Dayang Chem (Hangzhou) Co.,Ltd.
DayangChem exported this product to many countries and regions at best price. If you are looking for the material's manufacturer or supplier in China, DayangChem is your best choice. Pls contact with us freely for getting detailed product spe
Cas:75-26-3
Min.Order:1 Kilogram
FOB Price: $3.0
Type:Lab/Research institutions
inquirySimagchem Corporation
Welcome to Simagchem, your partner in China as a premier supply of bulk specialty chemicals for industry and life science. We introduce experienced quality product and exceptional JIT service with instant market intelligence in China to benefit our
Cas:75-26-3
Min.Order:1 Metric Ton
Negotiable
Type:Manufacturers
inquiryJinan Finer Chemical Co., Ltd
Product Description Product website: http://www.finerchem.com Product Name 2-Bromopropane CAS No.
Hebei yanxi chemical co.,LTD.
Hebei yanxi chemical co. LTD. has expanded a compositive entity from initially only as a small manufacturer. The company dedicated to the development, production and marketing of chemicals. After many years of efforts, we have established stable
Henan Allgreen Chemical Co.,Ltd
high quality Appearance:Colorless liquid Storage:Sealed, dry, microtherm , avoid light and smell. Package:According to the demand of customer Application:Organic synthesis Transportation:by air or by sea Port:shanghai
EAST CHEMSOURCES LIMITED
2-Bromopropane Molecular Formula: (CH3)2CHBr Molecular Weight: 122.99 Appearance: colorless liquid Melting point: -89°C Boiling point:
Ality Chemical Corporation
The above product is Ality Chemical's strong item with best price, good quality and fast supply. Ality Chemical has been focusing on the research and production of this field for over 14 years. At the same time, we are always committed to providi
Chemwill Asia Co., Ltd.
Our main production base is located in Xuzhou industry park. We are certified both to the ISO 9001 and ISO 14001 Standards, have a safety management system in place.Our R&D team masters core technology for process-design of target building block
Henan Tianfu Chemical Co., Ltd.
Our company was built in 2009 with an ISO certificate.In the past 5 years, we have grown up as a famous fine chemicals supplier in China and we had established stable business relationships with Samsung,LG,Merck,Thermo Fisher Scientific and so
LIDE PHARMACEUTICALS LIMITED
Advantage : LIDE PHARMACEUTICALS LTD. is a mid-small manufacturing-type enterprise, engaged in pharmaceutical intermediates of R&D, custom-made and production, and also involving trading chemicals for export. We have established the R&
Cas:75-26-3
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryHenan Sinotech Import&Export Corporation
CAS 75-26-3 Name 2-Bromopropane Appearance Colorless Liquid Application Pharmaceutical Intermediates Appearance: Colorless Transparent Liquid Storage:
Hubei Langyou International Trading Co., Ltd
Advantages: Hubei XinRunde Chemical Co., Ltd is a renowned pharmaceutical manufacturer. We can offer high quality products at competitive price in quick delivery with 100% custom pass guaranteed. Never stop striving to offer our best service is
Baoji Guokang Healthchem co.,ltd
Our company has been in existence for 10 years since its establishment. We have our own unique team. The company integrates independent research and development, production and sales. We have established famous brands at home and abroad. At presen
Cas:75-26-3
Min.Order:1 Kilogram
FOB Price: $60.0 / 80.0
Type:Trading Company
inquiryHenan Wentao Chemical Product Co., Ltd.
Henan Wentao Chemical Product Co.,Ltd is Located in Zhengzhou High-tech Development Zone with import and export license, We passed ISO 9001:2008 as well, Henan Wentao has developed more than 1000 compounds, which are widely used in the fields of prod
Lonwin Chemical Group Limited
2-bromopropane CAS:75-26-3 Specification Product Name 2-Bromopropane CAS No. 75-26-3 Appearance Colorless liquid Assay ≥99%
Triumph International Development Limilted
