Hebei Nengqian Chemical Import and Export Co., LTD
Our Advantage Rich Experience Our products are sold all over Europe,North&South America, Sino-East, Asia and pacific area as well as Africa,we establish long term. Quality service Company cooperates with research institutes. We strictly con
Cas:218600-44-3
Min.Order:1 Kilogram
FOB Price: $1.0 / 10.0
Type:Trading Company
inquiryZhuozhou Wenxi import and Export Co., Ltd
WITH US,YOUR MONEY IN SAFE,YOUR BUSINESS IN SAFE 1)Quick Response Within 12 hours; 2)Quality Guarantee: All products are strictly tested by our QC, confirmed by QA and approved by third party lab in China, USA, Canada, Germany, UK, Italy, France et
Cas:218600-44-3
Min.Order:10 Gram
FOB Price: $146.0 / 176.0
Type:Trading Company
inquiryHubei CuiRan Biotechnology Co., Ltd
Hubei CuiRan Biotechnology Co., Ltd is a leading company in the research, development, manufacture and marketing of High Quality Phytochemicals and Extracts(especially Active Ingredients from Traditional Chinese Medicine,Traditional Chinese Medicine)
Cas:218600-44-3
Min.Order:10 Milligram
Negotiable
Type:Lab/Research institutions
inquiryZibo Hangyu Biotechnology Development Co., Ltd
Zibo Hangyu Biotechnology Development Co., Ltd is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality pharmaceuticals, intermediates, special chemicals and OLED intermediates and other fine chemi
Cas:218600-44-3
Min.Order:10 Gram
Negotiable
Type:Lab/Research institutions
inquiryXiamen Jenny Chemical Technology Co., Ltd.
GMP standard, high purity, competitive price, in stock 1. Quick Response: within 6 hours after receiving your email. 2. Quality Guarantee: All products are strictly tested by our QC, confirmed by QA, and approved by a third-party lab in China, USA,
Henan Allgreen Chemical Co.,Ltd
high quality Storage:Sealed, dry, microtherm , avoid light and smell. Package:According to the demand of customer Application:Organic synthesis Transportation:by air or by sea
Cas:218600-44-3
Min.Order:0
Negotiable
Type:Manufacturers
inquiryAntimex Chemical Limied
factory?direct?sale Application:Fine chemical intermediates, used as the main raw material for the synthesis of various pesticides, medicines, surfactants, polymer monomers, and antifungal agents
Cas:218600-44-3
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryGuangdong Juda Chemical Industrial Co.,Limited
Factory supply high purity low priceAppearance:solid or liquid Storage:sealed in cool and dry place Package:As customer's requested Application:Pharma Intermediate Transportation:by courier/air/sea Port:Any port in China
Hangzhou Fandachem Co.,Ltd
Oleana-1,9(11)-dien-28-oic acid, 2-cyano-3,12-dioxo-Appearance:white crystalline powder Storage:Store in dry, dark and ventilated place Package:25KG drum Application:pharmaceutical intermediate Transportation:by air, by sea, by express
Cas:218600-44-3
Min.Order:0
Negotiable
Type:Other
inquiryBOC Sciences
BOC Sciences provides a wide range of research chemicals and biochemicals including inhibitors, building blocks, GMP Products, impurities and metabolites, APIs for Veterinary, Natural Compounds, ADCs, Stem Cell Molecule and chiral compounds. Package:
Shanghai Yuanye Bio-Technology Co., Ltd.
good quality, competitive price, thoughtful after sale serviceAppearance:white powder Storage:Keep it in dry,shady and cool place Package:5mg Application:Pharma;Industry;Agricultural;chemical reaserch Transportation:by express or by sea Port:Any port
Chemlyte Solutions
factory?direct?sale Application:Fine chemical intermediates, used as the main raw material for the synthesis of various pesticides, medicines, surfactants, polymer monomers, and antifungal agents
Cas:218600-44-3
Min.Order:0
Negotiable
Type:Other
inquiryChemsigma International Co.,Ltd.
bulk?production Application:Pharmaceutical intermediates
Cas:218600-44-3
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryAmadis Chemical Co., Ltd.
1.Professional synthesis laboratory and production base. 2.Strong synthesis team and service team. 3.Professional data management system. 4.We provide the professional test date and product information ,ex. HNMR ,CNMR,FNMR, HPLC/G
Cas:218600-44-3
Min.Order:10 Milligram
Negotiable
Type:Lab/Research institutions
inquiryHebei Luspharm Trading Co.,Ltd
1. Best prices with satisfied quality; 2. Clients can choose the package's Courier (EMS, DHL, FedEx, UPS,TNT); 3.Clients can choose the packing way for his produccts from many recent effective packing ways; 4.Specials are poss
Cas:218600-44-3
Min.Order:1 Gram
FOB Price: $1.0
Type:Trading Company
