Triumph International Development Limilted
Product name: sodium metabisulfite / sodium metabisulphite Molecular weight: 190.10 Specification: 25Kg woven bag Quality guarantee period: 6 months Appearance:white powder/crystal Storage:Store in a cool,dry place and keep away from direct
Cas:7647-15-6
Min.Order:10 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryKunshan Push Trading Co.,LTD
ADVANTAGE 1.Professional: More than 10 years chemical exporting experience. We have produced chemical more than fifteen years, 95% products are for export . More than 10 years chemical exporting experience. Good and stabilized factory price. 2.
COLORCOM LTD.
Colorcom is a global leader in industrial chemical manufacturing and is continuously innovating and transforming to exceed client expectations and industry standards. Colorcom prides itself on superior customer and technical focus, while focusing on
Jinan Finer Chemical Co., Ltd
Product Description Product website: http://www.finerchem.com/pro01en/id/2107.html Product Name Sodium bromide CAS No. 7647-15-6 Ap
Cas:7647-15-6
Min.Order:1 Gram
FOB Price: $2.0
Type:Lab/Research institutions
inquirytaicang liyuan chemical co,.ltd
Established in 2007,Taicang Liyuan Chemical Co.,ltd. is located in Taicang city with a rich cultural heritage in most developed economy area of China's Yangtze River Delta.The location of our company is superior with convenient transportation a
Yixing Zeyuan Chemical Technology Co Ltd
We can supply: 1..Place of Origin China with Super quality by reasonable price. 2. sample orders are welcome 3.Customer Satisfaction is our priority and guarantee for the future. 4.Fast delivery by safe express way to all the country. 5.Has a
Cas:7647-15-6
Min.Order:1 Kilogram
Negotiable
Type:Other
inquiryShanghai SMEC Trading Co., Ltd.
Used for making sensitive emulsion of photographic film and for making medicinal intermediates in medical-industries. E.g.diuretic.sedative etc. It is also used as bromine making complex material and complex dyes. Also for water treatment and oil p
Dayang Chem (Hangzhou) Co.,Ltd.
DayangChem exported this product to many countries and regions at best price. If you are looking for the material's manufacturer or supplier in China, DayangChem is your best choice. Pls contact with us freely for getting detailed product spe
Cas:7647-15-6
Min.Order:1 Kilogram
FOB Price: $3.0
Type:Lab/Research institutions
inquiryHangzhou Dingyan Chem Co., Ltd
Item Specifications Test Result Characteristics Colorless transparent liquid Conform Assay
Cas:7647-15-6
Min.Order:1 Kilogram
FOB Price: $3.0 / 10.0
Type:Manufacturers
inquirySimagchem Corporation
Welcome to Simagchem, your partner in China as a premier supply of bulk specialty chemicals for industry and life science. We introduce experienced quality product and exceptional JIT service with instant market intelligence in China to benefit our
Cas:7647-15-6
Min.Order:0 Metric Ton
Negotiable
Type:Manufacturers
inquiryHebei yanxi chemical co.,LTD.
Hebei yanxi chemical co., LTD is a professional research, development and production of lead acetate benzene acetamide enterprise backbone members by local well-known entrepreneurs and professional senior engineers in the party's "low carb
Hefei TNJ chemical industry co.,ltd
ISO Appearance:white powder Storage:Keep in a cool, well-ventilated area. Package:25kg/drum,,25kg/bag Application: used for making sensitive emulsion of photographic film and for making medicinal intermediates in medico-industries, e.g. diuretic ,
Henan Allgreen Chemical Co.,Ltd
T he company has advanced technology, as well as a large number of excellent R & D team, to provide customers from the grams to one hundred kilograms and tons of high-quality products, competitive prices and quality service Appearance:White
Ality Chemical Corporation
The above product is Ality Chemical's strong item with best price, good quality and fast supply. Ality Chemical has been focusing on the research and production of this field for over 14 years. At the same time, we are always committed to providi
EAST CHEMSOURCES LIMITED
The photosensitive industry is used to prepare photographic sensitizer. Pharmaceuticals are used for the production of diuretics and sedatives. The perfume industry is used for the production of synthetic flavors. Used as bromination agent in print
Cas:7647-15-6
Min.Order:1 Metric Ton
Negotiable
Type:Manufacturers
inquiryXi'an Xszo Chem Co., Ltd.
1. Factory price and high quality must be guaranteed, base on 8 years of production and R&D experience2. Free samples will be provided,ensure specifications and quality are right for customer3. Customers will receive the most professional technical s
Cas:7647-15-6
Min.Order:1 Gram
FOB Price: $0.1