Appearance:white or light yellow crystalline powder Storage:Store in a cool,dry place and keep away from direct strong light Package:As customer request Application:Used for research and industrial manufacture. Transportation:By
Wuhan Zenuo Biological Medicine Technology Co Ltd
Product Name: 2-Bromopropane Synonyms: 2-bromo-propan;2-Brompropan;bromo-2propane;iso-C3H7Br;propane,2-bromo-;sec-Propyl bromide;sec-propylbromide;2-bromopropane radical cation CAS: 75-26-3 MF: C3H7Br MW: 122.99 EINECS: 200-855-1 Produ
Cas:75-26-3
Min.Order:100 Gram
Negotiable
Type:Lab/Research institutions
inquiryHangzhou Keyingchem Co.,Ltd
Hangzhou KeyingChem Co., Ltd. exported this product to many countries and regions at best price. If you are looking for the material’s manufacturer or supplier in China, KeyingChem is your best choice. Pls contact with us freely for getting det
Hangzhou J&H Chemical Co., Ltd.
J&H CHEM R&D center can offer custom synthesis according to the contract research and development services for the fine chemicals, pharmaceutical, biotechnique and some of the other chemicals. J&H CHEM has some Manufacturing base in Jia
Cas:75-26-3
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquirySHANGHAI T&W PHARMACEUTICAL CO., LTD.
A substitute for perfluorooctanoic acid, mainly used as a surfactant, dispersant, additive, etc Appearance:White solid or Colorless liquid Purity:99.3 % We will ship the goods in a timely manner as required We can provide relevant documents acc
Zibo Hangyu Biotechnology Development Co., Ltd
Zibo Hangyu Biotechnology Development Co., Ltd is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality pharmaceuticals, intermediates, special chemicals and OLED intermediates and other fine chemi
Cas:75-26-3
Min.Order:10 Gram
FOB Price: $100.0
Type:Lab/Research institutions
inquiryZibo Dorne chemical technology co. LTD
Product Details Grade: pharmaceutical grade Purity:99%+ ProductionCapacity: 1000 Kilogram/Month Scope of use: For scientific research only(The product must be used legally) Our Advantage 1. Best quality with competitive price. 2. Quick shipping,
Cas:75-26-3
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryHangzhou Zhongqi chem Co.,Ltd.
(1) classified information Alias: iso-bromopropane, 2 - bromopropane, isopropyl bromide English name :2-Bromopropane, isopropyl bromide (IBP) Molecular formula: C3H7Br Molecular Weight: 122.99 CAS NO.: 75-26-3 UN Number: 2344 IMDG Code: 320
HANGZHOU YUNUO CHEMICAL CO.,LTD
Superior quality, moderate price & quick delivery. Appearance:colorless transparent liquid Storage:stored in a cool, dry and ventilated place to provent sun and rain Package:25kg/drum, or as per your request. Application:Used for Pesticide
Kono Chem Co.,Ltd
high purity lowest priceAppearance:solid or liquid Storage:in sealed air resistant place Package:drum and bag Application:for pharma use Transportation:by sea or air Port:Beijing or Guangzhou
JINHUA HUAYI CHEMICAL CO., LTD.
Jinhua huayi chemical co., ltd. is dedicated to the development, production and marketing of chemicals. On the basis of equality and mutual benefit, and under the principle of customer first, credit first, quality first, we are ready to join hands
Wuhan Fortuna Chemical Co.,Ltd
Best service,high quality and cheap price. Storage:Keep away from heat,sparks and flames. Package:25kg/50kg/200kg drum or Customer demand Application:Used as pharmaceutical intermediates Transportation:BY sea/air or by courier
Aecochem Corp.
Our clients, like BASF,CHEMO,Brenntag,ASR,Evonik,Merck and etc.Appearance:COA Storage:in stock Application:MSDS/TDS
Enke Pharma-tech Co.,Ltd. (Cangzhou, China )
Cangzhou Enke Pharma Tech Co.,ltd. is located in Cangzhou City, Hebei province ,where is a famous petroleum chemical industry city?in China. Enke Pharma a high-tech enterprise ,and we are dedicated to developing and manufacturing new api,?intermediat
Hunan chemfish Pharmaceutical co.,Ltd
Hunan chemfish Pharmaceutical co.,Ltd.located in Lugu High-tech industral park ,Hunan province . with its own R&D center and more than 10000㎡manufacture plant . Chemfish owns 40 reactors from 1000L to 8000L. With complete auxiliary equipment as
Synthetic route

