Shandong Xinhua Pharmaceutical Co.,Ltd
Best price,best quality Appearance:colorless or white cubic crystal ,no smell Storage:store in well-colosed conatiner,protected from light Package:25kgs Application:used as chemical materials ,medicine,photosensitive material,analytic reagent Port:
Dayang Chem (Hangzhou) Co.,Ltd.
Dayangchem's R&D center can offer custom synthesis according to the contract research and development services for the fine chemicals, pharmaceutical, biotechnique and some of the other chemicals. DayangChem can provide different quantities
Cas:7681-82-5
Min.Order:1 Kilogram
FOB Price: $3.0
Type:Lab/Research institutions
inquirySimagchem Corporation
Welcome to Simagchem, your partner in China as a premier supply of bulk specialty chemicals for industry and life science. We introduce experienced quality product and exceptional JIT service with instant market intelligence in China to benefit our
Hebei yanxi chemical co.,LTD.
hebei yanxi chemical co., LTD who registered capital of 10 million yuan, nearly to $2 million, we have a pharmaceutical raw materials factory production of pharmaceutical raw materials, and a reagent r&d center, and we do research and developm
Cas:7681-82-5
Min.Order:1 Metric Ton
FOB Price: $1.0 / 3.0
Type:Manufacturers
inquiryWuhan Fortuna Chemical Co.,Ltd
Unique advantages for Sodium iodide Cas7681-82-5 Guaranteed the purity High quality & competitive price Quality control Fast feedback Prompt shipment Appearance:White crystalline powder Storage:Store at RT Package:25kg/drum Applicati
Cas:7681-82-5
Min.Order:25 Kilogram
FOB Price: $70.0 / 77.0
Type:Trading Company
inquiryHefei TNJ chemical industry co.,ltd
1. Quality Our products meet MSDS safe standard and we have ISO and other certificate so yan can get high quality products from our company. 2. Price We are the company which is the joint of trade and industry so we cao provide the competiti
Cas:7681-82-5
Min.Order:25 Kilogram
Negotiable
Type:Trading Company
inquiryEAST CHEMSOURCES LIMITED
Sodium iodide CAS RN.: 7681-82-5 EINECS: 231-679-3 Molecular Weight: 149.89 Molecular Formula: NaI Density: 3.66 Melting Poi
Ality Chemical Corporation
The above product is Ality Chemical's strong item with best price, good quality and fast supply. Ality Chemical has been focusing on the research and production of this field for over 14 years. At the same time, we are always committed to providi
Xi'an Xszo Chem Co., Ltd.
1. Factory price and high quality must be guaranteed, base on 8 years of production and R&D experience2. Free samples will be provided,ensure specifications and quality are right for customer3. Customers will receive the most professional technical s
Cas:7681-82-5
Min.Order:1 Gram
FOB Price: $0.1
Type:Manufacturers
inquiryCOLORCOM LTD.
Colorcom is a global leader in industrial chemical manufacturing and is continuously innovating and transforming to exceed client expectations and industry standards. Colorcom prides itself on superior customer and technical focus, while focusing on
Chemwill Asia Co., Ltd.
Chemwill Asia co.,Ltd is one of the leading manufacturer in CHINA. Product quality, process, price and service Sodium iodide 7681-82-5 Factory IN STOCK CAS 7681-82-5 7681-82-5 CAS 7681-82-5 highly quality and immedaite delivery naio3
Cas:7681-82-5
Min.Order:5 Kiloliter
FOB Price: $3.5 / 5.0
Type:Manufacturers
inquiryHubei DiBo chemical co., LTD
Name:Sodium iodide CAS no:7681-82-5 Grade:Scientific research and export Molecular formula:NaI Molecular weight:149.89 Product Quality 12 years of chemical raw materials Mature operation of the industry System stability Data storage Secu
Cas:7681-82-5
Min.Order:25 Kilogram
FOB Price: $1.0 / 2.0
Type:Other
inquiryHenan Tianfu Chemical Co., Ltd.
Competitive Price High Quality Fast Delivery custom-made Welcome to Henan Tianfu Chemical Co., Ltd. website. Our company engages in Sodium Tripolyphosphate (STPP) and Sodium Hexametabphosphate (SHMP) production; development of noble me
Cas:7681-82-5
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryHebei Nengqian Chemical Import and Export Co., LTD
With our good experience, we offer detailed technical support and advice to assist customers. We communicate closely with customers to establish their quality requirements. Consistent Quality Our plant has strict quality control in each manufactu
Cas:7681-82-5
Min.Order:1 Kilogram
FOB Price: $1.0 / 10.0
Type:Trading Company
inquiryZhuozhou Wenxi import and Export Co., Ltd
WITH US,YOUR MONEY IN SAFE,YOUR BUSINESS IN SAFE 1)Quick Response Within 12 hours; 2)Quality Guarantee: All products are strictly tested by our QC, confirmed by QA and approved by third party lab in China, USA, Canada, Germany, UK, Italy, France et
Cas:7681-82-5
Min.Order:10 Gram
FOB Price: $146.0 / 176.0
Type:Trading Company
inquiryWuhan Han Sheng New Material Technology Co.,Ltd
