Dayang Chem (Hangzhou) Co.,Ltd.
DayangChem exported this product to many countries and regions at best price. If you are looking for the material's manufacturer or supplier in China, DayangChem is your best choice. Pls contact with us freely for getting detailed product spe
Cas:5419-55-6
Min.Order:1 Kilogram
FOB Price: $3.0
Type:Lab/Research institutions
inquirySimagchem Corporation
Welcome to Simagchem, your partner in China as a premier supply of bulk specialty chemicals for industry and life science. We introduce experienced quality product and exceptional JIT service with instant market intelligence in China to benefit our
Cas:5419-55-6
Min.Order:1 Kilogram
Negotiable
Type:Manufacturers
inquiryLIDE PHARMACEUTICALS LIMITED
LIDE PHARMACEUTICALS LIMITED is a professional chemicals and APIs leading manufacturer in China. Our core business line covers APIs, Intermediates, Herb extract, etc.
Cas:5419-55-6
Min.Order:1 Metric Ton
FOB Price: $0.9 / 1.0
Type:Lab/Research institutions
inquiryHefei TNJ chemical industry co.,ltd
Properties Triisopropyl Borate is a kind of Colorless clear liquid, with melting point -59 ℃,boiling point 140 ℃,flash point 62.6 °F. It is used for semiconductor and other organic synthesis intermediates,should be stored in a shady and dry
Cas:5419-55-6
Min.Order:1 Metric Ton
Negotiable
Type:Trading Company
inquiryHenan Allgreen Chemical Co.,Ltd
he company has advanced technology, as well as a large number of excellent R & D team, to provide customers from the grams to one hundred kilograms and tons of high-quality products, competitive prices and quality se T rvice Appearance:White or
Chemwill Asia Co., Ltd.
Cas:5419-55-6
Min.Order:1 Metric Ton
FOB Price: $1.0
Type:Manufacturers
inquiryHenan Tianfu Chemical Co., Ltd.
Product Name: Triisopropyl borate Synonyms: Triisopropyl borate Liquid;Tri-i-propylborate,min.98%;Triisopropyl borate ((CH3)2CHO)3B;Boron isopropoxide, Boric acid triisopropyl ester;Boric acid triisopropyl ester, Boron isopropoxide, Triisopropoxyb
Cas:5419-55-6
Min.Order:1 Kilogram
FOB Price: $500.0
Type:Lab/Research institutions
inquiryHenan Sinotech Import&Export Corporation
Product Name: Triisopropyl borate Synonyms: Isopropyl borate, ((C3H7O)3B);Triisopropoxyboron;triisopropylorthoborate;trisisopropoxyborane;Boric acid triisopropyl ester~Isopropyl borate;Isop;Triisopropyl bor
Cas:5419-55-6
Min.Order:5 Metric Ton
FOB Price: $1.0 / 2.0
Type:Other
inquiryShanghai Minstar Chemical Co., Ltd
Product Name: Triisopropyl borate Synonyms: ISOPROPYL BORATE;BORON TRI-I-PROPOXIDE;BORON ISOPROPOXIDE;BORIC ACID TRIISOPROPYL ESTER;TRI-I-PROPYLBORATE;TRIISOPROPOXYBORANE;TRIISOPROPYL BORATE;TIPB CAS: 5419-55-6 MF: C9H21BO3 MW: 188.07 EINECS
Cas:5419-55-6
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryWuhan Wonda Pharm Limited
1.high quality: quality is life. quality is the most important element for all goods. we have a lab doing research in wuhan china. hplc and nmr is available if needed. 2.reasonable price: we provide high quality products wi
Cas:5419-55-6
Min.Order:10 Gram
Negotiable
Type:Lab/Research institutions
inquiryHangzhou J&H Chemical Co., Ltd.
J&H CHEM R&D center can offer custom synthesis according to the contract research and development services for the fine chemicals, pharmaceutical, biotechnique and some of the other chemicals. J&H CHEM has some Manufacturing base in Jia
Cas:5419-55-6
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryTriumph International Development Limilted
Product name: sodium metabisulfite / sodium metabisulphite Molecular weight: 190.10 Specification: 25Kg woven bag Quality guarantee period: 6 months Appearance:white powder/crystal Storage:Store in a cool,dry place and keep away fr
Cas:5419-55-6
Min.Order:100 Metric Ton
Negotiable
Type:Lab/Research institutions
inquiryZibo Hangyu Biotechnology Development Co., Ltd
Zibo Hangyu Biotechnology Development Co., Ltd is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality pharmaceuticals, intermediates, special chemicals and OLED intermediates and other fine chemi
Cas:5419-55-6
Min.Order:10 Gram
FOB Price: $100.0
Type:Lab/Research institutions