Type:Manufacturers
inquiryWeifang Yukai Chemical Co.,Ltd.
Our factory was established in 2006, with rich production experience and 60% market share, serving the world's largest multinational corporations. The factory is located in the national economic development zone with stable production. Sodiu
Cas:7647-15-6
Min.Order:1 Metric Ton
Negotiable
Type:Manufacturers
inquiryShanxi Zorui Biotechnology Co.Ltd.
Zorui combines R&D, production and sales into its operations, While continuously providing high-quality raw materials, we also provide and optimize technical solutions for customers to achieve mutual benefit. We adhere to the "quality, integ
Cas:7647-15-6
Min.Order:1 Kilogram
Negotiable
Type:Other
inquiryChemwill Asia Co., Ltd.
Our main production base is located in Xuzhou industry park. We are certified both to the ISO 9001 and ISO 14001 Standards, have a safety management system in place.Our R&D team masters core technology for process-design of target building block
Hebei Nengqian Chemical Import and Export Co., LTD
With our good experience, we offer detailed technical support and advice to assist customers. We communicate closely with customers to establish their quality requirements. Consistent Quality Our plant has strict quality control in each manufactu
Cas:7647-15-6
Min.Order:1 Kilogram
FOB Price: $1.0 / 10.0
Type:Trading Company
inquiryLIDE PHARMACEUTICALS LIMITED
We are a professional chemicals, APIs and plant extract leading manufacturer in China. We are specialized in chemical synthesis, process development of pharmaceutical intermediates, active pharmaceutical ingredients (APIs), plant extract and rare c
Cas:7647-15-6
Min.Order:1 Gram
FOB Price: $1.0
Type:Lab/Research institutions
inquiryHenan Tianfu Chemical Co., Ltd.
Our advantage:Our company was built in 2009 with an ISO certificate.In the past 5 years, we have grown up as a famous fine chemicals supplier in China and we had established stable business relationships with Samsung,LG,Merck,Thermo Fisher Scient
Cas:7647-15-6
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryZhuozhou Wenxi import and Export Co., Ltd
WITH US,YOUR MONEY IN SAFE,YOUR BUSINESS IN SAFE 1)Quick Response Within 12 hours; 2)Quality Guarantee: All products are strictly tested by our QC, confirmed by QA and approved by third party lab in China, USA, Canada, Germany, UK, Italy, France et
Cas:7647-15-6
Min.Order:10 Gram
FOB Price: $146.0 / 176.0
Type:Trading Company
inquiryWuhan Han Sheng New Material Technology Co.,Ltd
Our Advantage: high quality with competitive price High quality standard: BP/USP/EP Enterprise standard All purity customized Fast and safe delivery We have reliable forwarder who can help us deliver our goods more fast and safe. We
Henan Sinotech Import&Export Corporation
Chemical Name: NaBr; Bromide salt of sodium; Sedoneural; Sodium bromide Molecular formula: NaBr Molecular weight: 102.9 CAS NO.: 7647-15-6 Use:This product is used for making sensitive emulsion of photographic film and for making medicinal i
Hubei Langyou International Trading Co., Ltd
Advantages: Hubei XinRunde Chemical Co., Ltd is a renowned pharmaceutical manufacturer. We can offer high quality products at competitive price in quick delivery with 100% custom pass guaranteed. Never stop striving to offer our best service is
Shanghai Upbio Tech Co.,Ltd
1.No Less 8 years exporting experience. Clients can 100% received goods 2.Lower Price with higher quality 3,Free sample 4,We are sincerely responsible for the "product quality" and "After Service" Upbio is Specialized
Cas:7647-15-6
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryXi'an Faithful Biotech Co., Ltd.
We are the manufacturers and suppliers of API in China, and warehouse in Germany and USA of California, which can quickly and safely deliver to your address 1.High quality and competitive price. 2.Free sample for your evaluation. 3.Promptly delivery
Cas:7647-15-6
Min.Order:10 Gram
FOB Price: $3.5
Type:Trading Company
inquiryHANWAYS CHEMPHARM CO.,LIMITED
Wuhan hanweishi Pharmchem Co., Ltd(Hanways chempharm) is a specialized company concentrating on the R&D, production, marketing and technical service of APIs and pharmaceutical intermediates. The leading products range are APIs, Hormones, Peptides
Cas:7647-15-6
Min.Order:100 Kilogram
FOB Price: $1.0 / 5.0
Type:Trading Company
inquiryBaoji Guokang Healthchem co.,ltd
Our company has been in existence for 10 years since its establishment. We have our own unique team. The company integrates independent research and development, production and sales. We have established famous brands at home and abroad. At presen
Cas:7647-15-6
Min.Order:1 Kilogram
FOB Price: $16.0 / 20.0
Type:Trading Company
inquirySynthetic route

