Hangzhou Think Chemical Co. Ltd
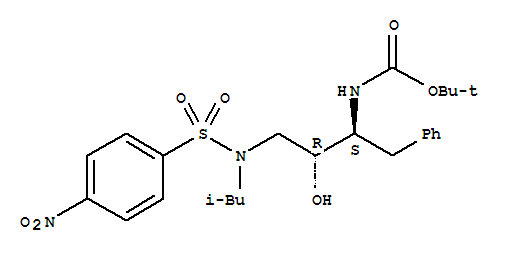
tert-Butyl [(1S,2R)-1-benzyl-2-hydroxy-3-[isobutyl[(4-nitrophenyl)sulfonyl]amino]propyl]carbamate CAS No.:191226-98-9 HANGZHOU THINK CHEMICAL CO., LTD. (THINKCHEM) is an integrative corporation of trade, research a
Cas:191226-98-9
Min.Order:1 Kilogram
FOB Price: $2.0
Type:Other
inquiryDayang Chem (Hangzhou) Co.,Ltd.
As a leading manufacturer and supplier of chemicals in China, DayangChem not only supply popular chemicals, but also DayangChem's R&D center offer custom synthesis services. DayangChem can provide different quantities of custom synthesis ch
Cas:191226-98-9
Min.Order:1 Kilogram
FOB Price: $3.0
Type:Lab/Research institutions
inquiryChemwill Asia Co., Ltd.
Our main production base is located in Xuzhou industry park. We are certified both to the ISO 9001 and ISO 14001 Standards, have a safety management system in place.Our R&D team masters core technology for process-design of target building block
Cas:191226-98-9
Min.Order:5 Kiloliter
FOB Price: $1.2 / 5.0
Type:Manufacturers
inquiryHenan Tianfu Chemical Co., Ltd.
Competitive Price High Quality Fast Delivery custom-made Welcome to Henan Tianfu Chemical Co., Ltd. website. Our company engages in Sodium Tripolyphosphate (STPP) and Sodium Hexametabphosphate (SHMP) production; development of noble me
Cas:191226-98-9
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryZhuozhou Wenxi import and Export Co., Ltd
WITH US,YOUR MONEY IN SAFE,YOUR BUSINESS IN SAFE 1)Quick Response Within 12 hours; 2)Quality Guarantee: All products are strictly tested by our QC, confirmed by QA and approved by third party lab in China, USA, Canada, Germany, UK, Italy, France et
Cas:191226-98-9
Min.Order:1 Kilogram
FOB Price: $139.0 / 210.0
Type:Trading Company
inquiryHenan Wentao Chemical Product Co., Ltd.
We are leading fine chemicals supplier in China and we had established stable business relationships with Samsung,LG,Merck,Thermo Fisher Scientific and so on.Our main business covers the fields below: 1.Noble Metal Catalysts (Pt.Pd...) 2.Orga
Cas:191226-98-9
Min.Order:1 Kilogram
FOB Price: $5.0
Type:Lab/Research institutions
inquiryHebei Nengqian Chemical Import and Export Co., LTD
Our Advantage Rich Experience Our products are sold all over Europe,North&South America, Sino-East, Asia and pacific area as well as Africa,we establish long term. Quality service Company cooperates with research institutes. We strictly con
Cas:191226-98-9
Min.Order:1 Kilogram
FOB Price: $15.0 / 50.0
Type:Trading Company
inquiryQingdao Beluga Import and Export Co., LTD
tert-Butyl [(1S,2R)-1-benzyl-2-hydroxy-3-[isobutyl[(4-nitrophenyl)sulfonyl]amino]propyl]carbamate CAS:191226-98-9 Qingdao Belugas Import and Export Co., Ltd. is a scientific and technological company integrating research and development, production
Cas:191226-98-9
Min.Order:1 Gram
Negotiable
Type:Lab/Research institutions
inquiryShandong Hanjiang Chemical Co., Ltd.
Hello, dear friend! I'm Hansen and Allen from China. Welcome to my lookchem mall! The following is a brief introduction of our company's products and services. If you are interested in our products, please contact us by emai
Cas:191226-98-9
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryXiamen Hisunny Chemical Co.,Ltd
Best quality & Attractive price & Professional service; Trial & Pilot & Commercial Hisunny Chemical is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality intermediates, specia
Cas:191226-98-9
Min.Order:0
Negotiable
Type:Manufacturers
inquiryAfine Chemicals Limited
Company Introduction 1. Established in 2005, with two independent business divisions: Fine chemicals division; Pharmaceutical division. 2. Main product: Optical brightener Textile auxiliary Dye stuff Pigments
Cas:191226-98-9
Min.Order:1 Gram
Negotiable
Type:Lab/Research institutions
inquirySHANGHAI T&W PHARMACEUTICAL CO., LTD.
A substitute for perfluorooctanoic acid, mainly used as a surfactant, dispersant, additive, etc Appearance:White solid or Colorless liquid Purity:99.3 % We will ship the goods in a timely manner as required We can provide relevant documents acc
Cas:191226-98-9
Min.Order:4 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryHenan Kanbei Chemical Co.,LTD
factory?direct?saleAppearance:White powder Storage:Sealed and preserved Package:200/Kilograms Application:healing drugs Transportation:By sea Port:Shanghai/tianjin
Cas:191226-98-9
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryZibo Hangyu Biotechnology Development Co., Ltd
Zibo Hangyu Biotechnology Development Co., Ltd is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality pharmaceuticals, intermediates, special chemicals and OLED intermediates and other fine chemi
Cas:191226-98-9
Min.Order:10 Gram
FOB Price: $100.0
Type:Lab/Research institutions
inquiryZibo Dorne chemical technology co. LTD
Product Details Grade: pharmaceutical grade Purity:99%+ ProductionCapacity: 1000 Kilogram/Month Scope of use: For scientific research only(The product must be used legally) Our Advantage 1. Best quality with competitive price. 2. Quick shipping,
