Jinan Finer Chemical Co., Ltd
Product description: Product name N,N-Diisopropylethylamine CAS number 7087-68-5 Assay ≥99% Appearance Colorless or yellowish liquid Capacity 500mt/year Application Pharmaceu
Cas:7087-68-5
Min.Order:1 Kilogram
FOB Price: $12.0
Type:Lab/Research institutions
inquiryHenan Tianfu Chemical Co., Ltd.
High purity CAS 7087-68-5 N,N-Diisopropylethylamine in stock Our main business covers the fields below: 1.Noble Metal Catalysts (Pt.Pd...) 2.Organic Phosphine Ligands (Tert-butyl-phosphine.Cyclohexyl-phosphine...) 3.OLED intermediates (
Cas:7087-68-5
Min.Order:1 Gram
FOB Price: $100.0
Type:Lab/Research institutions
inquirySynthetic route
-
-
56-34-8
tetraethylammonium chloride

-
-
108-18-9
diisopropylamine

-
-
7087-68-5
N-ethyl-N,N-diisopropylamine

| Conditions | Yield |
|---|---|
| With potassium hydroxide In butan-1-ol at 115℃; for 7h; Solvent; Reagent/catalyst; Temperature; | 97.2% |

| Conditions | Yield |
|---|---|
| With lithium chloride; zinc(II) chloride at 190℃; under 16501.7 Torr; for 10h; Catalytic behavior; Temperature; Pressure; Time; Reagent/catalyst; Autoclave; | 96.7% |

| Conditions | Yield |
|---|---|
| With iron(III) oxide; (triphenylphosphine)ruthenium acetate at 78℃; for 24h; Reagent/catalyst; Molecular sieve; | 92% |
| With ammonium acetate In aq. phosphate buffer at 34℃; for 10h; pH=8.5; Electrochemical reaction; | 28% |
| With Ru(OAc)2(1,1′-bis(diisopropylphosphino)ferrocene)(CO); trifluoroacetic acid at 78℃; for 24h; Inert atmosphere; Schlenk technique; |

| Conditions | Yield |
|---|---|
| With hydrogen; ZrO2-containing catalyst at 120 - 150℃; under 60006 Torr; | 80% |
| Stage #1: acetaldehyde; diisopropylamine With formic acid at 90 - 110℃; for 8.5h; Stage #2: With hydrogenchloride In water Stage #3: With sodium hydroxide In water | |
| With hydrogen; 5% Pd(II)/C(eggshell) at 100℃; under 18751.9 Torr; for 9h; Product distribution / selectivity; Autoclave; Inert atmosphere; | 95.2 %Chromat. |
| With 5% Pd/C; hydrogen at 120℃; under 18751.9 Torr; for 3h; Autoclave; |

| Conditions | Yield |
|---|---|
| With butanone |

| Conditions | Yield |
|---|---|
| at 140℃; | |
| at 140℃; |
-
-
104-01-8
4-Methoxyphenylacetic acid

-
-
13435-12-6
N-Trimethylsilylacetamide

-
-
7087-68-5
N-ethyl-N,N-diisopropylamine

| Conditions | Yield |
|---|---|
| In dichloromethane; N,N-dimethyl-formamide |


| Conditions | Yield |
|---|---|
| In dichloromethane; acetonitrile |

| Conditions | Yield |
|---|---|
| With N-ethyl-N,N-diisopropylamine In 4-(dicyanomethylene)-2-methyl-6-(p-dimethylaminostyryl)-4H-pyran; N,N-dimethyl-formamide |

-
-
75-07-0
acetaldehyde

-
-
108-18-9
diisopropylamine

-
A
-
64-17-5
ethanol

-
B
-
7087-68-5
N-ethyl-N,N-diisopropylamine

| Conditions | Yield |
|---|---|
| With hydrogen; 5% Pd(II)/C(eggshell) at 100℃; under 18751.9 Torr; Product distribution / selectivity; Autoclave; Inert atmosphere; Industry scale; | A 8.0 %Chromat. B 81.8 %Chromat. |