-
-
124-41-4
sodium methylate

-
A
-
74775-97-6
3-methoxy-7-n-butylaminomethyl-3-boracyclo[3.3.1]-nonane

-
B
-
7647-15-6
sodium bromide

| Conditions | Yield |
|---|---|
| In methanol (dry Ar); to the B-compound in MeOH added soln. of NaOCH3 in MeOH at 20°C; refluxed for 30 min; solvent removed in vacuo; ether added; NaBr filtered; washed with ether; ether sol. evapd. in vacuo; recrystd.; elem. anal.; | A 66% B 99.5% |

-
-
1067-10-3
Bromotriethylgermanium

-
-
4679-12-3, 61373-02-2
sodium 3,5-di-tert-butyl-o-benzosemiquinolate

-
A
-
3383-21-9
3,5-di-tert-butyl-o-benzoquinone

-
B
-
51639-37-3
C6H2(C(CH3)3)2(OGe(C2H5)3)2

-
C
-
7647-15-6
sodium bromide

| Conditions | Yield |
|---|---|
| In tetrahydrofuran mixing of solns. (20°C); removal of THF (vavc.), addn. of hexane, pptn. and sepn. of NaBr (filtration off), crystn. of quinone (low temp.); (t-Bu)2C6H2(OGeEt3)2 in mother liquor not isolated; GLC; | A 36% B 47% C 98% |


| Conditions | Yield |
|---|---|
| In dichloromethane Kinetics; Stirring of mixt. of starting complexes in CH2Cl2 at reflux under anhydrous conditions in a dry Ar atmosphere (24 h).; Filtn. of react. mixt., removal of solvent by trap-to-trap distn., crystn. of crude resulting material from CH2Cl2 under hexane, elem. anal., variable-temp. NMR spectroscopy.; | A 98% B n/a |

-
-
74789-86-9
3-methoxy-7-bromomethyl-3-borabicyclo[3.3.1]nonane

-
-
124-41-4
sodium methylate

-
A
-
74789-87-0
C8H13(CH2OCH3)B(OCH3)

-
B
-
7647-15-6
sodium bromide

| Conditions | Yield |
|---|---|
| In methanol (dry Ar); heated for 1.5 h at 50°C; MeOH vac. distd.; residue treated isopentane; ppt. NaBr filtered; filtrate distn.; elem. anal.; | A 84% B 98% |


| Conditions | Yield |
|---|---|
| In benzene (glovebox); addn. of a soln. of AlBr3 in C6H6 to a soln. of silicon compd. in C6H6 in vac. with stirring, sealing, heating to 40°C for 30min with shaking; centrifugation, decantation, evapn; elem. anal.; | A 98% B 98% |

-
-
13314-53-9
diethylgermanium dibromide

-
-
4679-12-3, 61373-02-2
disodium 3,5-di-t-butyl-1,2-pyrocatecholate

-
A
-
78100-52-4
2,2-diethyl(6,8-di-tert-butyl)-4,5-benzo-2-germa-1,3-dioxolane

-
B
-
7647-15-6
sodium bromide

| Conditions | Yield |
|---|---|
| In diethyl ether 3 h at 20°C; NaBr washed out (water); ether evapd.; recrystd. (acetonitrile); elem. anal.; | A 87% B 95% |


| Conditions | Yield |
|---|---|
| With sodium dithionite; sodium hydrogencarbonate In water; acetonitrile at 35 - 65℃; for 5.5h; Product distribution / selectivity; | A 94% B n/a |
| With disodium hydrogenphosphate; sodium dithionite In water; acetonitrile at 35 - 65℃; Product distribution / selectivity; | A 84% B n/a |

-
-
105260-39-7
triisopropylgermylmercuric bromide

-
B
-
24004-54-4
bis(triisopropylgermyl)mercury

-
C
-
7647-15-6
sodium bromide

| Conditions | Yield |
|---|---|
| In tetrahydrofuran; hexane addn. of Na-compound (prepared from (C5H5)Fe(CO)3 and excess 3% Na-amalgam) in THF to i-Pr3GeHgBr (prepared from (i-PrGe)2Hg and Br2) in hexane at -40°C; pptn. (NaBr), replacing of solvents in the mother-liquor by hexane, pptn. of Fe-compound, isolation of (i-Pr3Ge)2Hg by fractionation of the soln. in vac.; | A 90% B 93% C 71% |