| Conditions | Yield |
|---|---|
| With hydrogen bromide; oxygen at 20 - 23℃; under 1551.49 - 1603.2 Torr; for 4.5h; | A 97.8% B 1.57% |

| Conditions | Yield |
|---|---|
| With 2AlBr3*CBr4; bromine at -20℃; for 3h; | 96% |
| With antimony pentafluoride; 1,2-dibromomethane 1.) -78 deg C, 2 h, 2.) RT, 24 h; | 64% |
| With 2AlBr3*CBr4; bromine In various solvent(s) at -20℃; for 3h; | 48 % Turnov. |

| Conditions | Yield |
|---|---|
| With carbon tetrabromide; sodium bromide In N,N-dimethyl-formamide for 3h; Catalytic behavior; Irradiation; | 93% |
| With tetralin; bromine ueber mehrere Stufen; | |
| With sulfuric acid; potassium bromide |
-
-
116-17-6
triisopropyl phosphite

-
-
99-90-1
para-bromoacetophenone

-
A
-
106052-23-7
diisopropyl [4-(acetyl)phenyl]phosphonate

-
B
-
75-26-3
isopropyl bromide

| Conditions | Yield |
|---|---|
| With nickel dichloride at 180 - 190℃; for 0.0833333h; | A 91% B n/a |

-
-
108-20-3
di-isopropyl ether

-
-
201230-82-2
carbon monoxide

-
-
104-81-4
4-Methylbenzyl bromide

-
A
-
64450-64-2
isopropyl 2-p-tolylacetate

-
B
-
75-26-3
isopropyl bromide

| Conditions | Yield |
|---|---|
| 1,5-hexadienerhodium(I)-chloride dimer; potassium iodide at 75 - 90℃; under 735.5 Torr; overnight or in n-heptane; | A 78% B n/a |

-
-
116670-01-0
{(η5-C5Me5)Os(CO)(PMe2Ph)i-Pr}

-
-
7726-95-6
bromine

-
A
-
107087-80-9
{(η5-C5Me5)Os(CO)(PMe2Ph)Br}

-
B
-
187737-37-7
propene

-
C
-
75-26-3
isopropyl bromide

| Conditions | Yield |
|---|---|
| In dichloromethane-d2 (N2); electrophilic cleavage reaction by addn. of bromine to a soln. of Cp*Os(CO)(PMe2Ph)i-Pr in CD2Cl2;; not isolated, detected by NMR;; | A 77% B 9% C 50% |

-
-
116669-98-8
{(η5-C5Me5)Os(CO)2i-Pr}

-
A
-
81554-89-4
{(η5-C5Me5)Os(CO)2Br}

-
B
-
18819-83-5
isopropylmercury (1+); bromide

-
C
-
75-26-3
isopropyl bromide

| Conditions | Yield |
|---|---|
| In dichloromethane-d2 (N2); electrophilic cleavage reaction by addn. of HgBr2 to a soln. of Cp*Os(CO)2i-Pr in CD2Cl2;; not isolated, detected by NMR;; | A 77% B 61% C 11% |

-
-
116-17-6
triisopropyl phosphite

-
-
2142-63-4
1-(3-Bromophenyl)ethanone

-
A
-
127099-70-1
(3-Acetyl-phenyl)-phosphonic acid diisopropyl ester

-
B
-
75-26-3
isopropyl bromide

| Conditions | Yield |
|---|---|
| With nickel dichloride at 180 - 190℃; for 0.0833333h; | A 76% B n/a |

-
-
116669-98-8
{(η5-C5Me5)Os(CO)2i-Pr}

-
-
7726-95-6
bromine

-
A
-
81554-89-4
{(η5-C5Me5)Os(CO)2Br}

-
B
-
187737-37-7
propene

-
C
-
75-26-3
isopropyl bromide

| Conditions | Yield |
|---|---|
| In dichloromethane-d2 (N2); electrophilic cleavage reaction by addn. of bromine to a soln. of Cp*Os(CO)2i-Pr in CD2Cl2;; not isolated, detected by NMR;; | A 75% B 6% C 39% |

