-
Name
Vanillylacetone
- EINECS 204-548-3
- CAS No. 122-48-5
- Article Data43
- CAS DataBase
- Density 1.111 g/cm3
- Solubility
- Melting Point 40-41 °C(lit.)
- Formula C11H14O3
- Boiling Point 323 °C at 760 mmHg
- Molecular Weight 194.23
- Flash Point 123.7 °C
- Transport Information
- Appearance Clear slightly yellow liquid.
- Safety 26-36
- Risk Codes 36/37/38
-
Molecular Structure
- Hazard Symbols
- Synonyms Zingiberone;Zingerone;NSC 15335;Gingerone;4-Hydroxy-3-methoxybenzylacetone;4-(4-Hydroxy-3-methoxyphenyl)-2-butanone;3-Methoxy-4-hydroxybenzylacetone;(4-Hydroxy-3-methoxyphenyl)ethylmethyl ketone;
- PSA 46.53000
- LogP 1.92240
Synthetic route

| Conditions | Yield |
|---|---|
| With diphenyl sulfide; palladium 10% on activated carbon; hydrogen In methanol at 20℃; for 24h; | 94% |
| With N-benzylammonium trifluoroacetate; 1,4-dihydro-2,6-dimethyl-3,5-bis<(methylamino)carbonyl>pyridine In tetrahydrofuran at 70℃; for 16h; Sealed tube; | 93% |
| With palladium on activated charcoal; hydrogen; acetic acid In ethanol at 20℃; under 760.051 Torr; | 87% |
-
-
1080-12-2, 22214-42-2
(E)-4-(4-hydroxy-3-methoxyphenyl)but-3-ene-2-one

-
-
122-48-5
4-(4-hydroxy-3-methoxyphenyl)-2-butanone

| Conditions | Yield |
|---|---|
| With Rh/Al2O3; hydrogen In methanol at 20℃; under 750.075 Torr; for 2h; | 100% |
| With palladium 10% on activated carbon; hydrogen In methanol at 25℃; for 4h; Time; | 97% |
| With palladium 10% on activated carbon; hydrogen In methanol at 20℃; | 97% |
-
-
1080-12-2, 22214-42-2
(E)-4-(4-hydroxy-3-methoxyphenyl)but-3-ene-2-one

-
A
-
122-48-5
4-(4-hydroxy-3-methoxyphenyl)-2-butanone

-
B
-
39728-80-8
3-hydroxy-1-(4-hydroxy-3-methoxyphenyl)butane

| Conditions | Yield |
|---|---|
| With sodium tetrahydroborate; nickel(II) chloride hexahydrate; hydrogen In methanol at 0 - 30℃; under 760.051 Torr; Reflux; Inert atmosphere; | A 98% B 2% |
| With diphenyl diselenide; hypophosphorous acid In glycerol at 90℃; for 0.5h; Inert atmosphere; chemoselective reaction; | A 68% B 19% |
| With hydrogen; palladium on activated charcoal In ethyl acetate |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 63 percent / aq. NaOH / 48 h / Ambient temperature 2: 43 percent / H2 / 10percent Pd/C / 3 h / Ambient temperature View Scheme | |
| Multi-step reaction with 2 steps 1: aq. NaOH 2: H2 / Pd-C / CHCl3 View Scheme | |
| Multi-step reaction with 2 steps 1: aq. alkali 2: H2 / Raney-Ni View Scheme |

-
-
122-48-5
4-(4-hydroxy-3-methoxyphenyl)-2-butanone

| Conditions | Yield |
|---|---|
| With tetrabutyl ammonium fluoride; zinc(II) chloride In tetrahydrofuran; toluene at 20℃; for 0.75h; | 70% |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: aq. NaOH / 20 °C 2: aq. HCl / 20 °C 3: H2; AcOH / Pd/C / ethanol View Scheme | |
| Multi-step reaction with 2 steps 1: NaOH; water / 20 °C 2: H2 / Pd/C / ethyl acetate View Scheme | |
| Multi-step reaction with 2 steps 1: 83 percent / NaOH / H2O / 3 h 2: H2 / 10 percent Pd/C / ethyl acetate View Scheme |
-
-
1080-12-2
Dehydrozingerone