| Conditions | Yield |
|---|---|
| With 1,3-dioxolane In further solvent(s) (N2); salts mixt. (1/1 molar ratio) stirred at 25-74°C for 1-48 h in 1,3-dioxolane; heated (vac.); | A 92% B n/a |
| With 1,2-dimethoxyethane In 1,2-dimethoxyethane (N2); salts mixt. (1/1 molar ratio) stirred at 25-85°C for 1-36 h in monoglyme; heated (vac.); | A >99 B n/a |
| With C4H8O In tetrahydrofuran (N2); salts mixt. (1/1 molar ratio) stirred at 25-67°C for 1-48 h in THF; heated (vac.); | |
| With diethyl ether In diethyl ether (N2); salts mixt. (1/1 molar ratio) stirred at 25-35°C for 1-32 h in Et2O; heated (vac.); | A 6-100 B n/a |

-
-
13314-53-9
diethylgermanium dibromide

-
-
4679-12-3, 61373-02-2
sodium 3,5-di-tert-butyl-o-benzosemiquinolate

-
A
-
78100-52-4
2,2-diethyl(6,8-di-tert-butyl)-4,5-benzo-2-germa-1,3-dioxolane

-
B
-
3383-21-9
3,5-di-tert-butyl-o-benzoquinone

-
C
-
7647-15-6
sodium bromide

| Conditions | Yield |
|---|---|
| In diethyl ether Na-:Ge-compound 2:1, 3 h at 20°C; ether replaced with hexane; NaBr washed out (water); recrystd. (acetonitrile); | A 69% B 87% C 92% |

-
-
108-89-4
picoline

-
-
13465-09-3
indium tribromide

-
-
4679-12-3, 61373-02-2
sodium 3,5-di-tert-butyl-o-benzosemiquinolate

-
B
-
7647-15-6
sodium bromide

| Conditions | Yield |
|---|---|
| In tetrahydrofuran byproducts: H2; N2, blue soln. of semiquinone sodium salt added dropwise over 1 h to a soln. of In-salt, stirred for 2 h at room temp., NaBr removed from intermediate product by filtrn., excess 4-picoline added to the filtrate; collected, dried, recrystd. (toluene/THF); elem. anal.; intermediate and formn. of In(I) products also discussed; | A 50% B 92% |


-
-
57-13-6
urea

-
A
-
124-38-9
carbon dioxide

-
B
-
7727-37-9
nitrogen

-
C
-
7732-18-5
water

-
D
-
7647-15-6
sodium bromide

| Conditions | Yield |
|---|---|
| A 90% B 90% C 90% D 90% |

-
-
1067-10-3
Bromotriethylgermanium

-
-
4679-12-3, 61373-02-2
sodium 3,5-di-tert-butyl-o-benzosemiquinolate

-
A
-
78100-52-4
2,2-diethyl(6,8-di-tert-butyl)-4,5-benzo-2-germa-1,3-dioxolane

-
B
-
3383-21-9
3,5-di-tert-butyl-o-benzoquinone

-
C
-
51639-37-3
C6H2(C(CH3)3)2(OGe(C2H5)3)2

-
D
-
7647-15-6
sodium bromide

| Conditions | Yield |
|---|---|
| In diethyl ether 3 h at 20°C; ether replaced with hexane; NaBr washed out (water); recrystd. (hexane); | A 12% B 63% C 88% D 90% |

-
-
141-52-6
sodium ethanolate

-
-
60458-17-5
methyldibromobismuth(III)

-
-
60-24-2
2-hydroxyethanethiol

-
A
-
60458-10-8
CH3Bi(SCH2CH2OH)2

-
B
-
7647-15-6
sodium bromide

| Conditions | Yield |
|---|---|
| 20°C, equiv. amt. of HSCH2C6H5; NaCl filtered off, the solvent evapd. in vac.; | A 88% B n/a |

-
-
141-52-6
sodium ethanolate

-
-
60458-17-5
methyldibromobismuth(III)

-
A
-
60458-07-3
methyldiethoxybismutane

-
B
-
7647-15-6
sodium bromide

| Conditions | Yield |
|---|---|
| In ethanol abs. ethanol, 24 h, -25°C; | A 87% B n/a |

-
-
13755-29-8
sodium tetrafluoroborate

-
B
-
7647-15-6
sodium bromide

| Conditions | Yield |
|---|---|
| In water at 20℃; for 1h; | A 87% B n/a |


| Conditions | Yield |
|---|---|
| With sodium dithionite; sodium hydrogencarbonate In water; acetonitrile at 35 - 65℃; for 6h; Product distribution / selectivity; | A 86% B n/a |
-
-
141-52-6
sodium ethanolate

