 This product is a nationally controlled contraband, and the Lookchem platform doesn't provide relevant sales information.
This product is a nationally controlled contraband, and the Lookchem platform doesn't provide relevant sales information.
Synthetic route

-
-
7439-95-4
magnesium

| Conditions | Yield |
|---|---|
| With calcium oxide In neat (no solvent) distillation at 1350 °C within a period of 2 hours;; | 99% |
| With SiO; CaO In neat (no solvent) distillation at 1350 °C within a period of 2 hours;; | 99% |
| With silicon In neat (no solvent) |
-
-
84559-48-8
magnesium anthracene * 3 THF

-
-
7439-95-4
magnesium

| Conditions | Yield |
|---|---|
| In toluene byproducts: anthracene, THF; suspn. heated to 100°C with stirring (1h); hot suspn. filtered, washed (hot toluene), dried (70°C, in vacuo); | 96% |
| In neat (no solvent, solid phase) byproducts: anthracene, THF; solid magnesium anthracene*3 THF heated to 200°C (high vacuum) in several stages; | |
| In toluene byproducts: anthracene, THF; other Radiation; ultrasonic irradiation of suspn.; filtered, dried (in vacuo); | |
| In n-heptane byproducts: anthracene, THF; other Radiation; ultrasonic irradiation of suspn.; filtered, dried (in vacuo); | |
| In n-heptane byproducts: anthracene, THF; suspn. heated; filtered, dried (in vacuo); |

-
-
7439-95-4
magnesium

| Conditions | Yield |
|---|---|
| With calcium carbide In neat (no solvent) equimolar amts. of CaC2 and MgF2; 4 h, 0.5 Torr, 1200°C;; | 85% |
| With CaC2 In neat (no solvent) equimolar amts. of CaC2 and MgF2; 4 h, 0.5 Torr, 1200°C;; | 85% |
| With calcium carbide In neat (no solvent) equimolar amts. of CaC2 and MgF2; 4 h, 0.5 Torr, 1100°C;; | 84% |

| Conditions | Yield |
|---|---|
| In neat (no solvent) equimolar amts. of CaC2 and MgO; 1-2 Torr, 1200°C;; | A 80.05% B n/a |
| In neat (no solvent) equimolar amts. of CaC2 and MgO; 1-2 Torr, 1200°C;; | A 80.05% B n/a |
| In neat (no solvent) equimolar amts. of CaC2 and MgO; 1-2 Torr, 1100°C;; | A 30.52% B n/a |


| Conditions | Yield |
|---|---|
| In neat (no solvent, solid phase) heating at 850 °C; | A 11% B 11% C 38% D 36% E 4% |


| Conditions | Yield |
|---|---|
| In gaseous matrix deoxidn. using laser to raise temp. above 4000 K required for sepn. of Mg and O, argon gas flow prevent Mg from reoxidn.; CO2 or YAG laser, 100-1000 W, chamber pressure 500 Pa - 0.1 MPa; | A n/a B 20% |
| With magnesium In neat (no solvent) partial dissociation of MgO (in a mixt. with metallic Mg) in an iron tube heated to red heat; O2 evolution is observed;; | |
| In neat (no solvent) dissociation of solid MgO in metallic Mg and gaseous O2 at 2000-3000 K;; |

| Conditions | Yield |
|---|---|
| Electrolysis; |

| Conditions | Yield |
|---|---|
| titanium(III) chloride at 250 - 350℃; under 760.051 Torr; Product distribution / selectivity; Neat (no solvent); Balled milled; |