Cas:191226-98-9
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryKono Chem Co.,Ltd
Product Name: tert-Butyl [(1S,2R)-1-benzyl-2-hydroxy-3-[isobutyl[(4-nitrophenyl)sulfonyl]amino]propyl]carbamate Synonyms: [(1S,2R)-1-Benzyl-2-hydroxy…Appearance:Solid powder Storage:Sealed,light and oxygen resistant Package:aluminum foil bag,carton
Cas:191226-98-9
Min.Order:0
Negotiable
Type:Other
inquiryXiamen Jenny Chemical Technology Co., Ltd.
GMP standard, high purity, competitive price, in stock 1. Quick Response: within 6 hours after receiving your email. 2. Quality Guarantee: All products are strictly tested by our QC, confirmed by QA, and approved by a third-party lab in China, USA,
Cas:191226-98-9
Min.Order:1 Milligram
Negotiable
Type:Trading Company
inquiryKAISA GROUP INC
1.Applied in food field.it can improve the immune system and prolong life. 2.Appliedin cosmetic field.it can improve the skin care. 3.Applied in pharmaceutical field.it can treat various dieases. 4.Our product quality assurance will make our customer
Cas:191226-98-9
Min.Order:1 Metric Ton
FOB Price: $7.0 / 8.0
Type:Trading Company
inquirySuzhou Health Chemicals Co., Ltd.
High quality,stable supply chain.Appearance:white/off-white or light yellow Storage:Store in cool and dry place, keep away from strong light and heat. Package:aluminum bottle,glass bottle,PTFE bottle,cardboard drum Application:This product can be use
Cas:191226-98-9
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquirySAGECHEM LIMITED
SAGECHEM is a chemical R&D, manufacturing and distribution company in China since 2009, including pharmaceutical intermediates, agrochemical, dyestuff intermediates, organosilicone, API and etc. We also offer a full range of services in custom synthe
Cas:191226-98-9
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryGIHI CHEMICALS CO.,LIMITED
Lower price, sample is available,SDS test documents are available,large stock in warehouseAppearance:White powder Storage:Sealed and preserved Package:200/Kilograms Application:Fine chemical intermediates, used as the main raw material for the synthe
Cas:191226-98-9
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryWuxi TAA Chemical Industry Co.,LTD.
1.A strong technical force and advanced processing equipments. The quality of the products has been strictly inspected and all kinds of index have reached or exceeded domestic and international standards.2. Now we have established long-term stable re
Cas:191226-98-9
Min.Order:0
Negotiable
Type:Other
inquiryHANGZHOU YUNUO CHEMICAL CO.,LTD
Superior quality, moderate price & quick delivery. Appearance:white crystalline powder Storage:Sealed in a cool ,dry and microtherm place , avoid light . Package:100g/bottle,1kg/bottle,25kg/drum or as per your request Application:It is an impo
Cas:191226-98-9
Min.Order:100 Gram
Negotiable
Type:Trading Company
inquiryHenan Allgreen Chemical Co.,Ltd
high quality Storage:Sealed, dry, microtherm , avoid light and smell. Package:According to the demand of customer Application:Organic synthesis Transportation:by air or by sea
Cas:191226-98-9
Min.Order:0
Negotiable
Type:Manufacturers
inquiryAecochem Corp.
Our clients, like BASF,CHEMO,Brenntag,ASR,Evonik,Merck and etc.Appearance:COA Storage:in stock Application:MSDS/TDS
Cas:191226-98-9
Min.Order:0
Negotiable
Type:Manufacturers
inquiryHubei Taiho Chemical Co.,LTD
TAIHO Unique Advantages:1.We're factory2.Free samples available3.Commodity inspection can be done4.ISO9001,Kosher certifications5.10 years experiences Storage:Store in cool &dry place Package:aluminium foil bag/fiber can/plastic drum Application:comp
Cas:191226-98-9
Min.Order:0
Negotiable
Type:Manufacturers
inquiryXian Changyue Biological Technology Co., Ltd.
best seller Application:API
Cas:191226-98-9
Min.Order:0
Negotiable
Type:Manufacturers
inquiryAntimex Chemical Limied
Ansciep Chemical is a professional enterprise manufacturing and distributing fine chemicals and speciality chemicals. We have been dedicated to heterocycle compounds and phenyl rings for tens of years. This is our mature product for export. Our quali
Cas:191226-98-9
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryGuangdong Juda Chemical Industrial Co.,Limited
Factory supply high purity low priceAppearance:solid or liquid Storage:sealed in cool and dry place Package:As customer's requested Application:Pharma Intermediate Transportation:by courier/air/sea Port:Any port in China
Cas:191226-98-9
Min.Order:0
Negotiable
Type:Trading Company
inquiryWuhan Circle Star Chem-medical Technology co.,Ltd.
good quality, competitive price, thoughtful after sale serviceAppearance:White to Off-White Solid Storage:Keep it in dry,shady and cool place Package:as your requirement Application:Pharma;Industry;Agricultural;chemical reaserch Transportation:by Sea
Cas:191226-98-9
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquirySynthetic route
-
-
98-74-8
4-Nitrobenzenesulfonyl chloride

