Henan Wentao Chemical Product Co., Ltd.
We are leading fine chemicals supplier in China and we had established stable business relationships with Samsung,LG,Merck,Thermo Fisher Scientific and so on.Our main business covers the fields below: 1.Noble Metal Catalysts (Pt.Pd...)
Cas:1310-73-2
Min.Order:1 Kilogram
FOB Price: $5.0
Type:Lab/Research institutions
inquiryHangzhou Dingyan Chem Co., Ltd
Items Standard Result Appearance White Complies Assay ≥99% 99.02%
Cas:1310-73-2
Min.Order:1 Gram
FOB Price: $100.0 / 500.0
Type:Trading Company
inquiryHebei yanxi chemical co.,LTD.
Hebei yanxi chemical co., LTD is a professional research, development and production of lead acetate benzene acetamide enterprise backbone members by local well-known entrepreneurs and professional senior engineers in the party's "low carb
Cas:1310-73-2
Min.Order:1 Kilogram
FOB Price: $1.0 / 3.0
Type:Manufacturers
inquiryHefei TNJ chemical industry co.,ltd
Can be sent by ship and by train. If by train ,1500mt min,but good price. Appearance:white flake or white granule Storage:Store in cool,dry and ventilated place Package:25kg/bag,25mt/FCL,flake,27.5mt/FCL prilled Application:Chemical Pulping Tissu
Cas:1310-73-2
Min.Order:25 Metric Ton
Negotiable
Type:Trading Company
inquiryHenan Allgreen Chemical Co.,Ltd
he company has advanced technology, as well as a large number of excellent R & D team, to provide customers from the grams to one hundred kilograms and tons of high-quality products, competitive prices and quality se T rvice Appearance:Whit
EAST CHEMSOURCES LIMITED
Sodium Hydroxide(NaOH) Sodium hydroxide Chemical Properties mp 681 °C(lit.) bp 145 °C density
Cas:1310-73-2
Min.Order:1 Kilogram
FOB Price: $0.4 / 0.5
Type:Manufacturers
inquiryAlity Chemical Corporation
The above product is Ality Chemical's strong item with best price, good quality and fast supply. Ality Chemical has been focusing on the research and production of this field for over 14 years. At the same time, we are always committed to providi
Chemwill Asia Co., Ltd.
Chemwill Asia co.,Ltd is one of the leading manufacturer of Organic compounds in CHINA. Our main production base is located in Xuzhou industry park. We produce a wide range of organics including Active pharmaceutical ingredients(APIs), Veter
Cas:1310-73-2
Min.Order:1 Kilogram
FOB Price: $1.0
Type:Manufacturers
inquiryHenan Tianfu Chemical Co., Ltd.
Our company was built in 2009 with an ISO certificate.In the past 6 years, we have grown up as a famous fine chemicals supplier in China and we had established stable business relationships with Samsung,LG,Merck,Thermo Fisher Scientific and so on.O
Cas:1310-73-2
Min.Order:1 Kilogram
FOB Price: $1000.0
Type:Lab/Research institutions
inquiryHenan Sinotech Import&Export Corporation
CAS# Chemical Name Percent EINECS/ELINCS 1310-73-2 Sodium hydroxide 95-100 215-185-5
Hubei Langyou International Trading Co., Ltd
Advantages: Hubei XinRunde Chemical Co., Ltd is a renowned pharmaceutical manufacturer. We can offer high quality products at competitive price in quick delivery with 100% custom pass guaranteed. Never stop striving to offer our best s
Cas:1310-73-2
Min.Order:10 Gram
Negotiable
Type:Other
inquiryShanghai Upbio Tech Co.,Ltd
1,In No Less five years exporting experience. 2.Lower Price with higher quality 3,Free sample 4,We are sincerely responsible for the "product quality" and "After Service" Onchem specialized in APIs, chemical intermediate
Cas:1310-73-2
Min.Order:1 Metric Ton
Negotiable
Type:Lab/Research institutions
inquiryBaoji Guokang Healthchem co.,ltd
Our company has been in existence for 10 years since its establishment. We have our own unique team. The company integrates independent research and development, production and sales. We have established famous brands at home and abroad. At present
Cas:1310-73-2
Min.Order:1 Metric Ton
FOB Price: $835.0 / 900.0
Type:Trading Company
inquiryHebei Mojin Biotechnology Co.,Ltd
Company information: Hebei Mojin Biotechnology Co., Ltd, Our company is a professional in carbomer,carbopol,lead acetate,meglumine diphenyl ethylamine and other chemical raw materials and chemical reagents research and development production ente
