Dayang Chem (Hangzhou) Co.,Ltd.
As a leading manufacturer and supplier of chemicals in China, DayangChem not only supply popular chemicals, but also DayangChem's R&D center offer custom synthesis services. DayangChem can provide different quantities of custom synthesis ch
Cas:7758-09-0
Min.Order:1 Kilogram
FOB Price: $3.0
Type:Lab/Research institutions
inquirySimagchem Corporation
Welcome to Simagchem, your partner in China as a premier supply of bulk specialty chemicals for industry and life science. We introduce experienced quality product and exceptional JIT service with instant market intelligence in China to benefit our
Cas:7758-09-0
Min.Order:1 Kilogram
Negotiable
Type:Manufacturers
inquiryAlity Chemical Corporation
The above product is Ality Chemical's strong item with best price, good quality and fast supply. Ality Chemical has been focusing on the research and production of this field for over 14 years. At the same time, we are always committed to providi
Chemwill Asia Co., Ltd.
Our main production base is located in Xuzhou industry park. We are certified both to the ISO 9001 and ISO 14001 Standards, have a safety management system in place.Our R&D team masters core technology for process-design of target building block
Cas:7758-09-0
Min.Order:5 Kiloliter
FOB Price: $1.2 / 5.0
Type:Manufacturers
inquiryHenan Tianfu Chemical Co., Ltd.
Our company engages in Sodium Tripolyphosphate (STPP) and Sodium Hexametabphosphate (SHMP) production; development of noble metal catalysts, synthesis of electronic chemical materials and general chemicals Imp&Exp trading business. The company
Cas:7758-09-0
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryChangchun Artel lmport and Export trade company
Product Detail Minimum Order Qty. 10 Gram
Cas:7758-09-0
Min.Order:10 Gram
Negotiable
Type:Trading Company
inquiryZibo Hangyu Biotechnology Development Co., Ltd
Zibo Hangyu Biotechnology Development Co., Ltd is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality pharmaceuticals, intermediates, special chemicals and OLED intermediates and other fine chemi
Cas:7758-09-0
Min.Order:10 Gram
FOB Price: $100.0
Type:Lab/Research institutions
inquiryHANGZHOU YUNUO CHEMICAL CO.,LTD
Superior quality, moderate price & quick delivery. Appearance:white or light yellow powder Storage:Stored in cool, dry and ventilation place; Away from fire and heat Package:25kg/drum, or as per your request. Application:Used as Pharmaceutical
Zibo Dorne chemical technology co. LTD
Product Details Grade: pharmaceutical grade Purity:99%+ ProductionCapacity: 1000 Kilogram/Month Scope of use: For scientific research only(The product must be used legally) Our Advantage 1. Best quality with competitive price. 2. Quick shipping,
Cas:7758-09-0
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryHangzhou Dingyan Chem Co., Ltd
Appearance White or light yellow colum crystal Assay 98%min Moisture 2%max Water-insolubles 0.01%max Chlorides 0.01%max Sulphates 0.02%max Iron 0.005%max Heavy metal 0.005%max Package:1kg/foil alum bag,25kg net/paper drum Port:Shan
Cas:7758-09-0
Min.Order:0 Metric Ton
Negotiable
Type:Lab/Research institutions
inquiryGreenutra Resource Inc
Potassium nitriteAppearance:solid or liquid Storage:in sealed air resistant place Package:drum and bag Application:for pharma use Transportation:by sea or air
Kono Chem Co.,Ltd
high purity lowest priceAppearance:solid or liquid Storage:in sealed air resistant place Package:drum and bag Application:for pharma use Transportation:by sea or air Port:Beijing or Guangzhou
Henan Allgreen Chemical Co.,Ltd
high quality Storage:Sealed, dry, microtherm , avoid light and smell. Package:According to the demand of customer Application:Organic synthesis Transportation:by air or by sea
Aecochem Corp.
Our clients, like BASF,CHEMO,Brenntag,ASR,Evonik,Merck and etc.Appearance:COA Storage:in stock Application:MSDS/TDS
Zhuozhou Wenxi import and Export Co., Ltd
Product Description Description & Specification Category Pharmaceutical Raw Materials, Fine Chemicals, Bulk drug Standard Medical standard
Cas:7758-09-0
Min.Order:1 Kilogram