inquiryHangzhou Lingrui Chemical Co.,Ltd.
Triisopropyl borateAppearance:Pls see the Details Storage:Keep away of light, hot, water, Store in dry, dark and ventilated place Package:according to customers' requirements Application:Steroids, Cosmetics Ingredients, APIs, Intermediates, OLED&Batt
Zibo Dorne chemical technology co. LTD
Product Details Grade: pharmaceutical grade Purity:99%+ ProductionCapacity: 1000 Kilogram/Month Scope of use: For scientific research only(The product must be used legally) Our Advantage 1. Best quality with competitive price. 2. Quick shipping,
Cas:5419-55-6
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryHenan Wentao Chemical Product Co., Ltd.
We are leading fine chemicals supplier in China with ISO certificate, Our main business covers the fields below: 1.Noble Metal Catalysts (Pt.Pd...) 2.Organic Phosphine Ligands (Tert-butyl-phosphine.Cyclohexyl-phosphine...) 3.OLED
Cas:5419-55-6
Min.Order:1 Kilogram
FOB Price: $3.0
Type:Lab/Research institutions
inquiryHangzhou Zhongqi chem Co.,Ltd.
Stable offering high purity lower price Triisopropyl borate 5419-55-6 Located in Hangzhou National Hi-Tech Industrial Development Zone, zhongqichem is a technical company mainly focus on the Custom synthesis, manufacturing, sales of chemicals to
Cas:5419-55-6
Min.Order:1 Gram
Negotiable
Type:Other
inquiryHangzhou ZeErRui Chemical Co., Ltd.
Name: Triisopropylborate. Synonym:Boricacidtriisopropylester~Isopropylborate;Isop;Triisopropylborate, (Boricacidtriisopropylester;Triisorpopylborate;Triisoproylborate;TriisopropylBorate (TIPB); TRIISOPROPYLBORATE,98+%;Isopropylborate, ((C3H7O) 3B
Cas:5419-55-6
Min.Order:10 Gram
Negotiable
Type:Lab/Research institutions
inquiryHunan chemfish Pharmaceutical co.,Ltd
Appearance:95%+ Package:R&D,Pilot run Transportation:per client require Port:Express ,Air, Sea
HANGZHOU YUNUO CHEMICAL CO.,LTD
Superior quality, moderate price & quick delivery. Storage:Stored in cool, dry and ventilation place; Away from fire and heat Package:as per your request Application:Used as Pharmaceutical Intermediates,ect. Transportation:as per your request
Kono Chem Co.,Ltd
high purity lowest priceAppearance:solid or liquid Storage:in sealed air resistant place Package:drum and bag Application:for pharma use Transportation:by sea or air Port:Beijing or Guangzhou
JINHUA HUAYI CHEMICAL CO., LTD.
Jinhua huayi chemical co., ltd. is dedicated to the development, production and marketing of chemicals. On the basis of equality and mutual benefit, and under the principle of customer first, credit first, quality first, we are ready to join hands
Cas:5419-55-6
Min.Order:100 Gram
Negotiable
Type:Lab/Research institutions
inquiryAecochem Corp.
Our clients, like BASF,CHEMO,Brenntag,ASR,Evonik,Merck and etc.Appearance:COA Storage:in stock Application:MSDS/TDS
Enke Pharma-tech Co.,Ltd. (Cangzhou, China )
Cangzhou Enke Pharma Tech Co.,ltd. is located in Cangzhou City, Hebei province ,where is a famous petroleum chemical industry city?in China. Enke Pharma a high-tech enterprise ,and we are dedicated to developing and manufacturing new api,?intermediat
Zhuozhou Wenxi import and Export Co., Ltd
Product Description Description & Specification Category Pharmaceutical Raw Materials, Fine Chemicals, Bulk drug Standard Medical standard
Cas:5419-55-6
Min.Order:1 Kilogram
FOB Price: $112.0
Type:Trading Company
inquiryAntimex Chemical Limied
Ansciep Chemical is a professional enterprise manufacturing and distributing fine chemicals and speciality chemicals. We have been dedicated to heterocycle compounds and phenyl rings for tens of years. This is our mature product for export. Our quali
Cas:5419-55-6
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryXiamen AmoyChem Co.,Ltd
Amoychem is committed to providing the top-quality chemical products and services Internationally. We offer our customers with friendly, professional service and reliable, high performance products that have been manufactured according to the accredi
Hubei Langyou International Trading Co., Ltd
Triisopropyl Borate CAS NO.5419-55-6 Application:Triisopropyl Borate CAS NO.5419-55-6
Synthetic route