inquiryWuhan Sun-shine Bio-technology Corporation Limited
Wuhan Sun-shine Bio-technology Corporation Limited is specializing in the anticancer, antitumor,heart head blood-vessel,pharmaceutical intermediates,fine Chemicals production and customization. Company has strong ability of research and development
Cas:218600-44-3
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryChangzhou Extraordinary Pharmatech co.,LTD
Changzhou Extraordinary Pharmatech co., LTD. As a leading chemical manufacturer and supplier in China.DAS authentication is passed.We can provide the popular precursor chemicals, we have our own strong R & D team, have our own laboratories and fa
Wuhan New-Rise Biochem Co,. Ltd
1.High quality and competetive price 1)for prulifloxacin exported we can offer coa certificate and official invoice. 2)we are manufacturer with own lab and factory, can provide high quality products with factory price. 3)products purity is tes
Cas:218600-44-3
Min.Order:1 Kilogram
Negotiable
Type:Trading Company
inquirysuzhou BetterBioChem Co., Ltd.
Betterbiochem is specialized in APIs, pharmaceutical intermediates & fine chemicals. 1. The best quality, competitive price. 2. The best service before or after shipment. 3. Rich experience in export operation. 4. Rich experience in
Guangzhou PI & PI Biotech Inc. Ltd.
PI &PI Biotech Inc. provides great quality of APIs and Drug Impurities. Application:R & D
Cas:218600-44-3
Min.Order:0 Metric Ton
Negotiable
Type:Lab/Research institutions
inquiryU-Chemo Holding Co.,Limited
U-CHEMO Holding Co.,Limited is a very fast developing high-technology enterprise combining with manufacture and sales.U-CHEMO supply high quality products and excellent service to our customers.We make the insect pheromones, censor in the quality con
Shanghai united Scientific Co.,Ltd.
United Scientific Company Located in Shanghai of China , is a competitive player in the global specialty and fine chemical market. Fenghua has both the expertise and flexibility to produce a wide range of chemicals. Focusing on developing the innovat
Cas:218600-44-3
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryNanjing Chemlin Chemical Co., Ltd.
Nanjing Chemlin Chemical Industry Co., Ltd was established in Aug,1999, located in the industrial park of Nanjing University of Technology, It is a private enterprises in Jiangsu Province with 1200 square meters’ R&D center. Our R&D center has a well
Wuhan MoonZY Biological Technology Co.,Ltd
Superiority: 1. Quality: High purity, strict quality control, 10 years experience in chemical industry. 2. Price: Moderate price, more QTY more discount. 3. MOQ: Sample order is welcome to test quality. 4. Shipping time: In Stock, can be sh
Cas:218600-44-3
Min.Order:1 Gram
Negotiable
Type:Lab/Research institutions
inquiryWuhan Prominence Bio-technology Co., Ltd
Wuhan Prominence Bio-technology Co., Ltd is a leading provider and manufacturer of pharmaceutical and chemical products. Mainly involves API, pharmaceutical intermediates peditides and natural extract etc. We are capable to supply you with any quanti
Wuxi Morality Chemical Co., Ltd
Do best quality products, erect the morality model Application:please email us, thanks
Cas:218600-44-3
Min.Order:0
Negotiable
Type:Other
inquiryTianjin Tongde Biological Technology Co., Ltd.
2-cyano-3,12-dioxooleana-1,9(11)-dien-28-oic acid cas: 218600-44-3 Storage:hermetically sealed & avoid heat, moisture and light Application:2-cyano-3,12-dioxooleana-1,9(11)-dien-28-oic acid cas: 218600-44-3 Port:Tianjin
Cas:218600-44-3
Min.Order:0 Metric Ton
Negotiable
Type:Trading Company
inquiryShanghai Run-Biotech Co., Ltd.
1.We have rich experience in the development and production 2.Our factory is in Shanghai of China,We have convenient traffic conditions.3.We have advanced testing equipment and shipping line. Package:5g, 10g, 50g, 100g, 2kg Application:Pharmaceutical
Sino Rarechem Labs Co.Ltd
We have two R&D center both in Suzhou and Nanjing. The R&D Center owns labs with 2500m466 Production Base. And excellent staff, advanced equipments ,instruments, strict quality management system enhance our competitiveness.We are good at rare&hard to
Shanghai Caerulum Pharma Discovery Co.,Ltd
Shanghai Caerulum Pharma Discovery is the manufacturer of APIs and intermediates of some of the latest compounds in the market. Our focus is on cancer, diabetes and other therapeutic areas. Shanghai Caerulum Pharma Discovery (CPD) offers high qua
Cas:218600-44-3
Min.Order:25 Milligram
Negotiable
Type:Lab/Research institutions
inquirySynthetic route
-
-
218600-53-4
bardoxolone methyl

