Dayang Chem (Hangzhou) Co.,Ltd.
Dayangchem's R&D center can offer custom synthesis according to the contract research and development services for the fine chemicals, pharmaceutical, biotechnique and some of the other chemicals. DayangChem can provide different quantities
Cas:10102-43-9
Min.Order:1 Kilogram
FOB Price: $3.0
Type:Lab/Research institutions
inquiryAlity Chemical Corporation
The above product is Ality Chemical's strong item with best price, good quality and fast supply. Ality Chemical has been focusing on the research and production of this field for over 14 years. At the same time, we are always committed to providi
Chemwill Asia Co., Ltd.
Our main production base is located in Xuzhou industry park. We are certified both to the ISO 9001 and ISO 14001 Standards, have a safety management system in place.Our R&D team masters core technology for process-design of target building block
Hebei yanxi chemical co.,LTD.
D - glucuronic acid and N - acetyl glucosamine disaccharides unit composed of hyaluronic acid (Hyaluronan), also called uronic acid, hyaluronic acid, basic structure is made up of two disaccharides unit D - glucuronic acid and N - acetyl glucosamin
Zibo Hangyu Biotechnology Development Co., Ltd
Zibo Hangyu Biotechnology Development Co., Ltd is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality pharmaceuticals, intermediates, special chemicals and OLED intermediates and other fine chemi
Cas:10102-43-9
Min.Order:10 Gram
FOB Price: $100.0
Type:Lab/Research institutions
inquiryKono Chem Co.,Ltd
high purity lowest priceAppearance:solid or liquid Storage:in sealed air resistant place Package:drum and bag Application:for pharma use Transportation:by sea or air Port:Beijing or Guangzhou
Henan Allgreen Chemical Co.,Ltd
high quality Storage:Sealed, dry, microtherm , avoid light and smell. Package:According to the demand of customer Application:Organic synthesis Transportation:by air or by sea
Zhuozhou Wenxi import and Export Co., Ltd
Product Description Description & Specification Category Pharmaceutical Raw Materials, Fine Chemicals, Bulk drug Standard Medical standard
Cas:10102-43-9
Min.Order:1 Kilogram
FOB Price: $112.0
Type:Trading Company
inquiryAntimex Chemical Limied
Ansciep Chemical is a professional enterprise manufacturing and distributing fine chemicals and speciality chemicals. We have been dedicated to heterocycle compounds and phenyl rings for tens of years. This is our mature product for export. Our quali
Hunan Longxianng Runhui Trading Co.,Ltd
Nitric MonoxideAppearance:appearance White powder Storage:Dry and ventilated Package:1000 g/aluminum foil bag Application:Widely used in cosmetics, medicine, beauty health food and other industries Transportation:By express (Door to door) such as FED
Zhengzhou Kingorgchem Chemical Technology Co., Ltd.
1. Subsidiary of Institute of Chemistry, Henan Academy of Sciences, national research platform;2. About 30 years’ experience in this field since 1983;3. An experienced R&D team consisting of Doctors and Masters;4. Various types of analytical instrume
Cas:10102-43-9
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryHuBei Ipure Biotech CO.,ltd
HuBei ipure is a diversified product production and operation enterprise, with API, pharma intermediates, and other fine chemicals as well as R&D and pigments development and sales as one of the large enterprises, with more than 130 acres of pl
WUJI XINHUI CHEMICAL CO.,LTD
1. CAS No. 25212-86-6 2. Furan resin for foundry / casting 3. Leading manufacture since 2003 4. Free sample 1.Our advantages1. Fa…
Shanghai united Scientific Co.,Ltd.
United Scientific Company Located in Shanghai of China , is a competitive player in the global specialty and fine chemical market. Fenghua has both the expertise and flexibility to produce a wide range of chemicals. Focusing on developing the innovat
Shanghai Run-Biotech Co., Ltd.
Shanghai, Run-Biotech Co., Ltd is a leading domestic pharmaceutical, biopharmaceutical, and health care products R & D outsourcing services company. As an innovation-driven and customer-focused company, Run Biotech provides a broad and integrated por
changchun tuocai technology co.,ltld
Nitric MonoxideAppearance:appearance White powder Storage:Dry and ventilated Package:1000 g/aluminum foil bag Application:Widely used in cosmetics, medicine, beauty health food and other industries Transportation:shipping
Hebei Minshang Biotechnology Co., Ltd
Nitric Monoxide Application:Widely used in cosmetics, medicine, beauty health food and other industries
Nanjing Raymon Biotech Co., Ltd.
nitric oxide Storage:Store in dry and cool condition Package:25kg or according to cutomer's demand Application:Chemical research/Pharmaceutical intermediates Transportation:By Sea,by Air,By courier like DHL or Fedx. Port:Shanghai/Shenzhen
Asia Pacific Gas Enterprise Company Limited
Formula NO Application NO is produced on a massive scale as an intermediate in the Ostwald process for the synthesis of nitric acid from ammonia. Ni…
Chemical Co.Ltd
Nitrogen oxide (NO)Appearance:Off white to slight yellow solid Storage:Stored in shaded, cool and dry places Package:1L 5L 10L 25L bottle Application:pharma intermediate Transportation:Handle with cares to avoid damaging the packages. Protect them fr
NovaChemistry
high purity Application:Drug intermediates Materials intermediates and active molecules
Synthetic route
-
-
7632-00-0
sodium nitrite