-
-
160232-08-6
(2R,3S)-3-tert-butoxycarbonylamino-1-isobutylamino-4-phenyl-2-butanol

-
-
191226-98-9
[(1S,2R)-3-[(4-nitrophenylsulfonyl)(2-methylpropyl)amino]-2-hydroxy-1-(phenylmethyl)propyl]carbamic acid tert-butyl ester

| Conditions | Yield |
|---|---|
| With sodium hydrogencarbonate In dichloromethane; water at 20℃; for 21h; | 100% |
| With dmap; N-ethyl-N,N-diisopropylamine In tetrahydrofuran at 0 - 25℃; for 4h; | 98.2% |
| With sodium hydrogencarbonate In dichloromethane; water at 23℃; for 12h; | 96% |
-
-
78-81-9
isobutylamine

-
-
98760-08-8, 98818-34-9, 98818-35-0, 103127-56-6, 98737-29-2
(1-oxiranyl-2-phenylethyl)carbamic acid tert-butyl ester

-
-
98-74-8
4-Nitrobenzenesulfonyl chloride

-
-
191226-98-9
[(1S,2R)-3-[(4-nitrophenylsulfonyl)(2-methylpropyl)amino]-2-hydroxy-1-(phenylmethyl)propyl]carbamic acid tert-butyl ester

| Conditions | Yield |
|---|---|
| Stage #1: isobutylamine; (1-oxiranyl-2-phenylethyl)carbamic acid tert-butyl ester for 3h; Heating; Stage #2: 4-Nitrobenzenesulfonyl chloride With triethylamine In dichloromethane Reflux; | 92.88% |

-
-
98760-08-8, 98818-34-9, 98818-35-0, 103127-56-6, 98737-29-2
(1-oxiranyl-2-phenylethyl)carbamic acid tert-butyl ester

-
-
191226-98-9
[(1S,2R)-3-[(4-nitrophenylsulfonyl)(2-methylpropyl)amino]-2-hydroxy-1-(phenylmethyl)propyl]carbamic acid tert-butyl ester

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: ethanol / 3 h / 80 °C 2: aq. Na2CO3 / CH2Cl2 / 0 - 20 °C View Scheme | |
| Multi-step reaction with 2 steps 1: propan-2-ol / 2 h / 80 °C 2: 0.88 g / triethylamine / CH2Cl2 / 4 h / 20 °C View Scheme | |
| Multi-step reaction with 2 steps 1: 97 percent / propan-2-ol / 3 h / 80 °C 2: 90 percent / Et3N / CH2Cl2 / 16 h View Scheme |
-
-
326479-99-6
2(R)-((tert-butyloxycarbonyl)amino)-1,4-dichloro-3(S)-hydroxybutane

-
-
191226-98-9
[(1S,2R)-3-[(4-nitrophenylsulfonyl)(2-methylpropyl)amino]-2-hydroxy-1-(phenylmethyl)propyl]carbamic acid tert-butyl ester

| Conditions | Yield |
|---|---|
| Multi-step reaction with 6 steps 1.1: p-TsOH*H2O / CH2Cl2 2.1: NaH / tetrahydrofuran 3.1: CuBr*SMe2 / tetrahydrofuran 4.1: p-TsOH*H2O 4.2: KOH / methanol 5.1: 90 percent / propan-2-ol / 5 h / 50 °C 6.1: 88 percent / Et3N / CH2Cl2 / 12 h / 20 °C View Scheme | |
| Multi-step reaction with 6 steps 1: 100 percent / imidazole; DMAP / dimethylformamide / 12 h / 50 °C 2: 100 percent / NaH / tetrahydrofuran / 5 h / 0 °C 3: 75 percent / CuBr*Me2S / toluene; tetrahydrofuran / -78 - -20 °C 4: Bu4NF / tetrahydrofuran 5: 90 percent / propan-2-ol / 5 h / 50 °C 6: 88 percent / Et3N / CH2Cl2 / 12 h / 20 °C View Scheme | |
| Multi-step reaction with 6 steps 1.1: 100 percent / imidazole; DMAP / dimethylformamide / 12 h / 50 °C 2.1: 100 percent / NaH / tetrahydrofuran / 5 h / 0 °C 3.1: 82 percent / Et3N / methanol / 18 h / 20 °C 4.1: TBAF / tetrahydrofuran / 0.67 h / 20 °C 4.2: 75 percent / KOH / methanol / 2 h / 20 °C 5.1: 90 percent / propan-2-ol / 5 h / 50 °C 6.1: 88 percent / Et3N / CH2Cl2 / 12 h / 20 °C View Scheme |
-
-
326480-01-7
N-t-butyloxycarbonyl-2(S)-(1(S)-t-butyldimethylsilyloxy-2-chloroethyl)aziridine

-
-
191226-98-9
[(1S,2R)-3-[(4-nitrophenylsulfonyl)(2-methylpropyl)amino]-2-hydroxy-1-(phenylmethyl)propyl]carbamic acid tert-butyl ester

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: 75 percent / CuBr*Me2S / toluene; tetrahydrofuran / -78 - -20 °C 2: Bu4NF / tetrahydrofuran 3: 90 percent / propan-2-ol / 5 h / 50 °C 4: 88 percent / Et3N / CH2Cl2 / 12 h / 20 °C View Scheme | |
| Multi-step reaction with 4 steps 1.1: 82 percent / Et3N / methanol / 18 h / 20 °C 2.1: TBAF / tetrahydrofuran / 0.67 h / 20 °C 2.2: 75 percent / KOH / methanol / 2 h / 20 °C 3.1: 90 percent / propan-2-ol / 5 h / 50 °C 4.1: 88 percent / Et3N / CH2Cl2 / 12 h / 20 °C View Scheme |
-
-
357604-37-6
(S)-2-[(S)-2-Chloro-1-(tetrahydro-pyran-2-yloxy)-ethyl]-aziridine-1-carboxylic acid tert-butyl ester