-
-
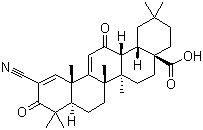
218600-44-3
bardoxolone

| Conditions | Yield |
|---|---|
| With lithium iodide In N,N-dimethyl-formamide | 71% |
| With lithium iodide In N,N-dimethyl-formamide | 71% |
| With lithium iodide In N,N-dimethyl-formamide at 153℃; for 12h; Inert atmosphere; | 70.3% |
| With lithium iodide In N,N-dimethyl-formamide for 4h; Heating; | 68% |
| With lithium iodide In N,N-dimethyl-formamide Inert atmosphere; Reflux; | 44% |
-
-
915710-94-0, 1244059-91-3, 1165931-54-3
tert‐butyl N‐[2‐(N‐hydroxycarbamimidoyl)ethyl]carbamate

-
-
443104-14-1
2-cyano-3,12-dioxooleana-1,9(11)-dien-28-oic acid chloride

-
B
-
218600-44-3
bardoxolone

| Conditions | Yield |
|---|---|
| Stage #1: tert‐butyl N‐[2‐(N‐hydroxycarbamimidoyl)ethyl]carbamate; 2-cyano-3,12-dioxooleana-1,9(11)-dien-28-oic acid chloride With triethylamine In dichloromethane at 20℃; for 2h; Inert atmosphere; Stage #2: With tetra(n-butyl)ammonium hydroxide In tetrahydrofuran; water at 20℃; for 5h; Inert atmosphere; | A 45% B n/a |

-
-
915710-94-0, 1244059-91-3, 1165931-54-3
tert‐butyl N‐[2‐(N‐hydroxycarbamimidoyl)ethyl]carbamate

-
-
443104-14-1
2-cyano-3,12-dioxooleana-1,9(11)-dien-28-oic acid chloride

-
B
-
218600-44-3
bardoxolone

| Conditions | Yield |
|---|---|
| With triethylamine In dichloromethane at 20℃; for 2h; Inert atmosphere; |

-
-
67015-15-0
3-methoxypropionamidoxime hydrochloride

-
-
443104-14-1
2-cyano-3,12-dioxooleana-1,9(11)-dien-28-oic acid chloride

-
B
-
218600-44-3
bardoxolone

| Conditions | Yield |
|---|---|
| With triethylamine; trimethylamine In dichloromethane at 20℃; Inert atmosphere; |

-
-
65023-20-3
methyl 3β-acetoxy-12-oxoolean-9(11)-en-28-oate

-
-
218600-44-3
bardoxolone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 7 steps 1: 97 percent / methanolic KOH / 0.5 h / Heating 2: 92 percent / Jones reagent / acetone / 0.17 h / 20 °C 3: 99 percent / NaOMe / benzene / 2 h / 20 °C 4: 66 percent / NH2OH*HCl / ethanol; H2O / 1 h / Heating 5: 100 percent / NaOMe / methanol; diethyl ether / 0.75 h / 20 °C 6: 92 percent / DDQ / benzene / 0.5 h / Heating 7: 68 percent / LiI / dimethylformamide / 4 h / Heating View Scheme | |
| Multi-step reaction with 7 steps 1: aq. KOH / methanol 2: CrO3, H2SO4 3: 100 percent / sodium methoxide / benzene 4: 61 percent / NH2OH*HCl / aq. ethanol 5: 100 percent / sodium methoxide / methanol; diethyl ether 6: 1.) phenylselenenyl chloride, 2.) 30 percent aq. H2O2 / 1.) ethyl acetate, 2.) THF 7: 71 percent / LiI / dimethylformamide View Scheme | |
| Multi-step reaction with 6 steps 1: potassium hydroxide; water / methanol 2: sodium methylate / benzene 3: hydroxylamine hydrochloride / water; ethanol 4: sodium methylate / methanol; diethyl ether 5: Phenylselenyl chloride / ethyl acetate 6: lithium iodide / N,N-dimethyl-formamide View Scheme | |
| Multi-step reaction with 5 steps 1.1: potassium hydroxide / methanol / 1 h / Reflux 2.1: chromium(VI) oxide; sulfuric acid / water; acetone / 0 - 20 °C 3.1: lithium diisopropyl amide / tetrahydrofuran; n-heptane; ethylbenzene / 0.33 h / -78 - 20 °C / Inert atmosphere 3.2: 0.08 h / -78 - 20 °C 4.1: 2,3-dicyano-5,6-dichloro-p-benzoquinone / benzene / 0.5 h / Reflux; Inert atmosphere 5.1: lithium iodide / N,N-dimethyl-formamide / Inert atmosphere; Reflux View Scheme |
-
-
218600-50-1
methyl 3,12-dioxoolean-9(11)-en-28-oate