Our Advantage: high quality with competitive price High quality standard: BP/USP/EP Enterprise standard All purity customized Fast and safe delivery We have reliable forwarder who can help us deliver our goods more fast and safe. We
Henan Sinotech Import&Export Corporation
Name: Sodium iodide (NaI) CAS No.: 7681-82-5 Formula: NaI Molecular Weight: 149.89 Synonyms: Sodiumiodide (8CI);Anayodin;Ioduril;NSC 77388;Sodium monoiodide;Soiodin;Sodium iodid bp grade;Sodiumiodide (NaI);Sodium iodide; EINECS: 231-679-3
Hubei Langyou International Trading Co., Ltd
1. Guaranteed purity; 2. Large quantity in stock; 3. Largest manufacturer; 4. Best service after shipment with email; 5. High quality & competitive price; Appearance:White fine Crystalline powder Storage:Store in sealed contai
Cas:7681-82-5
Min.Order:100 Gram
Negotiable
Type:Other
inquiryShanghai Upbio Tech Co.,Ltd
1.No Less 8 years exporting experience. Clients can 100% received goods 2.Lower Price with higher quality 3,Free sample 4,We are sincerely responsible for the "product quality" and "After Service" Upbio is Specialized
Cas:7681-82-5
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryHangzhou Fonlynn Health Technology Co., Ltd.
Packing: According to customer requirements Delivery time: In stock or depands Port of shipment: Ningbo/Shanghai/Qingdao OEM/ODM:Welcome Sample:We can offer our existing samples at once Appearance:white powder/ Refer to COA Storage:Refer to COA
Cas:7681-82-5
Min.Order:1 Kilogram
Negotiable
Type:Trading Company
inquiryBaoji Guokang Healthchem co.,ltd
Our company has been in existence for 10 years since its establishment. We have our own unique team. The company integrates independent research and development, production and sales. We have established famous brands at home and abroad. At present
Cas:7681-82-5
Min.Order:1 Kilogram
FOB Price: $20.0 / 35.0
Type:Trading Company
inquiryQingdao Beluga Import and Export Co., LTD
Sodium iodide CAS:7681-82-5 Qingdao Belugas Import and Export Co., Ltd. is a scientific and technological company integrating research and development, production and trade of chemical intermediates, specializing in high quality organic intermediate
Cas:7681-82-5
Min.Order:1 Gram
Negotiable
Type:Lab/Research institutions
inquiryHenan Wentao Chemical Product Co., Ltd.
Henan Wentao Chemical Product Co.,Ltd is Located in Zhengzhou High-tech Development Zone with import and export license, We passed ISO 9001:2008 as well, Henan Wentao has developed more than 1000 compounds, which are widely used in the fields of prod
Hebei Mojin Biotechnology Co.,Ltd
Hebei Mojin Biotechnology Co., Ltd, Our company is a professional in chemical raw materials and chemical reagents research and development production enterprises. Our business covers more than 30 countries, most of the big customers come from Europe,
Cas:7681-82-5
Min.Order:1 Kilogram
FOB Price: $300.0 / 350.0
Type:Trading Company
inquiryShandong Hanjiang Chemical Co., Ltd.
Hello, dear friend! I'm Hansen and Allen from China. Welcome to my lookchem mall! The following is a brief introduction of our company's products and services. If you are interested in our products, please contact us by emai
Cas:7681-82-5
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryLeader Biochemical Group
PRODUCT DETAILS
Cas:7681-82-5
Min.Order:100 Kilogram
FOB Price: $1.0 / 2.0
Type:Lab/Research institutions
inquiryTriumph International Development Limilted
Appearance:White Powder Storage:kept in a cool, dry and ventilated place Package:As customer request Application:Pharmaceutical Use Transportation:By Sea,by Air,By courier like DHL or Fedx Port:Qingdao/Shanghai,China
Cas:7681-82-5
Min.Order:0 Metric Ton
Negotiable
Type:Lab/Research institutions
inquiryHebei Quanhe Biotechnology Co. LTD
1. Timely and efficient service to ensure communication with customers 2. Produce products of different specifications and sizes according to your requirements. 3. Quality procedures and standards recognized by SGS. Advanced plant equipment ensures
Cas:7681-82-5
Min.Order:1 Kilogram
FOB Price: $30.0 / 50.0
Type:Lab/Research institutions
inquiryHenan Kanbei Chemical Co.,LTD
1. Well-equipped, stable and high-quality raw material supply chain, to provide customers with the best quality products. 2.Strict quality inspection process and professional certificate testing to ensure the quality level of the final product. 3.E
Cas:7681-82-5
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryHangzhou Keyingchem Co.,Ltd
Hangzhou KeyingChem Co., Ltd. exported this product to many countries and regions at best price. If you are looking for the material’s manufacturer or supplier in China, KeyingChem is your best choice. Pls contact with us freely for getting det
Cas:7681-82-5
Min.Order:0 Metric Ton
Negotiable
Type:Lab/Research institutions
inquirySynthetic route