| Conditions | Yield |
|---|---|
| In pentane byproducts: H2; (Ar (N2) or vac.); dropwise addn. of excess of i-PrOH to Zr-complex in pentane with stirring (60 min, room temp.); concn., sepn. by filtration, a) pptn. drying (vac.), b) distn. of filtrate gives B compd.; elem. anal.; | A 75% B 90% |


| Conditions | Yield |
|---|---|
| With calcium hydride In cyclohexane byproducts: H2; react. under N2, slow addn. of the alcohol to the trialkoxyboroxine and CaH2 in cyclohexane, cooling in a water bath, rapid gas evolution, stirring for 1 h at room temp., heating to 80-90°C overnight; distn. of the product under reduced pressure; | 88% |

| Conditions | Yield |
|---|---|
| In pentane byproducts: H2; (Ar (N2) or vac.); dropwise addn. of excess of i-PrOH to Hf-complex in pentane with stirring (60 min, room temp.); concn., filtration, drying (vac.); elem. anal.; | A n/a B 88% |


| Conditions | Yield |
|---|---|
| With calcium hydride In neat (no solvent) byproducts: H2; react. under N2, slow addn. of the alcohol via syringe to B(OH)3 (ratio 3:1) and CaH2, cooling in a water bath, slow heating to 80 - 90°C for 12 h after abating the H2 gas evolution; distn. of the volatile products under reduced pressure (distn. pot temp. <= 120°C); | 87% |
| With calcium hydride at 90℃; Inert atmosphere; Schlenk technique; | 66% |
| In benzene for 18h; Heating; | 48% |

| Conditions | Yield |
|---|---|
| With acetone In tetrahydrofuran addn. of acetone via microsyringe to U complex soln.; standing at room temp., 1h; reducing volume; addn. of pentane; cooling to -78°C; pptn.; rapid filtration, drying (vac.); evapn. of mother liquor, extn. in pentane: B compd.; | A n/a B 81% |
-
-
80493-41-0
1-[Amino(diisopropylamino)boryl]-2-[bis(diisopropylamino)boryl]-2-isopropenyl-1-isopropylhydrazin

-
-
67-63-0
isopropyl alcohol

-
-
5419-55-6
Triisopropyl borate

| Conditions | Yield |
|---|---|
| byproducts: i-Pr2NH, i-PrNHNC(CH3)2, NH3; acidimetric determined, chromd.; | 70% |
-
-
80493-42-1
2,3,4-Tris(2,6-dimethylpiperidino)-1,3,4,6,7,8,9,9a-octahydro-6,9a-dimethyl-2H-pyrido[1,2-e][1,3,5,2,4]triazadiborin

-
-
67-63-0
isopropyl alcohol

-
-
5419-55-6
Triisopropyl borate

| Conditions | Yield |
|---|---|
| byproducts: NH3; 8 h; further byproduct; chromd.; | 70% |
| byproducts: 2,6-dimethylpiperidine, 1-amino-2,6-dimethylpiperidine, 2,3,4,5-tetrahydro-2,6-dimethylpiperidine; 8 h; further byproduct; chromd.; | 70% |

| Conditions | Yield |
|---|---|
| With boric acid; benzene unter Entfernen des entstehenden Wassers; | |
| With boron trioxide at 130℃; | |
| With boron trioxide; toluene |

| Conditions | Yield |
|---|---|
| at 100-120 °C; | |
| With catalyst: Zeokar 2 In solid Kinetics; prepd. from predried reagents by mechanochemical activation in planetarycentrifuge mill at rotation frequency of 10-17 s**-1 for 1-30 min; equi librium reaction; analyzed by (1)H NMR spectroscopy and XRD; | |
| With catalyst: zeolite NaA In solid Kinetics; prepd. from predried reagents by mechanochemical activation in planetarycentrifuge mill at rotation frequency of 10-17 s**-1 for 1-30 min; equi librium reaction; analyzed by (1)H NMR spectroscopy and XRD; | |
| With catalyst: zeolite NaX In solid Kinetics; prepd. from predried reagents by mechanochemical activation in planetarycentrifuge mill at rotation frequency of 10-17 s**-1 for 1-30 min; equi librium reaction; analyzed by (1)H NMR spectroscopy and XRD; | |
| at 100-120 °C; |

| Conditions | Yield |
|---|---|
| 90°C, bomb tube; | |
| 90°C, bomb tube; |

-
-
75-77-4
chloro-trimethyl-silane

-
-
107-46-0
Hexamethyldisiloxane

-
-
11113-50-1
boric acid

-
-
67-63-0
isopropyl alcohol

-
C
-
5419-55-6
Triisopropyl borate

-
D
-
4325-85-3
tris(trimethylsilyl)borate

| Conditions | Yield |
|---|---|
| In neat (no solvent) Me3SiCl, Me3SiOSiMe3 and 2-propanol stirred for 30 min, boric acid added and stirred for 3 h; soln. filtered and concd., mixt. analysed by using combined gas chromatograph-mass spectroscopy; |


| Conditions | Yield |
|---|---|
| In toluene to BH3*SMe2 in anhyd. toluene under N2 added i-PrOH; soln. stirred for 10 min; not isolated; monitored by GC/MS; | |
| In neat (no solvent) byproducts: H2, S(CH3)2; react. in a round-bottom flask under N2, addn. of the alcohol to the borane complex at room temp. (stirred), refluxed (according to Brown, Krishnamurthy, J.Org.Chem. 1978, 43, 2731; with a slight modification); distn. from a small piece of K metal; |

| Conditions | Yield |
|---|---|
| lithium triethylborohydride In tetrahydrofuran byproducts: H2; at 20°C with and without catalyst; |

| Conditions | Yield |
|---|---|
| In neat (no solvent) at 0°C, molar ratio (OC)BH3:ROH=1:3;; | |
| In neat (no solvent) at 0°C, molar ratio (OC)BH3:ROH=1:3;; |
-
-
11113-50-1
boric acid