FOB Price: $112.0
Type:Trading Company
inquiryAntimex Chemical Limied
Ansciep Chemical is a professional enterprise manufacturing and distributing fine chemicals and speciality chemicals. We have been dedicated to heterocycle compounds and phenyl rings for tens of years. This is our mature product for export. Our quali
Hangzhou Ocean Chemical Co., Ltd.
Ocean inorganic department is a professional supplier and exporter engaging in inorganic chemical materials and metal organic compounds. Over the past years, our company is committed to improving the product quality and developing new products, in or
Xiamen AmoyChem Co.,Ltd
Amoychem is committed to providing the top-quality chemical products and services Internationally. We offer our customers with friendly, professional service and reliable, high performance products that have been manufactured according to the accredi
Hubei Langyou International Trading Co., Ltd
POTASSIUM NITRITE lowest price Application:POTASSIUM NITRITE lowest price
Wuhan ZeShanCheng Biomedical Technology Co., Ltd.
1,we produce and sell good chemicals around the world. 2,our success rate is about 95%. this means, if customer order is accepted, the probability that the customer will obtain the ordered substances, is 95%. 3,our staff consists of highl
Hunan Longxianng Runhui Trading Co.,Ltd
POTASSIUM NITRITE lowest priceAppearance:yellowish white crystalline Solid Storage:Store at +5°C to +30°C. Package:foil bags, drums or bottles Application:angiotensin-converting enzyme inhibitor (ACE-I)oncombustible but accelerates the burning of all
Cas:7758-09-0
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryBeantown Chemical
in stock Application:7758-09-0
Cas:7758-09-0
Min.Order:0
Negotiable
Type:Trading Company
inquiryHENAN SUNLAKE ENTERPRISE CORPORATION
POTASSIUM NITRITE Basic information Product Name: POTASSIUM NITRITE Synonyms:
Hangzhou Haiqiang Chemical Co.,Ltd.
Appearance White or light yellow colum crystal Assay 98%min Moisture 2%max Water-insolubles 0.01%max Chlorides 0.01%max Sulphates 0.02%max Iron 0.005%max Heavy metal 0.005%max Package:1kg/foil alum bag,25kg net/paper drum Port:
Shaanxi Mingqi Chemical Co., Ltd
Known for its best quality and competitve price, this chemicals we offered is widely appreciated by our customers. Our advantages:1, High quality with competitive price:1) Standard:BP/USP/EP/Enterprise standard2) All Purity≥99%4) We are manufactureAp
Cas:7758-09-0
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryXi'an Galaxy Chemicals CO., Ltd
Full experience of large numbers containers loading in Chinese sea port Fast shipment by reputed shipping line Packing with pallet as buyer's special request Best service after shipment with e mail Cargoes together with container sales seer
Cas:7758-09-0
Min.Order:1 Metric Ton
FOB Price: $3100.0
Type:Trading Company
inquiryHebei Ruishun Trade Co.,Ltd
Hebei ruishun trade co.,ltd, registered capital one million,have a production of pharmaceutical raw materials, pharmaceutical raw materials factory reagent r&d center,seek development by credit reputation.our products have a large price advanta
Cas:7758-09-0
Min.Order:10 Gram
FOB Price: $0.9 / 1.2
Type:Trading Company
inquiryQingdao Sigma Chemical Co., Ltd.
Qingdao Sigma Chemical Ltd is is a global chemical industry manufacturers and suppliers of pharmaceuticals and intermediates, peptide,Nootropis etc API, food and feed additives, herbal extracts, agrochemicals and fine chemicals etc. Our Labo
Cas:7758-09-0
Min.Order:1 Gram
FOB Price: $1.0
Type:Trading Company
inquiryXiamen BaiFuchem Co.,Ltd
BaiFuChem is a Professional chemical raw material supplier in China, our main products include Biochemical , Pharma Intermediate and Organic chemical etc. BaiFuChem have wealth of products,experience , expertise and state-of-the-art
JINAN SHIJITONGDA CHEMICAL CO., LTD.
potassium nitrite industrial grade 96%-98% cas 7758-09-0 high quality 1. Type: Prill, Crystalline, With anti-caking ag…Appearance:colorless crystal or white powder. Package:25 kg & 50 kg HDPE bags Application:It is mai
Synthetic route

