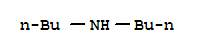Hebei Nengqian Chemical Import and Export Co., LTD
Our Advantage Rich Experience Our products are sold all over Europe,North&South America, Sino-East, Asia and pacific area as well as Africa,we establish long term. Quality service Company cooperates with research institutes. We strictly con
Cas:173937-91-2
Min.Order:1 Kilogram
FOB Price: $1.0 / 10.0
Type:Trading Company
inquiryHangzhou J&H Chemical Co., Ltd.
J&H CHEM R&D center can offer custom synthesis according to the contract research and development services for the fine chemicals, pharmaceutical, biotechnique and some of the other chemicals. J&H CHEM has some Manufacturing base in Jia
Zibo Hangyu Biotechnology Development Co., Ltd
Zibo Hangyu Biotechnology Development Co., Ltd is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality pharmaceuticals, intermediates, special chemicals and OLED intermediates and other fine chemi
Xiamen Jenny Chemical Technology Co., Ltd.
GMP standard, high purity, competitive price, in stock 1. Quick Response: within 6 hours after receiving your email. 2. Quality Guarantee: All products are strictly tested by our QC, confirmed by QA, and approved by a third-party lab in China, USA,
Xi`an Eastling Biotech Co., Ltd.
high purity lowest priceAppearance:solid or liquid Storage:in sealed air resistant place Package:Foil bag; Drum; Plastic bottle Application:Pharma;Industry;Agricultural Transportation:by sea or air Port:Beijing or Guangzhou
BOC Sciences
GMP factory Packing with pallet as buyer's special request Best service after shipment with e-mail Cargos together with container sales seervice available Api, inhibitor, and fine chemicals for more
Cas:173937-91-2
Min.Order:0 Metric Ton
Negotiable
Type:Trading Company
inquiryGuangdong Juda Chemical Industrial Co.,Limited
Factory supply high purity low priceAppearance:solid or liquid Storage:sealed in cool and dry place Package:As customer's requested Application:Pharma Intermediate Transportation:by courier/air/sea Port:Any port in China
Wuhan Circle Star Chem-medical Technology co.,Ltd.
good quality, competitive price, thoughtful after sale serviceAppearance:white powder Storage:Keep it in dry,shady and cool place Package:25kg,50kg,180kg,200kg,250kg,1000kg,customization Application:Pharma;Industry;Agricultural;chemical reaserch Tran
Hangzhou Fandachem Co.,Ltd
(2R,3R,4S)-4-(1,3-BENZODIOXOL-5-YL)-1-[2-(DIBUTYLAMINO)-2-OXOETHYL]-2-(METHOXYPHENYL)PYRROLIDINE-3-CARBOXYLIC ACIDCASAppearance:white crystalline powder Storage:Store in dry, dark and ventilated place Package:25KG drum Application:intermediate Transp
Cas:173937-91-2
Min.Order:0
Negotiable
Type:Other
inquiryChemlyte Solutions
Stock products, own laboratory Package:Grams, Kilograms Application:For R&D Transportation:According to customer request Port:Shanghai
ZHEJIANG JIUZHOU CHEM CO.,LTD
factory?direct?saleAppearance:White powder Storage:Sealed and preserved Package:200/Kilograms Application:healing drugs Transportation:By sea Port:Shanghai/tianjin
Amadis Chemical Co., Ltd.
1.Professional synthesis laboratory and production base. 2.Strong synthesis team and service team. 3.Professional data management system. 4.We provide the professional test date and product information ,ex. HNMR ,CNMR,FNMR, HPLC/G
Wuhan Sun-shine Bio-technology Corporation Limited
Wuhan Sun-shine Bio-technology Corporation Limited is specializing in the anticancer, antitumor,heart head blood-vessel,pharmaceutical intermediates,fine Chemicals production and customization. Company has strong ability of research and development
Cas:173937-91-2
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryChangzhou Extraordinary Pharmatech co.,LTD
Changzhou Extraordinary Pharmatech co., LTD. As a leading chemical manufacturer and supplier in China.DAS authentication is passed.We can provide the popular precursor chemicals, we have our own strong R & D team, have our own laboratories and fa
MENGNA
This chemical is called Atrasentan, and its systematic name is (2R,3R,4S)-4-(1,3-Benzodioxol-5-yl)-1-[2-(dibutylamino)-2-oxoethyl]-2-(4-methoxyphenyl)pyrrolidine-3-carboxylic acid. With the molecular formula of C29H38N2O6, its molecular weight is 5
Jinan Great Chemical Co.,Ltd.
Molecular Formula C29H38N2O6 Molecular Weight 510.62 CAS Registry Number 173937-91-2 AtrasentanSynonyms(2R,3R,4S)-4-(1,3-Benz…
Wuhan MoonZY Biological Technology Co.,Ltd
instock with good quality and wholesale price Storage:Keep in a cool & dry place Package:Packing material and QTY as your request Application:Pharma;Industry;other application Transportation:Express or as your request Port:Any port of China
Cas:173937-91-2
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquirycoolpharm Ltd
Known for its best quality and competitve price, this chemicals we offered is widely appreciated by our customers.Appearance:yellow to white solid Storage:keep sealed and keep from direct light Package:According client's requirements Application:Phar
Synthetic route