-
-
5419-55-6
Triisopropyl borate

| Conditions | Yield |
|---|---|
| With isopropanol In benzene byproducts: water; distilled out at 138-140°C, elem. anal.;; |

| Conditions | Yield |
|---|---|
| In tetrahydrofuran potassium tetraisopropoxyborohydride refluxed in THF for 18 h; cooled to room temp., not isolated, detected by NMR; |
-
-
6336-44-3, 7547-96-8, 7547-97-9, 88982-39-2
allylboronic acid

-
-
67-63-0
isopropyl alcohol

-
A
-
5419-55-6
Triisopropyl borate

-
B
-
51851-79-7
diisopropyl allylboronate

| Conditions | Yield |
|---|---|
| In benzene byproducts: H2O; under Ar, soln. of allylboronic acid in C6H6 treated with 2-propanol (2 equiv), heated to reflux until evolution of H2O completed (Dean-Stark trap); fractionally distd. (bp 62-64°C, 40 mmHg); |

-
-
925-90-6
ethylmagnesium bromide

-
-
91083-27-1
(E)-1-hexenyldiisopropoxyborane

-
A
-
74953-03-0
diethyl(isopropoxy)borane

-
B
-
5419-55-6
Triisopropyl borate

-
C
-
148058-18-8
ethyl-1-hexenylisopropoxyborane

| Conditions | Yield |
|---|---|
| With methanesulfonic acid In tetrahydrofuran dropwise addn. of EtMgBr (1-3 equiv.) to soln. of C6H11B(OCH(CH3)2)2 (2.5 mmol) in THF at -78°C via syringe; stirring at -78°C for 3 h; addn. of CH3SO3H at -78°C; warming slowly to room temp.; stirring (room temp.);; not isolated; product distribution is determined by 11B-NMR;; |

-
-
925-90-6
ethylmagnesium bromide

-
-
91083-27-1
(E)-1-hexenyldiisopropoxyborane

-
A
-
5419-55-6
Triisopropyl borate

-
B
-
148058-18-8
ethyl-1-hexenylisopropoxyborane

| Conditions | Yield |
|---|---|
| With acetyl chloride In diethyl ether dropwise addn. of EtMgBr (1-2.5 equiv.) to soln. of C6H11B(OCH(CH3)2)2 (2.5 mmol) in ether at -78°C via syringe; stirring at -78°C for 3 h; addn. of CH3COCl at -78°C; warming slowly to room temp.; stirring (room temp.);; not isolated; product distribution is determined by 11B-NMR;; |

-
-
1068-55-9
isopropylmagnesium chloride

-
-
91083-27-1
(E)-1-hexenyldiisopropoxyborane

-
A
-
5419-55-6
Triisopropyl borate

-
C
-
148058-19-9
isopropyl-1-hexenylisopropoxyborane

| Conditions | Yield |
|---|---|
| With chloro-trimethyl-silane In tetrahydrofuran dropwise addn. of i-PrMgBr (2 equiv.) to soln. of C6H11B(OCH(CH3)2)2 (2.5 mmol) in THF at -78°C via syringe; stirring at -78°C for 3 h; addn. of (CH3)3SiCl at -78°C; warming slowly to room temp.; stirring (room temp.);; not isolated; product distribution is determined by 11B-NMR;; |

-
-
926-62-5
isobutylmagnesium bromide

-
-
91083-27-1
(E)-1-hexenyldiisopropoxyborane

-
A
-
5419-55-6
Triisopropyl borate

-
B
-
148058-20-2
isobutyl-1-hexenylisopropoxyborane

| Conditions | Yield |
|---|---|
| With methanesulfonic acid In tetrahydrofuran dropwise addn. of i-BuMgBr (2 equiv.) to soln. of C6H11B(OCH(CH3)2)2 (2.5 mmol) in THF at -78°C via syringe; stirring at -78°C for 3 h; addn. of CH3SO3H at -78°C; warming slowly to room temp.; stirring at room temp.;; not isolated; product distribution is determined by 11B-NMR;; |

-
-
917-54-4
methyllithium

-
-
91083-27-1
(E)-1-hexenyldiisopropoxyborane

-
B
-
5419-55-6
Triisopropyl borate

-
D
-
148058-10-0
methyl-1-hexenylisopropoxyborane

-
E
-
86595-27-9
diisopropoxymethylborane

| Conditions | Yield |
|---|---|
| With methanesulfonic acid In hexane byproducts: (CH3)3B; dropwise addn. of CH3Li (1 equiv.) to soln. of C6H11B(OCH(CH3)2)2 (2.5 mmol) in hexane at -78°C via syringe; stirring at -78°C for 3 h; addn. of CH3SO3H at 0/25°C; warming slowly to room temp.; stirring at room temp.;; not isolated; product distribution (depending on temp. at which acid is added) is determined by 11B-NMR;; |