Cas:1310-73-2
Min.Order:1 Kilogram
FOB Price: $350.0 / 400.0
Type:Trading Company
inquiryJilin Artel New Materials Co., LTD
Product Detail Minimum Order Qty. 10 Gram
Cas:1310-73-2
Min.Order:10 Gram
Negotiable
Type:Trading Company
inquiryHangzhou J&H Chemical Co., Ltd.
J&H CHEM R&D center can offer custom synthesis according to the contract research and development services for the fine chemicals, pharmaceutical, biotechnique and some of the other chemicals. J&H CHEM has some Manufacturing base in Jia
Cas:1310-73-2
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryAfine Chemicals Limited
Our Services 1. New Molecules R&D 2. Own test center HPLC NMR GC LC-MS 3. API and Intermediates from China reputed manufacturers 4. Documents support COA MOA MSDS DMF open part Our advantages 1. Government awarded company. Top 100 enter
Lonwin Chemical Group Limited
Lonwin Industry group limited as a professional manufactor & exporter of chemical materials ,we totally haver more than 270 stuffs, we have been on this line for more than 9 years. Our chemical materials are exported to lot of countries and reg
Cas:1310-73-2
Min.Order:100 Kilogram
Negotiable
Type:Other
inquiryHangzhou Keyingchem Co.,Ltd
Hangzhou KeyingChem Co., Ltd. exported this product to many countries and regions at best price. If you are looking for the material’s manufacturer or supplier in China, KeyingChem is your best choice. Pls contact with us freely for getting det
Cas:1310-73-2
Min.Order:0 Metric Ton
Negotiable
Type:Lab/Research institutions
inquiryHangzhou JINLAN Pharm-Drugs Technology Co., Ltd
Name Sodium hydroxide Synonyms Caustic soda Molecular Formula NaOH Molecular Weight 40.00 CAS Registry Numbe
Zibo Hangyu Biotechnology Development Co., Ltd
Zibo Hangyu Biotechnology Development Co., Ltd is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality pharmaceuticals, intermediates, special chemicals and OLED intermediates and other fine chemi
Cas:1310-73-2
Min.Order:10 Gram
FOB Price: $100.0
Type:Lab/Research institutions
inquiryHangzhou Lingrui Chemical Co.,Ltd.
Sodium hydroxideAppearance:White solid Storage:2-8°C Package:according to customers' requirements Application:1310-73-2 Transportation:By air(EMS or EUB or FedEx or TNT ect...) or by sea(FOB or CIF or CNF ect...)or according to your demand. Port:Any
Siwei Development Group Ltd.
Product name: Sodium Hydroxide CAS No.: 1310-73-2 Molecule Formula:NaOH Molecule Weight:41.00 Purity: 99.0% Package: 25kg/drum Description: Colorless non-transparent solid Manufacture Standards:Enterprise Standard
Cas:1310-73-2
Min.Order:1 Kilogram
Negotiable
Type:Trading Company
inquiryHangzhou Zhongqi chem Co.,Ltd.
Located in Hangzhou National Hi-Tech Industrial Development Zone, zhongqichem is a technical company mainly focus on the Custom synthesis, manufacturing, sales of chemicals to various industries. Benefiting from the outstanding customer service and h
Hunan chemfish Pharmaceutical co.,Ltd
Appearance:95%+ Package:R&D,Pilot run Transportation:per client require Port:Express ,Air, Sea
JINHUA HUAYI CHEMICAL CO., LTD.
Jinhua huayi chemical co., ltd. is dedicated to the development, production and marketing of chemicals. On the basis of equality and mutual benefit, and under the principle of customer first, credit first, quality first, we are ready to join hands
Antimex Chemical Limied
Ansciep Chemical is a professional enterprise manufacturing and distributing fine chemicals and speciality chemicals. We have been dedicated to heterocycle compounds and phenyl rings for tens of years. This is our mature product for export. Our quali
Hunan Russell Chemicals Technology Co.,Ltd
high purity lowest priceAppearance:solid or liquid Storage:in sealed air resistant place Package:Foil bag; Drum; Plastic bottle Application:Pharma;Industry;Agricultural Transportation:by sea or air Port:any port in China
Xiamen AmoyChem Co.,Ltd
Amoychem is committed to providing the top-quality chemical products and services Internationally. We offer our customers with friendly, professional service and reliable, high performance products that have been manufactured according to the accredi
Synthetic route