-
-
60458-17-5
methyldibromobismuth(III)

-
-
100-53-8
phenylmethanethiol

-
A
-
60458-09-5
Bis-(benzylthio)-methylbismutin

-
B
-
7647-15-6
sodium bromide

| Conditions | Yield |
|---|---|
| 20°C, equiv. amt. of HSCH2C6H5; NaCl filtered off, the solvent evapd. in vac.; | A 83% B n/a |

-
-
141-52-6
sodium ethanolate

-
-
60458-17-5
methyldibromobismuth(III)

-
-
108-98-5
thiophenol

-
A
-
28609-60-1
Methyl-bis-(phenylthio)-bismutin

-
B
-
7647-15-6
sodium bromide

| Conditions | Yield |
|---|---|
| 20°C, equiv. amt. of thiophenol; NaCl filtered off, the solvent evapd. in vac.; | A 80% B n/a |

-
-
13755-29-8
sodium tetrafluoroborate

-
B
-
7647-15-6
sodium bromide

| Conditions | Yield |
|---|---|
| In water at 20℃; for 1h; | A 80% B n/a |

-
-
15602-39-8
sodium{(THF)2.3} hexacarbonylniobate

-
-
18820-82-1
Pyridine hydrobromide

-
A
-
112713-60-7
tetracarbonyl{1,2-bis(diphenylphosphino)ethane}bromoniobium

-
B
-
7647-15-6
sodium bromide

| Conditions | Yield |
|---|---|
| With C2H4(P(C6H5)2)2 In tetrahydrofuran; toluene byproducts: C5H5N, CO, H2; Treating of suspn. of diphos and (H-py)Br in THF with Nb-complex at room temp. under a CO atmosphere.; Immediate react. with evolution of CO and H2 (gas chromy.) obsd. Stirring (2 h, room temp.), removal of THF (vac., room temp.), dissolving of residue (toluene, CO), stirring (2 h), filtn., treating with n-heptane. Sepn. of microcryst., elem. anal.; | A 78% B n/a |


| Conditions | Yield |
|---|---|
| In tetrahydrofuran A mixt. of BrCH2C6H4CH2Br and Na(MO(cp)(CO)3) in THF is stirred at roomtemp. for 1.5 h.; The soln. is filtered to remove NaBr, concd. in vacuo and treated with methanol to give yellow crystals of (Mo(cp)(CO)3)CH2C6H4CH2((CO)3(cp)Mo); | A 76% B n/a |


| Conditions | Yield |
|---|---|
| In tetrahydrofuran inert atmosphere; stirring (room temp., 4 h, pptn.); filtration, washing (THF), extn. (MeCN), partial evapn. of CH3CN soln., pptn. on Et2O addn., washing (Et2O), drying (vac., 1 h); elem. anal.; | A 75% B n/a |


| Conditions | Yield |
|---|---|
| In tetrahydrofuran inert atmosphere; stirring (room temp., 4 h, pptn.); filtration, washing (THF), extn. (MeCN), partial evapn. of CH3CN soln., pptn. on Et2O addn., washing (Et2O), drying (vac., 1 h); | A 74% B n/a |


| Conditions | Yield |
|---|---|
| In tetrahydrofuran inert atmosphere; stirring (room temp., 4 h, pptn.); filtration, washing (THF), extn. (MeCN), partial evapn. of CH3CN soln., pptn. on Et2O addn., washing (Et2O), drying (vac., 1 h); elem. anal.; | A 71% B n/a |

-
-
108-89-4
picoline

-
-
115381-20-9
HONC(CH3)C(CH3)NOCH2ONC(CH3)C(CH3)NOCoBr2

-
-
256460-68-1
HONC(CH3)C(CH3)NOCH2ONC(CH3)C(CH3)NOCo(NC5H4CH3)Br(1+)*ClO4(1-)=[HONC(CH3)C(CH3)NOCH2ONC(CH3)C(CH3)NOCo(NC5H4CH3)Br]ClO4

-
B
-
7647-15-6
sodium bromide

| Conditions | Yield |
|---|---|
| In methanol; water addn. of satd. aq. soln. of NaClO4 to hot suspn. of Co complex in MeOH, addn. of stoichiom. amt. of substituted pyridine in MeOH (stirring), refluxing (water bath, 4 h), pptn. on volume reduction and standing (35°, 2 h); filtration, washing (cold water, MeOH, Et2O), drying (vac.); elem. anal.; | A 69.7% B n/a |