-
-
75-07-0
acetaldehyde

-
-
108-18-9
diisopropylamine

-
A
-
41781-44-6
N-butyldiisopropylamine

-
B
-
7087-68-5
N-ethyl-N,N-diisopropylamine

| Conditions | Yield |
|---|---|
| With hydrogen; 5% Pd(II)/C(eggshell) at 120℃; under 37503.8 Torr; Product distribution / selectivity; Autoclave; Inert atmosphere; Industry scale; | A 6.3 %Chromat. B 81.1 %Chromat. |


| Conditions | Yield |
|---|---|
| With alumina at 360℃; under 760.051 Torr; Temperature; Inert atmosphere; Gas phase; Green chemistry; |
-
-
56-34-8
tetraethylammonium chloride

-
-
108-18-9
diisopropylamine

-
-
7087-68-5
N-ethyl-N,N-diisopropylamine

| Conditions | Yield |
|---|---|
| With potassium hydroxide In butan-1-ol at 115℃; for 7h; Solvent; Reagent/catalyst; Temperature; | 97.2% |

| Conditions | Yield |
|---|---|
| With lithium chloride; zinc(II) chloride at 190℃; under 16501.7 Torr; for 10h; Catalytic behavior; Temperature; Pressure; Time; Reagent/catalyst; Autoclave; | 96.7% |

| Conditions | Yield |
|---|---|
| With iron(III) oxide; (triphenylphosphine)ruthenium acetate at 78℃; for 24h; Reagent/catalyst; Molecular sieve; | 92% |
| With ammonium acetate In aq. phosphate buffer at 34℃; for 10h; pH=8.5; Electrochemical reaction; | 28% |
| With Ru(OAc)2(1,1′-bis(diisopropylphosphino)ferrocene)(CO); trifluoroacetic acid at 78℃; for 24h; Inert atmosphere; Schlenk technique; |

| Conditions | Yield |
|---|---|
| With hydrogen; ZrO2-containing catalyst at 120 - 150℃; under 60006 Torr; | 80% |
| Stage #1: acetaldehyde; diisopropylamine With formic acid at 90 - 110℃; for 8.5h; Stage #2: With hydrogenchloride In water Stage #3: With sodium hydroxide In water | |
| With hydrogen; 5% Pd(II)/C(eggshell) at 100℃; under 18751.9 Torr; for 9h; Product distribution / selectivity; Autoclave; Inert atmosphere; | 95.2 %Chromat. |
| With 5% Pd/C; hydrogen at 120℃; under 18751.9 Torr; for 3h; Autoclave; |

| Conditions | Yield |
|---|---|
| With butanone |

| Conditions | Yield |
|---|---|
| at 140℃; | |
| at 140℃; |
-
-
104-01-8
4-Methoxyphenylacetic acid

-
-
13435-12-6
N-Trimethylsilylacetamide

-
-
7087-68-5
N-ethyl-N,N-diisopropylamine

| Conditions | Yield |
|---|---|
| In dichloromethane; N,N-dimethyl-formamide |


| Conditions | Yield |
|---|---|
| In dichloromethane; acetonitrile |

| Conditions | Yield |
|---|---|
| With N-ethyl-N,N-diisopropylamine In 4-(dicyanomethylene)-2-methyl-6-(p-dimethylaminostyryl)-4H-pyran; N,N-dimethyl-formamide |

-
-
75-07-0
acetaldehyde

-
-
108-18-9
diisopropylamine

-
A
-
64-17-5
ethanol

-
B
-
7087-68-5
N-ethyl-N,N-diisopropylamine

| Conditions | Yield |
|---|---|
| With hydrogen; 5% Pd(II)/C(eggshell) at 100℃; under 18751.9 Torr; Product distribution / selectivity; Autoclave; Inert atmosphere; Industry scale; | A 8.0 %Chromat. B 81.8 %Chromat. |

-
-
75-07-0
acetaldehyde

-
-
108-18-9
diisopropylamine

-
A
-
41781-44-6
N-butyldiisopropylamine

-
B
-
7087-68-5
N-ethyl-N,N-diisopropylamine

| Conditions | Yield |
|---|---|
| With hydrogen; 5% Pd(II)/C(eggshell) at 120℃; under 37503.8 Torr; Product distribution / selectivity; Autoclave; Inert atmosphere; Industry scale; | A 6.3 %Chromat. B 81.1 %Chromat. |


| Conditions | Yield |
|---|---|
| With alumina at 360℃; under 760.051 Torr; Temperature; Inert atmosphere; Gas phase; Green chemistry; |
-
-
114552-24-8
2-(5,8-Dimethoxy-quinolin-4-yl)-phenylamine