-
-
191226-98-9
[(1S,2R)-3-[(4-nitrophenylsulfonyl)(2-methylpropyl)amino]-2-hydroxy-1-(phenylmethyl)propyl]carbamic acid tert-butyl ester

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1.1: CuBr*SMe2 / tetrahydrofuran 2.1: p-TsOH*H2O 2.2: KOH / methanol 3.1: 90 percent / propan-2-ol / 5 h / 50 °C 4.1: 88 percent / Et3N / CH2Cl2 / 12 h / 20 °C View Scheme |
-
-
326480-00-6
2(S)-t-butyldimethylsilyloxy-3(R)-(t-butyloxycarbonyl)amino-1,4-dichlorobutane

-
-
191226-98-9
[(1S,2R)-3-[(4-nitrophenylsulfonyl)(2-methylpropyl)amino]-2-hydroxy-1-(phenylmethyl)propyl]carbamic acid tert-butyl ester

| Conditions | Yield |
|---|---|
| Multi-step reaction with 5 steps 1: 100 percent / NaH / tetrahydrofuran / 5 h / 0 °C 2: 75 percent / CuBr*Me2S / toluene; tetrahydrofuran / -78 - -20 °C 3: Bu4NF / tetrahydrofuran 4: 90 percent / propan-2-ol / 5 h / 50 °C 5: 88 percent / Et3N / CH2Cl2 / 12 h / 20 °C View Scheme | |
| Multi-step reaction with 5 steps 1.1: 100 percent / NaH / tetrahydrofuran / 5 h / 0 °C 2.1: 82 percent / Et3N / methanol / 18 h / 20 °C 3.1: TBAF / tetrahydrofuran / 0.67 h / 20 °C 3.2: 75 percent / KOH / methanol / 2 h / 20 °C 4.1: 90 percent / propan-2-ol / 5 h / 50 °C 5.1: 88 percent / Et3N / CH2Cl2 / 12 h / 20 °C View Scheme |
-
-
357604-36-5
[(1R,2S)-3-Chloro-1-chloromethyl-2-(tetrahydro-pyran-2-yloxy)-propyl]-carbamic acid tert-butyl ester

-
-
191226-98-9
[(1S,2R)-3-[(4-nitrophenylsulfonyl)(2-methylpropyl)amino]-2-hydroxy-1-(phenylmethyl)propyl]carbamic acid tert-butyl ester

| Conditions | Yield |
|---|---|
| Multi-step reaction with 5 steps 1.1: NaH / tetrahydrofuran 2.1: CuBr*SMe2 / tetrahydrofuran 3.1: p-TsOH*H2O 3.2: KOH / methanol 4.1: 90 percent / propan-2-ol / 5 h / 50 °C 5.1: 88 percent / Et3N / CH2Cl2 / 12 h / 20 °C View Scheme |
-
-
357604-38-7
[(1S,2S)-1-Benzyl-3-chloro-2-(tetrahydro-pyran-2-yloxy)-propyl]-carbamic acid tert-butyl ester

-
-
191226-98-9
[(1S,2R)-3-[(4-nitrophenylsulfonyl)(2-methylpropyl)amino]-2-hydroxy-1-(phenylmethyl)propyl]carbamic acid tert-butyl ester

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1.1: p-TsOH*H2O 1.2: KOH / methanol 2.1: 90 percent / propan-2-ol / 5 h / 50 °C 3.1: 88 percent / Et3N / CH2Cl2 / 12 h / 20 °C View Scheme |
-
-
326480-02-8
2(S)-t-butyldimethylsilyloxy-3(S)-(t-butyloxycarbonyl)amino-1-chloro-4-phenylbutane

-
-
191226-98-9
[(1S,2R)-3-[(4-nitrophenylsulfonyl)(2-methylpropyl)amino]-2-hydroxy-1-(phenylmethyl)propyl]carbamic acid tert-butyl ester

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: Bu4NF / tetrahydrofuran 2: 90 percent / propan-2-ol / 5 h / 50 °C 3: 88 percent / Et3N / CH2Cl2 / 12 h / 20 °C View Scheme |
-
-
357604-39-8
(3S)-t-butyldimethylsilyloxy-(2R)-(t-butyloxycarbonyl)amino-4-chloro-1-phenylthiobutane

-
-
191226-98-9
[(1S,2R)-3-[(4-nitrophenylsulfonyl)(2-methylpropyl)amino]-2-hydroxy-1-(phenylmethyl)propyl]carbamic acid tert-butyl ester

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1.1: TBAF / tetrahydrofuran / 0.67 h / 20 °C 1.2: 75 percent / KOH / methanol / 2 h / 20 °C 2.1: 90 percent / propan-2-ol / 5 h / 50 °C 3.1: 88 percent / Et3N / CH2Cl2 / 12 h / 20 °C View Scheme |
-
-
1229623-13-5
tert-butyl ((2S)-4-chloro-3-hydroxy-1-phenylbutan-2-yl)carbamate

-
-
191226-98-9
[(1S,2R)-3-[(4-nitrophenylsulfonyl)(2-methylpropyl)amino]-2-hydroxy-1-(phenylmethyl)propyl]carbamic acid tert-butyl ester

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1.1: sodium hydrogencarbonate / dichloromethane / 5 h / Reflux 2.1: triethylamine / dichloromethane / Reflux 2.2: Reflux View Scheme |
-
-
220871-52-3
1(R,S)-<1'-(S)-<(tert-butyloxycarbonyl)amino>-2-phenylethyl>oxirane

-
-
191226-98-9
[(1S,2R)-3-[(4-nitrophenylsulfonyl)(2-methylpropyl)amino]-2-hydroxy-1-(phenylmethyl)propyl]carbamic acid tert-butyl ester