-
-
76313-88-7
2-isopropyl-2-boraadamantane

-
A
-
76313-91-2
2-bromo-2-boraadamantane

-
D
-
10035-10-6, 12258-64-9
hydrogen bromide

-
E
-
75-26-3
isopropyl bromide

| Conditions | Yield |
|---|---|
| With ethanol; bromine In dichloromethane Irradiation (UV/VIS); under Ar, to soln. of educt in CH2Cl2 added soln. of Br2 in CH2Cl2 in portions during 1 h at -10°C under pressure of 170-200 mmHg with irrdn. with 200 W bulb, stirred for 30 min at -10°C, heated under vac. to 17°C; concd. in vac., cooled to -70°C, ethanol added, evapd. in vac. without heating; GLC anal. of products after oxidative hydrolysis; | A n/a B n/a C n/a D 73% E 4% |
| With ethanol; bromine In dichloromethane under Ar in the dark, to soln. of educt in CH2Cl2 added soln. of Br2 inCH2Cl2 in portions during 5.5 h at -10°C under pressure of 170-200 mmHg, stirred for 30 min at -10°C, heated under vac. to 17°C; concd. in vac., cooled to -70°C, ethanol added, evapd. in vac. without heating; GLC anal. of products after oxidative hydrolysis; | A n/a B n/a C n/a D 14% E 65% |
| With ethanol; bromine In dichloromethane under Ar, to soln. of educt in CH2Cl2 added soln. of Br2 in CH2Cl2 in portions during 2.5 h at -10°C under pressure of 170-200 mmHg, stirred for 30 min at -10°C, heated under vac. to 17°C; concd. in vac., cooled to -70°C, ethanol added, evapd. in vac. without heating; GLC anal. of products after oxidative hydrolysis; | A n/a B n/a C n/a D 23% E 38% |

-
-
116-17-6
triisopropyl phosphite

-
-
109-70-6
1,3-chlorobromopropane

-
A
-
63602-20-0
O,O-diisopropyl (3-chloropropyl)phosphonate

-
B
-
75-26-3
isopropyl bromide

| Conditions | Yield |
|---|---|
| at 135 - 140℃; for 3h; Arbuzov rearrangement; | A 28% B 68% |

-
-
104-92-7
1-bromo-4-methoxy-benzene

-
-
116-17-6
triisopropyl phosphite

-
A
-
106052-22-6
diisopropyl (4-methoxyphenyl)phosphonate

-
B
-
75-26-3
isopropyl bromide

| Conditions | Yield |
|---|---|
| With nickel dichloride at 185 - 195℃; for 0.0833333h; | A 67% B n/a |

-
-
108-86-1
bromobenzene

-
-
116-17-6
triisopropyl phosphite

-
A
-
7237-16-3
diisopropyl phenylphosphonate

-
B
-
75-26-3
isopropyl bromide

| Conditions | Yield |
|---|---|
| With nickel dichloride at 185 - 195℃; for 0.0833333h; | A 65.5% B n/a |

-
-
1638-86-4
diethyl phenylphosphonite

-
-
109-70-6
1,3-chlorobromopropane

-
A
-
75-26-3
isopropyl bromide

-
B
-
231610-93-8
ethyl (3-chloropropyl)phenylphosphinate

| Conditions | Yield |
|---|---|
| at 135 - 140℃; for 3h; Arbuzov rearrangement; | A n/a B 62% |

-
-
116-17-6
triisopropyl phosphite

-
-
578-57-4
2-bromoanisole

-
A
-
127099-69-8
(2-Methoxy-phenyl)-phosphonic acid diisopropyl ester

-
B
-
75-26-3
isopropyl bromide

| Conditions | Yield |
|---|---|
| With nickel dichloride at 190 - 200℃; for 0.0833333h; | A 61% B n/a |
| nickel dichloride at 190 - 200℃; |

-
-
116-17-6
triisopropyl phosphite

-
-
1122-91-4
4-bromo-benzaldehyde

-
A
-
127099-71-2
4-(diisopropoxyphosphoryl)benzaldehyde

-
B
-
75-26-3
isopropyl bromide

| Conditions | Yield |
|---|---|
| With nickel dichloride at 170 - 180℃; for 0.0833333h; | A 54% B n/a |


| Conditions | Yield |
|---|---|
| at 250 - 270℃; Equilibrium constant; | |
| at 250 - 270℃; Equilibrium constant; die Umlagerung wird durch Sauerstoff, HBr, HgBr2, Tetraaethylammoniumbromid und Dibutylamin beschleunigt; | |
| lagert sich im UV-Licht teilweise um; |

| Conditions | Yield |
|---|---|
| With bromine | |
| With hydrogen bromide | |
| With pyridine; tributyltin bromide at 125℃; Thermodynamic data; Equilibrium constant; Δ G; | 49 % Chromat. |
-
-
187737-37-7
propene

