Refine
 This product is a nationally controlled contraband, and the Lookchem platform doesn't provide relevant sales information.
This product is a nationally controlled contraband, and the Lookchem platform doesn't provide relevant sales information.
Synthetic route

-
-
7440-23-5
sodium
Conditions

| Conditions | Yield |
|---|---|
| In neat (no solvent) heating NaN3 to 275°C;; | 100% |
| In neat (no solvent) heating NaN3 to 275°C;; | 100% |
| In neat (no solvent) carbon support was impregnated with NaN3 following with drying procedureunder UHV and at 450°C; carbon support was used; |
Conditions

| Conditions | Yield |
|---|---|
| With pyrographite; calcium oxide In neat (no solvent) 110°C, 1-2 Torr;; | 94% |
| With coal; CaO In neat (no solvent) 110°C, 1-2 Torr;; | 94% |
| With pyrographite In neat (no solvent) 110°C, 1-2 Torr;; | 92% |
Conditions

| Conditions | Yield |
|---|---|
| With calcium carbide In neat (no solvent) byproducts: CaCl2, C; at 900°C, diminshed yield with increase of pressure (1-10 Torr);; | 92% |
| With calcium carbide; sodium fluoride In neat (no solvent) byproducts: CaCl2, C; powdered and pressed, 0.5 Torr, 6% NaF, 875°C;; | 82% |
| With calcium carbide In neat (no solvent) reduction at 900°C;; | 63% |
Conditions

| Conditions | Yield |
|---|---|
| With pyrographite In neat (no solvent) 110°C, 1-2 Torr;; | 54% |
| With coal In neat (no solvent) 110°C, 1-2 Torr;; | 54% |
| With pyrographite; calcium oxide In neat (no solvent) 110°C, 1-2 Torr;; | 83-95 |
Conditions

| Conditions | Yield |
|---|---|
| decompn. at 500°C, heating in N2- or H2-atm.; | A n/a B 45% C n/a |
| decompn. at 500°C, heating in N2- or H2-atm.; | A n/a B 45% C n/a |
...Expand

Conditions

| Conditions | Yield |
|---|---|
| Electrolysis; |
Conditions

| Conditions | Yield |
|---|---|
| In neat (no solvent) 1200°C;; simultaneous preparation of Na and Mg;; |
...Expand

Conditions

| Conditions | Yield |
|---|---|
| In gaseous matrix slow reaction with small amounts; vigorous reaction with greater amounts and at a certain temperature;; | |
| In gaseous matrix slow reaction with small amounts; vigorous reaction with greater amounts and at a certain temperature;; |
...Expand

Conditions

| Conditions | Yield |
|---|---|
| In neat (no solvent) byproducts: iron, carbon dioxide, carbon monoxide; reaction of FeC2 (70 parts metal, 30 parts carbon) with 3 parts NaOH in a cast iron crucible; passing in of CO is of advantage; further byproduct: hydrogene;; |
Conditions

| Conditions | Yield |
|---|---|
| In gas Kinetics; byproducts: O2; carrier gas (He); fast flow reactor; not isolated, detected by fluorescence; |
Conditions

| Conditions | Yield |
|---|---|
| With iron In neat (no solvent) preparation of NaFeO2 by heating powdered Fe2O3 and Na2CO3 (stoechiometric mixture) to 900°C in an alundum-vessel, mixing with Fe-powder, heating at 2 torr to 1250°C;; |
Conditions

| Conditions | Yield |
|---|---|
| With pyrographite In neat (no solvent) a mixture of dry Na2CO3, Ca(OH)2, Fe2O3 is heated;; |
...Expand

-
-
1333-84-2, 1344-28-1
aluminum oxide

-
-
497-19-8
sodium carbonate

-
-
7757-82-6
sodium sulfate

-
B
-
124-38-9
carbon dioxide

-
C
-
7440-23-5
sodium

-
D
-
7704-34-9
sulfur

-
E
-
7429-90-5
aluminium
Conditions

| Conditions | Yield |
|---|---|
| In melt Kinetics; byproducts: O2, CO; Electrolysis; solid, granular sodium salts placed in alumina crucible and tube, purgedwith argon for 3 h, heated at 840.+-.20°C; detd. by IR gas analyzer, electrochemical cell analyzer, capillary ion electrophoresis, inductively coupled plasma anal., acid titration; |
...Expand