| Conditions | Yield |
|---|---|
| In water 88-97 % KNO2 content after evapn. to dryness and recrystn.; NaCl removed by pptn. by AgNO2 or AgNO3; | 91% |
| In water 88-97 % KNO2 content after evapn. to dryness and recrystn.; NaCl removed by pptn. by AgNO2 or AgNO3; | 91% |

| Conditions | Yield |
|---|---|
| A <1 B 65.2% | |
| A <1 B 65.2% |

-
-
1493-05-6
difluoro-nitro-methane

-
-
865-47-4
potassium tert-butylate

-
B
-
7758-09-0
potassium nitrite

-
C
-
105-58-8
Diethyl carbonate

| Conditions | Yield |
|---|---|
| In tetrahydrofuran byproducts: NO3(1-); alkoxide soln. addn. to F2C(NO2)2 soln. (molar ratio F2C(NO2)2:nucleophile=1:2), reacting (-10°C for 10-15 min), stirring (0°C for 1 h, further at 20°C for 2 h); salts. ppt. sepn. (CH2Cl2 addn.) after 1 d; chem. and GLC anal.; | A 43% B n/a C 11% |


| Conditions | Yield |
|---|---|
| In N,N-dimethyl-formamide alkoxide soln. addn. to F2C(NO2)2 soln. (molar ratio F2C(NO2)2:nucleophile=1:2), reacting (-10°C for 10-15 min), stirring (-50°C,then at 0°C for 2 d); salts. ppt. sepn. (CH2Cl2 addn.); chem. and GLC anal.; | A 19% B 25% |


| Conditions | Yield |
|---|---|
| With clay; soda lime; oxygen 370°C;with air; | A 2.6% B 23.2% |
| With O2; soda lime; clay 370°C;with air; | A 2.6% B 23.2% |
-
-
1493-05-6
difluoro-nitro-methane

-
-
917-58-8
potassium ethoxide

-
B
-
7758-09-0
potassium nitrite

-
C
-
105-58-8
Diethyl carbonate

| Conditions | Yield |
|---|---|
| In ethanol; dichloromethane alkoxide soln. (EtOH) addn. to F2C(NO2)2 soln. (CH2Cl2) (molar ratio F2C(NO2)2:nucleophile=1:1), reacting (-10°C for 10-15 min), stirring (0°C for 1 h); salts. ppt. sepn. (CH2Cl2 addn.); chem. and GLC anal.; | A n/a B n/a C 8% |


| Conditions | Yield |
|---|---|
| With nitrous gas In water excluding air; nitrous gas, obtained by redn. of HNO3 by As2O3 or starch, introduced in concd. soln. until complete neutralization; NO in small excess compared to NO2; | |
| In water double reaction of Ba-, Ag- or K-Pb-nitrite with K2CO3; soln. concd. in vacuo, dried in H2 at ambient temp. then at 100 °C; | |
| In water double reaction of Ba-, Ag- or K-Pb-nitrite with K2CO3; soln. concd. in vacuo, dried in H2 at ambient temp. then at 100 °C; | |
| With nitrous gas In water excluding air; nitrous gas, obtained by redn. of HNO3 by As2O3 or starch, introduced in concd. soln. until complete neutralization; NO in small excess compared to NO2; |

| Conditions | Yield |
|---|---|
| In water for prepn. of large quantities; KNO2 content 85-99.8 %; | >=72 |
| In water for prepn. of large quantities; KNO2 content 85-99.8 %; | >=72 |

| Conditions | Yield |
|---|---|
| heating in iron crucible to glow; | |
| heating in iron crucible to glow; |

| Conditions | Yield |
|---|---|
| In water pure KNO2 if AgNO2 was prepd. from NaNO2 purified by recrystn.; | |
| In water pure KNO2 if AgNO2 was prepd. from NaNO2 purified by recrystn.; |

| Conditions | Yield |
|---|---|
| heating;; | |
| heating;; |


| Conditions | Yield |
|---|---|
| In melt byproducts: K2SO4, K2SO3; in melt at 330°C; |