-
-
75-26-3
isopropyl bromide

| Conditions | Yield |
|---|---|
| With hydrogen bromide bei Ausschluss von Luft und Zusatz von Oxydationsverzoegern wie Diphenylamin oder Thiokresol; | |
| With tetrachloromethane; hydrogen bromide | |
| With hydrogen bromide; iron(III) chloride im Dunkeln; |

| Conditions | Yield |
|---|---|
| With hydrogen iodide at 150℃; |
-
-
74-98-6
propane

-
A
-
78-75-1
1,2-Dibromopropane

-
B
-
106-94-5
propyl bromide

-
C
-
109-64-8
1,3-dibromo-propane

-
D
-
75-26-3
isopropyl bromide

| Conditions | Yield |
|---|---|
| at 300 - 330℃; Bromierung; weitere Produkte: 2.2-Dibrom-propan, Tribrompropan und Tetrabrompropan; |


| Conditions | Yield |
|---|---|
| With bromine at 300 - 330℃; | |
| With Bromoform; 1,2-dibromomethane In nitrogen at 525℃; for 0.0166667h; |
-
-
596-43-0
bromo-triphenyl-methane

-
-
2959-75-3
isopropyl-diphenyl-phosphine oxide

-
B
-
75-26-3
isopropyl bromide

| Conditions | Yield |
|---|---|
| at 100 - 200℃; |

-
-
596-43-0
bromo-triphenyl-methane

-
-
27350-46-5
isopropyl diphenylphosphinite

-
B
-
75-26-3
isopropyl bromide

| Conditions | Yield |
|---|---|
| at 100 - 200℃; |


| Conditions | Yield |
|---|---|
| With bromine |

| Conditions | Yield |
|---|---|
| With tetrabutylammomium bromide In acetone at 50℃; Kinetics; other reaction partners, other solvents, other temperatures; |

| Conditions | Yield |
|---|---|
| With bromine at 24.9℃; Kinetics; other temperatures; |
-
-
187737-37-7
propene

-
A
-
4283-80-1
2-bromo-2-methylpentane

-
B
-
106-94-5
propyl bromide

-
C
-
75-26-3
isopropyl bromide

| Conditions | Yield |
|---|---|
| With hydrogen bromide at -78℃; for 96h; Product distribution; | A 34 % Chromat. B 0.5 % Chromat. C 45 % Chromat. |
| With hydrogen bromide at -78℃; for 96h; | A 34 % Chromat. B 0.5 % Chromat. C 45 % Chromat. |


| Conditions | Yield |
|---|---|
| With tributyltin bromide In pyridine at 50℃; Thermodynamic data; Equilibrium constant; other temperatures, Δ G; |

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide | 100% |
| With potassium carbonate In N,N-dimethyl-formamide at 70℃; | 90% |
| In acetone | 88% |

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide at 20℃; for 48h; Inert atmosphere; | 100% |
| With potassium carbonate In N,N-dimethyl-formamide at 80℃; for 1h; | 100% |
| With potassium carbonate In dimethyl sulfoxide at 55℃; for 11h; Inert atmosphere; | 100% |
-
-
16532-79-9
4-Bromophenylacetonitrile

-
-
75-26-3
isopropyl bromide

-
-
51632-12-3
2-(4-bromophenyl)-3-methylbutanenitrile

| Conditions | Yield |
|---|---|
| Stage #1: 4-Bromophenylacetonitrile With sodium hydride In N,N-dimethyl-formamide; mineral oil at 0℃; Inert atmosphere; Stage #2: isopropyl bromide In N,N-dimethyl-formamide; mineral oil for 16h; Inert atmosphere; Heating; | 100% |
| Stage #1: 4-Bromophenylacetonitrile With sodium hydride In N,N-dimethyl-formamide; mineral oil for 0.25h; Stage #2: isopropyl bromide In N,N-dimethyl-formamide; mineral oil at 20℃; for 2h; | |
| Stage #1: 4-Bromophenylacetonitrile With sodium hydride In N,N-dimethyl-formamide; mineral oil at 0℃; for 0.25h; Stage #2: isopropyl bromide In N,N-dimethyl-formamide; mineral oil at 20℃; for 2h; | 21.3 g |
| Stage #1: 4-Bromophenylacetonitrile With lithium hexamethyldisilazane In tetrahydrofuran at -78℃; for 0.5h; Inert atmosphere; Stage #2: isopropyl bromide In tetrahydrofuran for 16h; Inert atmosphere; | |
| With sodium hydride |