-
A
-
122-48-5
4-(4-hydroxy-3-methoxyphenyl)-2-butanone

-
B
-
39728-80-8
3-hydroxy-1-(4-hydroxy-3-methoxyphenyl)butane

| Conditions | Yield |
|---|---|
| With palladium 10% on activated carbon; hydrogen; acetic acid In methanol at 20℃; for 24h; | A 58% B 37% |
| With ethanol; palladium Hydrogenation; |
-
-
78-94-4
methyl vinyl ketone

-
-
90-05-1
2-methoxy-phenol

-
-
122-48-5
4-(4-hydroxy-3-methoxyphenyl)-2-butanone

| Conditions | Yield |
|---|---|
| With amberlyst-15 In toluene at 20 - 40℃; | 36.6% |

-
-
122-48-5
4-(4-hydroxy-3-methoxyphenyl)-2-butanone

| Conditions | Yield |
|---|---|
| With trifluoroacetic acid In dichloromethane at 20℃; for 24h; Inert atmosphere; solid phase reaction; | 12 mg |
-
-
61152-59-8
4-(4-hydroxyphenyl-3-methoxy)-4-hydroxy-2-butanone

-
-
122-48-5
4-(4-hydroxy-3-methoxyphenyl)-2-butanone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: aq. HCl / 20 °C 2: H2; AcOH / Pd/C / ethanol View Scheme |
-
-
39886-78-7
4-[4-(benzyloxy)-3-methoxyphenyl]butan-2-one

-
-
122-48-5
4-(4-hydroxy-3-methoxyphenyl)-2-butanone

| Conditions | Yield |
|---|---|
| With hydrogen; palladium on activated charcoal In methanol |
-
-
911682-18-3
4-(4-((tert-butyldimethylsiloxy)-methyl)-3-methoxyphenyl)-but-3-ene-2-one

-
-
122-48-5
4-(4-hydroxy-3-methoxyphenyl)-2-butanone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: tetrabutylammonium fluoride / tetrahydrofuran 2: H2 / Pd/C View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 5 steps 1: imidazole 2: MnO2 / pentane 3: tetrabutylammonium fluoride / tetrahydrofuran 4: H2 / Pd/C View Scheme |
-
-
290820-47-2
(2E)-3-[4-[[tert-butyldimethylsilyl]oxy]-3-methoxyphenyl]acrylaldehyde

-
-
122-48-5
4-(4-hydroxy-3-methoxyphenyl)-2-butanone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: MnO2 / pentane 2: tetrabutylammonium fluoride / tetrahydrofuran 3: H2 / Pd/C View Scheme |
-
-
911682-17-2
4-[4-(tert-butyl-dimethyl-silanyloxy)-3-methoxy-phenyl]-but-3-en-2-ol

-
-
122-48-5
4-(4-hydroxy-3-methoxyphenyl)-2-butanone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: MnO2 / pentane 2: tetrabutylammonium fluoride / tetrahydrofuran 3: H2 / Pd/C View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 82 percent / 10percent aq. sodium hydroxide / 96 h 2: 86 percent / H2 / Raney nickel / acetone View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: NaBH4 / methanol 2: PBr3 / diethyl ether 3: (i) nBuLi, THF, (ii) /BRN= 2130929/, (iii) aq. NaIO4, MeOH 4: H2 / Pd-C / methanol View Scheme |
-
-
72724-00-6
1-(benzyloxy)-4-(bromomethyl)-2-methoxybenzene

-
-
122-48-5
4-(4-hydroxy-3-methoxyphenyl)-2-butanone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: (i) nBuLi, THF, (ii) /BRN= 2130929/, (iii) aq. NaIO4, MeOH 2: H2 / Pd-C / methanol View Scheme |
-
-
33693-48-0
4-benzyloxy-3-methoxybenzyl alcohol

-
-
122-48-5
4-(4-hydroxy-3-methoxyphenyl)-2-butanone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: PBr3 / diethyl ether 2: (i) nBuLi, THF, (ii) /BRN= 2130929/, (iii) aq. NaIO4, MeOH 3: H2 / Pd-C / methanol View Scheme |
-
-
1135-24-6
(E)-3-(4-hydroxy-3-methoxyphenyl)acrylic acid