-
-
10102-43-9
nitrogen(II) oxide

-
-
7440-09-7
potassium

-
A
-
7758-09-0
potassium nitrite

-
B
-
7727-37-9
nitrogen


| Conditions | Yield |
|---|---|
| ignition; | |
| In not given ignition of heated K in NO, formation of KNO2, KNO3;; |

-
-
7440-09-7
potassium

-
-
10024-97-2
dinitrogen monoxide

-
A
-
7758-09-0
potassium nitrite

-
B
-
7727-37-9
nitrogen

| Conditions | Yield |
|---|---|
| In not given formation of KNO2 by a long (enough) reactn.time; appearance of NO (forming by thermical decompn. of N2O);; |


| Conditions | Yield |
|---|---|
| inflammation of K at room temp.; | |
| In not given ignition of heated K in NO2, formation of KNO2, KNO3;; | |
| inflammation of K at room temp.; |


| Conditions | Yield |
|---|---|
| 1 part of KNO3 melted with 2 part of Pb; boiling the melt with H2O, KNO2 pptd. with alcohol; | |
| 1 part of KNO3 melted with 2 part of Pb; boiling the melt with H2O, KNO2 pptd. with alcohol; |
-
-
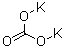
12775-96-1, 15158-11-9, 15721-63-8, 16941-75-6, 17493-86-6, 19498-52-3, 20499-83-6, 20499-84-7, 20499-85-8, 20499-86-9, 20573-10-8, 20573-11-9, 21595-51-7, 21595-52-8, 22206-52-6, 26445-28-3, 28959-95-7, 37362-93-9, 39417-05-5, 54603-16-6, 54603-23-5, 54603-32-6, 54603-40-6, 54603-48-4, 54603-81-5, 54603-89-3, 56316-56-4, 95985-91-4, 122297-32-9, 7440-50-8
copper

-
-
7758-09-0
potassium nitrite

| Conditions | Yield |
|---|---|
| melted with finely powd. Cu; | |
| With KOH more easily if Cu is of electrolytic quality; | |
| melted with finely powd. Cu; | |
| With potassium hydroxide more easily if Cu is of electrolytic quality; |

| Conditions | Yield |
|---|---|
| vac.; | |
| melted with much Fe filings to moderate glow; | |
| vac.; | |
| melted with much Fe filings to moderate glow; |

| Conditions | Yield |
|---|---|
| In water redn. of KNO3 in aq. soln. at normal temp. by Pd black, laden with H;; | |
| In water redn. of KNO3 in aq. soln. at normal temp. by Pd black, laden with H;; |


| Conditions | Yield |
|---|---|
| In water Kinetics; reaction of aq. solns. of starting materials at room temp.; IR-spectroscopy; |


| Conditions | Yield |
|---|---|
| With ammonia In water added NH3 to aq. soln. of KNO3 satd. at 30-40 °C then Zn under cooling; filtered, Zn pptd. by CO2, neutralized; KNO3 crystallizes first; | |
| With NH3 In water react. rate and yield increased by excess of NH3 and by holding temp. 20-25 °C; | |
| With NH3 In water added NH3 to aq. soln. of KNO3 satd. at 30-40 °C then Zn under cooling; filtered, Zn pptd. by CO2, neutralized; KNO3 crystallizes first; | |
| With ammonia In water react. rate and yield increased by excess of NH3 and by holding temp. 20-25 °C; |

| Conditions | Yield |
|---|---|
| In ethanol CP KNO2 with KOH (dissolved in alcohol) not in excess; | |
| In ethanol CP KNO2 with KOH (dissolved in alcohol) not in excess; |

| Conditions | Yield |
|---|---|
| at 320°C; | |
| at 320°C; |


| Conditions | Yield |
|---|---|
| With water | |
| With H2O |

| Conditions | Yield |
|---|---|
| heating in vacuum;; | |
| heating in vacuum;; |


| Conditions | Yield |
|---|---|
| byproducts: O2; Electrolysis; graphite electrodes; the graphite electrode is dissolved by the formed peroxide and K2CO3 and K2O are formed; with metal electrode is the yield of KNO2 lower; | |
| byproducts: O2; Electrolysis; aluminium or graphite cathode; the graphite electrode is dissolved by the formed peroxide and K2CO3 and K2O are formed; |


| Conditions | Yield |
|---|---|
| With K2SO4 In water Kinetics; Electrolysis; at -2.9 V versus Ag/AgCl at amb. temp.; |


