HANGZHOU YUNUO CHEMICAL CO.,LTD
Hangzhou Yunuo Chemical Co., Ltd is located in Hangzhou city China. As a global supplier in the chemical industry, Yunuochem is to create added value for customers around the world. Sticking to such business idea of "Good faith wins the busine
Hangzhou Hysen Pharma co.,Ltd.
We can provide high quality,cheap price,spot supply and good service. If you have any questions,pls do not hesitate to contact us. Hangzhou Hysen pharma CO.,LTD. is a professional Pharmaceutical company. We are specialized in manufacturing and
Cas:116649-85-5
Min.Order:10 Gram
Negotiable
Type:Lab/Research institutions
inquiryDayang Chem (Hangzhou) Co.,Ltd.
As a leading manufacturer and supplier of chemicals in China, DayangChem not only supply popular chemicals, but also DayangChem's R&D center offer custom synthesis services. DayangChem can provide different quantities of custom synthesis ch
Cas:116649-85-5
Min.Order:1 Kilogram
FOB Price: $3.0
Type:Lab/Research institutions
inquiryHangzhou Dingyan Chem Co., Ltd
Items Standard Result Assay (Ursolic acid) 98%min 98.22% ----------------------------------------------------------------
Cas:116649-85-5
Min.Order:1 Gram
FOB Price: $100.0 / 500.0
Type:Trading Company
inquirySimagchem Corporation
Welcome to Simagchem, your partner in China as a premier supply of bulk specialty chemicals for industry and life science. We introduce experienced quality product and exceptional JIT service with instant market intelligence in China to benefit our
Cas:116649-85-5
Min.Order:0 Metric Ton
Negotiable
Type:Manufacturers
inquiryHangzhou JINLAN Pharm-Drugs Technology Co., Ltd
Jinlan Pharm-Drugs Technology Co.,Limited (with its export company Hangzhou Royall Import & Export Co.,Ltd.)is located in Hangzhou, Zhejiang Province. Neighboring Ningbo port, Shanghai port, Hangzhou Xiaoshan Int’l Airport and Shangha
Chemwill Asia Co., Ltd.
Our main production base is located in Xuzhou industry park. We are certified both to the ISO 9001 and ISO 14001 Standards, have a safety management system in place.Our R&D team masters core technology for process-design of target building block
Hebei Nengqian Chemical Import and Export Co., LTD
Our advantages: 1, High quality with competitive price: 1) Standard:BP/USP/EP/Enterprise standard 2) All Purity≥99% 3) We are manufacturer and can provide high quality products with factory price. 2, Fast and safe delivery 1) Parcel can be
Cas:116649-85-5
Min.Order:1 Kilogram
FOB Price: $48.0 / 58.0
Type:Trading Company
inquiryZhuozhou Wenxi import and Export Co., Ltd
WITH US,YOUR MONEY IN SAFE,YOUR BUSINESS IN SAFE 1)Quick Response Within 12 hours; 2)Quality Guarantee: All products are strictly tested by our QC, confirmed by QA and approved by third party lab in China, USA, Canada, Germany, UK, Italy, France et
Cas:116649-85-5
Min.Order:1 Kilogram
FOB Price: $139.0 / 210.0
Type:Trading Company
inquiryShanghai Upbio Tech Co.,Ltd
1.No Less 8 years exporting experience. Clients can 100% received goods 2.Lower Price with higher quality 3,Free sample 4,We are sincerely responsible for the "product quality" and "After Service" Upbio is Specialized
Cas:116649-85-5
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryLeader Biochemical Group
About Product Details Items Specifications Test Results Appearance White to white crystalline powde
Cas:116649-85-5
Min.Order:1 Gram
FOB Price: $1.0 / 2.0
Type:Lab/Research institutions
inquiryBaoji Guokang Healthchem co.,ltd
Our company has been in existence for 10 years since its establishment. We have our own unique team. The company integrates independent research and development, production and sales. We have established famous brands at home and abroad. At present,
Qingdao Beluga Import and Export Co., LTD
Ramatroban CAS:116649-85-5 Qingdao Belugas Import and Export Co., Ltd. is a scientific and technological company integrating research and development, production and trade of chemical intermediates, specializing in high quality organic intermediates
Cas:116649-85-5
Min.Order:1 Gram
Negotiable
Type:Lab/Research institutions
inquiryShandong Hanjiang Chemical Co., Ltd.
Hello, dear friend! I'm Hansen and Allen from China. Welcome to my lookchem mall! The following is a brief introduction of our company's products and services. If you are interested in our products, please contact us by emai
Wuhan Wonda Pharm Limited
1.high quality: quality is life. quality is the most important element for all goods. we have a lab doing research in wuhan china. hplc and nmr is available if needed. 2.reasonable price: we provide high quality products wi