| Conditions | Yield |
|---|---|
| In water stirring Ca(OH)2-milk with Na2CO3-lye at 35°C with a turbo-mixer;; | A n/a B 96% |
| In water byproducts: Na2CO3*10H2O; caustification of concd. Na2CO3 lye with Ca(OH)2; cooling down to 0 to -15°C by pptn. of Na2CO3*10H2O;; lye containing 15 - 25% NaOH and not more than 1% Na2CO3;; | |
| In water PARNELL process;; |


| Conditions | Yield |
|---|---|
| With clay; sodium carbonate In water byproducts: NaAlO2; reaction of bricks of NaCl and clay with hot gases and overheated vapor in a shaft furnace; melting residue (acidic sodium silicon aluminate) with Na2CO3; product leached with water;; NaOH contains some Na2CO3 and NaAlO2;; | 90% |
| With clay; sodium carbonate In water byproducts: NaAlO2; reaction of bricks of NaCl and clay with hot gases and overheated vapor in a shaft furnace; melting residue (acidic sodium silicon aluminate) with Na2CO3; product leached with water;; NaOH contains some Na2CO3 and NaAlO2;; | 90% |
| With water byproducts: HCl; H2O steam; 1000°C; |

| Conditions | Yield |
|---|---|
| In neat (no solvent) reaction with steam gives 6% conversion in 1.5 h and 12.6% in 12 h;; | |
| silica gel In neat (no solvent) byproducts: HCl; at 810°C;; | |
| Electrolysis; electrolysis of moist NaCl;; |

-
-
7732-18-5
water

-
-
584-08-7
potassium carbonate

-
B
-
497-19-8
sodium carbonate

-
C
-
1310-73-2
sodium hydroxide

| Conditions | Yield |
|---|---|
| With tetrahydrofuran | A 100% B n/a C n/a |


| Conditions | Yield |
|---|---|
| With lead(II) oxide In water byproducts: basic lead chlorides; repeated grinding NaCl with PbO, Ca(OH)2 and a small amount of H2O;; evapn. of lye and sucking off pptd. NaCl; recycling basic lead chlorides (by heating to 170°C with boiling Ca(OH)2 soln. to form PbO);; | 50% |
| In water at ambient temp., equilibrium;; | 3% |
| With Ca-aluminate or bauxite In water byproducts: Ca-chloroaluminate; autoclave or 100°C, increase of yield with bauxite or Ca-aluminate;; |

| Conditions | Yield |
|---|---|
| With H2O In water hydrolysis equilibrium investigated;; |