-
-
917-54-4
methyllithium

-
-
91083-27-1
(E)-1-hexenyldiisopropoxyborane

-
B
-
5419-55-6
Triisopropyl borate

-
C
-
148058-10-0
methyl-1-hexenylisopropoxyborane

-
D
-
86595-27-9
diisopropoxymethylborane

| Conditions | Yield |
|---|---|
| With CH3SO3H or HCl or CH3COCl or ClCH2COCl or Cl3CCOCl In diethyl ether addn. of CH3Li to soln. of C6H11B(OCH(CH3)2)2 (2.5 mmol) in ether at -78°C via syringe; stirring at -78°C for 3 h; addn. of acid (further acids investigated: ClCH2COCl, (CH3)3SiCl); warming slowly to room temp.; stirring at room temp.;; not isolated; product distribution is determined by 11B-NMR for various conditions (temp. at which acid is added, amt. of CH3Li, amt. of acid);; | |
| With CH3SO3H or HCl or CH3COCl or ClCH2COCl or Cl3CCOCl In tetrahydrofuran addn. of CH3Li to soln. of C6H11B(OCH(CH3)2)2 (2.5 mmol) in THF at -78°C via syringe; stirring at -78°C for 3 h; addn. of acid (further acids investigated: ClCH2COCl, (CH3)3SiCl); warming slowly to room temp.; stirring at room temp.;; not isolated; product distribution is determined by 11B-NMR for various conditions (temp. at which acid is added, amt. of CH3Li, amt. of acid);; | |
| With methanesulfonic acid In hexane dropwise addn. of CH3Li (1 equiv.) to soln. of C6H11B(OCH(CH3)2)2 (2.5 mmol) in hexane at -78°C via syringe; stirring at -78°C for 3 h; addn. of CH3SO3H at -78°C; warming slowly to room temp.; stirring at room temp.;; not isolated; product distribution is determined by 11B-NMR;; |


| Conditions | Yield |
|---|---|
| In tetrahydrofuran byproducts: B-alkoxy-9-borabicyclo[3.3.1]nonane, KBr; 1 equivalent K(i-PrO)3BH, 25°C, 8.0 h; detected by 11B NMR; | |
| In tetrahydrofuran byproducts: B-alkoxy-9-borabicyclo[3.3.1]nonane, KBr; 1 equivalent K(i-PrO)3BH, 0°C, 0.25 h; detected by 11B NMR; |


-
-
70160-58-6
C8H14BClS(CH3)2

-
A
-
5419-55-6
Triisopropyl borate

-
B
-
76448-06-1
potassium (cyclooctane-1,5-diyl)dihydroborate

| Conditions | Yield |
|---|---|
| In tetrahydrofuran 2 equivalent K(i-PrO)3BH, 25°C, 0.25 h; detected by 11B NMR; |


| Conditions | Yield |
|---|---|
| In tetrahydrofuran byproducts: B-alkoxy-9-borabicyclo-[3.3.1]nonane, KCl; 1 equivalent K(i-PrO)3BH, 0°C, 0.25 h; detected by 11B NMR; |


| Conditions | Yield |
|---|---|
| With n-butyllithium In diethyl ether at 20℃; Substitution; | 100% |
| Stage #1: phenylacetylene With n-butyllithium In diethyl ether at -78℃; for 0.5h; Metallation; Stage #2: Triisopropyl borate In diethyl ether at -78℃; for 0.166667h; Addition; | |
| Stage #1: phenylacetylene With n-butyllithium In diethyl ether; hexane at -78℃; Metallation; Stage #2: Triisopropyl borate In diethyl ether; hexane at -78 - 20℃; Addition; Further stages.; |
-
-
5419-55-6
Triisopropyl borate

-
-
76782-82-6
tert-Butyldimethyl(prop-2-ynyloxy)silane

| Conditions | Yield |
|---|---|
| With n-butyllithium In diethyl ether at 20℃; Substitution; | 100% |

| Conditions | Yield |
|---|---|
| With n-butyllithium In diethyl ether at 20℃; Substitution; | 100% |
-
-
5419-55-6
Triisopropyl borate

-
-
18680-27-8
pinanediol

-
-
819816-59-6
(3aS,4s,6S,7aR)-hexahydro-3a,5,5-trimethyl-2-(1-methylethoxy)-4,6-methano-1,3,2-benzodioxaborole

| Conditions | Yield |
|---|---|
| In dichloromethane at 0 - 20℃; | 100% |
| In toluene for 0.5h; Heating; | 93% |
| In tetrahydrofuran at 0℃; for 2h; | 90% |
| In tetrahydrofuran at 0℃; for 2h; | 90% |
| In dichloromethane at 0 - 23℃; |
-
-
5419-55-6
Triisopropyl borate

| Conditions | Yield |
|---|---|
| Stage #1: 5-bromo-2-[(1-methylethyl)oxy]benzonitrile With n-butyllithium In tetrahydrofuran; hexane at -78℃; for 0.75h; Stage #2: Triisopropyl borate In tetrahydrofuran; hexane at -78 - -20℃; | 100% |
-
-
33070-32-5
5-bromo-2,2-difluoro-2H-1,3-benzodioxole