-
-
7087-68-5
N-ethyl-N,N-diisopropylamine

-
-
407-25-0
trifluoroacetic anhydride

-
-
114552-23-7
N-[2-(5,8-Dimethoxy-quinolin-4-yl)-phenyl]-2,2,2-trifluoro-acetamide

| Conditions | Yield |
|---|---|
| at 0℃; for 0.5h; | 100% |

-
-
7087-68-5
N-ethyl-N,N-diisopropylamine

-
-
536-74-3
phenylacetylene

-
-
451473-18-0
4-(1-methyl-2,4-dioxo-6-(2-phenylethynyl)-1,4-dihydro-2H-quinazolin-3-ylmethyl)-benzoic acid

| Conditions | Yield |
|---|---|
| CuI; dichlorobis(triphenylphosphine)palladium[II] | 100% |
-
-
24424-99-5
di-tert-butyl dicarbonate

-
-
7087-68-5
N-ethyl-N,N-diisopropylamine

-
-
217326-89-1
N-[(1,1-dimethylethoxy)carbonyl]-4-iodo-L-phenylalanine 1,1-dimethylethyl ester

| Conditions | Yield |
|---|---|
| In dichloromethane | 100% |


-
-
24424-99-5
di-tert-butyl dicarbonate

-
-
1991-81-7, 14173-41-2, 24250-85-9, 62561-75-5
4-iodo-L-phenylalanine

-
-
7087-68-5
N-ethyl-N,N-diisopropylamine

-
B
-
217326-89-1
N-[(1,1-dimethylethoxy)carbonyl]-4-iodo-L-phenylalanine 1,1-dimethylethyl ester

| Conditions | Yield |
|---|---|
| With hydrogenchloride; sodium hydroxide In dichloromethane; diethylene glycol dimethyl ether; ethyl acetate | A 100% B n/a |


-
-
38945-21-0
O-allylhydroxylamine hydrochloride

-
-
7087-68-5
N-ethyl-N,N-diisopropylamine

| Conditions | Yield |
|---|---|
| With benzotriazol-1-ol In dichloromethane | 100% |


-
-
7087-68-5
N-ethyl-N,N-diisopropylamine

-
-
217326-89-1
N-[(1,1-dimethylethoxy)carbonyl]-4-iodo-L-phenylalanine 1,1-dimethylethyl ester

| Conditions | Yield |
|---|---|
| With hydrogenchloride In dichloromethane | 100% |

-
-
672323-76-1
3-bromo-7-chloro-5-(2-chlorophenyl)pyrazolo[1,5-a]pyrimidine

-
-
7087-68-5
N-ethyl-N,N-diisopropylamine

-
-
144222-22-0
tert-butyl 4-(aminomethyl)piperidine-1-carboxylate

-
-
672316-65-3
4-{[3-BROMO-5-(2-CHLOROPHENYL)PYRAZOLO[1,5-a]PYRIMIDIN-7-YLAMINO]METHYL}PIPERIDINE-1-CARBOXYLIC ACID tert-BUTYL ESTER

| Conditions | Yield |
|---|---|
| In 1,4-dioxane | 100% |

-
-
10431-98-8
2-ethyl-4,5-dihydrooxazole

-
-
7087-68-5
N-ethyl-N,N-diisopropylamine

-
-
333-27-7
methyl trifluoromethanesulfonate

| Conditions | Yield |
|---|---|
| Stage #1: 2-ethyl-4,5-dihydrooxazole; methyl trifluoromethanesulfonate In chlorobenzene at 20 - 110℃; for 0.666667h; Stage #2: N-ethyl-N,N-diisopropylamine In chlorobenzene at 0 - 50℃; for 18h; | 100% |


| Conditions | Yield |
|---|---|
| Stage #1: (+/-)-1-[9-benzyl-3,9-diazabicyclo[4.2.1]non-9-yl]-propane-1-one; N-ethyl-N,N-diisopropylamine With propionic acid In tetrahydrofuran for 1h; Stage #2: sodium hydroxide In diethyl ether | 100% |


-
-
105832-38-0
O-(N-succinimidyl)-N,N,N',N'-tetramethyluronium tetrafluoroborate

-
-
7087-68-5
N-ethyl-N,N-diisopropylamine

| Conditions | Yield |
|---|---|
| In N,N-dimethyl-formamide at 20℃; for 2h; Inert atmosphere; | 100% |