-
-
122-48-5
4-(4-hydroxy-3-methoxyphenyl)-2-butanone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 6 steps 1.1: thionyl chloride / tetrahydrofuran / 2.5 h / 20 °C / Inert atmosphere 2.1: N-ethyl-N,N-diisopropylamine / tetrahydrofuran; dichloromethane / 24 h / 0 - 20 °C / Inert atmosphere 3.1: trimethylaluminum / dichloromethane; toluene / 15 h / 20 °C / Inert atmosphere 3.2: 2 h / 20 °C / Inert atmosphere 4.1: tetrahydrofuran / -78 °C / Inert atmosphere 5.1: hydrogenchloride / tetrahydrofuran; water / 5 h / Reflux 6.1: hydrogen; Rh/Al2O3 / methanol / 2 h / 20 °C / 750.08 Torr View Scheme |

-
-
122-48-5
4-(4-hydroxy-3-methoxyphenyl)-2-butanone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: tetrahydrofuran / -78 °C / Inert atmosphere 2: hydrogenchloride / tetrahydrofuran; water / 5 h / Reflux 3: hydrogen; Rh/Al2O3 / methanol / 2 h / 20 °C / 750.08 Torr View Scheme |

-
-
122-48-5
4-(4-hydroxy-3-methoxyphenyl)-2-butanone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: hydrogenchloride / tetrahydrofuran; water / 5 h / Reflux 2: hydrogen; Rh/Al2O3 / methanol / 2 h / 20 °C / 750.08 Torr View Scheme |

-
-
122-48-5
4-(4-hydroxy-3-methoxyphenyl)-2-butanone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 5 steps 1.1: N-ethyl-N,N-diisopropylamine / tetrahydrofuran; dichloromethane / 24 h / 0 - 20 °C / Inert atmosphere 2.1: trimethylaluminum / dichloromethane; toluene / 15 h / 20 °C / Inert atmosphere 2.2: 2 h / 20 °C / Inert atmosphere 3.1: tetrahydrofuran / -78 °C / Inert atmosphere 4.1: hydrogenchloride / tetrahydrofuran; water / 5 h / Reflux 5.1: hydrogen; Rh/Al2O3 / methanol / 2 h / 20 °C / 750.08 Torr View Scheme |

-
-
122-48-5
4-(4-hydroxy-3-methoxyphenyl)-2-butanone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1.1: trimethylaluminum / dichloromethane; toluene / 15 h / 20 °C / Inert atmosphere 1.2: 2 h / 20 °C / Inert atmosphere 2.1: tetrahydrofuran / -78 °C / Inert atmosphere 3.1: hydrogenchloride / tetrahydrofuran; water / 5 h / Reflux 4.1: hydrogen; Rh/Al2O3 / methanol / 2 h / 20 °C / 750.08 Torr View Scheme |

-
-
122-48-5
4-(4-hydroxy-3-methoxyphenyl)-2-butanone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: dmap / toluene / Inert atmosphere; Reflux 2: zinc(II) chloride; tetrabutyl ammonium fluoride / toluene; tetrahydrofuran / 0.75 h / 20 °C View Scheme |
-
-
69405-03-4
4-{[(tert-butyl)(diphenyl)silyl]oxy}-3-methoxybenzaldehyde

-
-
122-48-5
4-(4-hydroxy-3-methoxyphenyl)-2-butanone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: sodium tetrahydroborate / dichloromethane; ethanol / 0.75 h / 0 °C 2: dmap / toluene / Inert atmosphere; Reflux 3: zinc(II) chloride; tetrabutyl ammonium fluoride / toluene; tetrahydrofuran / 0.75 h / 20 °C View Scheme |

| Conditions | Yield |
|---|---|
| Stage #1: vanillin; acetone With sodium hydroxide at 20℃; for 12h; Stage #2: With hydrogen |

-
-
122-48-5
4-(4-hydroxy-3-methoxyphenyl)-2-butanone

| Conditions | Yield |
|---|---|
| With diethyl ether; hydrogen; platinum |
-
-
89082-79-1
(E)-ethyl 2-(4-hydroxy-3-methoxybenzylidene)-3-oxo-butanoate