-
-
10102-43-9
nitrogen(II) oxide

| Conditions | Yield |
|---|---|
| With acetic acid; potassium iodide In water Product distribution / selectivity; | 100% |
| With sulfuric acid; water at 10℃; under 375.038 Torr; for 1.38889E-05h; Reagent/catalyst; Temperature; Pressure; Time; | 99.99% |
| With maleic acid; ascorbic acid In water Product distribution / selectivity; |

| Conditions | Yield |
|---|---|
| In neat (no solvent) introduction of NO2 into an rapid streaming mixture of H-He;; spectrometric determination of OH concentration;; | A 100% B n/a |
| In gaseous matrix Kinetics; in argon carrier under 0.7 torr; followed by infrared chemiluminescence spectroscopy; | |
| In gas crossed molecular beam experiment; Laser fluorescence spectroscopy; |

-
-
7664-41-7
ammonia

-
-
80937-33-3
oxygen

-
A
-
7727-37-9
nitrogen

-
B
-
10102-43-9
nitrogen(II) oxide

-
C
-
10024-97-2
dinitrogen monoxide

| Conditions | Yield |
|---|---|
| With oxygen In neat (no solvent) Fe-ZSM-5 catalyst prepared by ion exchange and heat-treated at 400, 425or 450 °C, 100 % NH3 conversion, 100 % N2 selectivity, 1000 ppm NH3 in 2 % O2-contg. He; | A 100% B 0% C 0% |
| With catalyst:Fe-mordenite In neat (no solvent) Fe-mordenite catalyst prepared by ion exchange and heat-treated at 425 °C, 92 % NH3 conversion, 99 % N2 selectivity, 1000 ppm NH3 in 2 %O2-contg. He; | A 92% B n/a C 0% |
| With catalyst:Fe-ZSM-5 In neat (no solvent) Fe-ZSM-5 catalyst prepared by ion exchange and heat-treated at 375 °C, 90 % NH3 conversion, 99 % N2 selectivity, 1000 ppm NH3 in 2 % O2-contg. He; | A 90% B n/a C 0% |


-
-
10102-43-9
nitrogen(II) oxide

| Conditions | Yield |
|---|---|
| With cobaltocene In fluorobenzene for 5h; Inert atmosphere; | 100% |

-
-
10102-43-9
nitrogen(II) oxide

| Conditions | Yield |
|---|---|
| With water; nitric acid at 20℃; under 75.0075 Torr; for 2.77778E-05h; | 99.99% |
| In water decompn. in concd. soln. already at cool; | |
| In water decompn. in concd. soln. already at cool; |
-
-
7758-09-0
potassium nitrite

-
-
10102-43-9
nitrogen(II) oxide

| Conditions | Yield |
|---|---|
| With sulfuric acid; water at 20℃; under 375.038 Torr; for 2.22222E-05h; Pressure; Reagent/catalyst; Temperature; Time; | 99.98% |
| With sulfuric acid; potassium iodide In not given byproducts: H2O, I2; washing with NaOH soln.; | |
| above 500 °C, based on spect. results; |
-
-
7664-41-7
ammonia