| Conditions | Yield |
|---|---|
| With hydroxide In neat (no solvent) redn. with C or P and a hydroxide of the second-row elements by heating;; | |
| With H2 or H2/N2; nickel In neat (no solvent) byproducts: NH3; treatment with H2 or H2/N2-mixture at 100-800°C in presence of a catalyst; increase of yield with increase of H2-pressure;; | |
| With H2 or H2/N2; copper In neat (no solvent) byproducts: NH3; treatment with H2 or H2/N2-mixture at 100-800°C in presence of a catalyst; increase of yield with increase of H2-pressure;; |

| Conditions | Yield |
|---|---|
| With barium carbonate at 100℃; for 5h; |


| Conditions | Yield |
|---|---|
| In water Electrolysis; electrolysis of a Na2CO3 soln. in the cold with Hg cathode; decompn. of formed amalgam in paraffined tube in presence of CO2-free air and Pt with dest. H2O;; | |
| at 800°C; equiv. content 97.5% Na2CO3, 2.5% NaOH; | |
| In water Electrolysis; electrolysis of a concd. Na2CO3-soln., Hg-cathode, react. of the generated Na-amalgame with H2O, evapn. in vac. over H2SO4 at ambinet temp., dehydration of NaOH*H2o in vac. or in a dry N2-stream at 200-300°C;; pure NaOH; Na2CO3-content: 0.015-0.04%, H2O-content <0.1%;; | |
| In neat (no solvent) reaction of Na2CO3 with H2O-vapor;; | |
| at 800°C; equiv. content 97.5% Na2CO3, 2.5% NaOH; |

| Conditions | Yield |
|---|---|
| In neat (no solvent) adding at 175°C powdered NaNO3 to cautiously heated pure NaNH2 in Ni crucible;; | A 65.7% B n/a C n/a |
| In neat (no solvent) adding at 175°C powdered NaNO3 to cautiously heated pure NaNH2 in Ni crucible;; | A 65.7% B n/a C n/a |


| Conditions | Yield |
|---|---|
| With oxygen In not given byproducts: NH3; | |
| With O2 In not given byproducts: NH3; |

| Conditions | Yield |
|---|---|
| In water byproducts: BaSO4; cyclic process: reduction of BaSO4 to BaS, regeneration of Ba(OH)2 on boiling with H2O; 30% Na2SO4-soln., 3% excess Ba(OH)2;; 18% NaOH-soln.;; | 75% |
| In water byproducts: BaSO4; cyclic process: reduction of BaSO4 to BaS, regeneration of Ba(OH)2 on boiling with H2O; 40% Na2SO4-soln., 3% excess Ba(OH)2;; 27% NaOH-soln.;; | 70% |
| In water byproducts: BaSO4; |

| Conditions | Yield |
|---|---|
| With H2 In neat (no solvent) quantitative react. at 700-900°C;; | |
| With H2 In neat (no solvent) quantitative react. at 700-900°C;; |

| Conditions | Yield |
|---|---|
| With ammonia In not given ammonolysis of Na2O;; | |
| With NH3 In not given ammonolysis of Na2O;; |

| Conditions | Yield |
|---|---|
| With oxygen In ammonia in presence of NH4Br;; | |
| With O2 In ammonia NH3 (liquid); in presence of NH4Br;; |

| Conditions | Yield |
|---|---|
| With air In neat (no solvent) in moist air after a longer period of time;; | |
| With air In neat (no solvent) in moist air after a longer period of time;; |

| Conditions | Yield |
|---|---|
| With sodium peroxide In neat (no solvent) byproducts: Na2SO4; oxidation of Se on melting with Na2O2in a Ni crucible;; the formed melt contains NaOH and Na2CO3; isolation as mixture of Na2SeO4 and Na2SO4;; | A 58% B n/a C n/a |
| With Na2O2 In neat (no solvent) byproducts: Na2SO4; oxidation of Se on melting with Na2O2in a Ni crucible;; the formed melt contains NaOH and Na2CO3; isolation as mixture of Na2SeO4 and Na2SO4;; | A 58% B n/a C n/a |