| Conditions | Yield |
|---|---|
| With NaOH In water 20°C (24 h); hydrolysis; | A n/a B 99.5% C 100% |


| Conditions | Yield |
|---|---|
| With NaOH In toluene byproducts: H2; N2 atmospher; treatment of catechol and NaOH in toluene (stirring, 30 min), dropwise addn. of GaI in toluene (4 h); sepn. of NaI (filtration off, drying), collectin of Ga-complex by evapn. of filtrate; | A 97% B n/a |

-
-
108-89-4
picoline

-
-
13510-35-5
indium(III) iodide

-
-
4679-12-3, 61373-02-2
sodium 3,5-di-tert-butyl-o-benzosemiquinolate

-
B
-
7681-82-5
sodium iodide

| Conditions | Yield |
|---|---|
| With NaH In tetrahydrofuran byproducts: H2; N2, soln. of semiquinonate sodium salt added dropwise over 1 h to a soln. of In-salt, stirred for 2 h at room temp., NaI removed by filtrn., excess 4-picoline added to the filtrate; collected, dried, recrystd. (DMF); elem. anal.; further intermediates and other products. discussed; | A 50% B 92% |


| Conditions | Yield |
|---|---|
| In dichloromethane Kinetics; React. of starting complexes (CH2Cl2, 12 h, room temp., anhydrous conditions, dry Ar).; Crystn. from CH2Cl2 under hexane (-15°C), elem. anal., variable-temp. NMR spectroscopy.; | A 88% B n/a |


-
B
-
7681-82-5
sodium iodide

| Conditions | Yield |
|---|---|
| In tetrahydrofuran; toluene N2-atmosphere; stirring (room temp., overnight), evapn. (reduced pressure), toluene addn., stirring (1 h), filtering, concg.; n-hexane addn., crystn. (fridge); elem. anal.; | A 83% B n/a |

-
-
15602-40-1
sodium hexacarbonyltantalate*THF

-
-
18820-83-2
pyridine hydroiodide

-
A
-
112713-58-3
tetracarbonyl{1,2-bis(diphenylphosphino)ethane}iodotantalum

-
B
-
7681-82-5
sodium iodide

| Conditions | Yield |
|---|---|
| With C2H4(P(C6H5)2)2 In tetrahydrofuran; toluene byproducts: C5H5N, CO, H2; Treating of suspn. of diphos and (H-py)I in THF with Ta-complex at room temp. under a CO atmosphere.; Immediate react. with evolution of CO and H2 (gas chromy.) obsd. Stirring (2 h, room temp.), removal of THF (vac., room temp.), dissolving of residue (toluene, CO), stirring (2 h), filtn., treating with n-heptane. Sepn. of microcryst., elem. anal.; | A 79% B n/a |