-
-
122-48-5
4-(4-hydroxy-3-methoxyphenyl)-2-butanone

| Conditions | Yield |
|---|---|
| With sodium hydroxide; sodium amalgam erwaermt man das Reaktionsprodukt mit konz. Natronlauge und erhitzt die erhaltene Saeure im Vakuum; |

| Conditions | Yield |
|---|---|
| With 1:1 copper and nickel alloy on γ-Al2O3 at 245℃; for 3h; Activation energy; Temperature; Reagent/catalyst; Inert atmosphere; Autoclave; | 45.8% |
-
-
122-48-5
4-(4-hydroxy-3-methoxyphenyl)-2-butanone

-
-
39728-80-8
3-hydroxy-1-(4-hydroxy-3-methoxyphenyl)butane

| Conditions | Yield |
|---|---|
| With sodium tetrahydroborate In methanol at 20℃; for 2.5h; Solvent; Enzymatic reaction; | 100% |
| With sodium tetrahydroborate In methanol at 20℃; for 1.5h; | 96.6% |
| With sodium tetrahydroborate In ethanol | 91% |
| With hydrogen; palladium on activated charcoal In ethyl acetate | |
| Multi-step reaction with 2 steps 1: 84 percent / phenol, hexamethyldisilazane / benzene / Heating 2: 1.) lithium di-isopropylamide, hexanal, 2.) 2M HCl / 1.) -78 deg C, 1 h, THF, 2.) ether View Scheme |
-
-
110-87-2
3,4-dihydro-2H-pyran

-
-
122-48-5
4-(4-hydroxy-3-methoxyphenyl)-2-butanone

-
-
223801-42-1
4-(3-methoxy-4-(tetrahydro-2H-pyran-2-yloxy)phenyl)butan-2-one

| Conditions | Yield |
|---|---|
| With pyridinium p-toluenesulfonate In dichloromethane at 20℃; for 24h; | 99% |
| With pyridinium p-toluenesulfonate In dichloromethane |
-
-
122-48-5
4-(4-hydroxy-3-methoxyphenyl)-2-butanone

-
-
2835-77-0
(2-aminophenyl)(phenyl)methanone

-
-
1408335-48-7
2-methoxy-4-((2-methyl-4-phenylquinolin-3-yl)methyl)phenol

| Conditions | Yield |
|---|---|
| With trifluoroacetic acid at 100℃; for 0.5h; | 96% |
-
-
112-31-2
caprinaldehyde

-
-
122-48-5
4-(4-hydroxy-3-methoxyphenyl)-2-butanone

-
-
107257-18-1
5-hydroxy-1-(4-hydroxy-3-methoxyphenyl)tetradecan-3-one

| Conditions | Yield |
|---|---|
| With n-butyllithium; lithium diisopropyl amide In tetrahydrofuran; hexane at -78℃; for 6h; | 95% |
| Stage #1: 4-(4-hydroxy-3-methoxyphenyl)-2-butanone With lithium diisopropyl amide In tetrahydrofuran at -78℃; for 1h; Stage #2: caprinaldehyde In tetrahydrofuran at -78℃; for 3h; Time; | 32% |
| With lithium diisopropyl amide In tetrahydrofuran; hexane at -78℃; |
-
-
124-13-0
Octanal

-
-
122-48-5
4-(4-hydroxy-3-methoxyphenyl)-2-butanone

-
-
77398-92-6
5-hydroxy-1-(4-hydroxy-3-methoxyphenyl)dodecan-3-one

| Conditions | Yield |
|---|---|
| With n-butyllithium; lithium diisopropyl amide In tetrahydrofuran; hexane at -78℃; for 6h; | 95% |
| Stage #1: 4-(4-hydroxy-3-methoxyphenyl)-2-butanone With lithium diisopropyl amide In tetrahydrofuran at -78℃; for 1h; Stage #2: Octanal In tetrahydrofuran at -78℃; for 3h; Time; | 32% |
| With lithium diisopropyl amide In tetrahydrofuran; hexane at -78℃; |
-
-
122-48-5
4-(4-hydroxy-3-methoxyphenyl)-2-butanone