-
-
218600-44-3
bardoxolone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 5 steps 1: 99 percent / NaOMe / benzene / 2 h / 20 °C 2: 66 percent / NH2OH*HCl / ethanol; H2O / 1 h / Heating 3: 100 percent / NaOMe / methanol; diethyl ether / 0.75 h / 20 °C 4: 92 percent / DDQ / benzene / 0.5 h / Heating 5: 68 percent / LiI / dimethylformamide / 4 h / Heating View Scheme | |
| Multi-step reaction with 5 steps 1: 100 percent / sodium methoxide / benzene 2: 61 percent / NH2OH*HCl / aq. ethanol 3: 100 percent / sodium methoxide / methanol; diethyl ether 4: 1.) phenylselenenyl chloride, 2.) 30 percent aq. H2O2 / 1.) ethyl acetate, 2.) THF 5: 71 percent / LiI / dimethylformamide View Scheme | |
| Multi-step reaction with 5 steps 1: sodium methylate / benzene 2: hydroxylamine hydrochloride / water; ethanol 3: sodium methylate / methanol; diethyl ether 4: Phenylselenyl chloride / ethyl acetate 5: lithium iodide / N,N-dimethyl-formamide View Scheme | |
| Multi-step reaction with 3 steps 1.1: lithium diisopropyl amide / tetrahydrofuran; n-heptane; ethylbenzene / 0.33 h / -78 - 20 °C / Inert atmosphere 1.2: 0.08 h / -78 - 20 °C 2.1: 2,3-dicyano-5,6-dichloro-p-benzoquinone / benzene / 0.5 h / Reflux; Inert atmosphere 3.1: lithium iodide / N,N-dimethyl-formamide / Inert atmosphere; Reflux View Scheme |
-
-
65023-19-0
methyl 3β-hydroxy-12-oxoolean-9(11)-en-28-oate

-
-
218600-44-3
bardoxolone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 6 steps 1: 92 percent / Jones reagent / acetone / 0.17 h / 20 °C 2: 99 percent / NaOMe / benzene / 2 h / 20 °C 3: 66 percent / NH2OH*HCl / ethanol; H2O / 1 h / Heating 4: 100 percent / NaOMe / methanol; diethyl ether / 0.75 h / 20 °C 5: 92 percent / DDQ / benzene / 0.5 h / Heating 6: 68 percent / LiI / dimethylformamide / 4 h / Heating View Scheme | |
| Multi-step reaction with 6 steps 1: CrO3, H2SO4 2: 100 percent / sodium methoxide / benzene 3: 61 percent / NH2OH*HCl / aq. ethanol 4: 100 percent / sodium methoxide / methanol; diethyl ether 5: 1.) phenylselenenyl chloride, 2.) 30 percent aq. H2O2 / 1.) ethyl acetate, 2.) THF 6: 71 percent / LiI / dimethylformamide View Scheme | |
| Multi-step reaction with 4 steps 1.1: chromium(VI) oxide; sulfuric acid / water; acetone / 0 - 20 °C 2.1: lithium diisopropyl amide / tetrahydrofuran; n-heptane; ethylbenzene / 0.33 h / -78 - 20 °C / Inert atmosphere 2.2: 0.08 h / -78 - 20 °C 3.1: 2,3-dicyano-5,6-dichloro-p-benzoquinone / benzene / 0.5 h / Reflux; Inert atmosphere 4.1: lithium iodide / N,N-dimethyl-formamide / Inert atmosphere; Reflux View Scheme |
-
-
218600-52-3
methyl 12-oxoisoxazolo[4,5-b]olean-9(11)-en-28-oate

-
-
218600-44-3
bardoxolone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: 100 percent / NaOMe / methanol; diethyl ether / 0.75 h / 20 °C 2: 92 percent / DDQ / benzene / 0.5 h / Heating 3: 68 percent / LiI / dimethylformamide / 4 h / Heating View Scheme | |
| Multi-step reaction with 3 steps 1: 100 percent / sodium methoxide / methanol; diethyl ether 2: 1.) phenylselenenyl chloride, 2.) 30 percent aq. H2O2 / 1.) ethyl acetate, 2.) THF 3: 71 percent / LiI / dimethylformamide View Scheme | |
| Multi-step reaction with 3 steps 1: sodium methylate / methanol; diethyl ether 2: Phenylselenyl chloride / ethyl acetate 3: lithium iodide / N,N-dimethyl-formamide View Scheme |
-
-
1309236-68-7
2-cyano-3-hydroxy-12-oxoolean-2(3),9(11)-dien-28-oic acid

-
-
218600-44-3
bardoxolone

| Conditions | Yield |
|---|---|
| With 2,3-dicyano-5,6-dichloro-p-benzoquinone In benzene for 0.25h; Reflux; | 91% |
-
-
305818-39-7
methyl 2-hydroxymethylene-3,12-dioxoolean-9(11)-en-28-oate