-
-
15602-39-8
sodium{(THF)2.3} hexacarbonylniobate

-
-
18820-83-2
pyridine hydroiodide

-
A
-
112713-57-2
tetracarbonyl{1,2-bis(diphenylphosphino)ethane}iodoniobium

-
B
-
7681-82-5
sodium iodide

| Conditions | Yield |
|---|---|
| With C2H4(P(C6H5)2)2 In tetrahydrofuran; toluene byproducts: C5H5N, CO, H2; Treating of suspn. of diphos and (H-py)I in THF with Nb-complex at room temp. under a CO atmosphere.; Immediate react. with evolution of CO and H2 (gas chromy.) obsd. Stirring (2 h, room temp.), removal of THF (vac., room temp.), dissolving of residue (toluene, CO), stirring (2 h), filtn., treating with n-heptane. Sepn. of microcryst., elem. anal.; | A 76% B n/a |


-
C
-
7681-82-5
sodium iodide

| Conditions | Yield |
|---|---|
| In hexane dissolved in warm hexane (35°C); filtered, volatiles removed (vac.), extd. (toluene), cooled to -10°C overnight; elem. anal.; | A n/a B 72% C n/a |


| Conditions | Yield |
|---|---|
| In dimethoxyethane (DME) at 20℃; for 1h; | A n/a B n/a C 70% |

-
-
16940-66-2
sodium tetrahydroborate

-
-
33797-51-2
eschenmoser's salt

-
A
-
1231953-83-5
trimethylamine borane

-
B
-
7681-82-5
sodium iodide

| Conditions | Yield |
|---|---|
| In diethyl ether React. of starting materials in Et2O (vac. distd., -196°C), warming to 25°C, and stirring overnight.; Distn. of volatile materials into a -196°C U-trap and fractionalcondensation at -68 and -196°C yields Me3N*BH3 in the -68°C trap.; | A 45.6% B n/a |
| In neat (no solvent) Mixing of starting powders with vigorous stirring at 25°C under N2.; Condensing of volatile materials in a -196°C U-trap, results after 2 h in a small amt. of a solid.; |

-
-
15602-39-8
sodium hexacarbonylniobate*THF

-
A
-
112713-57-2
tetracarbonyl{1,2-bis(diphenylphosphino)ethane}iodoniobium

-
B
-
7681-82-5
sodium iodide

| Conditions | Yield |
|---|---|
| With C2H4(P(C6H5)2)2; I2 In tetrahydrofuran; toluene byproducts: CO; Treating of suspn. of starting compd. in THF with I2 (-78°C, N2 or Ar). Evolution of gas and formation of red soln. takes place upon mixing. Addn. of diphos at -50°C, stirring (room temp.).; Removal of solvent (vac.), dissolving of residue in toluene, filtn., treatment with n-heptane, cooling (-30°C), elem. anal.; | A 43% B n/a |
-
-
15602-40-1
sodium{(THF)2.8} hexacarbonyltantalate

-
A
-
112713-58-3
tetracarbonyl{1,2-bis(diphenylphosphino)ethane}iodotantalum

-
B
-
7681-82-5
sodium iodide

| Conditions | Yield |
|---|---|
| With C2H4(P(C6H5)2)2; I2 In tetrahydrofuran; toluene byproducts: CO; Treating of suspn. of starting compd. in THF with I2 (-78°C, N2 or Ar). Evolution of gas and formation of red soln. takes place upon mixing. Addn. of diphos at -50°C, stirring (1 h, room temp.).; Removal of solvent (vac.), dissolving of brown residue in toluene, filtn., treatment with n-heptane, cooling (-30°C), elem. anal.; | A 42% B n/a |
-
-
15602-41-2
sodium hexacarbonylvanadate