-
-
5419-55-6
Triisopropyl borate

-
-
190903-71-0
(2,2-difluoro-1,3-benzodioxol-5-yl)boronic acid

| Conditions | Yield |
|---|---|
| With n-butyllithium In tetrahydrofuran at -65 - -60℃; Inert atmosphere; | 100% |
| Stage #1: 5-bromo-2,2-difluoro-2H-1,3-benzodioxole With n-butyllithium In tetrahydrofuran; hexane for 0.5h; Inert atmosphere; Cooling with acetone-dry ice; Stage #2: Triisopropyl borate In tetrahydrofuran; hexane at 20℃; for 0.5h; Stage #3: With acetic acid In tetrahydrofuran; hexane at 20℃; for 0.166667h; | 73% |
| Stage #1: 5-bromo-2,2-difluoro-2H-1,3-benzodioxole With n-butyllithium In tetrahydrofuran; hexanes Stage #2: Triisopropyl borate In tetrahydrofuran; hexanes at 20℃; Stage #3: With acetic acid In tetrahydrofuran; hexanes at 20℃; for 0.166667h; | 73% |
-
-
5419-55-6
Triisopropyl borate

-
-
1310-73-2
sodium hydroxide

-
-
815581-69-2
N-(n-butyl)-2-(2-bromophenyl)-benzimidazole

| Conditions | Yield |
|---|---|
| With tert.-butyl lithium In diethyl ether; pentane a soln. of benzimidazole-compound in ether at -78°C under Ar was treated with t-BuLi in pentane over 15 min, stirred at -78.degre.C for 1h, treated with B(Oi-Pr)3, stirred for 48 h, allowed to warm to room te mp., NaOH was added, stirred for 1 h; ppt. was filtered, washed with ether, dried; | 100% |

-
-
5419-55-6
Triisopropyl borate

-
-
22348-32-9, 63401-04-7, 112022-88-5, 112068-01-6
(S)-diphenylprolinol

-
-
107-21-1
ethylene glycol

| Conditions | Yield |
|---|---|
| Stage #1: Triisopropyl borate; ethylene glycol In toluene Heating / reflux; Stage #2: (S)-diphenylprolinol In toluene at 80℃; under 20 Torr; for 1h; | 100% |
| In toluene at 80℃; for 1h; Inert atmosphere; | 100% |
| Stage #1: Triisopropyl borate; ethylene glycol In toluene Inert atmosphere; Reflux; Stage #2: (S)-diphenylprolinol In toluene at 80℃; for 1h; Inert atmosphere; Reflux; | 100% |
| Stage #1: Triisopropyl borate; ethylene glycol In toluene Inert atmosphere; Reflux; Stage #2: (S)-diphenylprolinol In toluene at 80℃; for 1h; Inert atmosphere; |


| Conditions | Yield |
|---|---|
| Stage #1: Triisopropyl borate; ethylene glycol In toluene Heating / reflux; Stage #2: 1,1-diphenyl-L-valinol In toluene at 80℃; under 20 Torr; for 1h; | 100% |


| Conditions | Yield |
|---|---|
| Stage #1: Triisopropyl borate; ethylene glycol In toluene for 0.05h; Heating / reflux; Stage #2: (1R,2S)-norephedrine In toluene at 20 - 80℃; for 1h; | 100% |

-
-
5419-55-6
Triisopropyl borate

-
-
60539-17-5, 79868-79-4, 129704-13-8
(R)-(+)-2-amino-1,1,2-triphenylethanol

-
-
107-21-1
ethylene glycol

| Conditions | Yield |
|---|---|
| Stage #1: Triisopropyl borate; ethylene glycol In toluene for 0.05h; Heating / reflux; Stage #2: (R)-(+)-2-amino-1,1,2-triphenylethanol In toluene at 20 - 80℃; for 1h; | 100% |

-
-
5419-55-6
Triisopropyl borate

-
-
1073633-66-5
7-bromo-2H-pyrano[3,2-b]pyridine

-
-
1073633-67-6
2H-pyrano[3,2-b]pyridin-7-ylboronic acid

| Conditions | Yield |
|---|---|
| Stage #1: 7-bromo-2H-pyrano[3,2-b]pyridine With n-butyllithium In hexanes; diethyl ether for 0.0833333h; Stage #2: Triisopropyl borate In hexanes; diethyl ether at -78 - 20℃; | 100% |

| Conditions | Yield |
|---|---|
| In tetrahydrofuran; toluene at -78 - 20℃; Inert atmosphere; Cooling with acetone-dry ice; | 100% |
| In tetrahydrofuran; toluene 1. triisopropylborate, toluene/THF 4/1; 2. n-C4H9Li over 90 min, -78°C to room temp.; | 99% |