-
A
-
7727-37-9
nitrogen

-
B
-
10102-43-9
nitrogen(II) oxide

-
C
-
10024-97-2
dinitrogen monoxide

| Conditions | Yield |
|---|---|
| With oxygen In neat (no solvent) Fe-mordenite catalyst prepared by ion exchange and heat-treated at 450 °C, 99 % NH3 conversion, 100 % N2 selectivity, 1000 ppm NH3 in 2 % O2-contg. He; | A 99% B 0% C 0% |
| With oxygen; platinum high excess O2,500 °C; | A <1 B n/a C n/a |
| With oxygen; platinum at 300 °C; | A n/a B <9 C n/a |

-
-
7664-41-7
ammonia

-
-
10102-43-9
nitrogen(II) oxide

| Conditions | Yield |
|---|---|
| With oxygen; platinum react. with air adding O2; | 99% |
| With oxygen; platinum optimal time of contact at very high temp.:some E-6 s; | 97% |
| With oxygen; Pt(90),Rh(10) (X%) 8 atm,high temp.; | 96% |
-
-
624-91-9
methyl nitrite

-
-
201230-82-2
carbon monoxide

-
A
-
553-90-2
Dimethyl oxalate

-
B
-
10102-43-9
nitrogen(II) oxide

| Conditions | Yield |
|---|---|
| With 0.5% Pd/C at 100℃; under 375.038 Torr; for 0.000555556h; Pressure; Reagent/catalyst; Temperature; Time; | A 98.9% B n/a |


| Conditions | Yield |
|---|---|
| Sr0.22La0.78CoO2.843 In neat (no solvent) byproducts: H2O; 800°C; | A n/a B 98.6% |
| Sr0.42La0.68CoO2.80 In neat (no solvent) byproducts: H2O; 800°C; | A n/a B 98.1% |
| Sr0.46La0.54CoO2.79 In neat (no solvent) byproducts: H2O; 800°C; | A n/a B 98% |

-
-
16649-19-7, 38586-93-5
chloro(N,N'-ethylenebis(salicylideneiminato))iron(III)

-
A
-
18601-34-8
(μ-oxo)bis[(1,2-ethanediamino-N,N'-bis(salicylidene))iron(III)]

-
B
-
10102-43-9
nitrogen(II) oxide

| Conditions | Yield |
|---|---|
| In acetonitrile under vac., equimolar mixt.; chromy.; | A n/a B 98% |
| With water In acetonitrile under vac., equimolar mixt.; chromy.; | A n/a B 63% |


-
A
-
7727-37-9
nitrogen

-
B
-
10102-43-9
nitrogen(II) oxide

-
C
-
10102-44-0
Nitrogen dioxide

-
D
-
10024-97-2
dinitrogen monoxide

| Conditions | Yield |
|---|---|
| at 220-260°C, nearly 98% N2O, 2% N2; troces of NO and NO2 (0.001%); | A 2% B n/a C n/a D 98% |

-
-
16921-91-8
nitrosyl hexafluorophosphate

-
-
12793-14-5
niobocene dichloride

-
B
-
10102-43-9
nitrogen(II) oxide

| Conditions | Yield |
|---|---|
| In liquid sulphur dioxide Reaction of Cp2NbCl2 with NOSbF6 in 1/1 molar ratio at room temp. under Ar.; sepn. of solvent and NO under vac., recrystn. (SO2), elem. anal.; | A 98% B n/a |


| Conditions | Yield |
|---|---|
| In acetonitrile Pd sponge (1.0 g) and NOBF4 (2.2 g) stirred in MeCN (50 mL) under vac. for 12 h with periodical removing of NO generated during the react.; filtration; anhydrous ether added to the filtrate; resulting ppt. washed (anhydrous ether) and dried (vac.); elem. anal.; | A 98% B n/a |

-
-
119030-00-1, 97337-05-8, 1228763-68-5
(N,N'-ethylenebis(salicylideneiminato))iron(III) nitrate

-
A
-
18601-34-8
(μ-oxo)bis[(1,2-ethanediamino-N,N'-bis(salicylidene))iron(III)]

-
B
-
10102-43-9
nitrogen(II) oxide

| Conditions | Yield |
|---|---|
| In acetonitrile under vac., equimolar mixt.; chromy.; | A n/a B 97% |


| Conditions | Yield |
|---|---|
| In gas Kinetics; High Pressure; the reagent ions were generated in an electron impact high-pressure ion source contg. NO2; the reagent ions were selected using a quadrupole mass filter before being injected into a drift region contg. helium carriergas; at 298 K; monitoring by quadrupole mass spectrometer at the end of the flow tube; | A 97% B 3% C n/a D n/a |


| Conditions | Yield |
|---|---|
| With tert.-butylnitrite; trifluoroacetic acid In chloroform Inert atmosphere; | A 95% B 97% |