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: isopropyl alcohol / 6 h / 80 °C 2: triethylamine / dichloromethane / 12 h / 0 - 20 °C View Scheme |

-
-
191226-98-9
[(1S,2R)-3-[(4-nitrophenylsulfonyl)(2-methylpropyl)amino]-2-hydroxy-1-(phenylmethyl)propyl]carbamic acid tert-butyl ester

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: sodium hydrogencarbonate / dichloromethane / 5 h / Reflux 2: triethylamine / dichloromethane / Reflux View Scheme |
-
-
98-74-8
4-Nitrobenzenesulfonyl chloride

-
-
191226-98-9
[(1S,2R)-3-[(4-nitrophenylsulfonyl)(2-methylpropyl)amino]-2-hydroxy-1-(phenylmethyl)propyl]carbamic acid tert-butyl ester

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 1,4-dioxane / 1 h / 10 - 25 °C 2: potassium carbonate; N-benzyl-N,N,N-triethylammonium chloride / 1,4-dioxane / 6 h / 80 - 85 °C View Scheme |
-
-
89840-80-2
N-isobutyl-4-nitrobenzenesulfonamide

-
-
98760-08-8, 98818-34-9, 98818-35-0, 103127-56-6, 98737-29-2
(1-oxiranyl-2-phenylethyl)carbamic acid tert-butyl ester

-
-
191226-98-9
[(1S,2R)-3-[(4-nitrophenylsulfonyl)(2-methylpropyl)amino]-2-hydroxy-1-(phenylmethyl)propyl]carbamic acid tert-butyl ester

| Conditions | Yield |
|---|---|
| With N-benzyl-N,N,N-triethylammonium chloride; potassium carbonate In 1,4-dioxane at 80 - 85℃; for 6h; | 10 g |
-
-
165727-45-7
tert-butyl ((2S,3S)-4-chloro-3-hydroxy-1-phenylbutan-2-yl)carbamate

-
-
191226-98-9
[(1S,2R)-3-[(4-nitrophenylsulfonyl)(2-methylpropyl)amino]-2-hydroxy-1-(phenylmethyl)propyl]carbamic acid tert-butyl ester

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: sodium carbonate / water / 3 h / 60 - 65 °C 2: triethylamine / dichloromethane / 40 - 45 °C View Scheme | |
| Multi-step reaction with 2 steps 1: potassium hydroxide / ethanol / 15 - 20 °C / Large scale 2: triethylamine / isopropyl alcohol / 40 - 60 °C / Large scale View Scheme |
-
-
910642-68-1
C15H20N3O3(1+)

-
-
191226-98-9
[(1S,2R)-3-[(4-nitrophenylsulfonyl)(2-methylpropyl)amino]-2-hydroxy-1-(phenylmethyl)propyl]carbamic acid tert-butyl ester

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: hydrogenchloride 2: aluminum isopropoxide / isopropyl alcohol / 3 h / Reflux 3: sodium carbonate / water / 3 h / 60 - 65 °C 4: triethylamine / dichloromethane / 40 - 45 °C View Scheme |
-
-
102123-74-0
(S)-tert-butyl (4-chloro-3-oxo-1-phenylbutan-2-yl)carbamate

-
-
191226-98-9
[(1S,2R)-3-[(4-nitrophenylsulfonyl)(2-methylpropyl)amino]-2-hydroxy-1-(phenylmethyl)propyl]carbamic acid tert-butyl ester

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: aluminum isopropoxide / isopropyl alcohol / 3 h / Reflux 2: sodium carbonate / water / 3 h / 60 - 65 °C 3: triethylamine / dichloromethane / 40 - 45 °C View Scheme | |
| Multi-step reaction with 3 steps 1: aluminum isopropoxide / isopropyl alcohol / 3 h / Reflux 2: sodium carbonate / water / 3 h / 60 - 65 °C 3: triethylamine / dichloromethane / 40 - 45 °C View Scheme | |
| Multi-step reaction with 3 steps 1: aluminum sec-butoxide / toluene; 2-methyl-propan-1-ol / 15 - 20 °C / Large scale 2: potassium hydroxide / ethanol / 15 - 20 °C / Large scale 3: triethylamine / isopropyl alcohol / 40 - 60 °C / Large scale View Scheme |
-
-
13734-34-4
N-tert-butoxycarbonyl-L-phenylalanine

-
-
191226-98-9
[(1S,2R)-3-[(4-nitrophenylsulfonyl)(2-methylpropyl)amino]-2-hydroxy-1-(phenylmethyl)propyl]carbamic acid tert-butyl ester

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: triethylamine 2: aluminum sec-butoxide / toluene; 2-methyl-propan-1-ol / 15 - 20 °C / Large scale 3: potassium hydroxide / ethanol / 15 - 20 °C / Large scale 4: triethylamine / isopropyl alcohol / 40 - 60 °C / Large scale View Scheme |
-
-
98760-08-8
(2R,3S)-3-[N-(tert-butyloxycarbonyl)amino]-1,2-epoxy-4-phenylbutane

-
-
191226-98-9
[(1S,2R)-3-[(4-nitrophenylsulfonyl)(2-methylpropyl)amino]-2-hydroxy-1-(phenylmethyl)propyl]carbamic acid tert-butyl ester

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: isopropyl alcohol / 20 - 30 °C 2: triethylamine / dichloromethane / 15 - 25 °C View Scheme |
-
-
191226-98-9
[(1S,2R)-3-[(4-nitrophenylsulfonyl)(2-methylpropyl)amino]-2-hydroxy-1-(phenylmethyl)propyl]carbamic acid tert-butyl ester