Cas:116649-85-5
Min.Order:10 Gram
Negotiable
Type:Lab/Research institutions
inquiryHenan Wentao Chemical Product Co., Ltd.
Henan Wentao Chemical Product Co.,Ltd is Located in Zhengzhou High-tech Development Zone with import and export license, We passed ISO 9001:2008 as well, Henan Wentao has developed more than 1000 compounds, which are widely used in the fields of prod
Taizhou Crene Biotechnology co.ltd
Our company provides one-stop services of research - development - production for a variety of special prouducts. Not only do we make effective use of our strong technological strength, but also establish of cooperative relations with several well-
Cas:116649-85-5
Min.Order:100 Milligram
Negotiable
Type:Lab/Research institutions
inquiryHangzhou J&H Chemical Co., Ltd.
J&H CHEM R&D center can offer custom synthesis according to the contract research and development services for the fine chemicals, pharmaceutical, biotechnique and some of the other chemicals. J&H CHEM has some Manufacturing base in Jia
Afine Chemicals Limited
Our Services 1. New Molecules R&D 2. Own test center HPLC NMR GC LC-MS 3. API and Intermediates from China reputed manufacturers 4. Documents support COA MOA MSDS DMF open part Our advantages 1. Government awarded company. Top 100 enter
Hefei Zhaobo Technology Co., Ltd.
Our Advantages Production: Advanced chemical equipment with years of experience.Staffs for producing various extract products. Quality Control:A complete set of Testing Professional and Analysis Equipment ensures the Quality Requirements and Speci
Cas:116649-85-5
Min.Order:1 Kilogram
FOB Price: $150.0 / 180.0
Type:Trading Company
inquiryZibo Hangyu Biotechnology Development Co., Ltd
Zibo Hangyu Biotechnology Development Co., Ltd is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality pharmaceuticals, intermediates, special chemicals and OLED intermediates and other fine chemi
Cas:116649-85-5
Min.Order:10 Gram
FOB Price: $100.0
Type:Lab/Research institutions
inquiryAlity Chemical Corporation
The above product is Ality Chemical's strong item with best price, good quality and fast supply. Ality Chemical has been focusing on the research and production of this field for over 14 years. At the same time, we are always committed to providing h
Jiangsu Qianyu Molecular Technology Co., LTD.
Our Advantages A. International Top level TechnologyOur company owned biomedicine experts are famous at home and abroad with rich experience in research and development in the field of efficient chiral functional molecules research and development an
Xiamen Jenny Chemical Technology Co., Ltd.
GMP standard, high purity, competitive price, in stock 1. Quick Response: within 6 hours after receiving your email. 2. Quality Guarantee: All products are strictly tested by our QC, confirmed by QA, and approved by a third-party lab in China, USA,
Shandong Mopai Biotechnology Co., LTD
Shandong Mopai Biotechnology Co., LTD is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality pharmaceuticals, intermediates, special chemicals and OLED intermediates and other fine chemicals. W
Suzhou Health Chemicals Co., Ltd.
High quality,stable supply chain.Appearance:white/off-white or light yellow Storage:Store in cool and dry place, keep away from strong light and heat. Package:aluminum bottle,glass bottle,PTFE bottle,cardboard drum Application:Active Pharmaceutical I
Golden Pharma Co., Limited
GOLDEN PHARMA CO.,LIMITED.is a professional pharmaceutical company,our team have more than 20years expereince in pharmaceutical production and sales. we are a professional technical enterprise specializing in the R & D, production,QA regulation
Win-Win chemical Co.Ltd
Stock products, own laboratory Package:Grams, Kilograms Application:For R&D Transportation:According to customer request Port:Shanghai
Cas:116649-85-5
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquirySenova Technology Company Limited
Good price, high purity, mature production technology, accept custom synthesis per customer's requirements Package:As per Customer‘ requirements Application:Pharmaceutical intermediate
Hubei Vanz Pharm Co.,Ltd
ISO/factory/goodqualityAppearance:off white Storage:Dry,cool place Package:drum Application:active pharmaceutical ingredients Transportation:by air/sea/express Port:shenzhen/shanghai
Synthetic route
-
-
118699-38-0
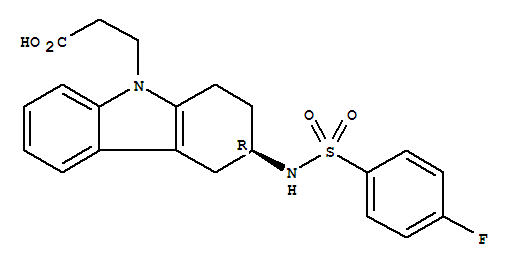
(R)-N-[9-(2-cyanoethyl)-2,3,4,9-tetrahydro-1H-carbazol-3-yl]-4-fluorobenzenesulfonamide