-
-
5419-55-6
Triisopropyl borate

-
-
187086-32-4
3-bromo-7,12-diphenylbenzo[k]fluoranthene

-
-
276249-59-3
7,12-diphenylbenzo[k]fluoranthen-3-ylboronic acid

| Conditions | Yield |
|---|---|
| Stage #1: 3-bromo-7,12-diphenylbenzo[k]fluoranthene With n-butyllithium In tetrahydrofuran at -80℃; for 0.25h; Stage #2: Triisopropyl borate In tetrahydrofuran | 100% |
| Stage #1: 3-bromo-7,12-diphenylbenzo[k]fluoranthene With n-butyllithium In tetrahydrofuran; toluene at -70℃; Inert atmosphere; Stage #2: Triisopropyl borate In tetrahydrofuran; toluene at -70 - 20℃; for 2h; | 88% |
| Stage #1: 3-bromo-7,12-diphenylbenzo[k]fluoranthene With n-butyllithium In tetrahydrofuran; toluene at -70℃; for 1h; Stage #2: Triisopropyl borate In tetrahydrofuran; toluene at 20℃; for 2h; | 88% |
| Stage #1: 3-bromo-7,12-diphenylbenzo[k]fluoranthene With n-butyllithium In tetrahydrofuran; toluene at -70℃; for 1h; Stage #2: Triisopropyl borate In tetrahydrofuran; toluene at -70 - 20℃; for 2h; Stage #3: With hydrogenchloride In tetrahydrofuran; water; toluene at 20℃; for 2h; | 88% |
| Stage #1: 3-bromo-7,12-diphenylbenzo[k]fluoranthene With n-butyllithium In tetrahydrofuran; hexane at -78℃; for 1h; Inert atmosphere; Stage #2: Triisopropyl borate In tetrahydrofuran; hexane at 20℃; |
-
-
5419-55-6
Triisopropyl borate

-
-
57598-34-2
3-Bromo-N-tert-butoxycarbonyl-N-methylaniline

-
-
887831-90-5
(3-((tert-butoxycarbonyl)(methyl)amino)phenyl)boronic acid

| Conditions | Yield |
|---|---|
| Stage #1: 3-Bromo-N-tert-butoxycarbonyl-N-methylaniline With n-butyllithium In tetrahydrofuran; hexane at -78℃; for 0.5h; Stage #2: Triisopropyl borate In tetrahydrofuran; hexane at -78℃; for 2h; Stage #3: With hydrogenchloride; water In tetrahydrofuran; hexane at -10℃; | 100% |
-
-
5419-55-6
Triisopropyl borate

-
-
13438-50-1
3-bromofluoranthene

-
-
359012-63-8
7,12-diphenylbenzo[k]fluoranthene-3-boronic acid

| Conditions | Yield |
|---|---|
| With n-butyllithium; water In tetrahydrofuran at -80℃; for 0.25h; | 100% |
| Stage #1: 3-bromofluoranthene With n-butyllithium In tetrahydrofuran at -100℃; for 2h; Inert atmosphere; Stage #2: Triisopropyl borate In tetrahydrofuran for 4h; Inert atmosphere; | 70% |
| Stage #1: 3-bromofluoranthene With n-butyllithium In tetrahydrofuran; hexane at -78℃; for 1h; Stage #2: Triisopropyl borate In tetrahydrofuran; hexane at -78℃; for 2h; | 60% |
| In tetrahydrofuran at -78℃; |
-
-
109-72-8, 29786-93-4
n-butyllithium

-
-
89488-29-9
2-bromo-4-methoxypyridine

-
-
5419-55-6
Triisopropyl borate

| Conditions | Yield |
|---|---|
| In tetrahydrofuran; toluene at -78 - 20℃; Inert atmosphere; Cooling with acetone-dry ice; | 100% |

-
-
109-72-8, 29786-93-4
n-butyllithium

-
-
5419-55-6
Triisopropyl borate

-
-
22918-01-0
2-bromo-4-chloropyridine

| Conditions | Yield |
|---|---|
| In tetrahydrofuran; toluene at -78 - 20℃; Inert atmosphere; Cooling with acetone-dry ice; | 100% |

-
-
5419-55-6
Triisopropyl borate

| Conditions | Yield |
|---|---|
| Stage #1: (S)-2-bromo-4-fluoro-1-(pent-4-en-2-yloxy)benzene With n-butyllithium In tetrahydrofuran; hexane at -78℃; for 1h; Stage #2: Triisopropyl borate In tetrahydrofuran; hexane for 0.166667h; | 100% |
| Stage #1: (S)-2-bromo-4-fluoro-1-(pent-4-en-2-yloxy)benzene With n-butyllithium In tetrahydrofuran; hexane at -78℃; for 0.5h; Stage #2: Triisopropyl borate In tetrahydrofuran; hexane at -78 - 20℃; for 0.5h; Stage #3: With hydrogenchloride In tetrahydrofuran; hexane; water |
-
-
5419-55-6
Triisopropyl borate

-
-
1620136-60-8
(S)-5-bromo-2-(2-(trifluoromethyl)pyrrolidin-1-yl)pyrimidine

| Conditions | Yield |
|---|---|
| With n-butyllithium In tetrahydrofuran; hexane at -78℃; for 2h; Inert atmosphere; | 100% |
| With n-butyllithium In tetrahydrofuran; hexane at -78℃; for 2h; Inert atmosphere; | 100% |
-
-
5419-55-6
Triisopropyl borate

-
-
79868-78-3
(S)-(-)-2-amino-1,1,3-triphenyl-1-propanol

-
-
107-21-1
ethylene glycol

| Conditions | Yield |
|---|---|
| In toluene at 80℃; for 1h; Inert atmosphere; | 100% |
| Stage #1: Triisopropyl borate; ethylene glycol In toluene Inert atmosphere; Reflux; Stage #2: (S)-(-)-2-amino-1,1,3-triphenyl-1-propanol In toluene at 80℃; for 1h; Inert atmosphere; Reflux; | 100% |