-
-
10467-10-4
ethylmagnesium iodide

-
-
7439-95-4
magnesium

| Conditions | Yield |
|---|---|
| In diethyl ether Electrolysis; current density = 1 A/dm^2, voltage = 8 V; room temperature; addition of dimethylaniline;; | |
| In diethyl ether Electrolysis; current density = 1 A/dm^2, voltage = 8 V; room temperature; addition of dimethylaniline;; |

| Conditions | Yield |
|---|---|
| In neat (no solvent) 1200°C;; simultaneous preparation of Na and Mg;; |


| Conditions | Yield |
|---|---|
| remarks on the technical circumstances; | |
| remarks on the technical circumstances; |

| Conditions | Yield |
|---|---|
| by heating; yields a mixt. of "amorphous" and "cryst." B; |

| Conditions | Yield |
|---|---|
| With sodium carbonate byproducts: sodiumzinccyanide; Mg2{Fe(CN)6} ignite with Na2CO3; resulting sodiummagnesiumcyanide reduced with Zn; |

| Conditions | Yield |
|---|---|
| at 1200-1500°C; |


-
-
7439-95-4
magnesium

| Conditions | Yield |
|---|---|
| With calcium carbide by heating; | |
| With iron In neat (no solvent) MgS is decomposed on Fe;; not isolated;; | |
| With iron In neat (no solvent) MgS is molten with iron shavings;; |

| Conditions | Yield |
|---|---|
| at 1050°C, under 2-4Hgmm; 3% MgS; |


| Conditions | Yield |
|---|---|
| at red heat; |

| Conditions | Yield |
|---|---|
| Electrolysis; |


-
-
7786-30-3
magnesium chloride

-
A
-
10025-67-9
disulfur dichloride

-
B
-
7704-34-9
sulfur

-
C
-
7439-95-4
magnesium

| Conditions | Yield |
|---|---|
| Electrolysis; |


-
-
7439-95-4
magnesium

| Conditions | Yield |
|---|---|
| With Al-Si-Fe alloy | |
| With Al-Si alloy | |
| With Al-Si-Fe alloy in the presence of alkali chloride, earth alkali chloride or fluoride; |

| Conditions | Yield |
|---|---|
| With pyrographite In neat (no solvent) Kinetics; reduction of magnesia with charcoal powder at 1743-1883 K; |

| Conditions | Yield |
|---|---|
| in the presence of C; |

| Conditions | Yield |
|---|---|
| In neat (no solvent) intimate mixture of stoiichiometric amounts of MgO and Al is heated in steel tube to ca. 1200 °C;; complete separation of Mg;; |


| Conditions | Yield |
|---|---|
| In melt solid MgO introduced in molten Al; Mg-Al-alloy separated in 2 layers of MgPb (MgCd) alloys and Al by molten Pb(Cd); | |
| at 1200°C in evacuated steel pipe; | |
| at >900°C; MgO and Al, powdwered, mixed and briquetted; |

| Conditions | Yield |
|---|---|
| at 1050°C, under 2-4 Hgmm; 0.5% S; |


| Conditions | Yield |
|---|---|
| With ferrosilicon in continuous process; |


| Conditions | Yield |
|---|---|
| In neat (no solvent) mixt. heated to 1300°C using high-frequency induction furnace; |


| Conditions | Yield |
|---|---|
| at 1900°C; | |
| 2.25 h at 1275°C; Mg distilles off; | >99 |
| 2.25 h at 1275°C; Mg distilles off; | >99 |

| Conditions | Yield |
|---|---|
| In neat (no solvent) Mg (purity: 99.5%) was heated in N2 (free of O2) at 800-850 °C for 4-5 h;; | 100% |
| In neat (no solvent) Mg was heated in a stream of N2 at 900 °C;; | 95% |

| Conditions | Yield |
|---|---|
| In water reduction with Mg in neutral or acidic soln.; | 100% |
| In water reduction with Mg in neutral or acidic soln.; | 100% |
| In water reaction in neutral and acid solutions complete;; |

| Conditions | Yield |
|---|---|
| In neat (no solvent) stoich. react. Mg, Th, and Te at 900°C with aid of Sn flux; | 100% |