-
-
116649-85-5
ramatroban

| Conditions | Yield |
|---|---|
| Stage #1: (R)-N-[9-(2-cyanoethyl)-2,3,4,9-tetrahydro-1H-carbazol-3-yl]-4-fluorobenzenesulfonamide With water; sodium hydroxide In isopropyl alcohol at 100℃; for 24h; Stage #2: With hydrogenchloride In water | 79% |
-
-
107-13-1
acrylonitrile

-
-
116650-36-3
(R)-4-fluoro-N-(2,3,4,9-tetrahydro-1H-carbazol-3-yl)benzenesulfonamide

-
-
116649-85-5
ramatroban

| Conditions | Yield |
|---|---|
| With potassium hydroxide; sodium hydride 1.) dimethylformamide, 2 h, room temperature, 2.) isopropanol, reflux, 16 h; Yield given. Multistep reaction; |
-
-
51145-61-0
1,2,3,4-tetrahydrocarbazol-3-one

-
-
116649-85-5
ramatroban

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: 2.) tetrabutylammonium borohydride, 3.) 2N sulfuric acid / 1.) benzene, reflux, 1 h, 2.) CH2Cl2, -50 deg C to room temperature, 3.) methanol, room temperature, 1 h 2: 1.) 2N NaOH, conc. HCl, 2.) ammonium formiate / 2.) 10percent Pd/C / 1.) methanol, ethyl acetate, 2.) dimethylformamide, reflux, 20 min 3: 84 percent / triethylamine / CH2Cl2 / 1 h / Ambient temperature 4: 1.) 80percent sodium hydride (in mineral oil), 2.) 10percent potassium hydroxide / 1.) dimethylformamide, 2 h, room temperature, 2.) isopropanol, reflux, 16 h View Scheme | |
| Multi-step reaction with 6 steps 1.1: D-glucose / water / 24 h / 30 °C / Enzymatic reaction 2.1: triphenylphosphine; diethylazodicarboxylate / tetrahydrofuran / 0.5 h / -20 °C / Inert atmosphere 2.2: 10 h / -25 - -15 °C 3.1: water; triphenylphosphine / 15 h / 65 °C 4.1: triethylamine / dichloromethane / 1 h / 20 °C 5.1: sodium hydride / N,N-dimethyl-formamide / 0.08 h / 20 °C 5.2: 1 h / 20 °C 6.1: water; sodium hydroxide / isopropyl alcohol / 24 h / 100 °C View Scheme | |
| Multi-step reaction with 7 steps 1.1: sodium tetrahydroborate / methanol / 1.5 h / 0 - 20 °C / Inert atmosphere 2.1: Candida antarctica Lipase A / tetrahydrofuran / 5 h / 30 °C / Enzymatic reaction 3.1: triphenylphosphine; diethylazodicarboxylate / tetrahydrofuran / 0.5 h / -20 °C / Inert atmosphere 3.2: 10 h / -25 - -15 °C 4.1: water; triphenylphosphine / 15 h / 65 °C 5.1: triethylamine / dichloromethane / 1 h / 20 °C 6.1: sodium hydride / N,N-dimethyl-formamide / 0.08 h / 20 °C 6.2: 1 h / 20 °C 7.1: water; sodium hydroxide / isopropyl alcohol / 24 h / 100 °C View Scheme |

-
-
116649-85-5
ramatroban

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: 1.) 2N NaOH, conc. HCl, 2.) ammonium formiate / 2.) 10percent Pd/C / 1.) methanol, ethyl acetate, 2.) dimethylformamide, reflux, 20 min 2: 84 percent / triethylamine / CH2Cl2 / 1 h / Ambient temperature 3: 1.) 80percent sodium hydride (in mineral oil), 2.) 10percent potassium hydroxide / 1.) dimethylformamide, 2 h, room temperature, 2.) isopropanol, reflux, 16 h View Scheme |
-
-
116650-33-0
(3R)-2,3,4,9-tetrahydro-1H-carbazol-3-amine

-
-
116649-85-5
ramatroban

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 84 percent / triethylamine / CH2Cl2 / 1 h / Ambient temperature 2: 1.) 80percent sodium hydride (in mineral oil), 2.) 10percent potassium hydroxide / 1.) dimethylformamide, 2 h, room temperature, 2.) isopropanol, reflux, 16 h View Scheme | |
| Multi-step reaction with 3 steps 1.1: triethylamine / dichloromethane / 1 h / 20 °C 2.1: sodium hydride / N,N-dimethyl-formamide / 0.08 h / 20 °C 2.2: 1 h / 20 °C 3.1: water; sodium hydroxide / isopropyl alcohol / 24 h / 100 °C View Scheme |
-
-
4746-97-8
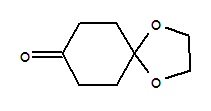
cyclohexanedione monoethylene ketal