-
A
-
10102-43-9
nitrogen(II) oxide

-
B
-
7440-57-5, 457905-15-6
gold

| Conditions | Yield |
|---|---|
| With [NO(1+),HMB]BF4(1-) In acetonitrile byproducts: CH4, C2H6; | A 96% B n/a |
-
-
12793-14-5
niobocene dichloride

-
B
-
10102-43-9
nitrogen(II) oxide

| Conditions | Yield |
|---|---|
| In liquid sulphur dioxide Reaction of Cp2NbCl2 with NOBF4 in 1/1 molar ratio at room temp. under Ar.; sepn. of solvent and NO under vac., recrystn. (SO2), elem. anal.; | A 96% B n/a |


-
-
603-35-0
triphenylphosphine

-
B
-
10102-43-9
nitrogen(II) oxide

| Conditions | Yield |
|---|---|
| With [NO(1+),HMB]BF4(1-) In acetonitrile | A 94% B 96% |

-
-
7783-99-5
silver(I) nitrite

-
-
119030-00-1, 97337-05-8, 1228763-68-5
(N,N'-ethylenebis(salicylideneiminato))iron(III) nitrate

-
A
-
18601-34-8
(μ-oxo)bis[(1,2-ethanediamino-N,N'-bis(salicylidene))iron(III)]

-
B
-
10102-43-9
nitrogen(II) oxide

| Conditions | Yield |
|---|---|
| In acetonitrile under vac., equimolar mixt.; chromy.; | A n/a B 95% |


-
-
76-05-1
trifluoroacetic acid

-
B
-
10102-43-9
nitrogen(II) oxide

| Conditions | Yield |
|---|---|
| In dichloromethane Kinetics; CF3COOH soln. added; UV; | A n/a B 95% |


-
-
64-19-7
acetic acid

-
B
-
10102-43-9
nitrogen(II) oxide

| Conditions | Yield |
|---|---|
| In dichloromethane gas chromy.; | A n/a B 95% |

-
-
34301-54-7
1-Adamantanethiol

-
B
-
10102-43-9
nitrogen(II) oxide

| Conditions | Yield |
|---|---|
| In diethyl ether at 20℃; for 0.25h; Solvent; | A 95% B n/a |

-
-
1282-37-7
ferrocenium(III) tetrafluoroborate

-
-
3695-77-0
triphenylmethanethiol

-
B
-
10102-43-9
nitrogen(II) oxide

| Conditions | Yield |
|---|---|
| With [PPN]2[S5Fe(μ-S)2FeS5] In tetrahydrofuran; acetonitrile at 20℃; for 12h; Inert atmosphere; Schlenk technique; | A 92% B n/a |

-
-
7440-62-2
vanadium

-
-
15969-55-8
dinitrogen tetroxide

-
A
-
15053-24-4
vanadium nitrate

-
B
-
10102-43-9
nitrogen(II) oxide

| Conditions | Yield |
|---|---|
| In acetonitrile acceleration with acetonitrile at o°C;; | A 91% B n/a |


| Conditions | Yield |
|---|---|
| In perchloric acid byproducts: H2O; nitric acid soln. addn. to Ag suspn. at 30-40°C, mixt. keeping at tis temp. for 15 min; | A n/a B 90% |

-
-
119030-00-1, 97337-05-8, 1228763-68-5
(N,N'-ethylenebis(salicylideneiminato))iron(III) nitrate

-
-
7632-00-0
sodium nitrite

-
A
-
18601-34-8
(μ-oxo)bis[(1,2-ethanediamino-N,N'-bis(salicylidene))iron(III)]

-
B
-
10102-43-9
nitrogen(II) oxide

| Conditions | Yield |
|---|---|
| In acetonitrile under vac., equimolar mixt.; chromy.; | A n/a B 90% |

-
-
122744-29-0
bis{octakis(dodecyloxy)phthalocyaninato}lutetium

-
A
-
122873-81-8
2(C12H25O)8C32H8N8(2-)*Lu(5+)*BF4(1-)={(C12H25O)8C32H8N8}2LuBF4

-
B
-
10102-43-9
nitrogen(II) oxide

| Conditions | Yield |
|---|---|
| In dichloromethane evapn., dissolved in heptane, filtered, recrystn. (ethyl acetate); elem. anal.; | A 90% B n/a |


| Conditions | Yield |
|---|---|
| In neat (no solvent) decompn. at 225-230°C;; | A 2-6 B 3-5 C n/a D 90% |
| In neat (no solvent) decompn. at 225-230°C;; | A 2-6 B 3-5 C n/a D 90% |