-
-
218600-44-3
bardoxolone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: 66 percent / NH2OH*HCl / ethanol; H2O / 1 h / Heating 2: 100 percent / NaOMe / methanol; diethyl ether / 0.75 h / 20 °C 3: 92 percent / DDQ / benzene / 0.5 h / Heating 4: 68 percent / LiI / dimethylformamide / 4 h / Heating View Scheme |
-
-
305818-40-0
1,2-dihydro-2-cyano-3,12-dioxooleana-1,9(11)-dien-28-oic acid methyl ester

-
-
218600-44-3
bardoxolone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 92 percent / DDQ / benzene / 0.5 h / Heating 2: 68 percent / LiI / dimethylformamide / 4 h / Heating View Scheme |

-
-
218600-44-3
bardoxolone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 1.) phenylselenenyl chloride, 2.) 30 percent aq. H2O2 / 1.) ethyl acetate, 2.) THF 2: 71 percent / LiI / dimethylformamide View Scheme | |
| Multi-step reaction with 2 steps 1: Phenylselenyl chloride / ethyl acetate 2: lithium iodide / N,N-dimethyl-formamide View Scheme | |
| Multi-step reaction with 2 steps 1: 2,3-dicyano-5,6-dichloro-p-benzoquinone / benzene / 0.5 h / Reflux; Inert atmosphere 2: lithium iodide / N,N-dimethyl-formamide / Inert atmosphere; Reflux View Scheme |
-
-
1309236-68-7
2-cyano-3-hydroxy-12-oxoolean-2(3),9(11)-dien-28-oic acid

-
A
-
1309943-46-1
2-cyano-3,12-dioxooleana-1,9(11)-diene-13β,28-lactone

-
B
-
218600-44-3
bardoxolone

| Conditions | Yield |
|---|---|
| With 2,3-dicyano-5,6-dichloro-p-benzoquinone In benzene for 24h; Reflux; | A 58% B 28% |
-
-
218600-51-2
(4aS,6aR,6bS,8aR,12aS,14aR,14bS)-11-Formyl-2,2,6a,6b,9,9,12a-heptamethyl-10,14-dioxo-1,3,4,5,6,6a,6b,7,8,8a,9,10,11,12,12a,14,14a,14b-octadecahydro-2H-picene-4a-carboxylic acid methyl ester

-
-
218600-44-3
bardoxolone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: 61 percent / NH2OH*HCl / aq. ethanol 2: 100 percent / sodium methoxide / methanol; diethyl ether 3: 1.) phenylselenenyl chloride, 2.) 30 percent aq. H2O2 / 1.) ethyl acetate, 2.) THF 4: 71 percent / LiI / dimethylformamide View Scheme | |
| Multi-step reaction with 4 steps 1: hydroxylamine hydrochloride / water; ethanol 2: sodium methylate / methanol; diethyl ether 3: Phenylselenyl chloride / ethyl acetate 4: lithium iodide / N,N-dimethyl-formamide View Scheme |
-
-
508-02-1
Oleanolic acid

-
-
218600-44-3
bardoxolone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 9 steps 1.1: triethylamine; dmap / dichloromethane / 12 h / 20 °C / Inert atmosphere 2.1: potassium carbonate / acetone / 24 h / 20 °C 3.1: 3-chloro-benzenecarboperoxoic acid / dichloromethane / 0 - 20 °C 4.1: hydrogen bromide; bromine / acetonitrile / 19 h / 35 °C 5.1: potassium hydroxide / methanol / 1 h / Reflux 6.1: chromium(VI) oxide; sulfuric acid / water; acetone / 0 - 20 °C 7.1: lithium diisopropyl amide / tetrahydrofuran; n-heptane; ethylbenzene / 0.33 h / -78 - 20 °C / Inert atmosphere 7.2: 0.08 h / -78 - 20 °C 8.1: 2,3-dicyano-5,6-dichloro-p-benzoquinone / benzene / 0.5 h / Reflux; Inert atmosphere 9.1: lithium iodide / N,N-dimethyl-formamide / Inert atmosphere; Reflux View Scheme | |
| Multi-step reaction with 9 steps 1.1: triethylamine; dmap / dichloromethane / 12 h / 20 °C / Inert atmosphere 2.1: potassium carbonate / acetone / 24 h / 20 °C 3.1: 3-chloro-benzenecarboperoxoic acid / dichloromethane / 12 h / 0 - 20 °C 4.1: hydrogen bromide; bromine / acetonitrile / 19 h / 35 °C 5.1: potassium hydroxide / methanol / 1 h / Reflux 6.1: chromium(VI) oxide; sulfuric acid / water; acetone / 0 - 20 °C 7.1: lithium diisopropyl amide / tetrahydrofuran; n-heptane; ethylbenzene / 0.33 h / -78 - 20 °C / Inert atmosphere 7.2: 0.08 h / -78 - 20 °C 8.1: 2,3-dicyano-5,6-dichloro-p-benzoquinone / benzene / 0.5 h / Reflux; Inert atmosphere 9.1: lithium iodide / N,N-dimethyl-formamide / Inert atmosphere; Reflux View Scheme | |
| Multi-step reaction with 5 steps 1.1: potassium carbonate / N,N-dimethyl-formamide / 24 h / 20 °C / Inert atmosphere 2.1: 1-hydroxy-3H-benz[d][1,2]iodoxole-1,3-dione / dimethyl sulfoxide; fluorobenzene / 24 h / 85 °C / Inert atmosphere 3.1: 3-chloro-benzenecarboperoxoic acid; hydrogen bromide / dichloromethane / 26 h / 20 °C 3.2: 24 h / 35 °C 4.1: potassium iodide / N,N-dimethyl-formamide / 30 h / 120 °C 5.1: lithium iodide / N,N-dimethyl-formamide / 12 h / 153 °C / Inert atmosphere View Scheme |
-
-
25493-69-0, 122798-61-2, 1721-57-9
(4aS,6aS,6bR,10S,12aR)-methyl 10-acetoxy-2,2,6a,6b,9,9,12a-heptamethyl-1,2,3,4,4a,5,6,6a,6b,7,8,8a,9,10,11,12,12a,12b,13,14b-icosahydropicene-4a-carboxylate