-
-
116649-85-5
ramatroban

| Conditions | Yield |
|---|---|
| Multi-step reaction with 10 steps 1.1: magnesium sulfate / dichloromethane / 2 h / 20 °C 1.2: 4 h / 110 °C 2.1: trifluoroacetic acid / acetone / 24 h / 65 °C 3.1: sodium tetrahydroborate / methanol / 1.5 h / 0 - 20 °C / Inert atmosphere 4.1: Candida antarctica Lipase A / tetrahydrofuran / 5 h / 30 °C / Enzymatic reaction 5.1: dmap; triethylamine / dichloromethane / 2 h / 0 - 20 °C / Inert atmosphere 6.1: sodium azide / N,N-dimethyl-formamide / 24 h / 70 °C 7.1: water; triphenylphosphine / 15 h / 65 °C 8.1: triethylamine / dichloromethane / 1 h / 20 °C 9.1: sodium hydride / N,N-dimethyl-formamide / 0.08 h / 20 °C 9.2: 1 h / 20 °C 10.1: water; sodium hydroxide / isopropyl alcohol / 24 h / 100 °C View Scheme | |
| Multi-step reaction with 10 steps 1.1: magnesium sulfate / dichloromethane / 2 h / 20 °C 1.2: 4 h / 110 °C 2.1: trifluoroacetic acid / acetone / 24 h / 65 °C 3.1: sodium tetrahydroborate / methanol / 1.5 h / 0 - 20 °C / Inert atmosphere 4.1: Pseudomonas cepacia lipase / tetrahydrofuran / 8 h / 30 °C / Enzymatic reaction 5.1: dmap; triethylamine / dichloromethane / 2 h / 0 - 20 °C / Inert atmosphere 6.1: sodium azide / N,N-dimethyl-formamide / 24 h / 70 °C 7.1: water; triphenylphosphine / 15 h / 65 °C 8.1: triethylamine / dichloromethane / 1 h / 20 °C 9.1: sodium hydride / N,N-dimethyl-formamide / 0.08 h / 20 °C 9.2: 1 h / 20 °C 10.1: water; sodium hydroxide / isopropyl alcohol / 24 h / 100 °C View Scheme | |
| Multi-step reaction with 10 steps 1.1: magnesium sulfate / dichloromethane / 2 h / 20 °C 1.2: 4 h / 110 °C 2.1: trifluoroacetic acid / acetone / 24 h / 65 °C 3.1: sodium tetrahydroborate / methanol / 1.5 h / 0 - 20 °C / Inert atmosphere 4.1: Candida antarctica lipase B / tetrahydrofuran / 8 h / 30 °C / Enzymatic reaction 5.1: dmap; triethylamine / dichloromethane / 2 h / 0 - 20 °C / Inert atmosphere 6.1: sodium azide / N,N-dimethyl-formamide / 24 h / 70 °C 7.1: water; triphenylphosphine / 15 h / 65 °C 8.1: triethylamine / dichloromethane / 1 h / 20 °C 9.1: sodium hydride / N,N-dimethyl-formamide / 0.08 h / 20 °C 9.2: 1 h / 20 °C 10.1: water; sodium hydroxide / isopropyl alcohol / 24 h / 100 °C View Scheme |
-
-
1374648-17-5
(S)-2,3,4,9-tetrahydro-1H-carbazol-3-yl methanesulfonate

-
-
116649-85-5
ramatroban

| Conditions | Yield |
|---|---|
| Multi-step reaction with 5 steps 1.1: sodium azide / dimethyl sulfoxide / 24 h / 20 °C 2.1: water; triphenylphosphine / 15 h / 65 °C 3.1: triethylamine / dichloromethane / 1 h / 20 °C 4.1: sodium hydride / N,N-dimethyl-formamide / 0.08 h / 20 °C 4.2: 1 h / 20 °C 5.1: water; sodium hydroxide / isopropyl alcohol / 24 h / 100 °C View Scheme | |
| Multi-step reaction with 5 steps 1.1: sodium azide / N,N-dimethyl-formamide / 24 h / 70 °C 2.1: water; triphenylphosphine / 15 h / 65 °C 3.1: triethylamine / dichloromethane / 1 h / 20 °C 4.1: sodium hydride / N,N-dimethyl-formamide / 0.08 h / 20 °C 4.2: 1 h / 20 °C 5.1: water; sodium hydroxide / isopropyl alcohol / 24 h / 100 °C View Scheme |
-
-
1374648-18-6
(R)-3-azido-2,3,4,9-tetrahydro-1H-carbazole