| Conditions | Yield |
|---|---|
| 100% | |
| With potassium carbonate In N,N-dimethyl-formamide at 18℃; for 19h; | 100% |
| With potassium carbonate In N,N-dimethyl-formamide at 80℃; for 1h; | 100% |

| Conditions | Yield |
|---|---|
| With sodium tetrahydroborate In 1,2-dimethoxyethane at 20℃; for 12h; Inert atmosphere; | 100% |
| With indium iodide In dichloromethane at 20℃; for 1h; | 80% |
| With N,N,N,N,N,N-hexamethylphosphoric triamide; samarium diiodide In tetrahydrofuran for 3h; Heating; | 78% |
| With triphenylphosphine; 1-pentyl-3-methylimidazolium bromide at 75℃; for 6h; | 72% |
-
-
15288-02-5
6-hydroxy-7-methoxy-1-tetralone

-
-
75-26-3
isopropyl bromide

-
-
98799-45-2
6-isopropoxy-7-methoxy-1-tetralone

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide Heating; | 100% |
-
-
3753-27-3
sodium 4-methylbenzenesulfonothioate

-
-
75-26-3
isopropyl bromide

-
-
53291-31-9
4-Methylbenzolthiosulfonsaeure-S-isopropylester

| Conditions | Yield |
|---|---|
| In N,N-dimethyl-formamide at 20℃; for 16h; Inert atmosphere; | 100% |
| With Amberlyst A-26 (Cl(1-) form) 1.) H2O, r.t., 18 h; 2.) benzene, reflux, 25 h; Yield given. Multistep reaction; |

| Conditions | Yield |
|---|---|
| With sodium bis(2-methoxyethoxy)aluminium dihydride In tetrahydrofuran at 65℃; for 6h; | 100% |
| With potassium hydroxide 1.) DMSO, 2.) 50 - 60 deg C, 1 h; Yield given. Multistep reaction; | |
| With potassium hydroxide 1) DMSO, 20 deg C, 2) DMSO, 50-60 deg C, 1.0 h; Yield given. Multistep reaction; |
-
-
14371-10-9
(E)-3-phenylpropenal

-
-
591-51-5
phenyllithium

-
-
75-26-3
isopropyl bromide

-
-
85267-90-9
(+/-)-4-methyl-1,3-diphenylpentan-1-one

| Conditions | Yield |
|---|---|
| Stage #1: (E)-3-phenylpropenal; phenyllithium In tetrahydrofuran at 20℃; for 7h; Stage #2: isopropyl bromide In tetrahydrofuran Further stages.; | 100% |
| Stage #1: (E)-3-phenylpropenal; phenyllithium In tetrahydrofuran at 20℃; for 7h; Addition; Stage #2: isopropyl bromide In tetrahydrofuran at 20℃; Alkylation; | 100 % Chromat. |


| Conditions | Yield |
|---|---|
| Stage #1: ethanethiol With sodium tetrahydroborate; sodium hydroxide In water at 5℃; Inert atmosphere; Stage #2: isopropyl bromide; tetrabutylammomium bromide In water at 50℃; Inert atmosphere; | 100% |
| With potassium hydroxide In ethanol for 0.5h; Heating; | 51% |
-
-
148-53-8
3-methoxy-2-hydroxybenzaldehyde

-
-
75-26-3
isopropyl bromide

-
-
75792-35-7
2-isopropoxy-3-methoxy-benzaldehyde

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide at 20 - 110℃; Industry scale; Inert atmosphere; | 100% |
| With potassium carbonate In N,N-dimethyl-formamide at 110℃; | 99% |
| Stage #1: 3-methoxy-2-hydroxybenzaldehyde With tetrabutylammomium bromide; potassium carbonate In acetone at 20℃; for 0.25h; Stage #2: isopropyl bromide In acetone at 20℃; for 24h; | 96% |
-
-
16611-84-0
6-Pentadecylsalicylic Acid