-
-
114395-23-2
Fe(CO)2(P(O-i-Pr)3)2I2

-
-
7439-95-4
magnesium

-
-
114395-22-1
μ-dinitrogenbis(dicarbonylbis(triisopropylphosphite)iron)

| Conditions | Yield |
|---|---|
| With N2 In diethyl ether protective gas atmosphere, reduction with Mg under N2 (-20°C, 8 h); filtn. (silica gel, -20°C), crystn. (pentane, -80°C); | 100% |

| Conditions | Yield |
|---|---|
| In water Mg (64mmol) was dissolved in a soln. of 85% phosphoric acid (128mmol) in water (50ml); after ending of evolving of gas, the solvent was removedin high vacuum with liquid N2;; after the evolving of gas has stopped, the solvent was removed in high vacuum (liq. N2), and the crystals were extracted in a Soxhlet apparatus(48h) and further dried in vacuum; elem. anal.;; | 100% |
-
-
7439-95-4
magnesium

-
-
106-95-6
allyl bromide

-
-
3433-80-5
1-Bromo-2-bromomethyl-benzene

-
-
71813-50-8
1-bromo-2-(but-3-enyl)benzene

| Conditions | Yield |
|---|---|
| Stage #1: magnesium; allyl bromide In diethyl ether Stage #2: 1-Bromo-2-bromomethyl-benzene In tetrahydrofuran | 100% |


| Conditions | Yield |
|---|---|
| at 20℃; for 15h; Inert atmosphere; Schlenk technique; | 100% |


| Conditions | Yield |
|---|---|
| at 700 - 1100℃; for 336h; Sealed tube; Inert atmosphere; | 100% |


| Conditions | Yield |
|---|---|
| With oxygen In dimethyl sulfoxide metal. Mg under O2 atm. treated with DMSO and triflic acid (2 equiv.) in3 portions, heated at 100°C for 17 h; | 99% |


| Conditions | Yield |
|---|---|
| With oxygen In dimethyl sulfoxide metal. Mg under O2 atm. treated with DMSO and triflimidic acid (2 equiv.) in 3 portions, heated at 100°C for 18 h; | 99% |


| Conditions | Yield |
|---|---|
| With calcium In neat (no solvent) 82kg YF3, 74kg CaCl2, 37.5 kg Ca (105 excess), 15.75kg Mg, Ti- or Zr-vessel, 800-960°C;; | 99% |
-
-
344-07-0
1-chloro-2,3,4,5,6-pentafluorobenzene

-
-
7439-95-4
magnesium

-
-
879-06-1
pentafluorophenylmagnesium chloride

| Conditions | Yield |
|---|---|
| In tetrahydrofuran reaction at -10°C, 1 hour;; | 99% |
| In tetrahydrofuran reaction at -10°C, 1.25 hours;; | 87% |
| In diethyl ether (CH2Br)2- activated Mg, reaction in boiling ether;; | 67% |

| Conditions | Yield |
|---|---|
| In ethylenediamine High Pressure; solvothermal synthesis; Mg, SbCl3, S, ethylenediamine heated in a sealedTeflon-lined stainless steel autoclave at 170°C for 10 d; coolin g to room temp.; filtered off; washed with H2O, EtOH, acetone; elem. anal.; | 99% |


| Conditions | Yield |
|---|---|
| With triethylaluminum In ethanol; n-heptane; toluene for 2h; Heating; | 99% |

| Conditions | Yield |
|---|---|
| Stage #1: magnesium; 6-bromo-1,4-benzodioxane; lithium chloride In tetrahydrofuran at 20℃; Inert atmosphere; Schlenk technique; Stage #2: zinc(II) chloride In tetrahydrofuran at 0 - 20℃; for 0.25h; Inert atmosphere; Schlenk technique; | 99% |


| Conditions | Yield |
|---|---|
| With N-chloro-succinimide at 40 - 75℃; under 760.051 Torr; for 4.16667h; Product distribution / selectivity; Inert atmosphere; | 98.3% |
| In neat (no solvent) with C2H5OH-vapor at 280 to 290.degree C.;; | |
| In ethanol dissolution in abs. ethylalcohol in presence of CCl4 or CHCl3, and in presence of air or N2 discussed;; |

| Conditions | Yield |
|---|---|
| In neat (no solvent) byproducts: MgCl2; heating at 900-1100°C;; | 98% |