-
-
66-25-1
hexanal

-
-
23513-14-6, 72749-01-0, 74027-33-1, 39886-76-5
(+/-)-[6]-gingerol

| Conditions | Yield |
|---|---|
| With n-butyllithium; lithium diisopropyl amide In tetrahydrofuran; hexane at -78℃; for 6h; | 95% |
| With potassium tert-butylate In tetrahydrofuran at -78℃; for 1.5h; | 76% |
| Stage #1: 4-(4-hydroxy-3-methoxyphenyl)-2-butanone With lithium diisopropyl amide In tetrahydrofuran at -78℃; for 1h; Stage #2: hexanal In tetrahydrofuran at -78℃; for 3h; Time; | 47% |
| With potassium tert-butylate In tetrahydrofuran at -78℃; for 1.5h; Inert atmosphere; | 30.73% |
| With lithium diisopropyl amide In tetrahydrofuran; hexane at -78℃; |
-
-
122-48-5
4-(4-hydroxy-3-methoxyphenyl)-2-butanone

-
-
74-88-4
methyl iodide

-
-
6302-60-9
4-(3,4-dimethoxy-phenyl)-butan-2-one

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetone Reflux; | 95% |
-
-
122-48-5
4-(4-hydroxy-3-methoxyphenyl)-2-butanone

-
-
1080-12-2, 22214-42-2
(E)-4-(4-hydroxy-3-methoxyphenyl)but-3-ene-2-one

| Conditions | Yield |
|---|---|
| With oxygen; palladium diacetate; trifluoroacetic acid In dimethyl sulfoxide at 80℃; for 9h; Sealed tube; | 95% |
-
-
122-48-5
4-(4-hydroxy-3-methoxyphenyl)-2-butanone

-
-
551-93-9
2-aminoacetophenone

-
-
1408335-47-6
4-((2,4-dimethylquinolin-3-yl)methyl)-2-methoxyphenol

| Conditions | Yield |
|---|---|
| With trifluoroacetic acid at 100℃; for 0.5h; | 90% |
-
-
122-48-5
4-(4-hydroxy-3-methoxyphenyl)-2-butanone

| Conditions | Yield |
|---|---|
| With ammonia; hydrogen In tetrahydrofuran at 120℃; for 15h; | 89% |
| With nickel(II) tetrafluoroborate hexahydrate; ammonia; hydrogen; bis(2-diphenylphosphinoethyl)phenylphosphine In 2,2,2-trifluoroethanol at 120℃; for 24h; chemoselective reaction; | 87% |
| With ammonia; hydrogen In methanol at 90℃; under 15001.5 Torr; for 4h; Autoclave; | 99 %Spectr. |
-
-
122-48-5
4-(4-hydroxy-3-methoxyphenyl)-2-butanone

-
-
719-59-5
2-Amino-5-chlorobenzophenone

-
-
1408335-50-1
4-((6-chloro-2-methyl-4-phenylquinolin-3-yl)methyl)-2-methoxyphenol

| Conditions | Yield |
|---|---|
| With trifluoroacetic acid at 100℃; for 0.5h; | 86% |

| Conditions | Yield |
|---|---|
| With triethylamine In dichloromethane at 0 - 20℃; for 13h; | 85% |
-
-
75-77-4
chloro-trimethyl-silane

-
-
122-48-5
4-(4-hydroxy-3-methoxyphenyl)-2-butanone

-
-
56700-87-9
4-(3-methoxy-4-trimethylsilyloxyphenyl)butan-2-one

| Conditions | Yield |
|---|---|
| With 1,1,1,3,3,3-hexamethyl-disilazane; phenol In benzene Heating; | 84% |
-
-
31643-49-9
4-Nitrophthalonitrile

-
-
122-48-5
4-(4-hydroxy-3-methoxyphenyl)-2-butanone

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide for 48h; Inert atmosphere; | 84% |

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at 0℃; for 2h; | 84% |
-
-
122-48-5
4-(4-hydroxy-3-methoxyphenyl)-2-butanone