-
-
141-86-6
2,6-Diaminopyridine

-
-
115381-20-9
HONC(CH3)C(CH3)NOCH2ONC(CH3)C(CH3)NOCoBr2

-
-
256460-77-2
HONC(CH3)C(CH3)NOCH2ONC(CH3)C(CH3)NOCo(N3C5H7)Br(1+)*ClO4(1-)=[HONC(CH3)C(CH3)NOCH2ONC(CH3)C(CH3)NOCo(N3C5H7)Br]ClO4

-
B
-
7647-15-6
sodium bromide

| Conditions | Yield |
|---|---|
| In methanol; water addn. of satd. aq. soln. of NaClO4 to hot suspn. of Co complex in MeOH, addn. of stoichiom. amt. of substituted pyridine in MeOH (stirring), refluxing (water bath, 4 h), pptn. on volume reduction and standing (35°, 2 h); filtration, washing (cold water, MeOH, Et2O), drying (vac.); elem. anal.; | A 69.5% B n/a |

-
-
109-06-8
α-picoline

-
-
115381-20-9
HONC(CH3)C(CH3)NOCH2ONC(CH3)C(CH3)NOCoBr2

-
-
256460-65-8
HONC(CH3)C(CH3)NOCH2ONC(CH3)C(CH3)NOCo(NC5H4CH3)Br(1+)*ClO4(1-)=[HONC(CH3)C(CH3)NOCH2ONC(CH3)C(CH3)NOCo(NC5H4CH3)Br]ClO4

-
B
-
7647-15-6
sodium bromide

| Conditions | Yield |
|---|---|
| In methanol; water addn. of satd. aq. soln. of NaClO4 to hot suspn. of Co complex in MeOH, addn. of stoichiom. amt. of substituted pyridine in MeOH (stirring), refluxing (water bath, 4 h), pptn. on volume reduction and standing (35°, 2 h); filtration, washing (cold water, MeOH, Et2O), drying (vac.); elem. anal.; | A 67.9% B n/a |

-
-
141-52-6
sodium ethanolate

-
-
60458-17-5
methyldibromobismuth(III)

-
-
75-08-1
ethanethiol

-
A
-
60458-08-4
Methyldi(ethylthio)bismutan

-
B
-
7647-15-6
sodium bromide

| Conditions | Yield |
|---|---|
| 20°C, equiv. amt. of C2H5SH; NaCl filtered off, the solvent evapd.in vac.; | A 67% B n/a |

-
-
15602-40-1
sodium hexacarbonyltantalate*THF

-
-
18820-82-1
Pyridine hydrobromide

-
A
-
112713-59-4
tetracarbonyl{1,2-bis(diphenylphosphino)ethane}bromotantalum

-
B
-
7647-15-6
sodium bromide

| Conditions | Yield |
|---|---|
| With C2H4(P(C6H5)2)2 In tetrahydrofuran; toluene byproducts: C5H5N, CO, H2; Treating of suspn. of diphos and (H-py)Br in THF with Ta-complex at room temp. under a CO atmosphere.; Immediate react. with evolution of CO and H2 (gas chromy.) obsd. Stirring (2 h, room temp.), removal of THF (vac., room temp.), dissolving of residue (toluene, CO), stirring (2 h), filtn., treating with n-heptane. Sepn. of microcryst., elem. anal.; | A 66% B n/a |


| Conditions | Yield |
|---|---|
| In sulfuric acid NaBr addn. to NH4ReO4 soln. in concd. H2SO4 (crystn.); extn. (toluene), distn. off of toluene; elem. anal.; | 100% |
| In sulfuric acid byproducts: HBr, Br2; dissoln. of NH4ReO4 in 98% H2SO4, dropwise addn. of 20-50 fold excess NaBr, stirring (20-30 min); other alkali metal bromides can be used instead of NaBr; extn. (toluene), distn. off of solvent (vac., room temp.); elem. anal.; | 99% |

-
-
7647-15-6
sodium bromide

-
-
128026-89-1, 128058-84-4
(1-hexadecyl-1,4,8,11,-tetra-azacyclotetradecane)nickel(II) bromide

| Conditions | Yield |
|---|---|
| In dichloromethane; water A CH2Cl2 layer of Ni(HNC2H4NHC3H6NHC2H4N(C16H33)C3H6)(ClO4)2 is equilibrated with an aq. layer of NaBr.; The ClO4(1-) ions are quantitatively replaced by the I(1-) ions forming the complex Ni(HNC2H4NHC3H6NHC2H4N(C16H33)C3H6)Br2.; | 100% |
-
-
721944-47-4
chloro{2-(dimethylaminomethyl)phenyl}phenylbismuthine