| Conditions | Yield |
|---|---|
| In neat (no solvent) mixing Mg and autocatalytic amts. of MgH2 in an autoclave, evacuating to 133 Pa, addn. of H2 to a pressure of 0.51 MPa, heating to 350°C with continous stirring, permanent H2-pressure: 0.58 MPa, react. time: 7 h; hydrid content: 7.1% by gas volumetry; | 98% |
| With chromium(III) oxide In neat (no solvent) mechanical grinding of mixt. of Mg and Cr2O3 under H2 atm.(up to 1.1 MPa) using planetary ball miller or vibratory miller, at 5 min-10 h; annealed at 200°C for 5 h or at 300°C for 5 h or at 330°Cfor 15 h; powder XRD, SEM; | |
| In neat (no solvent) Mg film reacted with H2 at room temp. and 100 kPa; |

| Conditions | Yield |
|---|---|
| byproducts: NH3, H2O, H2; at room temp.; EtOH added; elem. anal.; | 98% |

-
-
64-17-5
ethanol

-
-
7783-61-1
silicon tetrafluoride

-
-
7439-95-4
magnesium

-
B
-
78-10-4
tetraethoxy orthosilicate

| Conditions | Yield |
|---|---|
| Stage #1: ethanol; magnesium With iodine at 20℃; for 3.5h; Inert atmosphere; Reflux; Stage #2: silicon tetrafluoride for 2.5h; Inert atmosphere; Reflux; Stage #3: at 300℃; for 2h; Catalytic behavior; Reagent/catalyst; Temperature; Calcination; | A 98% B 82% |

-
-
67-56-1
methanol

-
-
7783-61-1
silicon tetrafluoride

-
-
7439-95-4
magnesium

-
B
-
681-84-5
tetramethylorthosilicate

| Conditions | Yield |
|---|---|
| Stage #1: methanol; magnesium With iodine at 20℃; for 1.5h; Inert atmosphere; Reflux; Stage #2: silicon tetrafluoride Inert atmosphere; Stage #3: at 300℃; for 2h; Catalytic behavior; Reagent/catalyst; Temperature; Calcination; | A 98% B 85% |
| Stage #1: methanol; magnesium at 20℃; for 3h; Stage #2: silicon tetrafluoride at 20℃; for 0.5h; Temperature; |


| Conditions | Yield |
|---|---|
| at 700 - 1100℃; for 336h; Sealed tube; Inert atmosphere; | 98% |


| Conditions | Yield |
|---|---|
| at 700 - 1100℃; for 336h; Sealed tube; Inert atmosphere; | 98% |


| Conditions | Yield |
|---|---|
| With triethylaluminum In ethanol; n-heptane; toluene for 2h; Heating; | 98% |

| Conditions | Yield |
|---|---|
| Stage #1: 1-bromo-4-methoxy-benzene; magnesium; lithium chloride In tetrahydrofuran at 25℃; Inert atmosphere; Schlenk technique; Stage #2: zinc(II) chloride In tetrahydrofuran at 0 - 25℃; for 0.25h; Inert atmosphere; Schlenk technique; | 98% |


| Conditions | Yield |
|---|---|
| Stage #1: bromochlorobenzene; magnesium; lithium chloride In tetrahydrofuran at 20℃; Inert atmosphere; Schlenk technique; Stage #2: zinc(II) chloride In tetrahydrofuran at 0 - 20℃; for 0.25h; Inert atmosphere; Schlenk technique; | 98% |


| Conditions | Yield |
|---|---|
| at 700 - 1100℃; for 336h; Sealed tube; Inert atmosphere; | 97% |


| Conditions | Yield |
|---|---|
| With sodium chloride for 0.0333333h; Time; | 97% |

| Conditions | Yield |
|---|---|
| for 0.0333333h; Time; | 97% |

Related products
Downstream Products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View




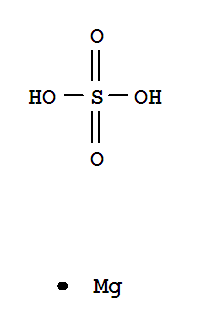











 F,
F, Xn
Xn