-
-
10102-43-9
nitrogen(II) oxide

-
-
7727-37-9
nitrogen

| Conditions | Yield |
|---|---|
| With Pd supported ZrO2-CeO2 catalyst at 175 - 400℃; for 1h; Reagent/catalyst; Temperature; Inert atmosphere; | 100% |
| With H2; O2; catalyst: 0.1 wtpercent Pt/La0.7Sr0.2Ce0.1FeO3 In neat (no solvent, gas phase) Kinetics; NO:H2:O2 = 1:0.25:5% gas mixt. with 5% H2O added in feed stream; at 140°C for 20 h; H2O as reagent; detd. by mass spectrometry, gas chromy.; | 93% |
| With (Y0.90Pr0.10)2O(3+x) at 900℃; Temperature; Inert atmosphere; | 79% |
-
-
201230-82-2
carbon monoxide

-
-
10102-43-9
nitrogen(II) oxide

| Conditions | Yield |
|---|---|
| In neat (no solvent) 85 atm CO, 15 atm NO, 200°C;; | 100% |


| Conditions | Yield |
|---|---|
| In neat (no solvent) 105°C, humidity as catalyst;; sublimation in N2-stream at 115°C;; | 100% |
| In neat (no solvent) 105°C, humidity as catalyst;; sublimation in N2-stream at 115°C;; | 100% |
| In ethanol | |
| In ethanol |
-
-
201230-82-2
carbon monoxide

-
-
10102-43-9
nitrogen(II) oxide

| Conditions | Yield |
|---|---|
| In neat (no solvent) 85 atm CO, 15 atm NO, 200°C;; | 100% |


| Conditions | Yield |
|---|---|
| 1.5h, 250°C; at room temp. slowly; | 100% |
| 1.5h, 250°C; at room temp. slowly; | 100% |
| With oxygen In water | |
| With oxygen In water |

| Conditions | Yield |
|---|---|
| In neat (no solvent) 2g dried CoI2 at 105°C reacts with NO gas during 15-20 h;; sublimation in N2 stream at 115°C;; | 100% |
| In neat (no solvent) byproducts: I2; 70-80°C;; | |
| With copper In neat (no solvent) at 140°C;; | 0% |
| With nickel In neat (no solvent) |
-
-
70936-44-6
Fe(tetraphenylporphyrinate)C6H5

-
-
10102-43-9
nitrogen(II) oxide

-
-
89672-69-5
{(C20H8(C6H5)4N4)Fe(C6H5)(NO)}

| Conditions | Yield |
|---|---|
| reaction of a solid complex in NO atm. or a soln. of complex in toluenesaturated with NO at -28°C; | 100% |
| In neat (no solvent) exposure of degassed solid ((C20H8(C6H5)4N4)Fe(C6H5)) to 1 atm of nitric oxide;; elem. anal.;; | >99 |

-
-
10102-43-9
nitrogen(II) oxide

-
B
-
100-47-0
benzonitrile

| Conditions | Yield |
|---|---|
| With NaClO4; CH2Cl2 In dichloromethane room temp., 15 min; | A 80% B 100% |
| With CH2Cl2 In dichloromethane room temp., 15 min; | A n/a B 73% |


| Conditions | Yield |
|---|---|
| In neat (no solvent) under 760.051 Torr; for 0.166667h; | 100% |

| Conditions | Yield |
|---|---|
| In benzene Inert atmosphere; | 100% |
-
-
277309-56-5
[(1,3-bis[(di-tert-butylphosphino)methyl)-4,6-dimethylbenzene(-1H))Rh(N2)]