-
-
66-25-1
hexanal

-
-
23513-13-5, 104186-09-6, 555-66-8
[6]-shogaol

| Conditions | Yield |
|---|---|
| With DIMCARB at 25℃; | 80% |
| With DIMCARB at 20℃; for 72h; | 55% |
| Stage #1: 4-(4-hydroxy-3-methoxyphenyl)-2-butanone With pyrrolidine In dichloromethane at 20℃; for 0.25h; Stage #2: hexanal In dichloromethane for 18h; | 45.52 g |
-
-
122-48-5
4-(4-hydroxy-3-methoxyphenyl)-2-butanone

-
-
104-86-9
4-chlorobenzylamine

-
-
1247013-23-5
4-(3-(4-chlorobenzylamino)butyl)-2-methoxyphenol

| Conditions | Yield |
|---|---|
| Stage #1: 4-(4-hydroxy-3-methoxyphenyl)-2-butanone; 4-chlorobenzylamine In toluene for 16h; Reflux; Inert atmosphere; Stage #2: With methanol; sodium tetrahydroborate In toluene at 0℃; for 16.5h; Reflux; | 75% |
| Stage #1: 4-(4-hydroxy-3-methoxyphenyl)-2-butanone; 4-chlorobenzylamine With sodium tetrahydroborate In toluene at 0℃; for 16h; Inert atmosphere; Dean-Stark; Reflux; Stage #2: With sodium tetrahydroborate In methanol; toluene for 16.5h; Reflux; Inert atmosphere; | 75% |
| Stage #1: 4-(4-hydroxy-3-methoxyphenyl)-2-butanone; 4-chlorobenzylamine In toluene for 16h; Reflux; Inert atmosphere; Dean-Stark; Stage #2: With sodium tetrahydroborate In methanol; toluene at 0℃; for 16.5h; Reflux; | 75% |
-
-
122-48-5
4-(4-hydroxy-3-methoxyphenyl)-2-butanone

-
-
3300-51-4
p-Trifluoromethylbenzylamine

-
-
1247013-24-6
2-methoxy-4-(3-(4-(trifluoromethyl)benzylamino)butyl)phenol

| Conditions | Yield |
|---|---|
| Stage #1: 4-(4-hydroxy-3-methoxyphenyl)-2-butanone; p-Trifluoromethylbenzylamine In toluene for 16h; Reflux; Inert atmosphere; Stage #2: With methanol; sodium tetrahydroborate In toluene at 0℃; for 16.5h; Reflux; | 74% |
| Stage #1: 4-(4-hydroxy-3-methoxyphenyl)-2-butanone; p-Trifluoromethylbenzylamine In toluene at 0℃; for 16h; Inert atmosphere; Reflux; Dean-Stark; Stage #2: With sodium tetrahydroborate In methanol; toluene for 16.5h; Reflux; Inert atmosphere; | 74% |
| Stage #1: 4-(4-hydroxy-3-methoxyphenyl)-2-butanone; p-Trifluoromethylbenzylamine In toluene for 16h; Reflux; Inert atmosphere; Dean-Stark; Stage #2: With sodium tetrahydroborate In methanol; toluene at 0℃; for 16.5h; Reflux; | 74% |
-
-
122-48-5
4-(4-hydroxy-3-methoxyphenyl)-2-butanone

-
-
51762-67-5
3-nitrobenzene-1,2-dicarbonitrile

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide for 48h; Inert atmosphere; | 72% |
-
-
122-48-5
4-(4-hydroxy-3-methoxyphenyl)-2-butanone

-
-
1775-95-7
2-amino-5-nitrobenzophenone

-
-
1408335-49-8
2-methoxy-4-((2-methyl-6-nitro-4-phenylquinolin-3-yl)methyl)phenol

| Conditions | Yield |
|---|---|
| With trifluoroacetic acid at 100℃; for 1.5h; | 70% |
-
-
122-48-5
4-(4-hydroxy-3-methoxyphenyl)-2-butanone

-
-
80638-48-8
3-(3'-methoxy-4'-hydroxyphenyl)propionaldehyde

-
-
79067-88-2, 81569-02-0, 128700-97-0
(E)-1,7-bis(4-hydroxy-3-methoxyphenyl)-4-hepten-3-one

| Conditions | Yield |
|---|---|
| With DIMCARB at 20℃; for 96h; | 70% |
-
-
122-48-5
4-(4-hydroxy-3-methoxyphenyl)-2-butanone