-
-
1228231-19-3
C11H13ClN4

-
-
36476-78-5
3-azetidinecarboxylic acid

-
-
7087-68-5
N-ethyl-N,N-diisopropylamine

-
-
1228231-28-4
1-(7-methyl-6,7,8,9-tetrahydro-5H-1,4,7,10a-tetraaza-cyclohepta[f]inden-10-yl)-azetidine-3-carboxylic acid diisopropylethylamine salt

| Conditions | Yield |
|---|---|
| In ethanol at 75℃; for 4h; | 100% |

-
-
791834-68-9
(6S,7S)-6-((4-phenylpiperazin-1-yl)carbonyl)-5-azaspiro(2,5)octane-7-carboxylic acid

-
-
7087-68-5
N-ethyl-N,N-diisopropylamine

| Conditions | Yield |
|---|---|
| Stage #1: (6S,7S)-6-((4-phenylpiperazin-1-yl)carbonyl)-5-azaspiro(2,5)octane-7-carboxylic acid; O-benzylhydroxylamine hydrochloride With (benzotriazo-1-yloxy)tris(dimethylamino)phosphonium hexafluorophosphate In N,N-dimethyl-formamide at 0℃; for 0.0833333h; Stage #2: N-ethyl-N,N-diisopropylamine In N,N-dimethyl-formamide at 20℃; for 3h; | 100% |


| Conditions | Yield |
|---|---|
| In chloroform-d1; d(4)-methanol at 20℃; | 100% |


| Conditions | Yield |
|---|---|
| In dichloromethane at 0 - 20℃; for 18h; | A n/a B 100% |

-
-
7087-68-5
N-ethyl-N,N-diisopropylamine

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at -20℃; | 99% |
-
-
7087-68-5
N-ethyl-N,N-diisopropylamine

-
-
250215-21-5
N2-Z-N5-acetyl-N5-O-acetyl-L-ornithyl-N5-acetyl-N5-O-acetyl-L-ornithyl-N5-acetyl-N5-O-acetyl-L-ornithine

| Conditions | Yield |
|---|---|
| In methanol at 20℃; for 12h; Deacetylation; | 99% |
-
-
486436-98-0
4-(4'-trifluoromethyl-biphenyl-2-carbonylamino)-1-methyl-pyrrole-2-carboxylic acid

-
-
486437-37-0
4-(pyrimidin-4-yl-carbonylamino)-benzylamine

-
-
7087-68-5
N-ethyl-N,N-diisopropylamine

-
-
486435-54-5
N-[4-(pyrimidin-4-yl-carbonylamino)-phenylmethyl]-4-(4'-trifluoromethylbiphenyl-2-carbonylamino)-1-methyl-pyrrole-2-carboxylic acid amide

| Conditions | Yield |
|---|---|
| With O-(benzotriazol-1-yl)-N,N,N',N'-tetramethyluronium tetrafluoroborate In N-methyl-acetamide | 99% |


-
-
830-03-5
4-nitrophenol acetate

-
-
7087-68-5
N-ethyl-N,N-diisopropylamine

-
-
176249-60-8
1-(2-methoxybenzyl)-2,4-dimethyl-4-(1H-indol-3-ylmethyl)-2-imidazoline

| Conditions | Yield |
|---|---|
| In tetrahydrofuran; ethyl acetate | 99% |

-
-
630424-75-8
pyridin-2-yl (1,1,1-trifluoro-2-methylpropan-2-yl) carbonate

-
-
3850-31-5
methyl tert-leucinate

-
-
7087-68-5
N-ethyl-N,N-diisopropylamine

-
-
1028801-63-9
(S)-3,3-Dimethyl-2-(2,2,2-trifluoro-1,1-dimethyl-ethoxycarbonylamino)-butyric acid methyl ester

| Conditions | Yield |
|---|---|
| With sodium hydroxide In 4-(dicyanomethylene)-2-methyl-6-(p-dimethylaminostyryl)-4H-pyran | 99% |