-
A
-
36643-83-1
tetracarbonyl{1,2-bis(diphenylphosphino)ethane}iodovanadium

-
B
-
7681-82-5
sodium iodide

| Conditions | Yield |
|---|---|
| With C2H4(P(C6H5)2)2; I2 In tetrahydrofuran; toluene byproducts: CO; Treating of suspn. of starting compd. in THF with I2 (-78°C, N2 or Ar). Evolution of gas and formation of red soln. takes place upon mixing. Addn. of diphos at -50°C, slow warming of mixt. up to -10°C.; Removal of solvent (vac., -10°C), dissolving of residue in toluene, filtn. (-10°C). Addn. of cold n-heptane causes sepn. of brown solid. Isolation by filtn., recrystn. from toluene/n-heptane, elem. anal.; | A 24% B n/a |

| Conditions | Yield |
|---|---|
| In not given complete reaction on twofold or threefold evaporating Na2SO4 with a mixture of NH4I and NH4Br;; | |
| In not given complete reaction on twofold or threefold evaporating Na2SO4 with a mixture of NH4I and NH4Br;; |


| Conditions | Yield |
|---|---|
| byproducts: PH3, Na5P3O10; at 380.degrre.C in sealed glass tube, in 40-60 min; |


| Conditions | Yield |
|---|---|
| hydrogen sulfide In water Kinetics; catalysts: solid anorganic sulfids and in acids insol. sulfids or H2S in mineralwater; | |
| mercaptoacetic acid In water | |
| pentathionate(2-) In water Kinetics; |


| Conditions | Yield |
|---|---|
| With sodium sulfide In not given byproducts: N2; adding Na2S*9H2O to neutral or acetic acidic soln. of J2 and NaN3;; | |
| With sodium thiosulfate In not given byproducts: N2; adding sodium thiosulfate to neutral or acetic acidic soln. of J2 and NaN3;; | |
| With Na2S*9H2O In not given byproducts: N2; adding Na2S*9H2O to neutral or acetic acidic soln. of J2 and NaN3;; | |
| With sodium thiosulfate In not given byproducts: N2; adding sodium thiosulfate to neutral or acetic acidic soln. of J2 and NaN3;; |

| Conditions | Yield |
|---|---|
| With charcoal; double silicate In neat (no solvent) reduction with charcoal in I2 atmosphere at elevated temperature;; | |
| With pyrographite In neat (no solvent) reduction with charcoal in I2 atmosphere at elevated temperature;; |

| Conditions | Yield |
|---|---|
| In water sulfate precipitates or can be removed with BaI2; no formation of iodate;; | |
| In water sulfate precipitates or can be removed with BaI2; no formation of iodate;; |


| Conditions | Yield |
|---|---|
| In water byproducts: F2; presence of Na2CO3;; |

| Conditions | Yield |
|---|---|
| In not given on presence of water;; |

| Conditions | Yield |
|---|---|
| With Na-amlgam | |
| preparation of very pure crystals; purification by iron iodide method; | |
| In water; Petroleum ether treatment of I2 in petroleum ether (by extractn. of I2-containing solns. generated by electrolysis) with aq. Na2CO3;; | |
| With Na2S2O3 In not given byproducts: Na2SO4, H2O; treating soln. of Na2S2O3 alternating with excess of both I2 and NaOH while heating;; fractionate crystallisation;; | |
| With sodium thiosulfate In not given byproducts: Na2SO4, H2O; treating soln. of Na2S2O3 alternating with excess of both I2 and NaOH while heating;; fractionate crystallisation;; |

| Conditions | Yield |
|---|---|
| In not given treating soln. of NaOH with I2 and heating residue with charcoal;; | |
| In not given treating soln. of NaOH with I2 and heating residue with charcoal;; |


| Conditions | Yield |
|---|---|
| With sulfur dioxide In methanol byproducts: CH3OSO3H; | A >99 B >99 |
| With SO2 In methanol byproducts: CH3OSO3H; | A >99 B >99 |


| Conditions | Yield |
|---|---|
| With sodium hydrogensulfite; sodium sulfite In water boiling I2-containing absorption-coal with Na2CO3-soln., formation of a iodide/iodate-mixture, reduction of iodate with Na2SO3 and NaHSO3;; | |
| With formic acid In neat (no solvent) heating Na2CO3 and I2 in presence of HCO2H;; product free from iodate;; | |
| With sodium sulfite; starch In water absorption of I2 with starch from I2-containing H2O, treatment with Na2CO3 and Na2SO3;; |