-
-
7758-09-0
potassium nitrite

| Conditions | Yield |
|---|---|
| hydrated tin dioxide In water Irradiation (UV/VIS); redn. of KNO3 to KNO2 in aq. soln. by sunlight in presence of colloid tin(IV) oxide hydrate;; | |
| byproducts: O2; dissocn. pressures are given at various temp.; | |
| byproducts: O2; strong heating; |
-
-
7758-09-0
potassium nitrite

-
-
10102-43-9
nitrogen(II) oxide

| Conditions | Yield |
|---|---|
| With sulfuric acid; water at 20℃; under 375.038 Torr; for 2.22222E-05h; Pressure; Reagent/catalyst; Temperature; Time; | 99.98% |
| With sulfuric acid; potassium iodide In not given byproducts: H2O, I2; washing with NaOH soln.; | |
| above 500 °C, based on spect. results; |
-
-
7758-09-0
potassium nitrite

| Conditions | Yield |
|---|---|
| With HClO4 In water byproducts: KClO4; addn. of soln. of KNO2 to soln. of complex over 10 min, addn. of 1 M HClO4 up to pH 3; filtering after 3 d, evapn. in vac. until pptn. of KClO4, addn. of EtOH, filtration, concg., addn. of dioxane, filtration, addn. of dioxane and EtOH, addn. of ether with stirring, filtering ppt., washing with ether, drying in vac. over P2O5; elem. anal.; | 97% |

| Conditions | Yield |
|---|---|
| In water cooling down to 0°C, shaking; recrystn.; | 97% |


| Conditions | Yield |
|---|---|
| With CH3COOH In water byproducts: CH3COOK, NO, H2O; Co(NO3)2 and KNO2 (1:7) combined; acetic acid added (Co(NO3)2:CH3COOH=1:2) at 70+/-5°C under stirring; mixt. stirred at a rate of 500+/-50 rpm for 30 min; ppt. allowed to settle for 15-16 h; crystals washed (H2O) until pH about 4.5; solid filtered; dried at 90+/-%°C for 32 h; elem. anal.; | 97% |

| Conditions | Yield |
|---|---|
| In water suspn. of Pd powder in distd. water was treated with Cl2 until all Pd dissolved, purged with N2, filtered, KNO2 was added; crystd. at 278 K; elem. anal.; | 95% |


-
-
7758-09-0
potassium nitrite

-
-
159310-08-4
[ruthenium(II)(chloride)(η6-p-cymene)(2,2’-bipyridine)][hexafluorophosphate]

| Conditions | Yield |
|---|---|
| In methanol byproducts: AgCl; under inert atm. to Ru complex and AgNO3 in MeOH KNO2 was added and stirred for 18 h; excess NH4PF6 was added and cooled in ice bath, ppt. was filtered and stirred in acetone for 10 min, soln. was filtered and concd. in vacuo, Et2O was added, ppt. was filtered and washed with Et2O; elem. anal.; | 93.1% |

-
-
7758-09-0
potassium nitrite

| Conditions | Yield |
|---|---|
| In water addn. of KNO2 in H2O to a soln. of Pt-complex in H2O (molar ratio complex:KNO2=1:5), pptn. after 1-2 min, cooled in an ice bath; filtered, washed (several drops of icy water, EtOH and ether); elem. anal.; | 93% |

| Conditions | Yield |
|---|---|
| In water anion exchange in aq. soln.; molar ratio (C5H5Ni(P(C4H9-n)3)2)(1+)Cl(1-):KNO2 = 1:3; between O°C and room temp.; product dissolved in benzene; recrystn. from hexane or benzene/hexane in the presence of P(C4H9-n)3; | 91% |
| In water |
-
-
7758-09-0
potassium nitrite

-
-
10049-08-8
ruthenium(III)chloride

| Conditions | Yield |
|---|---|
| With Na(1+); Cl2 In hydrogenchloride heating on a water bath for 30 min; filtn. and dissolving in a minimum of water; elem. anal.; | 90% |
-
-
7758-09-0
potassium nitrite