| Conditions | Yield |
|---|---|
| With water In water decompn.;; | |
| With H2O In water decompn.;; |

| Conditions | Yield |
|---|---|
| With H2 or H2/N2; iron In neat (no solvent) byproducts: NH3; treatment with H2 or H2/N2-mixture at 100-800°C in presence of a catalyst; increase of yield with increase of H2-pressure;; | |
| With H2 or H2/N2; nickel In neat (no solvent) byproducts: NH3; treatment with H2 or H2/N2-mixture at 100-800°C in presence of a catalyst; increase of yield with increase of H2-pressure;; | |
| With H2 or H2/N2; copper In neat (no solvent) byproducts: NH3; treatment with H2 or H2/N2-mixture at 100-800°C in presence of a catalyst; increase of yield with increase of H2-pressure;; |

| Conditions | Yield |
|---|---|
| In water Electrolysis; formation of PbSO4 on Pb-anode; electrolysis of PbSO4; | A 91% B 91% |
| With H2O Electrolysis; | |
| In water Electrolysis; |

| Conditions | Yield |
|---|---|
| In not given react. of Ba(OH)2 lye with Na2SO4;; reprocessing of BaSO4;; | |
| In not given dissolving recrystallized Na2SO4; addition of the soln. to a soln. of recrystallizaed Ba(OH)2;; filtration of fromed BaSO4; crystn. of NaOH on evaporating;; |


| Conditions | Yield |
|---|---|
| In neat (no solvent) exothermic react., react. enthalpy given;; | |
| In neat (no solvent) exothermic react., react. enthalpy given;; |


| Conditions | Yield |
|---|---|
| With sodium nitrate absent of air; NaOH-melt; | |
| With H2O(gas) In melt byproducts: HI(gas); hydrolytic equilibrium (1100 and 1200 K); emf control of cell with gaseous electrodes and molten salt mixt.; |

| Conditions | Yield |
|---|---|
| With water; oxygen In melt Electrochem. Process; molten Na is used as anode on a layer of molten NaOH, the cathode is a metal covered with a layer of oxide by influence of oxygen, or which is covered by higher Mn-Oxides. Introduction of H2O vapor causes electrochemical formation of NaOH;; molten NaOH is continuously separated from the reaction mixture;; | |
| With potassium hydroxide In neat (no solvent) byproducts: K; equilibrium;; | |
| In not given |

| Conditions | Yield |
|---|---|
| With methyllithium In water equilibrium of causticizing react. investigated;; | |
| With iron(III) oxide In water byproducts: Fe(OH)3, CO2; heating of soda with metal oxide, especially Fe2O3; formation of CO2 and residue NaOH;; | |
| With iron(III) oxide In water byproducts: Fe(OH)3; evaporation of Na2CO3 soln. with Fe2O3 to dryness; mixture heating to red heat (in revolving furnace); formation of CO2 and Na-ferrite, which is leached with cold and then with hot water; use of pptd. Fe(OH)3 as educt;; concd. NaOH (d=1.32 - 1.38);; |

| Conditions | Yield |
|---|---|
| With iron oxide In water reaction with iron oxide in water;; | |
| With water byproducts: H2S, O2; between 300 and 1400°C; | |
| With sodium sulfide; silica gel In melt leaching out of melt;; |

| Conditions | Yield |
|---|---|
| With manganese(IV) oxide; oxygen In water oxidation of aq. Na2S with MnO2 forming Na2S2O3, NaOH and Mn(OH)2; oxidation of Mn(OH)2 by O2 reprocessing MnO2;; | |
| With oxygen In water oxidation of aq. Na2S on air forming Na2S2O3;; | |
| With MnO2; O2 In water oxidation of aq. Na2S with MnO2 forming Na2S2O3, NaOH and Mn(OH)2; oxidation of Mn(OH)2 by O2 reprocessing MnO2;; | |
| With O2 In water oxidation of aq. Na2S on air forming Na2S2O3;; |