-
-
218600-44-3
bardoxolone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 7 steps 1.1: 3-chloro-benzenecarboperoxoic acid / dichloromethane / 0 - 20 °C 2.1: hydrogen bromide; bromine / acetonitrile / 19 h / 35 °C 3.1: potassium hydroxide / methanol / 1 h / Reflux 4.1: chromium(VI) oxide; sulfuric acid / water; acetone / 0 - 20 °C 5.1: lithium diisopropyl amide / tetrahydrofuran; n-heptane; ethylbenzene / 0.33 h / -78 - 20 °C / Inert atmosphere 5.2: 0.08 h / -78 - 20 °C 6.1: 2,3-dicyano-5,6-dichloro-p-benzoquinone / benzene / 0.5 h / Reflux; Inert atmosphere 7.1: lithium iodide / N,N-dimethyl-formamide / Inert atmosphere; Reflux View Scheme | |
| Multi-step reaction with 7 steps 1.1: 3-chloro-benzenecarboperoxoic acid / dichloromethane / 12 h / 0 - 20 °C 2.1: hydrogen bromide; bromine / acetonitrile / 19 h / 35 °C 3.1: potassium hydroxide / methanol / 1 h / Reflux 4.1: chromium(VI) oxide; sulfuric acid / water; acetone / 0 - 20 °C 5.1: lithium diisopropyl amide / tetrahydrofuran; n-heptane; ethylbenzene / 0.33 h / -78 - 20 °C / Inert atmosphere 5.2: 0.08 h / -78 - 20 °C 6.1: 2,3-dicyano-5,6-dichloro-p-benzoquinone / benzene / 0.5 h / Reflux; Inert atmosphere 7.1: lithium iodide / N,N-dimethyl-formamide / Inert atmosphere; Reflux View Scheme |
-
-
4339-72-4
oleanolic acid 3-acetate

-
-
218600-44-3
bardoxolone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 8 steps 1.1: potassium carbonate / acetone / 24 h / 20 °C 2.1: 3-chloro-benzenecarboperoxoic acid / dichloromethane / 0 - 20 °C 3.1: hydrogen bromide; bromine / acetonitrile / 19 h / 35 °C 4.1: potassium hydroxide / methanol / 1 h / Reflux 5.1: chromium(VI) oxide; sulfuric acid / water; acetone / 0 - 20 °C 6.1: lithium diisopropyl amide / tetrahydrofuran; n-heptane; ethylbenzene / 0.33 h / -78 - 20 °C / Inert atmosphere 6.2: 0.08 h / -78 - 20 °C 7.1: 2,3-dicyano-5,6-dichloro-p-benzoquinone / benzene / 0.5 h / Reflux; Inert atmosphere 8.1: lithium iodide / N,N-dimethyl-formamide / Inert atmosphere; Reflux View Scheme | |
| Multi-step reaction with 8 steps 1.1: potassium carbonate / acetone / 24 h / 20 °C 2.1: 3-chloro-benzenecarboperoxoic acid / dichloromethane / 12 h / 0 - 20 °C 3.1: hydrogen bromide; bromine / acetonitrile / 19 h / 35 °C 4.1: potassium hydroxide / methanol / 1 h / Reflux 5.1: chromium(VI) oxide; sulfuric acid / water; acetone / 0 - 20 °C 6.1: lithium diisopropyl amide / tetrahydrofuran; n-heptane; ethylbenzene / 0.33 h / -78 - 20 °C / Inert atmosphere 6.2: 0.08 h / -78 - 20 °C 7.1: 2,3-dicyano-5,6-dichloro-p-benzoquinone / benzene / 0.5 h / Reflux; Inert atmosphere 8.1: lithium iodide / N,N-dimethyl-formamide / Inert atmosphere; Reflux View Scheme |
-
-
25493-94-1
methyl 3-O-β-acetyl-11-dehydro-12-oxo-18β-oleanolate