-
-
7647-15-6
sodium bromide

-
-
721944-48-5
bromo{2-(dimethylaminomethyl)phenyl}phenylbismuthine

| Conditions | Yield |
|---|---|
| In water; benzene Ar; NaBr in water added to Cl(C6H4CH2N(CH3)2)Bi(C6H5)Cl in benzene; organic layer extd. with ethyl acetate; ext. evapd. under reduced pressure; crystn. from hexane/CH2Cl2 gave crystals; | 100% |

| Conditions | Yield |
|---|---|
| With tetrafluoroboric acid In water; toluene under Ar; a soln. of the starting complex in toluene is added to NaBr in aq. HBF4, mixt. stirred for 30 min; the toluene layer is separated, evapd.; the solid is treated with water-toluene, the organic layer is separated, evapd.; the residue is dissolved in toluene, hexane is added; elem. anal.; | 100% |

-
-
7647-15-6
sodium bromide

| Conditions | Yield |
|---|---|
| In N,N-dimethyl-formamide mixt. of Pd-complexes and NaBr in DMF was stirred for 12 h at 120°C under Ar, cooled to ambient temp.; diluted with Et2O, washed with H2O, dried over MgSO4, filtered, concd., silica gel with cyclohexane/AcOEt; elem. anal.; | 100% |


-
-
7647-15-6
sodium bromide

| Conditions | Yield |
|---|---|
| With AgBF4 In methanol; dichloromethane | 100% |

-
-
7647-15-6
sodium bromide

-
-
1447423-46-2
[(2,6-bis-(di-tert-butylphosphinomethyl)pyridine)RhBr]

| Conditions | Yield |
|---|---|
| In acetone at 20℃; for 1h; Inert atmosphere; Schlenk technique; Glovebox; | 100% |
-
-
113214-83-8
cis-dichlorobis(diphenylvinylphosphine)palladium(II)

-
-
7647-15-6
sodium bromide

-
-
113214-84-9
trans-dibromobis(diphenylvinylphosphine)palladium(II)

| Conditions | Yield |
|---|---|
| In methanol; dichloromethane; water Addn. of a CH3OH/H2O soln. of NaBr to a CH2Cl2 soln. of dichlorobis(diphenylvinylphosphine)palladium(II).; Elem.anal.; | 99.5% |
-
-
198629-52-6
[PtClMe(2,9-dimethyl-1,10-phenanthroline)(CH2=CHOH)]

-
-
7647-15-6
sodium bromide

-
-
198629-59-3
[PtBrMe(2,9-dimethyl-1,10-phenanthroline)(CH2=CHOH)]

| Conditions | Yield |
|---|---|
| In chloroform slight excess of NaBr, stirring at room temp. for 1 h; sepn. of org. layer, washing (water), filtration over Na2SO4, solvent removal (vac.); | 99% |
-
-
579490-55-4
chloro[bis(2-diphenylphosphinophenyl)amide]nickel(II)

-
-
7647-15-6
sodium bromide

-
-
881020-83-3
[N(o-C6H4PPh2)2]NiBr

| Conditions | Yield |
|---|---|
| In acetone under N2 atm. suspn. NaBr in acetone was added to soln. Ni complex in acetone and stirred at room temp. for 24 h; react. mixt. was evapd. to dryness in vacuo, residue was extd. with CH2Cl2, filtered through Celite, and evapd. in vacuo; elem. anal.; | 99% |

-
-
7647-15-6
sodium bromide

| Conditions | Yield |
|---|---|
| In not given dissoln. of Cr-complex in 0.01 M HCl (30°C), pptn. on addn. of satd. NaBr and slow cooling to 0°C; elem. anal.; | 99% |

-
-
7647-15-6
sodium bromide

| Conditions | Yield |
|---|---|
| In dichloromethane 1) NaBr (excess), 2) (Cp2Fe)PF6 (2 equiv), room temp.; | 99% |


-
-
7647-15-6
sodium bromide

| Conditions | Yield |
|---|---|
| In methanol addn. of NaBr to methanolic soln. of ruthenium compd., heating at 50°C for 20 h; NMR; | 99% |

-
-
1070260-80-8
[Pd(C6H2(CH2SPh)-2,6-(OH)-4)(MeCN)]BF4

-
-
7647-15-6
sodium bromide

-
-
1070260-64-8
[PdBr(C6H2(CH2SPh)-2,6-(OH)-4)]

| Conditions | Yield |
|---|---|
| In water; acetonitrile (under N2, Schlenk); aq. soln. of NaBr added to MeCN soln. of Pd-complex, stirred for 2 h at ambient temp.; concd. to removed volatiles, residue washed with H2O, dried in vacuo; elem. anal.; | 99% |