| Conditions | Yield |
|---|---|
| In neat (no solvent) byproducts: O2; heating Na2O2 in I2 vapor;; | |
| In neat (no solvent) heating;; | |
| In water concd. aq. soln.;; | |
| In neat (no solvent) byproducts: O2; heating Na2O2 in I2 vapor;; |


| Conditions | Yield |
|---|---|
| In water reaction of aq. Na2S with solid I2;; | |
| In water reaction of aq. Na2S with I2 solution;; | |
| In water reaction of aq. Na2S with I2 solution;; | |
| In water reaction of aq. Na2S with solid I2;; |


| Conditions | Yield |
|---|---|
| In not given in concd. HCl soln., sulfate formed, too; |

-
-
7790-99-0
Iodine monochloride

-
A
-
7757-82-6
sodium sulfate

-
B
-
7681-82-5
sodium iodide

-
C
-
7647-14-5
sodium chloride

| Conditions | Yield |
|---|---|
| In not given in concd. HCl soln.; |


| Conditions | Yield |
|---|---|
| With phosgene In acetone at 20℃; for 0.25h; Reagent/catalyst; | 100% |
| With Fe3+-montmorillonite; propionic acid at 25℃; for 6h; | |
| With propionic acid; K 10-montmorillonite; FeCl3; mixture of, dried at 120 degrees C, grounded at 25℃; for 6h; |
-
-
7681-82-5
sodium iodide

-
-
14900-04-0
triiodide ion

| Conditions | Yield |
|---|---|
| With sodium acetate; acetic acid In ethanol; N,N-dimethyl-formamide Kinetics; byproducts: C6H9(OH)(CON(CH3)C6H10N), H2O; aerobic, 30°C; detected spectrophotometrically; | 100% |
| With methyl α-hydroperoxy-α,α-diphenylacetate; sodium acetate; acetic acid In ethanol; N,N-dimethyl-formamide Kinetics; byproducts: (C6H5)2C(COOCH3)OH, H2O; aerobic, 30°C; detected spectrophotometrically; | 100% |
| With sodium persulfate In decane; water Kinetics; investigation of oxidn. of NaI with Na2S2O8 in H2O/decane/sodium bis(2-ethylhexyl)sulfosuccinate microemulsion followed spectrophotometrically at 355 nm (298 °K, various concns. and ionic strength); |
-
-
97128-27-3
Os(η(6)-p-cymene)Cl2(PMe3)

-
-
7681-82-5
sodium iodide

-
-
97128-36-4
(η(6)-p-cymene)OsI2(trimethylphosphine)

| Conditions | Yield |
|---|---|
| In methanol; dichloromethane 25°C, 6 h, CH2Cl2:CH3OH=20:1; elem. anal.; | 100% |

-
-
7681-82-5
sodium iodide

| Conditions | Yield |
|---|---|
| In acetone The rhodium dichloride complex and NaI (10-fold excess) are dissolved in acetone and refluxed for 0.5 h.; solvent removal under vac., addn. of CH2Cl2, filtn., residual NaI is washed (CH2Cl2), the combined washings are concentrated and layered (hexane), elem. anal.; | 100% |

-
-
7681-82-5
sodium iodide

| Conditions | Yield |
|---|---|
| In dichloromethane; water A CH2Cl2 layer of Ni(HNC2H4NHC3H6NHC2H4N(C16H33)C3H6)(ClO4)2 is equilibrated with an aq. layer of NaI.; The ClO4(1-) ions are quantitatively replaced by the I(1-) ions forming the complex Ni(HNC2H4NHC3H6NHC2H4N(C16H33)C3H6)I2.; | 100% |
-
-
81353-33-5
m-carboran-9-yl(phenyl)iodonium tetrafluoroborate

-
-
7681-82-5
sodium iodide

-
-
81353-34-6
phenyl-9-m-carboranyliodonium iodide

| Conditions | Yield |
|---|---|
| In water; acetone The precipitated iodide was washed with water and ether and dried in the dark. Elem. anal.; | 100% |
-
-
81353-28-8
phenyl-9-o-carboranyliodonium tetrafluoroborate