-
-
251105-80-3
N-(3S-amino-2R-hydroxy-4-phenylbutyl)-N-isobutyl-4-nitro-benzenesulfonamide hydrochloride

| Conditions | Yield |
|---|---|
| With trifluoroacetic acid In dichloromethane at 0 - 20℃; for 3h; | 100% |
| With trifluoroacetic acid In dichloromethane for 3h; | 83% |
| With trifluoroacetic acid In dichloromethane at 25℃; for 2h; | 69.2% |
-
-
191226-98-9
[(1S,2R)-3-[(4-nitrophenylsulfonyl)(2-methylpropyl)amino]-2-hydroxy-1-(phenylmethyl)propyl]carbamic acid tert-butyl ester

-
-
169280-56-2
4-amino-N-(2R,3S)(3-amino-2-hydroxy-4-phenylbutyl)-N-isobutylbenzenesulfonamide

| Conditions | Yield |
|---|---|
| Stage #1: [(1S,2R)-3-[(4-nitrophenylsulfonyl)(2-methylpropyl)amino]-2-hydroxy-1-(phenylmethyl)propyl]carbamic acid tert-butyl ester With palladium 10% on activated carbon; ammonium formate; acetic acid In tetrahydrofuran at 15 - 20℃; Large scale; Stage #2: With hydrogenchloride In tetrahydrofuran; water at 35 - 45℃; Large scale; | 99.4% |
| Stage #1: [(1S,2R)-3-[(4-nitrophenylsulfonyl)(2-methylpropyl)amino]-2-hydroxy-1-(phenylmethyl)propyl]carbamic acid tert-butyl ester With triethanolamine; hydrogen; palladium 10% on activated carbon In methanol at 40 - 45℃; for 2h; Stage #2: With hydrogenchloride; water for 2h; Reflux; Stage #3: With sodium hydroxide In water; isopropyl alcohol at 20℃; for 10h; pH=9 - 10; | 95% |
| Multi-step reaction with 2 steps 1: 95 percent / H2 / Pd/C / ethyl acetate / 11 h / 23 °C 2: CF3COOH / CH2Cl2 / 0.67 h / 23 °C View Scheme |
-
-
191226-98-9
[(1S,2R)-3-[(4-nitrophenylsulfonyl)(2-methylpropyl)amino]-2-hydroxy-1-(phenylmethyl)propyl]carbamic acid tert-butyl ester

-
-
183004-94-6
3-S-(N-tert-butyloxyformamido)-[2R-hydroxy-1-[(4-aminophenylsulfonyl)(2-methylpropyl)]amino]-4-phenylbutane

| Conditions | Yield |
|---|---|
| With hydrogen; palladium on activated charcoal In ethanol at 22 - 30℃; Product distribution / selectivity; | 97% |
| With hydrogen; palladium dihydroxide In ethyl acetate for 4h; | 95% |
| With hydrogen; palladium on activated charcoal In ethyl acetate at 23℃; for 11h; | 95% |
-
-
144-62-7
oxalic acid

-
-
191226-98-9
[(1S,2R)-3-[(4-nitrophenylsulfonyl)(2-methylpropyl)amino]-2-hydroxy-1-(phenylmethyl)propyl]carbamic acid tert-butyl ester

| Conditions | Yield |
|---|---|
| Stage #1: [(1S,2R)-3-[(4-nitrophenylsulfonyl)(2-methylpropyl)amino]-2-hydroxy-1-(phenylmethyl)propyl]carbamic acid tert-butyl ester With hydrogen In isopropyl alcohol at 25 - 35℃; under 3750.38 - 4500.45 Torr; Stage #2: oxalic acid In toluene at 0 - 30℃; for 3h; Solvent; Reagent/catalyst; | 90% |
-
-
138499-08-8
1-({[(3S)-tetrahydro-3-furanyloxy]carbonyl}oxy)-2,5-pyrrolidinedione

-
-
191226-98-9
[(1S,2R)-3-[(4-nitrophenylsulfonyl)(2-methylpropyl)amino]-2-hydroxy-1-(phenylmethyl)propyl]carbamic acid tert-butyl ester

-
-
160231-69-6
4-nitro-N-((2R(syn),3S)-2-hydroxy-4-phenyl-3-((S)-tetrahydrofuran-3-yloxycarbonylamino)-butyl)-N-isobutylbenzenesulfonamide

| Conditions | Yield |
|---|---|
| Stage #1: [(1S,2R)-3-[(4-nitrophenylsulfonyl)(2-methylpropyl)amino]-2-hydroxy-1-(phenylmethyl)propyl]carbamic acid tert-butyl ester With hydrogenchloride In dichloromethane for 1.5h; Stage #2: 1-({[(3S)-tetrahydro-3-furanyloxy]carbonyl}oxy)-2,5-pyrrolidinedione With triethylamine In dichloromethane at 20℃; for 4h; Further stages.; | 85% |
-
-
191226-98-9
[(1S,2R)-3-[(4-nitrophenylsulfonyl)(2-methylpropyl)amino]-2-hydroxy-1-(phenylmethyl)propyl]carbamic acid tert-butyl ester

-
-
244634-31-9
[(1S,2R)-3-[(4-nitrophenylsulfonyl)(2-methylpropyl)amino]-2-hydroxy-1-(phenylmethyl)propyl]amine hydrochloride

| Conditions | Yield |
|---|---|
| With hydrogenchloride In 1,4-dioxane at 0 - 20℃; | 85% |
| With hydrogenchloride In ethanol | 80% |
| With hydrogenchloride In ethanol; water at 75 - 80℃; for 1h; | 173 g |
| With hydrogenchloride In ethanol; dichloromethane; water at 5 - 80℃; for 2h; Solvent; | 330 g |
-
-
5070-13-3
bis-(p-nitrophenyl) carbonate