-
-
116649-85-5
ramatroban

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1.1: water; triphenylphosphine / 15 h / 65 °C 2.1: triethylamine / dichloromethane / 1 h / 20 °C 3.1: sodium hydride / N,N-dimethyl-formamide / 0.08 h / 20 °C 3.2: 1 h / 20 °C 4.1: water; sodium hydroxide / isopropyl alcohol / 24 h / 100 °C View Scheme |
-
-
116650-12-5
4-fluoro-N-(2,3,4,9-tetrahydro-1H-carbazol-3-yl)benzenesulfonamide

-
-
116649-85-5
ramatroban

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1.1: sodium hydride / N,N-dimethyl-formamide / 20 °C 1.2: 20 °C 2.1: chiralpak IC / hexane; isopropyl alcohol / 40 °C / Resolution of racemate 3.1: water; sodium hydroxide / isopropyl alcohol / 24 h / 100 °C View Scheme | |
| Multi-step reaction with 3 steps 1.1: chiralpak IC / hexane; isopropyl alcohol / 30 °C / Resolution of racemate 2.1: sodium hydride / N,N-dimethyl-formamide / 0.08 h / 20 °C 2.2: 1 h / 20 °C 3.1: water; sodium hydroxide / isopropyl alcohol / 24 h / 100 °C View Scheme |

-
-
116649-85-5
ramatroban

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: chiralpak IC / hexane; isopropyl alcohol / 40 °C / Resolution of racemate 2: water; sodium hydroxide / isopropyl alcohol / 24 h / 100 °C View Scheme |

-
-
116649-85-5
ramatroban

| Conditions | Yield |
|---|---|
| Multi-step reaction with 6 steps 1.1: sodium azide / 24 h 2.1: chiralcel OD / hexane; isopropyl alcohol / 30 °C / Resolution of racemate 3.1: water; triphenylphosphine / 15 h / 65 °C 4.1: triethylamine / dichloromethane / 1 h / 20 °C 5.1: sodium hydride / N,N-dimethyl-formamide / 0.08 h / 20 °C 5.2: 1 h / 20 °C 6.1: water; sodium hydroxide / isopropyl alcohol / 24 h / 100 °C View Scheme | |
| Multi-step reaction with 6 steps 1.1: sodium azide / 24 h 2.1: palladium on activated charcoal; hydrogen / methanol / 2 h / 20 °C 3.1: triethylamine / dichloromethane / 20 °C 4.1: sodium hydride / N,N-dimethyl-formamide / 20 °C 4.2: 20 °C 5.1: chiralpak IC / hexane; isopropyl alcohol / 40 °C / Resolution of racemate 6.1: water; sodium hydroxide / isopropyl alcohol / 24 h / 100 °C View Scheme | |
| Multi-step reaction with 6 steps 1.1: sodium azide / 24 h 2.1: palladium on activated charcoal; hydrogen / methanol / 2 h / 20 °C 3.1: triethylamine / dichloromethane / 20 °C 4.1: chiralpak IC / hexane; isopropyl alcohol / 30 °C / Resolution of racemate 5.1: sodium hydride / N,N-dimethyl-formamide / 0.08 h / 20 °C 5.2: 1 h / 20 °C 6.1: water; sodium hydroxide / isopropyl alcohol / 24 h / 100 °C View Scheme | |
| Multi-step reaction with 6 steps 1.1: chiralcel OD / hexane; isopropyl alcohol / 40 °C / Resolution of racemate 2.1: sodium azide / dimethyl sulfoxide / 24 h / 20 °C 3.1: water; triphenylphosphine / 15 h / 65 °C 4.1: triethylamine / dichloromethane / 1 h / 20 °C 5.1: sodium hydride / N,N-dimethyl-formamide / 0.08 h / 20 °C 5.2: 1 h / 20 °C 6.1: water; sodium hydroxide / isopropyl alcohol / 24 h / 100 °C View Scheme | |
| Multi-step reaction with 6 steps 1.1: chiralcel OD / hexane; isopropyl alcohol / 40 °C / Resolution of racemate 2.1: sodium azide / N,N-dimethyl-formamide / 24 h / 70 °C 3.1: water; triphenylphosphine / 15 h / 65 °C 4.1: triethylamine / dichloromethane / 1 h / 20 °C 5.1: sodium hydride / N,N-dimethyl-formamide / 0.08 h / 20 °C 5.2: 1 h / 20 °C 6.1: water; sodium hydroxide / isopropyl alcohol / 24 h / 100 °C View Scheme |
-
-
1374648-14-2
(S)-(-)-2,3,4,9-tetrahydro-1H-carbazol-3-ol