-
-
218600-44-3
bardoxolone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 6 steps 1.1: hydrogen bromide; bromine / acetonitrile / 19 h / 35 °C 2.1: potassium hydroxide / methanol / 1 h / Reflux 3.1: chromium(VI) oxide; sulfuric acid / water; acetone / 0 - 20 °C 4.1: lithium diisopropyl amide / tetrahydrofuran; n-heptane; ethylbenzene / 0.33 h / -78 - 20 °C / Inert atmosphere 4.2: 0.08 h / -78 - 20 °C 5.1: 2,3-dicyano-5,6-dichloro-p-benzoquinone / benzene / 0.5 h / Reflux; Inert atmosphere 6.1: lithium iodide / N,N-dimethyl-formamide / Inert atmosphere; Reflux View Scheme |
-
-
1724-17-0, 73584-64-2
oleanolic acid methyl ester

-
-
218600-44-3
bardoxolone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1.1: 1-hydroxy-3H-benz[d][1,2]iodoxole-1,3-dione / dimethyl sulfoxide; fluorobenzene / 24 h / 85 °C / Inert atmosphere 2.1: 3-chloro-benzenecarboperoxoic acid; hydrogen bromide / dichloromethane / 26 h / 20 °C 2.2: 24 h / 35 °C 3.1: potassium iodide / N,N-dimethyl-formamide / 30 h / 120 °C 4.1: lithium iodide / N,N-dimethyl-formamide / 12 h / 153 °C / Inert atmosphere View Scheme |
-
-
69660-90-8
(4aS,6aS,6bR,8aR,12aR,12bR,14bS)-methyl2,2,6a,6b,9,9,12a-heptamethyl-10-oxo-1,2,3,4,4a,5,6,6a,6b,7,8,8a,9,10,12a,12b,13,14b-octadecahydropicene-4a-carboxylate

-
-
218600-44-3
bardoxolone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1.1: 3-chloro-benzenecarboperoxoic acid; hydrogen bromide / dichloromethane / 26 h / 20 °C 1.2: 24 h / 35 °C 2.1: potassium iodide / N,N-dimethyl-formamide / 30 h / 120 °C 3.1: lithium iodide / N,N-dimethyl-formamide / 12 h / 153 °C / Inert atmosphere View Scheme |

-
-
218600-44-3
bardoxolone

| Conditions | Yield |
|---|---|
| With 2,3-dicyano-5,6-dichloro-p-benzoquinone In benzene for 0.333333h; Reflux; |
-
-
218600-44-3
bardoxolone

-
-
443104-14-1
2-cyano-3,12-dioxooleana-1,9(11)-dien-28-oic acid chloride

| Conditions | Yield |
|---|---|
| With oxalyl dichloride In dichloromethane | 100% |
| With oxalyl dichloride In dichloromethane at 20℃; | 100% |
| With oxalyl dichloride In dichloromethane | 100% |
-
-
218600-44-3
bardoxolone

-
-
1192122-92-1
(4aS,6aR,6bS,8aR,12aS,14aR,14bS)-11-cyano-2,2,6a,6b,9,9,12a-heptamethyl-10,14-dioxo-1,2,3,4,4a,5,6,6a,6b,7,8,8a,9,10,12a,14,14a,14b-octadecahydropicene-4a-carbonyl azide

| Conditions | Yield |
|---|---|
| With diphenyl phosphoryl azide; triethylamine at 0 - 20℃; | 90% |
| With diphenyl phosphoryl azide; triethylamine In toluene at 20℃; for 6h; | 90% |
-
-
218600-44-3
bardoxolone

-
-
572-09-8
2,3,4,6-tetra-O-acetyl-α-D-glucopyranosyl bromide

| Conditions | Yield |
|---|---|
| With Aliquat 336; potassium carbonate In dichloromethane | 75% |
| With potassium carbonate; Aliquat 336 In dichloromethane; water | 75% |
-
-
218600-44-3
bardoxolone

-
-
1191917-53-9
(4aS,6aR,6bS,8aR,12aS,14aR,14bS)-11-cyano-2,2,6a,6b,9,9,12a-heptamethyl-10,14-dioxo-1,2,3,4,4a,5,6,6a,6b,7,8,8a,9,10,12a,14,14a,14b-octadecahydropicene-4a-carbonyl fluoride

| Conditions | Yield |
|---|---|
| With diethylamino-sulfur trifluoride In chloroform at 20℃; for 6.5h; | 94% |
-
-
218600-44-3
bardoxolone

| Conditions | Yield |
|---|---|
| With dihydrogen peroxide In methanol; water; acetonitrile at 20℃; for 30h; | 90% |

| Conditions | Yield |
|---|---|
| Stage #1: bardoxolone With oxalyl dichloride; N,N-dimethyl-formamide In dichloromethane at 20℃; for 1.16667h; Cooling with ice; Stage #2: methanol In dichloromethane for 1h; | 96% |

