin(IV) chloride at 50℃; for 0.75h; Product distribution / selectivity; | A 95.6% B 95.6% C 95.6% |
| With dibutyl ether; tin(IV) chloride at 50 - 100℃; for 0.75h; Product distribution / selectivity; | A 89% B 89% C 89% |


| Conditions | Yield |
|---|---|
| In dibutyl ether 70°C; | 98% |
| In dibutyl ether 70°C; | 98% |
| In dibutyl ether 70°C; | 98% |
-
-
1469537-96-9
N,N,N-trimethyl-2-naphthalenaminium trifluoromethanesulfonate

-
-
1070-00-4
trioctylaluminum

-
-
2876-44-0
2-(n-octyl)napthalene

| Conditions | Yield |
|---|---|
| With 1,3-bis[(diphenylphosphino)propane]dichloronickel(II) In 1,4-dioxane at 110℃; for 24h; Schlenk technique; Inert atmosphere; | 97% |
-
-
1351445-39-0
N,N,N-trimethyl-1-naphthalenaminium trifluoromethanesulfonate

-
-
1070-00-4
trioctylaluminum

-
-
2876-51-9
1-octylnaphthalene

| Conditions | Yield |
|---|---|
| With 1,3-bis[(diphenylphosphino)propane]dichloronickel(II) In 1,4-dioxane at 110℃; for 24h; Schlenk technique; Inert atmosphere; | 94% |

| Conditions | Yield |
|---|---|
| With copper(l) iodide; sodium methylate; lithium chloride In N,N-dimethyl-formamide at 120℃; for 12h; Inert atmosphere; Glovebox; | 91% |

| Conditions | Yield |
|---|---|
| In n-heptane at 0 - 20℃; for 1h; Inert atmosphere; diastereoselective reaction; | 91% |
-
-
111-66-0
oct-1-ene

-
-
15337-08-3
iso-C4H9SnH3

-
-
1070-00-4
trioctylaluminum

-
-
16257-00-4
(C8H17)3Sn-iso-C4H9

| Conditions | Yield |
|---|---|
| In cyclohexane reaction at 100°C for 36 h;; | 89% |
| In cyclohexane reaction at 100°C for 36 h;; | 89% |
| In cyclohexane reaction at 100°C for 36 h;; | 59% |
| In cyclohexane reaction at 100°C for 36 h;; | 59% |

-
-
1258862-63-3
5-bromo-2-(4-bromo-2-dibromoborylphenyl)pyridine

-
-
1070-00-4
trioctylaluminum

-
-
1258862-75-7
C27H40BBr2N

| Conditions | Yield |
|---|---|
| In dichloromethane; toluene at 20℃; for 3h; | 88% |
| In dichloromethane; toluene at 20℃; for 0.166667h; Inert atmosphere; | 88% |
| In dichloromethane; toluene at 20℃; for 6h; | 34% |

| Conditions | Yield |
|---|---|
| Stage #1: 2-Methoxynaphthalene With bis(1,5-cyclooctadiene)nickel (0); 1,2-bis-(dicyclohexylphosphino)ethane In di-isopropyl ether; toluene at 20℃; for 0.0833333h; Glovebox; Inert atmosphere; Stage #2: trioctylaluminum In di-isopropyl ether; toluene at 120℃; for 72h; Glovebox; Inert atmosphere; | 85% |

| Conditions | Yield |
|---|---|
| In n-heptane at 0 - 20℃; for 1h; Inert atmosphere; diastereoselective reaction; | 82% |

| Conditions | Yield |
|---|---|
| With 1,3-bis[(diphenylphosphino)propane]dichloronickel(II) In 1,4-dioxane at 110℃; for 24h; Schlenk technique; Inert atmosphere; | 79% |

| Conditions | Yield |
|---|---|
| With copper(l) iodide; sodium methylate; lithium chloride In N,N-dimethyl-formamide at 120℃; for 12h; Inert atmosphere; Glovebox; | 78% |
-
-
1893-38-5
methyl 2,2,3,3-tetrafluoropropionate

-
-
1070-00-4
trioctylaluminum

-
-
147123-74-8
1,1,2,2-tetrafluoroundecanonanol-3

| Conditions | Yield |
|---|---|
| In dichloromethane for 12h; Ambient temperature; | 77.5% |
| With hydrogen cation 1) CH2Cl2, -20 to 5 deg C, 2) H2O; Multistep reaction; |

| Conditions | Yield |
|---|---|
| With dilithium tetrachlorocuprate; triphenylphosphine In octane at 50℃; for 5h; | 74% |

-
-
1070-00-4
trioctylaluminum

| Conditions | Yield |
|---|---|
| In toluene (under Ar, Schlenk); B-compound placed in tube, Al(CH2)7CH3)3 in tolueneadded, heated at 120-130°C for 4 d, cooled to room temp., poured into mixt. of ice, CsOH, Na2EDTA; extd. with CH2Cl2, organic layer washed with CsCl, extd. with Et2O, organic phase dried over Cs2CO3, evapd. to dryness, recrystd. from H2O, filtered, washed with H2O, pentane, vac. dried; | 72% |

| Conditions | Yield |
|---|---|
| With aluminium trichloride In dichloromethane -5 deg C, then 20 deg C, 1 h; | 68% |

| Conditions | Yield |
|---|---|
| Stage #1: Nonanoyl chloride With aluminium trichloride In dichloromethane at -78 - 20℃; Stage #2: trioctylaluminum In hexane; dichloromethane at -78 - 20℃; Further stages.; | 67% |