-
-
173937-91-2
atrasentan

| Conditions | Yield |
|---|---|
| With lithium hydroxide; dihydrogen peroxide In tetrahydrofuran for 0.5h; | 78% |
-
-
1485-00-3, 22568-48-5
5-((E)-2-nitroethenyl)-1,3-benzodioxole

-
-
173937-91-2
atrasentan

| Conditions | Yield |
|---|---|
| Multi-step reaction with 10 steps 1: 78 percent / DBU / tetrahydrofuran; propan-2-ol / 4 h / Ambient temperature 2: 12.34 g / H2 / Raney nickel / ethanol / 3040 Torr 3: 1.) sodium cyanoborohydride, 2.) bromocresol green, conc. HCl / 1.) THF, EtOH, 2.) THF, EtOH, 20 min 4: 2.775 g / C2H5ONa / ethanol / Heating 5: 91 percent / CH2Cl2 / 1 h / Ambient temperature 6: 95 percent / NaOH 7: 1.) Et3N, t-BuCOCl, 2.) n-BuLi 8: 4 N aq. HCl / dioxane 9: iPr2NEt / acetonitrile 10: 78 percent / aq. LiOH, 30percent aq. H2O2 / tetrahydrofuran / 0.5 h View Scheme | |
| Multi-step reaction with 9 steps 1: 78 percent / DBU / tetrahydrofuran; propan-2-ol / 4 h / Ambient temperature 2: 12.34 g / H2 / Raney nickel / ethanol / 3040 Torr 3: 1.) sodium cyanoborohydride, 2.) bromocresol green, conc. HCl / 1.) THF, EtOH, 2.) THF, EtOH, 20 min 4: 91 percent / CH2Cl2 / 1 h / Ambient temperature 5: 95 percent / NaOH 6: 1.) Et3N, t-BuCOCl, 2.) n-BuLi 7: 4 N aq. HCl / dioxane 8: iPr2NEt / acetonitrile 9: 78 percent / aq. LiOH, 30percent aq. H2O2 / tetrahydrofuran / 0.5 h View Scheme |
-
-
40124-27-4
N,N-dibutylbromoacetamide

-
-
173937-91-2
atrasentan

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: iPr2NEt / acetonitrile 2: 78 percent / aq. LiOH, 30percent aq. H2O2 / tetrahydrofuran / 0.5 h View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: 95 percent / 1,2-dichloro-ethane / 0.83 h / -20 - 20 °C 2: iPr2NEt / acetonitrile 3: 78 percent / aq. LiOH, 30percent aq. H2O2 / tetrahydrofuran / 0.5 h View Scheme |
-
-
173864-46-5
Ethyl 2-(4-methoxyphenyl)-4-(1,3-benzodioxol-5-yl)-4.5-dihydro-3H-pyrrole-3-carboxylate