-
-
117581-24-5
potassium chloro(pyridine-2,6-dicarboxylato)platinate(II) hydrate

-
-
119695-31-7
potassium nitro(pyridine-2,6-dicarboxylato)platinate(II) dihydrate

| Conditions | Yield |
|---|---|
| In water byproducts: AgCl; soln. of KNO2 added slowly to the soln. of Pt-complex (70°C), stirring (1 h); addn. of AgNO3, AgCl filtered off, soln. cooled slowly to room temp.; crystals filtered off and dried (vac.); elem. anal.; | 90% |

-
-
7758-09-0
potassium nitrite

-
-
84330-57-4, 112711-85-0
[Pt(O2CCF3)(C6Cl5)(P(C6H5)3)2]

| Conditions | Yield |
|---|---|
| In acetone slight excess of KNO2 added to soln. of Pt complex in acetone, left at room temp. for 4 h; concd. in vac., recrystd. from CH2Cl2/MeOH; elem. anal.; | 90% |
-
-
7758-09-0
potassium nitrite

| Conditions | Yield |
|---|---|
| In water addn. of a satd. soln. of KNO2 to that of the Pt-compd. (ratio 2 : 1, 80°C, 30 min); ppt. filtered off, washed (H2O) and dried (over P2O5); elem. anal.; | 90% |
-
-
7758-09-0
potassium nitrite

| Conditions | Yield |
|---|---|
| In water byproducts: KCl; satd. solns. (1 : 2), 80°C (30 min); ppt. filtered off, washed (H2O) and dried (over P2O5); elem. anal.; | 90% |
| In water addn. of a satd. soln. of KNO2 to that of the Pt-compd. (ratio 2 : 1, 80°C, 30 min); ppt. filtered off, washed (H2O) and dried (over P2O5); elem. anal.; | 90% |
-
-
1202-34-2
di(pyridin-2-yl)amine

-
-
7758-09-0
potassium nitrite

-
-
6046-93-1
copper(II) acetate monohydrate

| Conditions | Yield |
|---|---|
| In methanol; water aq. soln. of Cu salt added to soln. of org. compd. in MeOH, then added aq. soln. of KNO2; allowed to stand for 1 wk, filtered, washed with mother liquor, air-dried; elem. anal.; | 90% |

-
-
7758-09-0
potassium nitrite

-
-
215306-19-7
[2-(N,N-dimethylaminomethyl)phenyl]di-n-butyltin(IV) chloride

| Conditions | Yield |
|---|---|
| In tetrahydrofuran; benzene at 30℃; for 48h; Inert atmosphere; Schlenk technique; | 90% |
-
-
7758-09-0
potassium nitrite

-
A
-
7727-37-9
nitrogen

-
B
-
10024-97-2
dinitrogen monoxide

-
C
-
19467-31-3, 173728-04-6
trans-hyponitrous acid

| Conditions | Yield |
|---|---|
| In not given at room temp. or upon boiling from equimolar amounts in 3% solns.; no formation of H2N2O2 even in concd. solns. at low temps.; mole ratio of N2O:N2 depends on the mole ratio nitrite:hydrazonium salt;; | A 11% B 89% C 0% |


| Conditions | Yield |
|---|---|
| In tetrahydrofuran; benzene at 30℃; for 48h; Inert atmosphere; Schlenk technique; | 88% |
-
-
7758-09-0
potassium nitrite

| Conditions | Yield |
|---|---|
| In acetone N2-atmosphere; refluxing of excess NaNO2 with Rh-complex (15 h); filtration, washing (acetone), solvent removal from filtrate (vac.), crystn. (CH2Cl2/hexane); elem. anal.; | 86% |

| Conditions | Yield |
|---|---|
| With hydrogenchloride; sulfur dioxide In water byproducts: H2SO4; react. in water contg. ice and K-acetate leading SO2 into it,mixing,at 0°C;filtering,washing with 0.5 n HCl,hydrolysis at 100°C,for 2h; pptg. of H2SO4 with BaCl2,filtering,evapn.; | 85% |

| Conditions | Yield |
|---|---|
| With hydrogenchloride In hydrogenchloride; water byproducts: KCl, H2O; KNO2 in H2O was added dropwise to aq. HCl (37.8%) soln. of B-compd. at 0°C (temp. was not allowed to rise above 5-7 °C, mixt. was kept at 0. degree.C for 1 h and at room temp. until soln. was not conta ined NO2(1-)-ion; concn. (vac., 30 °C), EtOH was added, filtn. and evapn. in vac. at 30 °C, drying at room temp. in high-vac. for 1 h, MeCN was added, chromy. (silica, MeCN-CHCl3=1:1) (2 fraction), evapn., H2O was added,Bu4NCl was added in H2O, elem. anal.; | A 85% B 24% |