-
-
218600-44-3
bardoxolone

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetone at 45℃; for 4h; | 68% |

-
-
218600-44-3
bardoxolone

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetone at 45℃; for 4h; | 74% |
-
-
218600-44-3
bardoxolone

| Conditions | Yield |
|---|---|
| With palladium 10% on activated carbon; hydrogen In tetrahydrofuran | 84% |

| Conditions | Yield |
|---|---|
| With 1,8-diazabicyclo[5.4.0]undec-7-ene In toluene Heating / reflux; | 83% |

| Conditions | Yield |
|---|---|
| Stage #1: isosorbide mononitrate; bardoxolone With potassium carbonate In N,N-dimethyl-formamide at 20℃; Stage #2: propionyl chloride In N,N-dimethyl-formamide | 35% |


| Conditions | Yield |
|---|---|
| Stage #1: isosorbide mononitrate; bardoxolone With potassium carbonate In N,N-dimethyl-formamide at 20℃; Stage #2: acetyl chloride In N,N-dimethyl-formamide for 6h; | 35% |

-
-
218600-44-3
bardoxolone

-
-
1192122-95-4
(4aR,6aS,6bR,8aS,12aS,12bR,14bS)-8a-isocyanato-4,4,6a,6b,11,11,14b-heptamethyl-3,13-dioxo-3,4,4a,5,6,6a,6b,7,8,8a,9,10,11,12,12a,12b,13,14b-octadecahydropicene-2-carbonitrile

| Conditions | Yield |
|---|---|
| Stage #1: bardoxolone With diphenyl phosphoryl azide; triethylamine In toluene at 0 - 20℃; Stage #2: In benzene at 80℃; for 2h; | |
| Multi-step reaction with 2 steps 1: diphenyl phosphoryl azide; triethylamine / 0 - 20 °C 2: benzene / 2 h / 80 °C View Scheme |
-
-
218600-44-3
bardoxolone

-
A
-
1192122-92-1
(4aS,6aR,6bS,8aR,12aS,14aR,14bS)-11-cyano-2,2,6a,6b,9,9,12a-heptamethyl-10,14-dioxo-1,2,3,4,4a,5,6,6a,6b,7,8,8a,9,10,12a,14,14a,14b-octadecahydropicene-4a-carbonyl azide

-
B
-
1192122-95-4
(4aR,6aS,6bR,8aS,12aS,12bR,14bS)-8a-isocyanato-4,4,6a,6b,11,11,14b-heptamethyl-3,13-dioxo-3,4,4a,5,6,6a,6b,7,8,8a,9,10,11,12,12a,12b,13,14b-octadecahydropicene-2-carbonitrile

| Conditions | Yield |
|---|---|
| With diphenyl phosphoryl azide; triethylamine In toluene at 0 - 20℃; Overall yield = 94 %; Overall yield = 19.7 g; |
-
-
218600-44-3
bardoxolone

-
-
359860-27-8
N-(2-(2-(2-(2-amidoethoxy)ethoxy)ethoxy)ethoxy)biotinamide

| Conditions | Yield |
|---|---|
| With 2`,3`-dideoxycytidine; dmap In dichloromethane at 20℃; for 48h; | 79% |
-
-
218600-44-3
bardoxolone

-
-
443104-14-1
2-cyano-3,12-dioxooleana-1,9(11)-dien-28-oic acid chloride

-
-
1224947-97-0
2-cyano-3,10-dioxooleana-1,9(11)-dien-28-oic acid anhydride

| Conditions | Yield |
|---|---|
| With triethylamine In tetrahydrofuran at 20℃; for 1.5h; | 96% |
-
-
218600-44-3
bardoxolone

| Conditions | Yield |
|---|---|
| With 1,8-diazabicyclo[5.4.0]undec-7-ene In toluene Heating; | 97% |
-
-
218600-44-3
bardoxolone

| Conditions | Yield |
|---|---|
| With 1,8-diazabicyclo[5.4.0]undec-7-ene In toluene Heating; | 100% |
-
-
218600-44-3
bardoxolone

| Conditions | Yield |
|---|---|
| With 1,8-diazabicyclo[5.4.0]undec-7-ene In toluene Heating; | 89% |
-
-
218600-44-3
bardoxolone

| Conditions | Yield |
|---|---|
| With 1,8-diazabicyclo[5.4.0]undec-7-ene In toluene Heating; | 81% |
-
-
218600-44-3
bardoxolone

| Conditions | Yield |
|---|---|
| With 1,8-diazabicyclo[5.4.0]undec-7-ene In toluene Heating; | 74% |
Related products
Raw Materials
Downstream Products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View