-
-
116649-85-5
ramatroban

| Conditions | Yield |
|---|---|
| Multi-step reaction with 5 steps 1.1: triphenylphosphine; diethylazodicarboxylate / tetrahydrofuran / 0.5 h / -20 °C / Inert atmosphere 1.2: 10 h / -25 - -15 °C 2.1: water; triphenylphosphine / 15 h / 65 °C 3.1: triethylamine / dichloromethane / 1 h / 20 °C 4.1: sodium hydride / N,N-dimethyl-formamide / 0.08 h / 20 °C 4.2: 1 h / 20 °C 5.1: water; sodium hydroxide / isopropyl alcohol / 24 h / 100 °C View Scheme | |
| Multi-step reaction with 6 steps 1.1: dmap; triethylamine / dichloromethane / 2 h / 0 - 20 °C / Inert atmosphere 2.1: sodium azide / N,N-dimethyl-formamide / 24 h / 70 °C 3.1: water; triphenylphosphine / 15 h / 65 °C 4.1: triethylamine / dichloromethane / 1 h / 20 °C 5.1: sodium hydride / N,N-dimethyl-formamide / 0.08 h / 20 °C 5.2: 1 h / 20 °C 6.1: water; sodium hydroxide / isopropyl alcohol / 24 h / 100 °C View Scheme | |
| Multi-step reaction with 6 steps 1.1: dmap; triethylamine / dichloromethane / 2 h / 0 - 20 °C / Inert atmosphere 2.1: sodium azide / dimethyl sulfoxide / 24 h / 20 °C 3.1: water; triphenylphosphine / 15 h / 65 °C 4.1: triethylamine / dichloromethane / 1 h / 20 °C 5.1: sodium hydride / N,N-dimethyl-formamide / 0.08 h / 20 °C 5.2: 1 h / 20 °C 6.1: water; sodium hydroxide / isopropyl alcohol / 24 h / 100 °C View Scheme |
-
-
128432-38-2, 14384-34-0
(+/-)-2,3,4,9-tetrahydro-1H-carbazol-3-ol

-
-
116649-85-5
ramatroban

| Conditions | Yield |
|---|---|
| Multi-step reaction with 6 steps 1.1: Candida antarctica Lipase A / tetrahydrofuran / 5 h / 30 °C / Enzymatic reaction 2.1: triphenylphosphine; diethylazodicarboxylate / tetrahydrofuran / 0.5 h / -20 °C / Inert atmosphere 2.2: 10 h / -25 - -15 °C 3.1: water; triphenylphosphine / 15 h / 65 °C 4.1: triethylamine / dichloromethane / 1 h / 20 °C 5.1: sodium hydride / N,N-dimethyl-formamide / 0.08 h / 20 °C 5.2: 1 h / 20 °C 6.1: water; sodium hydroxide / isopropyl alcohol / 24 h / 100 °C View Scheme | |
| Multi-step reaction with 6 steps 1.1: Candida antarctica lipase B / tetrahydrofuran / 8 h / 30 °C / Enzymatic reaction 2.1: triphenylphosphine; diethylazodicarboxylate / tetrahydrofuran / 0.5 h / -20 °C / Inert atmosphere 2.2: 10 h / -25 - -15 °C 3.1: water; triphenylphosphine / 15 h / 65 °C 4.1: triethylamine / dichloromethane / 1 h / 20 °C 5.1: sodium hydride / N,N-dimethyl-formamide / 0.08 h / 20 °C 5.2: 1 h / 20 °C 6.1: water; sodium hydroxide / isopropyl alcohol / 24 h / 100 °C View Scheme | |
| Multi-step reaction with 6 steps 1.1: Pseudomonas cepacia lipase / tetrahydrofuran / 8 h / 30 °C / Enzymatic reaction 2.1: triphenylphosphine; diethylazodicarboxylate / tetrahydrofuran / 0.5 h / -20 °C / Inert atmosphere 2.2: 10 h / -25 - -15 °C 3.1: water; triphenylphosphine / 15 h / 65 °C 4.1: triethylamine / dichloromethane / 1 h / 20 °C 5.1: sodium hydride / N,N-dimethyl-formamide / 0.08 h / 20 °C 5.2: 1 h / 20 °C 6.1: water; sodium hydroxide / isopropyl alcohol / 24 h / 100 °C View Scheme |
-
-
349-88-2
4-Fluorobenzenesulfonyl chloride

-
-
116649-85-5
ramatroban

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1.1: triethylamine / dichloromethane / 1 h / 20 °C 2.1: sodium hydride / N,N-dimethyl-formamide / 0.08 h / 20 °C 2.2: 1 h / 20 °C 3.1: water; sodium hydroxide / isopropyl alcohol / 24 h / 100 °C View Scheme |