-
-
75-26-3
isopropyl bromide

-
-
440094-90-6
isopropyl-2-isopropoxy-6-pentadecylbenzoate

| Conditions | Yield |
|---|---|
| With tetra(n-butyl)ammonium hydrogensulfate; potassium carbonate In 4-methyl-2-pentanone for 24h; Reflux; | 100% |
| With potassium carbonate In various solvent(s) for 36h; Heating; | |
| With benzyltri(n-butyl)ammonium chloride; potassium carbonate In 4-methyl-2-pentanone for 8h; Heating / reflux; | |
| With benzyltri(n-butyl)ammonium chloride; potassium carbonate In 4-methyl-2-pentanone for 8h; Heating / reflux; |
-
-
304-06-3
3-phenylsalicylic acid

-
-
75-26-3
isopropyl bromide

-
-
566162-84-3
2-isopropoxy-biphenyl-3-carboxylic acid isopropyl ester

| Conditions | Yield |
|---|---|
| With potassium carbonate at 50℃; for 12h; | 100% |
-
-
118-93-4
o-hydroxyacetophenone

-
-
75-26-3
isopropyl bromide

-
-
70201-54-6
1-(2-(1-methylethoxy)phenyl)ethanone

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide at 70℃; for 3h; Inert atmosphere; | 100% |
| With potassium carbonate In N,N-dimethyl-formamide at 70℃; | 94% |
| With potassium carbonate; potassium iodide In acetone Heating; | 91% |
-
-
75-26-3
isopropyl bromide

-
-
178626-86-3
2,3-dimercaptobenzoic acid methyl ester

-
-
547765-68-4
2,3-bis-isopropylsulfanyl-benzoic acid methyl ester

| Conditions | Yield |
|---|---|
| With lithium methanolate In methanol for 20h; Heating; | 100% |
-
-
7368-78-7
4-bromoguaiacol

-
-
75-26-3
isopropyl bromide

-
-
138505-27-8
4-bromo-1-isopropoxy-2-methoxy-benzene

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide at 20 - 90℃; | 100% |
| With potassium carbonate In dimethyl sulfoxide at 20 - 55℃; for 2h; | 94% |
| With potassium carbonate In dimethyl sulfoxide at 55℃; for 2h; | 84% |
-
-
73751-11-8
ethyl 5-bromo-4-hydroxy-3-methylbenzofuran-2-carboxylate

-
-
75-26-3
isopropyl bromide

-
-
857081-69-7
ethyl 5-bromo-4-isopropoxy-3-methyl-1-benzofuran-2-carboxylate

| Conditions | Yield |
|---|---|
| With potassium carbonate; bis-triphenylphosphine-palladium(II) chloride In N,N-dimethyl-formamide at 20℃; for 12h; | 100% |
-
-
2973-76-4
5-bromo-4-hydroxy-3-methoxybenzaldehyde

-
-
75-26-3
isopropyl bromide

-
-
400070-31-7
3-bromo-4-isopropyloxy-5-methoxybenzaldehyde

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide at 80℃; | 100% |
-
-
898830-67-6
2,2'-dihydroxybiphenyl-3,3'-dicarboxylic acid bisdiethyl amide

-
-
75-26-3
isopropyl bromide

-
-
898830-75-6
2,2'-diisopropoxy-biphenyl-3,3'-dicarboxylic acid bis-diethylamide

| Conditions | Yield |
|---|---|
| With sodium hydride In N,N-dimethyl-formamide | 100% |

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetone for 72h; Inert atmosphere; Reflux; | 100% |
| With potassium carbonate In acetone for 72h; Inert atmosphere; Reflux; | 100% |
| With potassium carbonate In acetone at 85℃; for 72h; Sealed tube; | 94.7% |
| With sodium hydride In N,N-dimethyl-formamide at 60℃; | 80% |
-
-
124702-80-3
5,6-dihydroxy-indan-1-one