-
-
7758-09-0
potassium nitrite

| Conditions | Yield |
|---|---|
| With 18-crown-6 ether In N,N-dimethyl-formamide at 20℃; for 2h; Inert atmosphere; Schlenk technique; | 85% |
-
-
7758-09-0
potassium nitrite

| Conditions | Yield |
|---|---|
| With ferrocenium hexafluorophosphate In tetrahydrofuran at 0℃; Schlenk technique; | A 85% B n/a |

-
-
7758-09-0
potassium nitrite

-
-
85116-30-9
chloronitro(bipyridyl)(N,N'-dinitroso-1,2-ethanediaminato)platinum(II) hemihydrate

| Conditions | Yield |
|---|---|
| In water soln. of KNO2 in boiling water added to a soln. of the Pt-complex in boiling water, kept on a steam bath for 3 h; cooled to room temp., pptn. filtered off, washed with hot water, EtOH, and then ether, dissolved in boiling DMF, recrystd. by cooling, evapd., elem. anal.; | 84% |
-
-
7758-09-0
potassium nitrite

-
-
101420-28-4
tris<2-(1-methylimidazolyl)>methoxymethane

-
-
178316-11-5
bis(nitrito-O,O')[tris[2-(1-methyl)imidazolyl]methoxymethane]copper(II)

| Conditions | Yield |
|---|---|
| In acetone addn. of 1 equiv. ligand and 2 equiv. KNO2 to Cu-salt, stirring for 30 min; filtration, crystn. on evapn., collection (filtration), drying in air; | 82% |


| Conditions | Yield |
|---|---|
| In not given in acidic soln. from equimolar amounts of components; mole ratio N2O : N2 depends on the mole ratio of nitrite : hydrazonium salt;; | A 20% B 80% |
| In not given in acidic soln.; mole ratio N2O : N2 = 2:1 at mole ratio of nitrite : hydrazonium salt = 2:1;; |

-
-
7758-09-0
potassium nitrite

| Conditions | Yield |
|---|---|
| In methanol under N2 addn. of MeOH and KNO2 to complex, 50-60°C, 4 h, pptn.,evapn. in vac.; extn. with CH2Cl2, filtration, addn. of MeOH to ext., concn. in vac., recrystn. from CH2Cl2/MeOH, not sepn., detectation by IR, 31P NMR; | 80% |
-
-
7758-09-0
potassium nitrite

-
-
78331-77-8
[Ni(pentachlorophenyl)(pyridine)3]ClO4

| Conditions | Yield |
|---|---|
| In acetone excess KNO2, stirring (6 h, pptn.); filtration, washing (water), recrystn. (CH2Cl2/MeOH); elem. anal.; | 80% |

| Conditions | Yield |
|---|---|
| With triethyl formate In ethanol byproducts: KCl; LaCl3*7.1H2O in ethanol dehydrated by boiling with triethyl formate in presence OP(NMe2)3 for 15 min; filtering beaker with KNO2 mounted on backflow condenser, mixt. refluxed to complete dissoln. of KNO3; KCl filtered off; solvents distd. off in vac. at 300°K; filtered(acetone soln.), acetone distd. off in vac., crystd. after treatment with OP(NMe2)3 three times; washed (anhydrous diethyl ether), dried in vac.; elem. anal., XRD; | 80% |


| Conditions | Yield |
|---|---|
| Stage #1: dihydrogen hexachloroplatinate tetrahydrate; sodium nitrite In water for 2h; Stage #2: potassium nitrite | 80% |

Related products
Raw Materials
Downstream Products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View





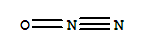


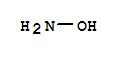





 O,
O, T,
T, N
N