-
-
7087-68-5
N-ethyl-N,N-diisopropylamine

-
-
95-01-2
2,4-Dihydroxybenzaldehyde

-
-
3188-13-4
ethyl chloromethyl ether

-
-
128837-29-6
2,4-bis(ethoxymethoxy)benzaldehyde

| Conditions | Yield |
|---|---|
| In N,N-dimethyl-formamide | 99% |

-
-
1658-27-1
1,5-dioxaspiro[5.5]undecane-2,4-dione

-
-
880-78-4
pentafluoronitrobenzen

-
-
7087-68-5
N-ethyl-N,N-diisopropylamine

| Conditions | Yield |
|---|---|
| In acetonitrile at 30℃; for 0.333333h; | 99% |

-
-
7087-68-5
N-ethyl-N,N-diisopropylamine

| Conditions | Yield |
|---|---|
| With 2,2,2-Trifluoroacetophenone; dihydrogen peroxide; acetonitrile In tert-butyl alcohol at 20℃; for 18h; Green chemistry; chemoselective reaction; | 98% |
| With 2,2,6,6-tetramethylpiperidin-1-oxoammonium chloride Ambient temperature; | |
| With sodium hydroxide; dihydrogen peroxide; N-tosylimidazole In methanol for 1.5h; | |
| With C19H16FeN4O5(1-); dihydrogen peroxide at 20℃; for 2h; pH=10; aq. buffer; | 40.2 %Chromat. |
| With dihydrogen peroxide In water at 0 - 30℃; |
-
-
123-75-1
pyrrolidine

-
-
68837-59-2
4-bromo-2-methylbenzoic acid

-
-
125700-67-6
O-(benzotriazol-1-yl)-N,N,N',N'-tetramethyluronium tetrafluoroborate

-
-
7087-68-5
N-ethyl-N,N-diisopropylamine

-
-
276678-35-4
(4-bromo-2-methylphenyl)(pyrrolidin-1-yl)methanone

| Conditions | Yield |
|---|---|
| In tetrahydrofuran; water | 98% |


-
-
24424-99-5
di-tert-butyl dicarbonate

-
-
7087-68-5
N-ethyl-N,N-diisopropylamine

-
-
345957-94-0
2-[4-(N-benzyl-methylamino)-3-nitro-phenyl]-2-methyl-3-tert.butyloxycarbonylamino-1-pyrrolidino-propan-1-one

| Conditions | Yield |
|---|---|
| In 1,4-dioxane | 98% |


| Conditions | Yield |
|---|---|
| With sodium hydrogencarbonate; citric acid In N,N-dimethyl-formamide | 98% |


| Conditions | Yield |
|---|---|
| With iodine; potassium bromide In dichloromethane; acetonitrile addn. of CoCl2 in MeCN to soln. of org. ligand and EtN(i-Pr)2 in CH2Cl2, stirring at room temp. overnight, evapn., dissolving residue in CH2Cl2 and aq. satd. soln. of KBr with stirring, addn. of I2 in CH2Cl2; extn. of aq. phase (CH2Cl2), washing (satd. KBr soln., H2O), filtration (silica gel, CH2Cl2/MeOH 100:3), drying in vac. at room temp.; | 98% |


| Conditions | Yield |
|---|---|
| In toluene at 20℃; for 2h; | 98% |

-
-
7087-68-5
N-ethyl-N,N-diisopropylamine

| Conditions | Yield |
|---|---|
| Stage #1: tert-butyl (23-((bis(benzyloxy)phosphoryl)oxy)-11-oxo-3,6,9,15,18,21-hexaoxa-12-azatricosyl)carbamate With palladium 10% on activated carbon; hydrogen In methanol for 1.5h; Inert atmosphere; Stage #2: N-ethyl-N,N-diisopropylamine Inert atmosphere; | 98% |

-
-
7087-68-5
N-ethyl-N,N-diisopropylamine

-
-
298212-99-4
2-(4-iodophenyl)-1,8a-dihydroazulene-1,1-dicarbonitrile

| Conditions | Yield |
|---|---|
| With copper(l) iodide In toluene at 20℃; for 48h; | 98% |

-
-
1658-27-1
1,5-dioxaspiro[5.5]undecane-2,4-dione

-
-
771-69-7
2,3,4-trifluoronitrobenzene

-
-
7087-68-5
N-ethyl-N,N-diisopropylamine

| Conditions | Yield |
|---|---|
| In acetonitrile at 45℃; for 20h; | 98% |

Related products
Downstream Products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View



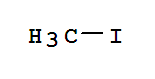









 F,
F,  C
C