-
-
7681-82-5
sodium iodide

-
-
81353-29-9
phenyl-9-0-carboranyliodonium iodide

| Conditions | Yield |
|---|---|
| In water; acetone The precipitated iodide was washed with water and ether and dried in the dark. Elem. anal.; | 100% |

-
-
7681-82-5
sodium iodide

-
-
103093-04-5
[C5H5Rh(P(CH3)3)2I](1+)*BF4(1-)=[C5H5Rh(P(CH3)3)2I]BF4

| Conditions | Yield |
|---|---|
| In methanol N2; stirring (55°C, 5 h), cooling (room temp.); solvent removal, dissolving (MeNO2), filtration, pptn. on ether addn., filtration, washing (ether), drying (vac.); | 100% |
-
-
55839-76-4, 173829-27-1, 173829-26-0
diag-(η(5)-C5H4Me)Re(CO)2Br2

-
-
7681-82-5
sodium iodide

-
-
185566-67-0, 185630-64-2
diag-(η(5)-C5H4Me)Re(CO)2I2

| Conditions | Yield |
|---|---|
| In acetone N2-atmosphere; excess NaI; solvent removal, drying (vac., 25°C, then 100-105°C, 18 h); | 100% |

| Conditions | Yield |
|---|---|
| In methanol N2 atmosphere, addn. of NaI to soln. of Ru complex, stirring (room temp., 1 h); evapn. of solvent, dissolution (CH2Cl2), evapn. of solvent, filtration, R(Ru),R(C)/S(Ru),R(C) diastereomers in a ratio of 88:12; elem. anal.; | 100% |

-
-
7681-82-5
sodium iodide

| Conditions | Yield |
|---|---|
| In N,N-dimethyl-formamide mixt. of Pd-complexes and NaI in DMF was stirred for 12 h at 120°C, cooled to ambient temp. under Ar; Et2O was added, washed with H2O, dried over MgSO4, filtered, concd., silica gel with cyclohexane/AcOEt, crystd. by slow evapn. in CH2Cl2 and cyclohexane; | 100% |


| Conditions | Yield |
|---|---|
| In methanol N2 atmosphere, addn. of NaI to soln. of Ru complex, stirring (room temp., 1 h); evapn. of solvent, dissolution (CH2Cl2), evapn. of solvent, filtration, R(Ru),R(C)/S(Ru),R(C) diastereomers in a ratio of 65:35; elem. anal.; | 100% |
-
-
1040922-31-3
Pd(CH3COO)(C5H5N)(C5H4NC6H4NSO2C6H4NO2)

-
-
7681-82-5
sodium iodide

-
-
1176325-74-8
C22H17IN4O4PdS

| Conditions | Yield |
|---|---|
| In ethanol at 23℃; for 0.5h; | 100% |

| Conditions | Yield |
|---|---|
| In acetone (N2); using Schlenk techniques; suspending of (RuI2(η6-C6H5(CH2)4SO3(i-Bu)))2 in acetone with NaI, stirring at reflux; pptn., filtration, washing with EtOH, drying in vac.; | 100% |

-
-
7681-82-5
sodium iodide

| Conditions | Yield |
|---|---|
| In methanol; chloroform at 20℃; for 24h; Inert atmosphere; | 100% |

-
-
15857-57-5
tetraphosphorus decasulfide

-
-
7681-82-5
sodium iodide

| Conditions | Yield |
|---|---|
| In melt at 650℃; for 20h; Sealed tube; | 100% |


-
-
7681-82-5
sodium iodide

| Conditions | Yield |
|---|---|
| In acetone at 20℃; for 22h; Inert atmosphere; Schlenk technique; Glovebox; | 100% |

-
-
7681-82-5
sodium iodide

| Conditions | Yield |
|---|---|
| In dichloromethane byproducts: NaCl; Ar-atmosphere; stirring for 24 h;; chromd. (silica gel); evapd. dried for 14 h in vacuo; elem. anal.;; | 99% |

-
-
7681-82-5
sodium iodide

| Conditions | Yield |
|---|---|
| In tetrahydrofuran for 3h; Reflux; | 99% |
| In acetone The rhodium dichloride complex and NaI (10-fold excess) are dissolved in acetone and refluxed.; |
-
-
130214-50-5
trans-{PdCl2(1C-diphenylphosphinomethyl-2C-methyl-o-carborane)2