-
-
173937-91-2
atrasentan

| Conditions | Yield |
|---|---|
| Multi-step reaction with 8 steps 1: 1.) sodium cyanoborohydride, 2.) bromocresol green, conc. HCl / 1.) THF, EtOH, 2.) THF, EtOH, 20 min 2: 2.775 g / C2H5ONa / ethanol / Heating 3: 91 percent / CH2Cl2 / 1 h / Ambient temperature 4: 95 percent / NaOH 5: 1.) Et3N, t-BuCOCl, 2.) n-BuLi 6: 4 N aq. HCl / dioxane 7: iPr2NEt / acetonitrile 8: 78 percent / aq. LiOH, 30percent aq. H2O2 / tetrahydrofuran / 0.5 h View Scheme | |
| Multi-step reaction with 7 steps 1: 1.) sodium cyanoborohydride, 2.) bromocresol green, conc. HCl / 1.) THF, EtOH, 2.) THF, EtOH, 20 min 2: 91 percent / CH2Cl2 / 1 h / Ambient temperature 3: 95 percent / NaOH 4: 1.) Et3N, t-BuCOCl, 2.) n-BuLi 5: 4 N aq. HCl / dioxane 6: iPr2NEt / acetonitrile 7: 78 percent / aq. LiOH, 30percent aq. H2O2 / tetrahydrofuran / 0.5 h View Scheme |
-
-
173864-45-4
Ethyl 2-(4-methoxybenzoyl)-3-(3,4-methylenedioxy-phenyl)-4-nitro-butanoate

-
-
173937-91-2
atrasentan

| Conditions | Yield |
|---|---|
| Multi-step reaction with 9 steps 1: 12.34 g / H2 / Raney nickel / ethanol / 3040 Torr 2: 1.) sodium cyanoborohydride, 2.) bromocresol green, conc. HCl / 1.) THF, EtOH, 2.) THF, EtOH, 20 min 3: 2.775 g / C2H5ONa / ethanol / Heating 4: 91 percent / CH2Cl2 / 1 h / Ambient temperature 5: 95 percent / NaOH 6: 1.) Et3N, t-BuCOCl, 2.) n-BuLi 7: 4 N aq. HCl / dioxane 8: iPr2NEt / acetonitrile 9: 78 percent / aq. LiOH, 30percent aq. H2O2 / tetrahydrofuran / 0.5 h View Scheme | |
| Multi-step reaction with 8 steps 1: 12.34 g / H2 / Raney nickel / ethanol / 3040 Torr 2: 1.) sodium cyanoborohydride, 2.) bromocresol green, conc. HCl / 1.) THF, EtOH, 2.) THF, EtOH, 20 min 3: 91 percent / CH2Cl2 / 1 h / Ambient temperature 4: 95 percent / NaOH 5: 1.) Et3N, t-BuCOCl, 2.) n-BuLi 6: 4 N aq. HCl / dioxane 7: iPr2NEt / acetonitrile 8: 78 percent / aq. LiOH, 30percent aq. H2O2 / tetrahydrofuran / 0.5 h View Scheme |

-
-
173937-91-2
atrasentan

| Conditions | Yield |
|---|---|
| Multi-step reaction with 5 steps 1: 95 percent / NaOH 2: 1.) Et3N, t-BuCOCl, 2.) n-BuLi 3: 4 N aq. HCl / dioxane 4: iPr2NEt / acetonitrile 5: 78 percent / aq. LiOH, 30percent aq. H2O2 / tetrahydrofuran / 0.5 h View Scheme |

-
-
173937-91-2
atrasentan

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: 4 N aq. HCl / dioxane 2: iPr2NEt / acetonitrile 3: 78 percent / aq. LiOH, 30percent aq. H2O2 / tetrahydrofuran / 0.5 h View Scheme |

-
-
173937-91-2
atrasentan

| Conditions | Yield |
|---|---|
| Multi-step reaction with 7 steps 1: 2.775 g / C2H5ONa / ethanol / Heating 2: 91 percent / CH2Cl2 / 1 h / Ambient temperature 3: 95 percent / NaOH 4: 1.) Et3N, t-BuCOCl, 2.) n-BuLi 5: 4 N aq. HCl / dioxane 6: iPr2NEt / acetonitrile 7: 78 percent / aq. LiOH, 30percent aq. H2O2 / tetrahydrofuran / 0.5 h View Scheme |