-
-
10102-43-9
nitrogen(II) oxide

| Conditions | Yield |
|---|---|
| In tetrahydrofuran Inert atmosphere; | 100% |

| Conditions | Yield |
|---|---|
| With catalyst: Ni(2+)Y zeolite In neat (no solvent) reduction of very dild. mixt. of NO/NO2 in gas mixt. of N2/H2 on zeolite catalyst (300°C reaction temp.); gas chromy. (dimethylsulfolane coated diatomite); | 99% |
| With catalyst: industrial nickel methanation catalyst In neat (no solvent) reduction of very dild. mixt. of NO/NO2 in gas mixt. of N2/H2 on zeolite catalyst (300°C reaction temp.); gas chromy. (dimethylsulfolane coated diatomite); | 99% |
| With catalyst: phthalocyanineNiY zeolite In neat (no solvent) reduction of very dild. mixt. of NO/NO2 in gas mixt. of N2/H2 on zeolite catalyst (230°C reaction temp.); gas chromy. (dimethylsulfolane coated diatomite); | 94% |
| With catalyst: NiY zeolite In neat (no solvent) reduction of very dild. mixt. of NO/NO2 in gas mixt. of N2/H2 on zeolite catalyst (450°C reaction temp.); gas chromy. (dimethylsulfolane coated diatomite); | 84% |


-
-
10102-43-9
nitrogen(II) oxide

-
-
79299-90-4
[(1,1',2,2'-bisdiphenylphosphinoethylene)Pt(CF3)(NO2)]

| Conditions | Yield |
|---|---|
| In tetrahydrofuran byproducts: HNO2; | 99% |
| In tetrahydrofuran; diethyl ether suspended in THF/Et2O mixture, evacuated and saturated with NO, stirredat 25°C for 1.5 h, N2 bubbled; filtered, washed with Et2O, dried, recrystd. from CH2Cl2/MeOH; |

| Conditions | Yield |
|---|---|
| With perchloric acid; sodium perchlorate In water Kinetics; byproducts: H2O; HClO4 was added to soln. of Fe complex in aq. NaClO4 to pH 3; soln. was deoxygenated with N2; NO was bubbled; UV monitoring; | 99% |
| With perchloric acid; sodium perchlorate; sodium thiocyanide In perchloric acid; water Kinetics; deoxygenated soln. of NaSCN in aq. HClO4 was satd. with NO and mixed with deoxygenated soln. of Fe complex in aq. NaClO4 (pH 3) at 25°C; UV monitoring; | |
| With 1,4-pyrazine; perchloric acid; sodium perchlorate In perchloric acid; water Kinetics; deoxygenated soln. of pyrazine in aq. HClO4 was satd. with NO and mixed with deoxygenated soln. of Fe complex in aq. NaClO4 (pH 3) at 25°C; UV monitoring; |
-
-
114581-86-1
[CuI(tris(2-pyridylmethyl)amine)(CH3CN)]2+

-
-
10102-43-9
nitrogen(II) oxide

-
A
-
133578-96-8
[Cu(tris(2-pyridylmethyl)amine)(H2O)](2+)

-
B
-
10024-97-2
dinitrogen monoxide

| Conditions | Yield |
|---|---|
| In water room temp., pH = 7.0; reaction followed by gas chromy.; | A 99% B n/a |


-
-
10102-43-9
nitrogen(II) oxide

| Conditions | Yield |
|---|---|
| In tetrahydrofuran byproducts: HNO2; | 99% |

| Conditions | Yield |
|---|---|
| In tetrahydrofuran byproducts: HNO2; | 99% |

| Conditions | Yield |
|---|---|
| In diethyl ether reaction at 25°C; | 99% |
-
-
205433-03-0
[(η(3)-cyclooctenyl)Co(Pr(i)2PCH2CH2PPr(i)2)](+)BF4(-)

-
-
10102-43-9
nitrogen(II) oxide

-
-
205446-13-5
[(η(3)-cyclooctenyl)Co(Pr(i)2PCH2CH2PPr(i)2)NO](+)BF4(-)

| Conditions | Yield |
|---|---|
| In acetone Ar-atmosphere; excess NO, 4 h; collection (filtration), washing (Et2O), drying (vac.); elem. anal.; | 99% |

-
-
10102-43-9
nitrogen(II) oxide

| Conditions | Yield |
|---|---|
| In further solvent(s) NO passed for 30 min through a CH2Cl2/tetra-n-butylammonium perchlorate soln. of Ru complex; the soln. purged with N2 until dry, washed (acetone); elem. anal.; | 99% |

| Conditions | Yield |
|---|---|
| In diethyl ether reaction at 25°C; | 99% |

| Conditions | Yield |
|---|---|
| In diethyl ether reaction at 25°C; | 99% |
Related products
Downstream Products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View



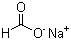




 O,
O,  T
T