-
-
1310-73-2
sodium hydroxide

| Conditions | Yield |
|---|---|
| With ZnO or Zn In potassium hydroxide byproducts: ZnS; Na2S was treated with alkaline soln. of ZnO or Zn, filtration of the hot soln., crystn. of NaOH from the filtrate;; | |
| With Cu oxides | |
| With Zn- or Sn- or Pb- or Ni- or Mn- or Cu- or; Fe-metallate In water treatment of Na2S with a Na-metallate;; thermal decompn. of the metal-sulfide in oxide and SO2;; |

-
-
1310-73-2
sodium hydroxide

| Conditions | Yield |
|---|---|
| With ammonia In neat (no solvent) byproducts: H2O, Na, N; careful heating, formation of NaNH2 and H2O, further heating to 200 - 300°C formation of NaOH and Na, N and H;; | |
| With carbon monoxide; water In neat (no solvent) heating glass in a reductive flame: redn. of Na2O by CO, react. of Na with H2O;; | |
| With water In water dissolving in H2O;; not isolated;; | |
| With H2O In water dissolving in H2O;; not isolated;; | |
| With NH3 In neat (no solvent) byproducts: H2O, Na, N; careful heating, formation of NaNH2 and H2O, further heating to 200 - 300°C formation of NaOH and Na, N and H;; |

| Conditions | Yield |
|---|---|
| In water N2H4*H2O (80%) added to aq. soln. of NiCl2*6H2O (temp. increased to 65°C) under vigorous stirring, cooling to 50°C, aq. NaOH (50%)added (temp. increased to 54°C), 1 h; ppt. washed with water and dried at room temp. for 16 h; | 100% |
| In water; ethylene glycol byproducts: N2, H2O; other Radiation; soln. of Ni compd. in ethylene glycol treated with hydrazine hydrate andNaOH under external magnetic field (0.13-0.35 T) with vigorous stirring , react at 60°C; pptd., septd., washed (ethanol), dried (vac., 40°C, 24 h), SEM, XRD; |


| Conditions | Yield |
|---|---|
| In water pure SO2 is introduced into alkaline Na2S soln.; | A n/a B 100% |


| Conditions | Yield |
|---|---|
| In water; isopropyl alcohol soln. of Ni salt added to hot soln. of NaOH at a rate of 100 drops/min at stirring within 1.5-2.0 h; i-PrOH added; mixt. stirred for 30 min at 80°C; ppt. washed (H2O); dried at 50°C; ground in agate mortar; | 100% |
| In water pptn. by dropwise addn. of 0.5 M NaOH to aq. soln. of Ni salt; washed, dried at 130°C overnight; | |
| In water soln. of Ni(NO3)2 in water was added to soln. of NaOH in water, stirring, pptn. of apple-green solid, stirring for 5 min.; filtration, washing with water, dried for 12 h at 80 ° C; |

| Conditions | Yield |
|---|---|
| In sodium hydroxide High Pressure; 2 M Zn(NO3)2 neutralized with 2 M NaOH to pH 11; heated with microwave treatment (100°C, 15 psi, 2 min); solid sepd. (centrifugation), washed (deionized H2O, EtOH), dried; XRD; | 100% |
| In sodium hydroxide Sonication; 2 M Zn(NO3)2 neutralized with 2 M NaOH to pH 12; heated with microwave treatment (100°C, 15 or 30 min); solid sepd. (centrifugation), washed (deionized H2O, EtOH), dried; XRD; | 99.7% |
| In sodium hydroxide High Pressure; 2 M Zn(NO3)2 neutralized with 2 M NaOH to pH 12; heated with microwave treatment (100°C, 15 psi, 2 min); solid sepd. (centrifugation), washed (deionized H2O, EtOH), dried; XRD; | 98.3% |

| Conditions | Yield |
|---|---|
| In water at ratio OH(1-):Cd(2+)=1.56; | 100% |
| In water pptn.; | |
| In water pptn.; |

| Conditions | Yield |
|---|---|
| In sodium hydroxide heating in 8% NaOH solution; precipitate contents NaOH; a small amount of Zr phosphate remains in filtrate;; | 100% |

| Conditions | Yield |
|---|---|
| In water filtration, titration (pH 5.5), crystn.; IR spectroscopy; | 100% |
-
-
7722-84-1
dihydrogen peroxide

