ZHANGJIAGANG HUAMAO FINE CHEMICAL CO.,LTD
Zhangjiagang Huamao Fine Chemical Co., Ltd. was founded in 2006. It is a Sino-US joint venture engaged in research and production. The company is situated in Yangtze River International Chemical Industrial Park, Free Trade Zone, Zhangjiagang. Based o
Dayang Chem (Hangzhou) Co.,Ltd.
DayangChem exported this product to many countries and regions at best price. If you are looking for the material's manufacturer or supplier in China, DayangChem is your best choice. Pls contact with us freely for getting detailed product spe
Cas:26171-23-3
Min.Order:1 Kilogram
FOB Price: $3.0
Type:Lab/Research institutions
inquirySimagchem Corporation
Welcome to Simagchem, your partner in China as a premier supply of bulk specialty chemicals for industry and life science. We introduce experienced quality product and exceptional JIT service with instant market intelligence in China to benefit our
Cas:26171-23-3
Min.Order:1 Kilogram
Negotiable
Type:Manufacturers
inquiryChemwill Asia Co., Ltd.
Our main production base is located in Xuzhou industry park. We are certified both to the ISO 9001 and ISO 14001 Standards, have a safety management system in place.Our R&D team masters core technology for process-design of target building block
Cas:26171-23-3
Min.Order:5 Kiloliter
FOB Price: $1.2 / 5.0
Type:Manufacturers
inquiryHebei Nengqian Chemical Import and Export Co., LTD
With our good experience, we offer detailed technical support and advice to assist customers. We communicate closely with customers to establish their quality requirements. Consistent Quality Our plant has strict quality control in each manufacturin
Cas:26171-23-3
Min.Order:1 Kilogram
FOB Price: $2.0 / 5.0
Type:Trading Company
inquiryZhuozhou Wenxi import and Export Co., Ltd
WITH US,YOUR MONEY IN SAFE,YOUR BUSINESS IN SAFE 1)Quick Response Within 12 hours; 2)Quality Guarantee: All products are strictly tested by our QC, confirmed by QA and approved by third party lab in China, USA, Canada, Germany, UK, Italy, France et
Cas:26171-23-3
Min.Order:1 Kilogram
FOB Price: $139.0 / 210.0
Type:Trading Company
inquiryLIDE PHARMACEUTICALS LIMITED
Advantage : LIDE PHARMACEUTICALS LTD. is a mid-small manufacturing-type enterprise, engaged in pharmaceutical intermediates of R&D, custom-made and production, and also involving trading chemicals for export. We have established the
Cas:26171-23-3
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryShanghai Upbio Tech Co.,Ltd
1.In No Less 10 years exporting experience. you can 100% received goods 2.Lower Price with higher quality 3,Free sample 4,We are sincerely responsible for the "product quality" and "After Service" Upbio is Specializ
Cas:26171-23-3
Min.Order:1 Gram
Negotiable
Type:Lab/Research institutions
inquiryHenan Wentao Chemical Product Co., Ltd.
Henan Wentao Chemical Product Co.,Ltd is Located in Zhengzhou High-tech Development Zone with import and export license, We passed ISO 9001:2008 as well, Henan Wentao has developed more than 1000 compounds, which are widely used in the fields of prod
Cas:26171-23-3
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryAfine Chemicals Limited
Our Services 1. New Molecules R&D 2. Own test center HPLC NMR GC LC-MS 3. API and Intermediates from China reputed manufacturers 4. Documents support COA MOA MSDS DMF open part Our advantages 1. Government awarded company. Top 100 enter
Hangzhou J&H Chemical Co., Ltd.
J&H CHEM R&D center can offer custom synthesis according to the contract research and development services for the fine chemicals, pharmaceutical, biotechnique and some of the other chemicals. J&H CHEM has some Manufacturing base in Jia
Cas:26171-23-3
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryShanghai Massive Chemical Technology Co., Ltd.
Massive Chemical is certified with ISO9001 and ISO14001 manufacturer for this product. We will offer all documents as requirement for the materials which includes, Certificate of Analysis, Material Safety Data Sheet, and Method of Analysis and
Cas:26171-23-3
Min.Order:1 Gram
FOB Price: $1.0
Type:Lab/Research institutions
inquiryZibo Dorne chemical technology co. LTD
Product Details Grade: pharmaceutical grade Purity:99%+ ProductionCapacity: 1000 Kilogram/Month Scope of use: For scientific research only(The product must be used legally) Our Advantage 1. Best quality with competitive price. 2. Quick shipping,
Cas:26171-23-3
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryXiamen Jenny Chemical Technology Co., Ltd.
GMP standard, high purity, competitive price, in stock 1. Quick Response: within 6 hours after receiving your email. 2. Quality Guarantee: All products are strictly tested by our QC, confirmed by QA, and approved by a third-party lab in China, USA,