-
-
116649-85-5
ramatroban

| Conditions | Yield |
|---|---|
| Multi-step reaction with 5 steps 1.1: palladium on activated charcoal; hydrogen / methanol / 2 h / 20 °C 2.1: triethylamine / dichloromethane / 20 °C 3.1: sodium hydride / N,N-dimethyl-formamide / 20 °C 3.2: 20 °C 4.1: chiralpak IC / hexane; isopropyl alcohol / 40 °C / Resolution of racemate 5.1: water; sodium hydroxide / isopropyl alcohol / 24 h / 100 °C View Scheme | |
| Multi-step reaction with 5 steps 1.1: palladium on activated charcoal; hydrogen / methanol / 2 h / 20 °C 2.1: triethylamine / dichloromethane / 20 °C 3.1: chiralpak IC / hexane; isopropyl alcohol / 30 °C / Resolution of racemate 4.1: sodium hydride / N,N-dimethyl-formamide / 0.08 h / 20 °C 4.2: 1 h / 20 °C 5.1: water; sodium hydroxide / isopropyl alcohol / 24 h / 100 °C View Scheme | |
| Multi-step reaction with 5 steps 1.1: chiralcel OD / hexane; isopropyl alcohol / 30 °C / Resolution of racemate 2.1: water; triphenylphosphine / 15 h / 65 °C 3.1: triethylamine / dichloromethane / 1 h / 20 °C 4.1: sodium hydride / N,N-dimethyl-formamide / 0.08 h / 20 °C 4.2: 1 h / 20 °C 5.1: water; sodium hydroxide / isopropyl alcohol / 24 h / 100 °C View Scheme |
-
-
61894-99-3, 116650-33-0, 116650-34-1, 134525-54-5
2,3,4,9-tetrahydro-1H-carbazol-3-yl-amine

-
-
116649-85-5
ramatroban

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1.1: triethylamine / dichloromethane / 20 °C 2.1: sodium hydride / N,N-dimethyl-formamide / 20 °C 2.2: 20 °C 3.1: chiralpak IC / hexane; isopropyl alcohol / 40 °C / Resolution of racemate 4.1: water; sodium hydroxide / isopropyl alcohol / 24 h / 100 °C View Scheme | |
| Multi-step reaction with 4 steps 1.1: triethylamine / dichloromethane / 20 °C 2.1: chiralpak IC / hexane; isopropyl alcohol / 30 °C / Resolution of racemate 3.1: sodium hydride / N,N-dimethyl-formamide / 0.08 h / 20 °C 3.2: 1 h / 20 °C 4.1: water; sodium hydroxide / isopropyl alcohol / 24 h / 100 °C View Scheme |
-
-
100-63-0
phenylhydrazine

-
-
116649-85-5
ramatroban

| Conditions | Yield |
|---|---|
| Multi-step reaction with 10 steps 1.1: magnesium sulfate / dichloromethane / 2 h / 20 °C 1.2: 4 h / 110 °C 2.1: trifluoroacetic acid / acetone / 24 h / 65 °C 3.1: sodium tetrahydroborate / methanol / 1.5 h / 0 - 20 °C / Inert atmosphere 4.1: Candida antarctica Lipase A / tetrahydrofuran / 5 h / 30 °C / Enzymatic reaction 5.1: dmap; triethylamine / dichloromethane / 2 h / 0 - 20 °C / Inert atmosphere 6.1: sodium azide / N,N-dimethyl-formamide / 24 h / 70 °C 7.1: water; triphenylphosphine / 15 h / 65 °C 8.1: triethylamine / dichloromethane / 1 h / 20 °C 9.1: sodium hydride / N,N-dimethyl-formamide / 0.08 h / 20 °C 9.2: 1 h / 20 °C 10.1: water; sodium hydroxide / isopropyl alcohol / 24 h / 100 °C View Scheme | |
| Multi-step reaction with 10 steps 1.1: magnesium sulfate / dichloromethane / 2 h / 20 °C 1.2: 4 h / 110 °C 2.1: trifluoroacetic acid / acetone / 24 h / 65 °C 3.1: sodium tetrahydroborate / methanol / 1.5 h / 0 - 20 °C / Inert atmosphere 4.1: Pseudomonas cepacia lipase / tetrahydrofuran / 8 h / 30 °C / Enzymatic reaction 5.1: dmap; triethylamine / dichloromethane / 2 h / 0 - 20 °C / Inert atmosphere 6.1: sodium azide / N,N-dimethyl-formamide / 24 h / 70 °C 7.1: water; triphenylphosphine / 15 h / 65 °C 8.1: triethylamine / dichloromethane / 1 h / 20 °C 9.1: sodium hydride / N,N-dimethyl-formamide / 0.08 h / 20 °C 9.2: 1 h / 20 °C 10.1: water; sodium hydroxide / isopropyl alcohol / 24 h / 100 °C View Scheme | |
| Multi-step reaction with 10 steps 1.1: magnesium sulfate / dichloromethane / 2 h / 20 °C 1.2: 4 h / 110 °C 2.1: trifluoroacetic acid / acetone / 24 h / 65 °C 3.1: sodium tetrahydroborate / methanol / 1.5 h / 0 - 20 °C / Inert atmosphere 4.1: Candida antarctica lipase B / tetrahydrofuran / 8 h / 30 °C / Enzymatic reaction 5.1: dmap; triethylamine / dichloromethane / 2 h / 0 - 20 °C / Inert atmosphere 6.1: sodium azide / N,N-dimethyl-formamide / 24 h / 70 °C 7.1: water; triphenylphosphine / 15 h / 65 °C 8.1: triethylamine / dichloromethane / 1 h / 20 °C 9.1: sodium hydride / N,N-dimethyl-formamide / 0.08 h / 20 °C 9.2: 1 h / 20 °C 10.1: water; sodium hydroxide / isopropyl alcohol / 24 h / 100 °C View Scheme |
-
-
116650-36-3
(R)-4-fluoro-N-(2,3,4,9-tetrahydro-1H-carbazol-3-yl)benzenesulfonamide