-
-
191226-98-9
[(1S,2R)-3-[(4-nitrophenylsulfonyl)(2-methylpropyl)amino]-2-hydroxy-1-(phenylmethyl)propyl]carbamic acid tert-butyl ester

-
-
229495-94-7
Carbonic acid (1R,2S)-2-tert-butoxycarbonylamino-1-{[isobutyl-(4-nitro-benzenesulfonyl)-amino]-methyl}-3-phenyl-propyl ester 4-nitro-phenyl ester

| Conditions | Yield |
|---|---|
| With lithium hydroxide In N,N-dimethyl-formamide at 20℃; for 2h; | 80% |
-
-
2937-50-0
Allyl chloroformate

-
-
191226-98-9
[(1S,2R)-3-[(4-nitrophenylsulfonyl)(2-methylpropyl)amino]-2-hydroxy-1-(phenylmethyl)propyl]carbamic acid tert-butyl ester

-
-
599173-46-3
Carbonic acid allyl ester (1R,2S)-2-tert-butoxycarbonylamino-1-{[isobutyl-(4-nitro-benzenesulfonyl)-amino]-methyl}-3-phenyl-propyl ester

| Conditions | Yield |
|---|---|
| With n-butyllithium In tetrahydrofuran at 0 - 20℃; | 36% |
-
-
79-22-1
methyl chloroformate

-
-
191226-98-9
[(1S,2R)-3-[(4-nitrophenylsulfonyl)(2-methylpropyl)amino]-2-hydroxy-1-(phenylmethyl)propyl]carbamic acid tert-butyl ester

-
-
599173-45-2
Carbonic acid (1R,2S)-2-tert-butoxycarbonylamino-1-{[isobutyl-(4-nitro-benzenesulfonyl)-amino]-methyl}-3-phenyl-propyl ester methyl ester

| Conditions | Yield |
|---|---|
| With n-butyllithium In tetrahydrofuran at 0 - 20℃; for 3h; | 13% |
-
-
191226-98-9
[(1S,2R)-3-[(4-nitrophenylsulfonyl)(2-methylpropyl)amino]-2-hydroxy-1-(phenylmethyl)propyl]carbamic acid tert-butyl ester

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: 95 percent / H2 / Pd/C / ethyl acetate / 11 h / 23 °C 2: CF3COOH / CH2Cl2 / 0.67 h / 23 °C 3: 75 percent / iPr2NEt / acetonitrile / 8 h / 23 °C View Scheme |
-
-
191226-98-9
[(1S,2R)-3-[(4-nitrophenylsulfonyl)(2-methylpropyl)amino]-2-hydroxy-1-(phenylmethyl)propyl]carbamic acid tert-butyl ester

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: TFA / CH2Cl2 / 1 h 2: Et3N / tetrahydrofuran / 20 °C 3: SnCl2*2H2O / ethyl acetate / 80 °C View Scheme |
-
-
191226-98-9
[(1S,2R)-3-[(4-nitrophenylsulfonyl)(2-methylpropyl)amino]-2-hydroxy-1-(phenylmethyl)propyl]carbamic acid tert-butyl ester

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: TFA / CH2Cl2 / 1 h 2: Et3N / tetrahydrofuran / 20 °C 3: SnCl2*2H2O / ethyl acetate / 80 °C View Scheme |
-
-
191226-98-9
[(1S,2R)-3-[(4-nitrophenylsulfonyl)(2-methylpropyl)amino]-2-hydroxy-1-(phenylmethyl)propyl]carbamic acid tert-butyl ester

-
-
919081-52-0
2-oxo-3-phenyl-oxazolidine-5-carboxylic acid {1-benzyl-2-hydroxy-3-[isobutyl-(4-nitro-benzenesulfonyl)-amino]-propyl}-amide

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: TFA / CH2Cl2 / 1 h 2: Et3N / tetrahydrofuran / 20 °C View Scheme |
-
-
191226-98-9
[(1S,2R)-3-[(4-nitrophenylsulfonyl)(2-methylpropyl)amino]-2-hydroxy-1-(phenylmethyl)propyl]carbamic acid tert-butyl ester

-
-
919081-33-7
2-oxo-3-phenyl-oxazolidine-5-carboxylic acid {1-benzyl-2-hydroxy-3-[isobutyl-(4-nitro-benzenesulfonyl)-amino]-propyl}-amide

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: TFA / CH2Cl2 / 1 h 2: Et3N / tetrahydrofuran / 20 °C View Scheme |
-
-
191226-98-9
[(1S,2R)-3-[(4-nitrophenylsulfonyl)(2-methylpropyl)amino]-2-hydroxy-1-(phenylmethyl)propyl]carbamic acid tert-butyl ester

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: TFA / CH2Cl2 / 1 h 2: Et3N / tetrahydrofuran / 20 °C 3: SnCl2*2H2O / ethyl acetate / 80 °C View Scheme |
-
-
191226-98-9
[(1S,2R)-3-[(4-nitrophenylsulfonyl)(2-methylpropyl)amino]-2-hydroxy-1-(phenylmethyl)propyl]carbamic acid tert-butyl ester

-
-
919081-34-8
3-(3-fluoro-phenyl)-2-oxo-oxazolidine-5-carboxylic acid {1-benzyl-2-hydroxy-3-[isobutyl-(4-nitro-benzenesulfonyl)-amino]-propyl}-amide

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: TFA / CH2Cl2 / 1 h 2: Et3N / tetrahydrofuran / 20 °C View Scheme |
-
-
191226-98-9
[(1S,2R)-3-[(4-nitrophenylsulfonyl)(2-methylpropyl)amino]-2-hydroxy-1-(phenylmethyl)propyl]carbamic acid tert-butyl ester