Hunan chemfish Pharmaceutical co.,Ltd
Hunan chemfish Pharmaceutical co.,Ltd.located in Lugu High-tech industral park ,Hunan province . with its own R&D center and more than 10000㎡manufacture plant . Chemfish owns 40 reactors from 1000L to 8000L. With complete auxiliary equipment as
Cas:26171-23-3
Min.Order:0 Metric Ton
Negotiable
Type:Manufacturers
inquiryHangzhou Huarong Pharm Co., Ltd.
Hangzhou Huarong Pharm Co., Ltd.established since 2006 , has been actively developing specialty products for Finished Dosages, APIs, Intermediates, and Fine chemicals markets in North America, Europe, Korea, Japan, Mid-East and all over the World. Hu
KAISA GROUP INC
1.Applied in food field.it can improve the immune system and prolong life. 2.Appliedin cosmetic field.it can improve the skin care. 3.Applied in pharmaceutical field.it can treat various dieases. 4.Our product quality assurance will make our customer
Cas:26171-23-3
Min.Order:1 Metric Ton
FOB Price: $7.0 / 8.0
Type:Trading Company
inquiryHenan Tianfu Chemical Co., Ltd.
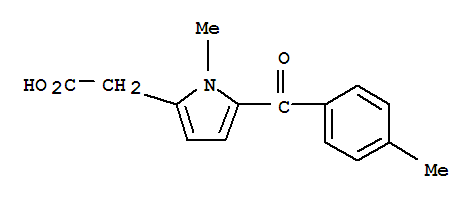
Product Name: Tolmetin Synonyms: Tolmetin;Tolmetine;TOLMETINUM;2-[1-Methyl-5-(4-methylbenzoyl)-pyrrol-2-yl]acetic acid;1H-Pyrrole-2-acetic acid, 1-methyl-5-(4-methylbenzoyl)-;1-Methyl-5-p-toluoylpyrrole-2-acetic acid;5-[(p-Tolyl)carbonyl]-1-methyl
Bluecrystal chem-union
We are a Union of chemistry in China, consists of chemists,engineers, laboratories,factories in China. We organize surplus capacity of R&D and production as well as custom synthesis for chemical products and chemical business project. We are supp
Cas:26171-23-3
Min.Order:0
Negotiable
Type:Trading Company
inquiryHubei Vanz Pharm Co.,Ltd
ISO Factory/Good qualityAppearance:off white Storage:dry and cool place Package:10G/BAG; 1KG/BAG ;25KG/DRUM Application:Active pharaceutical ingredients Transportation:BY SEA,AIR,EXPRESS Port:SHANGHAI;BEIJING
GIHI CHEMICALS CO.,LIMITED
Lower price, sample is available,SDS test documents are available,large stock in warehouseAppearance:White powder Storage:Sealed and preserved Package:200/Kilograms Application:Fine chemical intermediates, used as the main raw material for the synthe
Hangzhou Dingyan Chem Co., Ltd
R & D enterprises have their own stock in stockAppearance:To be subject to the object Package:Customized Application:pharmaceutical intermediates Transportation:Air Port:Shanghai;Guangzhou
Cas:26171-23-3
Min.Order:0
Negotiable
Type:Manufacturers
inquirySinoway Industrial Co., Ltd.
Why is SINOWAY:1) Specialized in pharmaceutical and healthcare industrial since 19872) ISO 9001:2015 & SGS audited supplier . 3) Accept various payment terms : T.T 30-60 days.4) We have warehouse in USA with quickly shipment . Application:API
Cas:26171-23-3
Min.Order:0
Negotiable
Type:Trading Company
inquiryShanghai Acmec Biochemical Technology Co., Ltd.
Acmec is a leading manufacturer and supplier of biochemical reagents and life science products. We have over 40,000 items in stock (real-time inventory) and offer discounted prices to registered members of the online store ( www.acmec.com.cn ) Appea
Cas:26171-23-3
Min.Order:1 bottle
Negotiable
Type:Lab/Research institutions
inquiryHenan Allgreen Chemical Co.,Ltd
high quality Storage:Sealed, dry, microtherm , avoid light and smell. Package:According to the demand of customer Application:Organic synthesis Transportation:by air or by sea
Hubei Taiho Chemical Co.,LTD
TAIHO Unique Advantages:1.We're factory2.Free samples available3.Commodity inspection can be done4.ISO9001,Kosher certifications5.10 years experiences Storage:Store in cool &dry place Package:aluminium foil bag/fiber can/plastic drum Application:comp
Antimex Chemical Limied
CAS:26171-23-3 C15H15NO3Appearance:white poweder Storage:under the cool and dry area Package:Plastic vacuum packaging bag or bucket Application:CAS:26171-23-3 C15H15NO3 Transportation:by express or by sea
Cas:26171-23-3
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryHangzhou Ocean Chemical Co., Ltd.
★.Best quality according to requirement ★.Competitive price in China market ★.Mature Technical support ★.Professional logistic support
HANGZHOU TIANYE CHEMICALS CO., LTD.
We product this chemical more than 10 years . We are very experience to export it to many countries, Our superior & stable quality , competitive price gain warm reception from our customers. Application:Pharm intermediate
BOC Sciences
We are committed to providing our customers with the best products and services at the most competitive prices.Appearance:off-white powder Storage:Room temperature with sealed well Package:according to the clients requirement Application:Use as prima
Synthetic route
-
-
52074-58-5
ethyl 2-(1-methyl-5-(4-methylbenzoyl)-1H-pyrrol-2-yl)acetate