-
-
173937-91-2
atrasentan

| Conditions | Yield |
|---|---|
| Multi-step reaction with 6 steps 1: 91 percent / CH2Cl2 / 1 h / Ambient temperature 2: 95 percent / NaOH 3: 1.) Et3N, t-BuCOCl, 2.) n-BuLi 4: 4 N aq. HCl / dioxane 5: iPr2NEt / acetonitrile 6: 78 percent / aq. LiOH, 30percent aq. H2O2 / tetrahydrofuran / 0.5 h View Scheme |

-
-
173937-91-2
atrasentan

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: 1.) Et3N, t-BuCOCl, 2.) n-BuLi 2: 4 N aq. HCl / dioxane 3: iPr2NEt / acetonitrile 4: 78 percent / aq. LiOH, 30percent aq. H2O2 / tetrahydrofuran / 0.5 h View Scheme |
-
-
40124-27-4
N,N-dibutylbromoacetamide

-
-
178739-03-2
ethyl (2R,3R,4S)-5-(4-methoxyphenyl)-3-(3,4-(methylenedioxy)phenyl)pyrrolidine-3-carboxylate

-
-
173937-91-2
atrasentan

| Conditions | Yield |
|---|---|
| With N-ethyl-N,N-diisopropylamine In acetonitrile |
-
-
195708-14-6
ethyl (2R,3R,4S)-(+)-2-(4-methoxyphenyl)-4-(1,3-benzodioxol-5-yl)pyrrolidine-3-carboxylate, (S)-(+)-mandelate

-
-
40124-27-4
N,N-dibutylbromoacetamide

-
-
173937-91-2
atrasentan

| Conditions | Yield |
|---|---|
| Stage #1: ethyl (2R,3R,4S)-(+)-2-(4-methoxyphenyl)-4-(1,3-benzodioxol-5-yl)pyrrolidine-3-carboxylate, (S)-(+)-mandelate With sodium hydrogencarbonate In water; ethyl acetate at 25℃; Stage #2: N,N-dibutylbromoacetamide With N-ethyl-N,N-diisopropylamine In acetonitrile at 10℃; for 12h; Stage #3: With hydrogenchloride; sodium hydroxide more than 3 stages; |
-
-
403614-49-3
ethyl (2R,3R,4S)-(+)-2-(4-methoxyphenyl)-4-(1,3-benzodioxol-5-yl)pyrrolidine-3-carboxylate, (S)-(+) mandelate

-
-
40124-27-4
N,N-dibutylbromoacetamide

-
-
7087-68-5
N-ethyl-N,N-diisopropylamine

-
-
173937-91-2
atrasentan

| Conditions | Yield |
|---|---|
| Stage #1: ethyl (2R,3R,4S)-(+)-2-(4-methoxyphenyl)-4-(1,3-benzodioxol-5-yl)pyrrolidine-3-carboxylate, (S)-(+) mandelate With sodium hydrogencarbonate In water; ethyl acetate at 25℃; Stage #2: N,N-dibutylbromoacetamide; N-ethyl-N,N-diisopropylamine In ethyl acetate; acetonitrile at 110℃; for 16h; |

-
-
173937-91-2
atrasentan

| Conditions | Yield |
|---|---|
| With hydrogenchloride In ethanol; water; ethyl acetate for 0.25h; | |
| With hydrogenchloride In water; ethyl acetate at 0℃; for 2 - 3h; Product distribution / selectivity; | |
| With hydrogenchloride In ethanol; water; ethyl acetate for 0.25h; Product distribution / selectivity; | |
| With hydrogenchloride In ethyl acetate at 0 - 45℃; for 26.5 - 39.5h; |

| Conditions | Yield |
|---|---|
| In methanol; water at 50℃; |

| Conditions | Yield |
|---|---|
| In methanol; water at 40℃; Temperature; |

| Conditions | Yield |
|---|---|
| In water at 50℃; for 0.25h; Sonication; |
Related products
Raw Materials
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View