-
-
919081-38-2
3-(4-acetyl-phenyl)-2-oxo-oxazolidine-5-carboxylic acid {1-benzyl-2-hydroxy-3-[isobutyl-(4-nitro-benzenesulfonyl)-amino]-propyl}-amide

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: TFA / CH2Cl2 / 1 h 2: Et3N / tetrahydrofuran / 20 °C View Scheme |
-
-
191226-98-9
[(1S,2R)-3-[(4-nitrophenylsulfonyl)(2-methylpropyl)amino]-2-hydroxy-1-(phenylmethyl)propyl]carbamic acid tert-butyl ester

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: TFA / CH2Cl2 / 1 h 2: Et3N / tetrahydrofuran / 20 °C 3: SnCl2*2H2O / ethyl acetate / 80 °C View Scheme |
-
-
191226-98-9
[(1S,2R)-3-[(4-nitrophenylsulfonyl)(2-methylpropyl)amino]-2-hydroxy-1-(phenylmethyl)propyl]carbamic acid tert-butyl ester

-
-
919081-37-1
3-(3-acetyl-phenyl)-2-oxo-oxazolidine-5-carboxylic acid {1-benzyl-2-hydroxy-3-[isobutyl-(4-nitro-benzenesulfonyl)-amino]-propyl}-amide

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: TFA / CH2Cl2 / 1 h 2: Et3N / tetrahydrofuran / 20 °C View Scheme |
-
-
191226-98-9
[(1S,2R)-3-[(4-nitrophenylsulfonyl)(2-methylpropyl)amino]-2-hydroxy-1-(phenylmethyl)propyl]carbamic acid tert-butyl ester

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: TFA / CH2Cl2 / 1 h 2: Et3N / tetrahydrofuran / 20 °C 3: SnCl2*2H2O / ethyl acetate / 80 °C View Scheme |
-
-
191226-98-9
[(1S,2R)-3-[(4-nitrophenylsulfonyl)(2-methylpropyl)amino]-2-hydroxy-1-(phenylmethyl)propyl]carbamic acid tert-butyl ester

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: TFA / CH2Cl2 / 1 h 2: Et3N / tetrahydrofuran / 20 °C 3: SnCl2*2H2O / ethyl acetate / 80 °C View Scheme |
-
-
191226-98-9
[(1S,2R)-3-[(4-nitrophenylsulfonyl)(2-methylpropyl)amino]-2-hydroxy-1-(phenylmethyl)propyl]carbamic acid tert-butyl ester

-
-
919081-35-9
3-(3,4-difluoro-phenyl)-2-oxo-oxazolidine-5-carboxylic acid {1-benzyl-2-hydroxy-3-[isobutyl-(4-nitro-benzenesulfonyl)-amino]-propyl}-amide

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: TFA / CH2Cl2 / 1 h 2: Et3N / tetrahydrofuran / 20 °C View Scheme |
-
-
191226-98-9
[(1S,2R)-3-[(4-nitrophenylsulfonyl)(2-methylpropyl)amino]-2-hydroxy-1-(phenylmethyl)propyl]carbamic acid tert-butyl ester

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: TFA / CH2Cl2 / 1 h 2: Et3N / tetrahydrofuran / 20 °C 3: SnCl2*2H2O / ethyl acetate / 80 °C View Scheme |
-
-
191226-98-9
[(1S,2R)-3-[(4-nitrophenylsulfonyl)(2-methylpropyl)amino]-2-hydroxy-1-(phenylmethyl)propyl]carbamic acid tert-butyl ester

-
-
919081-36-0
2-oxo-3-(3-trifluoromethyl-phenyl)-oxazolidine-5-carboxylic acid {1-benzyl-2-hydroxy-3-[isobutyl-(4-nitro-benzenesulfonyl)-amino]-propyl}-amide

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: TFA / CH2Cl2 / 1 h 2: Et3N / tetrahydrofuran / 20 °C View Scheme |
-
-
191226-98-9
[(1S,2R)-3-[(4-nitrophenylsulfonyl)(2-methylpropyl)amino]-2-hydroxy-1-(phenylmethyl)propyl]carbamic acid tert-butyl ester

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: H2 / Pd(OH)2 on carbon / ethyl acetate / 1 h 2: 0.75 g / trifluoroacetic acid / CH2Cl2 / 20 °C 3: 45 percent / 3-dimethoxyphosphoryloxy-1,2,3-benzotriazine-4(3H)-one; triethylamine / tetrahydrofuran / 4 h View Scheme |
-
-
191226-98-9
[(1S,2R)-3-[(4-nitrophenylsulfonyl)(2-methylpropyl)amino]-2-hydroxy-1-(phenylmethyl)propyl]carbamic acid tert-butyl ester

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: H2 / Pd(OH)2 on carbon / ethyl acetate / 1 h 2: 0.75 g / trifluoroacetic acid / CH2Cl2 / 20 °C 3: 51 percent / N-hydroxysuccinimide; 1,3-dicyclohexylcarbodiimide; N-methylmorpholine / CH2Cl2 / 18 h View Scheme |
-
-
191226-98-9
[(1S,2R)-3-[(4-nitrophenylsulfonyl)(2-methylpropyl)amino]-2-hydroxy-1-(phenylmethyl)propyl]carbamic acid tert-butyl ester

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: H2 / Pd(OH)2 on carbon / ethyl acetate / 1 h 2: 0.75 g / trifluoroacetic acid / CH2Cl2 / 20 °C 3: N-hydroxysuccinimide; 1,3-dicyclohexylcarbodiimide; N-methylmorpholine / CH2Cl2 / 18 h View Scheme |
Related products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View