-
-
75-26-3
isopropyl bromide

-
-
760995-28-6
5,6-diisopropoxyindan-1-one

| Conditions | Yield |
|---|---|
| With caesium carbonate In DMF (N,N-dimethyl-formamide) at 60℃; for 8h; | 100% |
-
-
3470-49-3
5-hydroxy-2,3-dihydro-1H-indene-1-one

-
-
75-26-3
isopropyl bromide

-
-
760995-38-8
5-isopropoxyindan-1-one

| Conditions | Yield |
|---|---|
| With caesium carbonate In DMF (N,N-dimethyl-formamide) at 60℃; | 100% |
-
-
127724-13-4
methyl 4-hydroxybenzofuran-2-carboxylate

-
-
75-26-3
isopropyl bromide

-
-
857080-64-9
methyl 4-isopropoxy-1-benzofuran-2-carboxylate

| Conditions | Yield |
|---|---|
| With potassium carbonate In DMF (N,N-dimethyl-formamide) at 20℃; | 100% |
-
-
99246-81-8
5-chloro-4-hydroxy-3-methyl-benzofuran-2-carboxylic acid ethyl ester

-
-
75-26-3
isopropyl bromide

-
-
857080-97-8
5-chloro-4-isopropoxy-3-methyl-benzofuran-2-carboxylic acid ethyl ester

| Conditions | Yield |
|---|---|
| With potassium carbonate In DMF (N,N-dimethyl-formamide) at 20℃; | 100% |
-
-
1092447-34-1
2-(3,5-dichloro-2-hydroxy-benzenesulfonylamino)-indan-2-carboxylic acid ethyl ester

-
-
75-26-3
isopropyl bromide

-
-
1092447-35-2
2-(3,5-dichloro-2-isopropoxy-benzenesulfonylamino)-indan-2-carboxylic acid ethyl ester

| Conditions | Yield |
|---|---|
| With caesium carbonate; potassium iodide In N,N-dimethyl-formamide at 110℃; for 2.5h; Microwave irradiation; | 100% |

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide at 80℃; for 2h; | 100% |
| With potassium carbonate In N,N-dimethyl-formamide at 80℃; for 2h; |

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetonitrile Reflux; | 100% |
| With potassium carbonate In acetonitrile at 20℃; for 24h; Reflux; | 87% |
| With potassium carbonate In N,N-dimethyl-formamide at 60℃; for 2.5h; | 58% |
| With potassium carbonate In acetonitrile at 80℃; for 16h; | 47% |
-
-
1072-63-5
1-vinylimidazole

-
-
75-26-3
isopropyl bromide

-
-
1020109-60-7
1-vinyl-3-isopropylimidazolium bromide

| Conditions | Yield |
|---|---|
| for 16h; Reflux; | 100% |
-
-
5010-37-7
2-(4-bromo-phenyl)-5-hydroxy-benzofuran-3-carboxylic acid ethyl ester

-
-
75-26-3
isopropyl bromide

-
-
691856-79-8
ethyl 2-(4-bromophenyl)-5-isopropoxybenzofuran-3-carboxylate

| Conditions | Yield |
|---|---|
| With caesium carbonate In 1-methyl-pyrrolidin-2-one at 60℃; for 4h; | 100% |
| Stage #1: 2-(4-bromo-phenyl)-5-hydroxy-benzofuran-3-carboxylic acid ethyl ester With caesium carbonate In 1-methyl-pyrrolidin-2-one at 20℃; for 0.333333h; Stage #2: isopropyl bromide In 1-methyl-pyrrolidin-2-one at 80℃; for 4h; | 95% |

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide at 20℃; | 100% |
| With potassium carbonate In acetonitrile for 2h; Reflux; | 72% |
| With potassium carbonate In acetone for 18h; Inert atmosphere; Reflux; |
Related products
Downstream Products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View


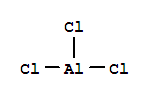



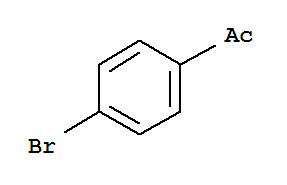



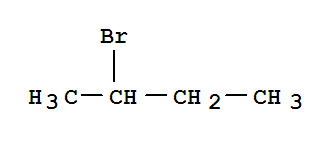





 F,
F, T
T