-
-
1333-84-2, 1344-28-1
aluminum oxide

-
-
497-19-8
sodium carbonate

-
-
7757-82-6
sodium sulfate

-
B
-
124-38-9
carbon dioxide

-
C
-
7440-23-5
sodium

-
D
-
7704-34-9
sulfur

-
E
-
7429-90-5
aluminium
Conditions

| Conditions | Yield |
|---|---|
| In melt Kinetics; byproducts: O2, CO; Electrolysis; solid, granular sodium salts placed in alumina crucible and tube, purgedwith argon for 3 h, heated at 840.+-.20°C; detn. by IR gas analyzer, electrochemical cell analyzer, capillary ion electrophoresis, inductively coupled plasma anal., acid titration; |
...Expand

Conditions

| Conditions | Yield |
|---|---|
| In melt Electrolysis; Al2O3-cryolite melt;; Na permeates through the carbon-bottom-plate of the bath and can be periodically removed;; | |
| In melt Electrolysis; Al2O3-cryolite melt;; Na permeates through the carbon-bottom-plate of the bath and can be periodically removed;; |
...Expand

Conditions

| Conditions | Yield |
|---|---|
| In neat (no solvent) decompn. upon heating; |
Conditions

| Conditions | Yield |
|---|---|
| In solid Irradiation (UV/VIS); photolysis of up to 5.0 GPa compressed solid NaN3; | |
| In neat (no solvent) Irradiation (UV/VIS); λ=2537 Å; -268.93°C, next heating to 600 K; | |
| In neat (no solvent) decompn. in vac. at 275°C; |
Conditions

| Conditions | Yield |
|---|---|
| In neat (no solvent) heating a mixture of Na and Bi (molar ratio 3 : 1) at 775°C under Ar;; | 100% |
| determined by thermal analysis;; | |
| determined by chemical methods;; |
Conditions

| Conditions | Yield |
|---|---|
| In neat (no solvent) Na was warmed in high vacuum and added to liquid Te in N2 atmosphere, NH3 was condensed over the mixture, the NH3 was slowly evaporated;; tempered at 400-500°C, stored under N2;; | 100% |
| In neat (no solvent) Na was warmed in high vacuum and added to liquid Te in N2 atmosphere, NH3 was condensed over the mixture, the NH3 was slowly evaporated;; tempered at 400-500°C, stored under N2;; | 100% |
| In neat (no solvent) Te heated to 620 .-+. 20°C was added in small portions to 300°C hot Na, excess of Na was distilled off;; | 80% |
-
-
7439-96-5
manganese

-
-
7440-23-5
sodium

-
-
12775-96-1, 15158-11-9, 15721-63-8, 16941-75-6, 17493-86-6, 19498-52-3, 20499-83-6, 20499-84-7, 20499-85-8, 20499-86-9, 20573-10-8, 20573-11-9, 21595-51-7, 21595-52-8, 22206-52-6, 26445-28-3, 28959-95-7, 37362-93-9, 39417-05-5, 54603-16-6, 54603-23-5, 54603-32-6, 54603-40-6, 54603-48-4, 54603-81-5, 54603-89-3, 56316-56-4, 95985-91-4, 122297-32-9, 7440-50-8
copper
Conditions

| Conditions | Yield |
|---|---|
| In neat (no solvent, solid phase) (inert gas), mixed, heated to 500°C for 24 h, stayed at 500°C for 2 days, heated to 800°C for 2 days, held at 800°C for 10 days; slowly cooled to room temp., elem. anal., XRD; | 100% |
...Expand

Conditions

| Conditions | Yield |
|---|---|
| In neat (no solvent, solid phase) (inert gas), mixed, heated to 500°C for 24 h, stayed at 500°C for 2 days, heated to 800°C for 2 days, held at 800°C for 10 days; slowly cooled to room temp., elem. anal., XRD; | 100% |
...Expand

Conditions

| Conditions | Yield |
|---|---|
| In neat (no solvent) byproducts: RbSb; molar ratio Na:Rb:Sb stoichiometric to 2:1:1; heating (12 h to 873 K; 120 h, 873 K), cooling (48 h, room temp.); XRD; | 100% |
...Expand

Conditions

| Conditions | Yield |
|---|---|
| In N,N,N,N,N,N-hexamethylphosphoric triamide (Ar); react. metal carbonyl with Na in HMPA for 1 h at 20°C; | 99% |
Conditions

| Conditions | Yield |
|---|---|
| In N,N,N,N,N,N-hexamethylphosphoric triamide (Ar); react. metal carbonyl with Na in HMPA for 1 h at 20°C; | 99% |
| In ammonia NH3 (liquid); NH3 distilled onto Na/Ru mixture (acetone/dry ice bath) under vac., slowly warmed to 235 K, vigorously stirred for ca. 60 min, warmed to 260 K; dried (vac., 260 K, 3.5 h); |
Related products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View
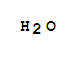

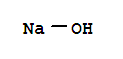













 C,
C, F,
F, T
T