-
-
7681-82-5
sodium iodide

| Conditions | Yield |
|---|---|
| In propan-1-ol refluxing Pd-complex and 10-fold excess of NaJ in propanol until soln. fading, pptn. on cooling; filtration, washing (water, MeOH, hexane), drying; elem. anal.; isomer product mixt. not sepd.; | 99% |

| Conditions | Yield |
|---|---|
| In tetrahydrofuran under inert atm.; soln. of complex and NaI stirred for 12 h; evapd. (vac.), dissolved in CH2Cl2, filtered, concd., addn. of hexane, crystn.; | 99% |

| Conditions | Yield |
|---|---|
| In acetone inert atmosphere; refluxed; 15 h;; evapd.; dissolved in CH2Cl2; chromd. (SiO2, CH2Cl2, then acetone); HPLC (Chiracel, hexane/i-propanol);; | A n/a B 1% C 99% |


-
-
7681-82-5
sodium iodide

-
-
112317-58-5, 85931-02-8
cyclopentadienyl-1,2-diphenylvinyl-iodid-triisopropylphosphine-rhodium(III)

| Conditions | Yield |
|---|---|
| In acetone under Ar, NaI mixed in acetone with Rh complex; | 99% |
-
-
158414-97-2, 16985-53-8
carbonylchlorodiiodobis(triethylphosphine)rhodium(III)

-
-
7681-82-5
sodium iodide

-
-
158367-94-3
carbonyltriiodobis(triethylphosphine)rhodium(III)

| Conditions | Yield |
|---|---|
| In acetone inert atmosphere; stirring Rh-complex with 5 equiv. of NaI (room temp., 15 min); concn., addn. of light petroleum, filtration, concn., addn. of Et2O, crystn. (-30°C, 48 h), filtration at -40°C; elem. anal.; | 99% |
-
-
23895-60-5
tri-n-butylstannyldibromomethane

-
-
7681-82-5
sodium iodide

-
-
213203-63-5
tri-n-butyl(diiodomethyl)stannane

| Conditions | Yield |
|---|---|
| In acetone Ar; NaI addn. to stirred Sn-compd. soln. at room temp., mixt. evapn. after 18 h (reduced pressure); residue diln. (hexane), filtration, concn. (reduced pressure), diln. (CH2Cl2), filtration, concn. (reduced pressure); | 99% |
-
-
194346-40-2
cis-[bis(1,3,5-triaza-7-phosphaadamantane)dichloroplatinum(II)]

-
-
7681-82-5
sodium iodide

-
-
330805-47-5, 852447-80-4
trans-diiodobis(1,3,5-triaza-7-phosphaadamantane)platinum(II)

| Conditions | Yield |
|---|---|
| In water addn. of aq. soln. of NaI to aq. soln. of platinum complex; crystn. by slow evapn. of solvent; | 99% |
-
-
198629-52-6
[PtClMe(2,9-dimethyl-1,10-phenanthroline)(CH2=CHOH)]

-
-
7681-82-5
sodium iodide

-
-
198629-60-6
[PtIMe(2,9-dimethyl-1,10-phenanthroline)(CH2=CHOH)]

| Conditions | Yield |
|---|---|
| In chloroform slight excess of NaI, stirring at room temp. for 1 h; sepn. of org. layer, washing (water), filtration over Na2SO4, solvent removal (vac.); | 99% |

-
-
7681-82-5
sodium iodide

| Conditions | Yield |
|---|---|
| In not given byproducts: NaCl, pyridine; under Ar or N2; within 2 h at 25°C; | 99% |

-
-
7681-82-5
sodium iodide

| Conditions | Yield |
|---|---|
| In N,N-dimethyl-formamide (N2); stirring (1 h), filtration, layering with ether, standing (-20°C, overnight), filtration; extn. (CH3CN), taking to dryness, recrystn. (DMF, diethyl ether); elem. anal.; | 99% |
Related products
Raw Materials
Downstream Products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View



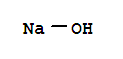


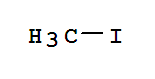

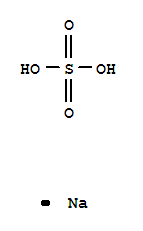
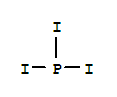




 Xi
Xi