-
-
1070-00-4
trioctylaluminum

| Conditions | Yield |
|---|---|
| In toluene Inert atmosphere; | A 67% B n/a |


| Conditions | Yield |
|---|---|
| With aluminium trichloride In dichloromethane | 65% |
-
-
7446-70-0, 7784-13-6
aluminum (III) chloride

-
-
10026-11-6
zirconium(IV) chloride

-
-
1070-00-4
trioctylaluminum

-
-
7429-90-5
aluminium

-
-
71-43-2
benzene

| Conditions | Yield |
|---|---|
| Stage #1: aluminum (III) chloride; zirconium(IV) chloride; aluminium; benzene With 1,3,5-trimethyl-benzene for 3h; Reflux; Stage #2: trioctylaluminum In hexane | 65% |


-
-
1070-00-4
trioctylaluminum

| Conditions | Yield |
|---|---|
| In toluene Inert atmosphere; | 65% |

| Conditions | Yield |
|---|---|
| With bis(1,5-cyclooctadiene)nickel (0); tricyclohexylphosphine In 1,4-dioxane at 120℃; for 24h; | 64% |

| Conditions | Yield |
|---|---|
| With potassium fluoride; benzylnixantphos; iron(II) acetate In tetrahydrofuran at 0 - 40℃; for 44h; Negishi Coupling; Inert atmosphere; | 60% |
-
-
1240637-92-6
(1R)-2,3,4-tri-O-acetyl-1-fluoro-5N,6O-oxomethylidene-1-deoxynojirimycin

-
-
1070-00-4
trioctylaluminum

-
A
-
1240637-95-9
(1R)-2,3,4-tri-O-acetyl-1-octyl-5N,6O-oxomethylidene-1-deoxynojirimycin

-
B
-
1240638-01-0
(1S)-2,3,4-tri-O-acetyl-1-octyl-5N,6O-oxomethylidene-1-deoxynojirimycin

| Conditions | Yield |
|---|---|
| In toluene Inert atmosphere; | A 58% B n/a |
| In toluene at 0℃; for 3h; Inert atmosphere; optical yield given as %de; |

-
-
98-08-8
α,α,α-trifluorotoluene

-
-
1070-00-4
trioctylaluminum

-
A
-
111-66-0
oct-1-ene

-
B
-
1081-77-2
n-nonylbenzene

-
C
-
1197344-24-3
C31H56

-
D
-
20216-92-6
1-octyl-1-phenylnonane

| Conditions | Yield |
|---|---|
| In hexane; 1,2-dichloro-ethane at 50℃; for 42h; | A 57% B 11% C 8% D 34% |


| Conditions | Yield |
|---|---|
| In n-heptane; toluene for 3h; | 57% |
Tri-n-octylaluminum Specification
The CAS registry number of Tri-n-octylaluminum is 1070-00-4. The IUPAC name is trioctylalumane. In addition, the molecular formula is C24H51Al. It is stable at common pressure and temperature, and it should be stored in a cool and dry place. What's more, it is sensitive to moisture air and water.
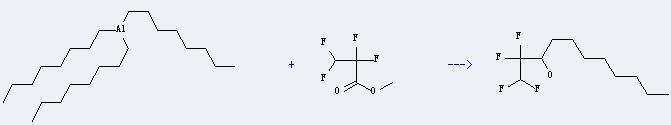
Uses of Tri-n-octylaluminum: it can react with 2,2,3,3-tetrafluoro-propionic acid methyl ester to get 1,1,2,2-tetrafluoro-undecan-3-ol. This reaction will need solvent CH2Cl2. The reaction time is 12 hours with ambient temperature. The yield is about 77.5%.
When you are using this chemical, please be cautious about it as the following:
It is highly flammable and can cause severe burns, so you should keep it away from sources of ignition - No smoking. And it will react violently with water. It is harmful by inhalation, in contact with skin and if swallowed. In addition, it has the danger of serious damage to health by prolonged exposure. Its vapours may cause drowsiness and dizziness. Moreover, it is harmful as it may cause lung damage if swallowed. It is also harmful as it has the danger of serious damage to health by prolonged exposure through inhalation. Besides, it possibly has the risk of impaired fertility. It is toxic to aquatic organisms. When using it, you need wear suitable protective clothing, gloves and eye/face protection. You should not breathe gas/fumes/vapour/spray (appropriate wording to be specified by the manufacturer). In case of contact with eyes, you should rinse immediately with plenty of water and seek medical advice. And in case of accident or if you feel unwell seek medical advice immediately (show the label where possible). It should be avoided releasing to the environment just refering to special instructions/safety data sheet. In addition, you should keep the container in a well-ventilated place after using it.
You can still convert the following datas into molecular structure:
(1)SMILES: CCCCCCCC[Al](CCCCCCCC)CCCCCCCC
(2)InChI: InChI=1/3C8H17.Al/c3*1-3-5-7-8-6-4-2;/h3*1,3-8H2,2H3;/rC24H51Al/c1-4-7-10-13-16-19-22-25(23-20-17-14-11-8-5-2)24-21-18-15-12-9-6-3/h4-24H2,1-3H3
(3)InChIKey: LFXVBWRMVZPLFK-AQKGURAUAT
Related Products
- Tri-n-octylaluminum
- 107001-49-0
- 1070-03-7
- 107007-99-8
- 107008-28-6
- 1070-10-6
- 1070-11-7
- 107015-83-8
- 107016-79-5
- 107-01-7
- 107017-72-1
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View