-
-
116649-85-5
ramatroban

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1.1: sodium hydride / N,N-dimethyl-formamide / 0.08 h / 20 °C 1.2: 1 h / 20 °C 2.1: water; sodium hydroxide / isopropyl alcohol / 24 h / 100 °C View Scheme |
-
-
54621-12-4
1,2,4,9-tetrahydrospiro[3H-carbazole-3,2*-[1,3]doxolane]

-
-
116649-85-5
ramatroban

| Conditions | Yield |
|---|---|
| Multi-step reaction with 7 steps 1.1: trifluoroacetic acid / acetone / 24 h / 65 °C 2.1: D-glucose / water / 24 h / 30 °C / Enzymatic reaction 3.1: triphenylphosphine; diethylazodicarboxylate / tetrahydrofuran / 0.5 h / -20 °C / Inert atmosphere 3.2: 10 h / -25 - -15 °C 4.1: water; triphenylphosphine / 15 h / 65 °C 5.1: triethylamine / dichloromethane / 1 h / 20 °C 6.1: sodium hydride / N,N-dimethyl-formamide / 0.08 h / 20 °C 6.2: 1 h / 20 °C 7.1: water; sodium hydroxide / isopropyl alcohol / 24 h / 100 °C View Scheme | |
| Multi-step reaction with 8 steps 1.1: trifluoroacetic acid / acetone / 24 h / 65 °C 2.1: D-glucose / water / 24 h / 30 °C / Enzymatic reaction 3.1: dmap; triethylamine / dichloromethane / 2 h / 0 - 20 °C / Inert atmosphere 4.1: sodium azide / N,N-dimethyl-formamide / 24 h / 70 °C 5.1: water; triphenylphosphine / 15 h / 65 °C 6.1: triethylamine / dichloromethane / 1 h / 20 °C 7.1: sodium hydride / N,N-dimethyl-formamide / 0.08 h / 20 °C 7.2: 1 h / 20 °C 8.1: water; sodium hydroxide / isopropyl alcohol / 24 h / 100 °C View Scheme | |
| Multi-step reaction with 8 steps 1.1: trifluoroacetic acid / acetone / 24 h / 65 °C 2.1: sodium tetrahydroborate / methanol / 1.5 h / 0 - 20 °C / Inert atmosphere 3.1: Candida antarctica Lipase A / tetrahydrofuran / 5 h / 30 °C / Enzymatic reaction 4.1: triphenylphosphine; diethylazodicarboxylate / tetrahydrofuran / 0.5 h / -20 °C / Inert atmosphere 4.2: 10 h / -25 - -15 °C 5.1: water; triphenylphosphine / 15 h / 65 °C 6.1: triethylamine / dichloromethane / 1 h / 20 °C 7.1: sodium hydride / N,N-dimethyl-formamide / 0.08 h / 20 °C 7.2: 1 h / 20 °C 8.1: water; sodium hydroxide / isopropyl alcohol / 24 h / 100 °C View Scheme |
-
-
67-56-1
methanol

-
-
116649-85-5
ramatroban

-
-
141307-19-9
(R)-methyl 3-[3-(4-fluorophenylsulfonamido)-3,4-dihydro-1H-carbazol-9(2H)-yl]propanoate

| Conditions | Yield |
|---|---|
| With sulfuric acid In water at 65℃; for 15h; | 60% |
-
-
116649-85-5
ramatroban

| Conditions | Yield |
|---|---|
| With sodium hydroxide |
Related products
Raw Materials
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View