-
-
5419-55-6
Triisopropyl borate

-
-
130798-48-0, 127986-84-9
(S)-(-)-α,α-di(2-naphthyl)-2-pyrrolidinemethanol

-
-
107-21-1
ethylene glycol

| Conditions | Yield |
|---|---|
| In toluene at 80℃; for 1h; Inert atmosphere; | 100% |
| Stage #1: Triisopropyl borate; ethylene glycol In toluene Inert atmosphere; Reflux; Stage #2: (S)-(-)-α,α-di(2-naphthyl)-2-pyrrolidinemethanol In toluene at 80℃; for 1h; Inert atmosphere; Reflux; | 100% |


| Conditions | Yield |
|---|---|
| In toluene at 80℃; for 1h; Inert atmosphere; | 100% |
| Stage #1: Triisopropyl borate; ethylene glycol In toluene Inert atmosphere; Reflux; Stage #2: (R)-α,α-diphenylprolinol In toluene at 80℃; for 1h; Inert atmosphere; Reflux; | 100% |
| Stage #1: Triisopropyl borate; ethylene glycol In toluene Inert atmosphere; Reflux; Stage #2: (R)-α,α-diphenylprolinol In toluene at 80℃; for 1h; Inert atmosphere; | 90% |

-
-
76-09-5
2,3-dimethyl-2,3-butane diol

-
-
5419-55-6
Triisopropyl borate

-
-
22348-32-9, 63401-04-7, 112022-88-5, 112068-01-6
(S)-diphenylprolinol

| Conditions | Yield |
|---|---|
| In toluene at 80℃; for 1h; Inert atmosphere; | 100% |
| Stage #1: 2,3-dimethyl-2,3-butane diol; Triisopropyl borate In toluene Inert atmosphere; Reflux; Stage #2: (S)-diphenylprolinol In toluene at 80℃; for 1h; Inert atmosphere; Reflux; | 100% |

-
-
5419-55-6
Triisopropyl borate

-
-
22348-32-9, 63401-04-7, 112022-88-5, 112068-01-6
(S)-diphenylprolinol

-
-
120-80-9
benzene-1,2-diol

| Conditions | Yield |
|---|---|
| In toluene at 80℃; for 1h; Inert atmosphere; | 100% |
| Stage #1: Triisopropyl borate; benzene-1,2-diol In toluene Inert atmosphere; Reflux; Stage #2: (S)-diphenylprolinol In toluene at 80℃; for 1h; Inert atmosphere; Reflux; | 100% |

-
-
5419-55-6
Triisopropyl borate

-
-
107-21-1
ethylene glycol

-
-
167316-27-0
N-[(1S,2S)-2-amino-1,2-diphenylethyl]-4-methylbenzenesulfonamide

| Conditions | Yield |
|---|---|
| Stage #1: Triisopropyl borate; ethylene glycol In toluene Inert atmosphere; Reflux; Stage #2: N-[(1S,2S)-2-amino-1,2-diphenylethyl]-4-methylbenzenesulfonamide In toluene at 80℃; for 1h; Inert atmosphere; Reflux; | 100% |

-
-
5419-55-6
Triisopropyl borate

-
-
130798-48-0
(R)-(+)-α,α-di-(2-naphthyl)-2-pyrrolidinemethanol

-
-
107-21-1
ethylene glycol

| Conditions | Yield |
|---|---|
| Stage #1: Triisopropyl borate; ethylene glycol In toluene Inert atmosphere; Reflux; Stage #2: (R)-(+)-α,α-di-(2-naphthyl)-2-pyrrolidinemethanol In toluene at 80℃; for 1h; Inert atmosphere; Reflux; | 100% |

-
-
5419-55-6
Triisopropyl borate

-
-
19111-87-6
2-bromobenzo[9,10]phenanthrene

-
-
654664-63-8
benzo[l]phenanthrene-2-boronic acid

| Conditions | Yield |
|---|---|
| Stage #1: 2-bromobenzo[9,10]phenanthrene With n-butyllithium In tetrahydrofuran for 0.25h; Heating; Stage #2: Triisopropyl borate In tetrahydrofuran | 100% |
-
-
5419-55-6
Triisopropyl borate

| Conditions | Yield |
|---|---|
| With n-butyllithium In tetrahydrofuran; toluene at -78 - 20℃; Inert atmosphere; | 100% |
-
-
5419-55-6
Triisopropyl borate

| Conditions | Yield |
|---|---|
| Stage #1: 2-tert-butyl-4-iodo-1-(2-methoxy-ethoxymethoxy)-benzene With n-butyllithium In tetrahydrofuran; hexane at -78℃; for 1h; Stage #2: Triisopropyl borate In tetrahydrofuran; hexane at -78 - 20℃; for 2h; | 100% |
Related products
Downstream Products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View

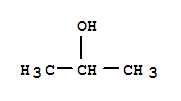














 F,
F, Xi
Xi