-
-
7782-50-5
chlorine

-
-
1310-73-2
sodium hydroxide

-
-
7782-44-7
oxygen

-
B
-
7647-14-5
sodium chloride

| Conditions | Yield |
|---|---|
| In water H2O2 soln. poured onto porous glass plate within the singlet O2 generator (closed glass system, under Ar), spent liquid discarded into the lower flask, Cl2 (1-2 kPa) admitted, alkali soln. poured in, evolution of singlet O2 over a period of 12-16 min.; luminiscence spectroscopy, calorimetry; | A 100% B n/a |


| Conditions | Yield |
|---|---|
| In sodium hydroxide aq. NaOH; dissolving 0.01mol Na4P4O12 in 85ml hot H2O, addn. of 0.02mol NaOH in a small amount of H2O after cooling, standing in vac. over H2SO4 at 40°C for 100h;; | 100% |

| Conditions | Yield |
|---|---|
| In water High Pressure; soln. of NaOH added to soln. of HfOCl2*8H2O; heated in autoclave at 120°C for 792 h at pH 7 or for 24 h at pH 13; washed twice with water; dried at 60°C for 24 h; | 100% |
| In water High Pressure; soln. of NaOH added to soln. of HfOCl2*8H2O; heated in autoclave at 90°C, 336 h, pH 9.5; washed twice with water; dried at 60°C for 24 h; | 78% |

-
-
1310-73-2
sodium hydroxide

| Conditions | Yield |
|---|---|
| In acetone byproducts: Mg(OH)2; addn. of an aq. soln. of NaOH to a soln. of ((Os(PhNNC(NO)CH3)3)2Mg) inacetone, stirring at 298 K for 0.5 h, color turns green; filtered from pptd. Mg(OH)2, evapn. (vac.), recrystn. from acetonitrile/water; | 100% |

| Conditions | Yield |
|---|---|
| In sodium hydroxide aq. NaOH; High Pressure; mixt. of V2O3 and GeO2 in 1.05 M aq. NaOH in sealed OD silver tubes heated at 580°C in a Tuttle "cold sealed" autoclave for 3 d; | 100% |


-
-
1310-73-2
sodium hydroxide

| Conditions | Yield |
|---|---|
| In water Ar atm.; 1 N NaOH, 60°C, 1 h; | 100% |

-
-
1310-73-2
sodium hydroxide

| Conditions | Yield |
|---|---|
| In water stirring (48 h, pptn.), centrifugation; washing (water, centrifugation), drying in vacuum; elem. anal.; | 100% |
-
-
5419-55-6
Triisopropyl borate

-
-
1310-73-2
sodium hydroxide

-
-
815581-69-2
N-(n-butyl)-2-(2-bromophenyl)-benzimidazole

| Conditions | Yield |
|---|---|
| With tert.-butyl lithium In diethyl ether; pentane a soln. of benzimidazole-compound in ether at -78°C under Ar was treated with t-BuLi in pentane over 15 min, stirred at -78.degre.C for 1h, treated with B(Oi-Pr)3, stirred for 48 h, allowed to warm to room te mp., NaOH was added, stirred for 1 h; ppt. was filtered, washed with ether, dried; | 100% |


-
-
1310-73-2
sodium hydroxide

| Conditions | Yield |
|---|---|
| In water Ar atm.; 1 N NaOH, 60°C, 1 h; | 100% |