-
-
139152-08-2
4,5-dichlorophthalonitrile

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide for 48h; Inert atmosphere; | 68% |
-
-
122-48-5
4-(4-hydroxy-3-methoxyphenyl)-2-butanone

-
-
1112137-79-7
4,4'-(6,6'-dihydroxy-5,5'-dimethoxy-[1,1'-biphenyl]-3,3'-diyl)bis(butan-2-one)

| Conditions | Yield |
|---|---|
| With methyltri(butyl)ammonium permanganate In dichloromethane at 20℃; | 65% |
| With methyltri(butyl)ammonium permanganate In dichloromethane at 20℃; for 1h; Inert atmosphere; | 65% |
| With MTBAP In dichloromethane at 20℃; for 1h; | |
| With Colletotrichum gloeosporioides In water; dimethyl sulfoxide at 20℃; for 360h; Microbiological reaction; |
-
-
122-48-5
4-(4-hydroxy-3-methoxyphenyl)-2-butanone

-
-
139-85-5
3,4-dihydroxybenzaldehyde

-
-
1443545-73-0
(E)-1-(3,4-dihydroxyphenyl)-5-(4-hydroxy-3-methoxyphenyl)-pent-1-en-3-one

| Conditions | Yield |
|---|---|
| With pyrrolidine; acetic acid In tetrahydrofuran Reflux; | 63% |

| Conditions | Yield |
|---|---|
| With Amberlyst15; air In water at 20℃; for 24h; Irradiation; Green chemistry; | 60% |
| With methanesulfonic acid In acetonitrile at 20℃; for 0.25h; Irradiation; | 52% |

| Conditions | Yield |
|---|---|
| With phosphoric acid In acetonitrile at 20℃; for 12h; Irradiation; | 60% |
Vanillylacetone Chemical Properties
Product Name: Vanillyl acetone (122-48-5)
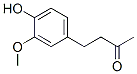
Molecular Formula: C11H14O3
Molecular Weight: 194.23g/mol
Mol File: 122-48-5.mol
Einecs: 204-548-3
Melting Point: 40-41 °C(lit.)
Boiling point: 141 °C0.5 mm Hg(lit.)
Flash Point: >230 °F
Density: 1.14 g/mL at 25 °C(lit.)
Refractive index: n20/D 1.541(lit.)
Product Categories: Miscellaneous Natural Products
Synonyms of Vanillyl acetone (122-48-5): (0)-paradol ; [0]-Paradol ; 3-Methoxy-4-hydroxybenzylacetone ; 3-methoxy-4-hydroxy-benzylacetone ; 4-(3-methoxy-4-hydroxyphenyl)-2-butanone ; 4-(4-hydroxy-3 ; 4-(4-hydroxy-3-methoxyphenyl)-2-butanon ; Gingerone .
Vanillylacetone Toxicity Data With Reference
Moderately toxic by ingestion. A skin irritant. A flammable liquid. When heated to decomposition it emits acrid smoke and irritating fumes.
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| rabbit | LD50 | skin | > 5gm/kg (5000mg/kg) | Food and Chemical Toxicology. Vol. 20, Pg. 851, 1982. | |
| rat | LD50 | oral | 2580mg/kg (2580mg/kg) | Food and Chemical Toxicology. Vol. 20, Pg. 851, 1982. |
Vanillylacetone Consensus Reports
Reported in EPA TSCA Inventory.
Vanillylacetone Safety Profile
Safety Information of Vanillyl acetone (122-48-5):
Risk Statements: 36/37/38
36: Irritating to the eyes
37: Irritating to the respiratory system
38: Irritating to the skin
Safety Statements: 26-36
26: In case of contact with eyes, rinse immediately with plenty of water and seek medical advice
36: Wear suitable protective clothing
Vanillylacetone Standards and Recommendations
DOT Classification: 3; Label: Flammable Liquid
Related Products
- Vanillylacetone
- 1225-01-0
- 1225053-32-6
- 122509-72-2
- 122-51-0
- 12251-32-0
- 1225199-26-7
- 122520-12-1
- 122520-86-9
- 122520-90-5
- 122-52-1
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View