-
-
7647-15-6
sodium bromide

| Conditions | Yield |
|---|---|
| In dichloromethane; acetone (under Ar, Schlenk); NaBr added to soln. of Ru-complex in CH2Cl2-acetone1:1, stirred for 4 h at room temp.; volatiles evapd., silica column chromy. with CH2Cl2-acetone 95:5, recrystd. from CH2Cl2-pentane at -20°C; elem. anal.; | 99% |
-
-
110-86-1
pyridine

-
-
1450713-40-2
(S)-4-benzyl-3-(biphenyl-4-ylmethyl)-1-(2,6-diisopropylphenyl)-4,5-dihydro-1H-imidazol-3-ium bromide

-
-
7647-15-6
sodium bromide

| Conditions | Yield |
|---|---|
| With potassium carbonate at 80℃; for 12h; Inert atmosphere; | 99% |


| Conditions | Yield |
|---|---|
| In dimethyl sulfoxide at 90℃; | 99% |


| Conditions | Yield |
|---|---|
| In dimethyl sulfoxide at 90℃; | 99% |


| Conditions | Yield |
|---|---|
| In methanol Ni-salt in methanol was added to a soln. of phenolate in methanol, Na-acetate and NaBr in methanol was added; pptn. with diethyl ether, filtered off, dried; | 98% |


-
-
7647-15-6
sodium bromide

| Conditions | Yield |
|---|---|
| In methanol N2, an excess of Na compd., stirred for 1 h at room temp., solvent removed, residue dissolved (CH2Cl2), filtered (celite), solvent removed (repeated 3 times); elem. anal.; | 98% |
-
-
1356935-23-3
cis-PtCl2[Ph2PNH(C6H3(3,5-CO2Me)2)](Ph2POMe)

-
-
7647-15-6
sodium bromide

-
-
1356935-26-6
cis-PtBr2[Ph2PNH(C6H3(3,5-CO2Me)2)](Ph2POMe)

| Conditions | Yield |
|---|---|
| In methanol; acetone Schlenk techniques; room temp.; elem. anal.; | 98% |
-
-
1026780-30-2
1-benzyl-3-isopropyl-benzimidazolium bromide

-
-
3375-31-3
palladium diacetate

-
-
7647-15-6
sodium bromide

| Conditions | Yield |
|---|---|
| In dimethyl sulfoxide at 90℃; | 98% |

-
-
1315478-57-9
C6H4N2CH(CH2CH2CH2CH3)(CH2C6H5)(1+)*I(1-)=C6H4N2CH(CH2CH2CH2CH3)(CH2C6H5)I

-
-
3375-31-3
palladium diacetate

-
-
7647-15-6
sodium bromide

| Conditions | Yield |
|---|---|
| In dimethyl sulfoxide at 90℃; | 98% |

-
-
98651-97-9, 114030-80-7
dichlorobis(diphenylvinylphosphine)platinum(II)

-
-
7647-15-6
sodium bromide

-
-
118207-85-5, 130324-15-1, 122170-80-3
dibromobis(diphenylvinylphosphine)platinum(II)

| Conditions | Yield |
|---|---|
| In methanol; dichloromethane; water metathesis of chloride complex in CH2Cl2/CH3OH/H2O for 3 days with a mixture of NaBr and complex in 6:1 molar ratio under N2; elem. anal.; | 97.8% |
-
-
777855-06-8
rhodium(III) κC,κN,κN'-bis(N-tert-butyl)isophthalaldimin-2-yl dichloride

-
-
777855-07-9
rhodium(III) κC,κN,κN'-bis(N-tert-butyl)isophthalaldimin-2-yl chloride bromide

-
-
7647-15-6
sodium bromide

-
-
777855-08-0
rhodium(III) κC,κN,κN'-bis(N-tert-butyl)isophthalaldimin-2-yl dibromide

| Conditions | Yield |
|---|---|
| In acetone under N2; addn. of NaBr to mixt. of rhodium complexes in acetone, stirring overnight, evapn., extn. with chloroform, evapn., react. with NaBr, concn., stored on air for 3 d; ppt.; elem. anal.; | 97% |

Related products
Downstream Products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View
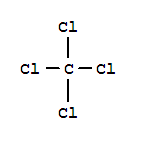






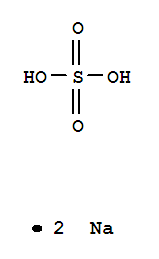




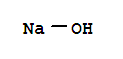


 Xi
Xi