-
-
10138-52-0
gadolinium(III) chloride

-
-
1310-73-2
sodium hydroxide

| Conditions | Yield |
|---|---|
| In water equimolar ratio Gd:ligand, pH 7, stirring; evapn. (vac.), drying (3 h, 200°C), ; elem. anal.; | 100% |


| Conditions | Yield |
|---|---|
| In ethanol soln. of ZnCl2 (0.19 mmol) in EtOH added to soln. of ligand (0.08 mmol) and 0.3 ml 1M NaOH in EtOH; stirred for 75 min; ppt. filtered off; washed (EtOH); dried (vac., 56°C); elem. anal.; | 100% |


| Conditions | Yield |
|---|---|
| In water at 100℃; for 72h; Autoclave; High pressure; | 100% |


| Conditions | Yield |
|---|---|
| In methanol at 20℃; Darkness; | 100% |

-
-
66-22-8
uracil

-
-
1310-73-2
sodium hydroxide

-
-
42563-71-3, 53006-96-5, 59245-40-8
sodium pyrimidin-1-ide-2,4-dione

| Conditions | Yield |
|---|---|
| In ethanol; water at 20℃; for 0.25h; | 100% |

| Conditions | Yield |
|---|---|
| With polyethylene glycol 20,000 In water at 160℃; for 12h; Autoclave; Sealed tube; | 100% |

| Conditions | Yield |
|---|---|
| at 80℃; for 18h; pH=8; | 100% |


-
-
1310-73-2
sodium hydroxide

-
-
1616588-24-9
Na[Mn(N-tosyl-N',N",N"-diethylenetriaminetriacetate)]

| Conditions | Yield |
|---|---|
| In water pH=6.4 - 6.6; | 100% |


-
-
1422257-76-8
4-vinyl(pyridine-2,6-diyl)bis(methyleneamino)-N,N,N',N'-tetrakis(acetic acid)

-
-
1310-73-2
sodium hydroxide

| Conditions | Yield |
|---|---|
| In water pH=7.2; | 100% |


-
-
1310-73-2
sodium hydroxide

| Conditions | Yield |
|---|---|
| Stage #1: gadolinium(III) chloride hexahydrate; C75H77N15O24Ru(2+)*2C2F3O2(1-) In water at 20℃; for 1h; Stage #2: sodium hydroxide In water at 20℃; for 18h; pH=5 - 6; | 100% |


-
-
1310-73-2
sodium hydroxide

| Conditions | Yield |
|---|---|
| Stage #1: europium(III) chloride hexahydrate; C75H77N15O24Ru(2+)*2C2F3O2(1-) In water at 20℃; for 1h; Stage #2: sodium hydroxide In water at 20℃; for 18h; pH=5 - 6; | 100% |


-
-
1310-73-2
sodium hydroxide

| Conditions | Yield |
|---|---|
| Stage #1: neodymium(III) chloride hexahydrate; C75H77N15O24Ru(2+)*2C2F3O2(1-) In water at 20℃; for 1h; Stage #2: sodium hydroxide In water at 20℃; for 18h; pH=5 - 6; | 100% |


-
-
1310-73-2
sodium hydroxide

| Conditions | Yield |
|---|---|
| Stage #1: ytterbium(III) chloride hexahydrate; C75H77N15O24Ru(2+)*2C2F3O2(1-) In water at 20℃; for 1h; Stage #2: sodium hydroxide In water at 20℃; for 18h; pH=5 - 6; | 100% |

Related products
Raw Materials
Downstream Products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View






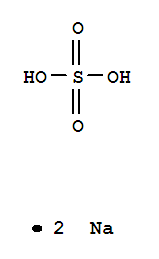






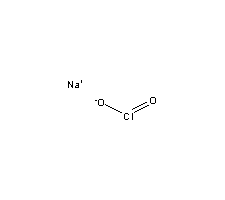
 C,
C, Xi
Xi