-
-
26171-23-3
1-methyl-5-(p-toluoyl)pyrrole-2-acetic acid

| Conditions | Yield |
|---|---|
| With sodium hydroxide In methanol; water for 2h; Reflux; | 100% |
| With sodium hydroxide for 0.5h; Heating; | 95.2% |
| With water; sodium hydroxide at 60℃; for 2h; Temperature; | 78.8% |
| With water at 70℃; for 24h; Alkaline conditions; | 57.4 mg |
-
-
26171-22-2
1-Methyl-5-(4-methylbenzoyl)pyrrole-2-acetonitrile

-
-
26171-23-3
1-methyl-5-(p-toluoyl)pyrrole-2-acetic acid

| Conditions | Yield |
|---|---|
| Stage #1: 1-Methyl-5-(4-methylbenzoyl)pyrrole-2-acetonitrile With water; sodium hydroxide In ethanol for 6h; Reflux; Stage #2: With hydrogenchloride at 20℃; | 98% |
| With sodium hydroxide In ethanol for 15h; Heating; | 74% |
| With sodium hydroxide; sodium chloride In ethanol | |
| In sodium hydroxide; ethanol | |
| With sodium hydroxide; sodium chloride In ethanol |
-
-
33369-52-7
methyl 1-Methyl-5-(4-methylbenzoyl)-1H-pyrrole-2-acetate

-
-
26171-23-3
1-methyl-5-(p-toluoyl)pyrrole-2-acetic acid

| Conditions | Yield |
|---|---|
| With lithium hydroxide In tetrahydrofuran; water at 20℃; | 98% |
| With water; sodium hydroxide at 60℃; for 2h; Temperature; | 82.2% |
| Stage #1: methyl 1-Methyl-5-(4-methylbenzoyl)-1H-pyrrole-2-acetate With water; sodium hydroxide at 20℃; for 48h; Inert atmosphere; Stage #2: With hydrogenchloride In water at 20℃; Inert atmosphere; | 78% |
-
-
35711-34-3
tolmetin sodium

-
-
26171-23-3
1-methyl-5-(p-toluoyl)pyrrole-2-acetic acid

| Conditions | Yield |
|---|---|
| With hydrogenchloride In water | 95% |
| With water at 20 - 25℃; for 1h; pH=1.5 - 4.0; Product distribution / selectivity; Acidic aqueous solution; | |
| With water at 20℃; Acidic conditions; |

-
-
74-93-1
methylthiol

-
-
26171-23-3
1-methyl-5-(p-toluoyl)pyrrole-2-acetic acid

| Conditions | Yield |
|---|---|
| With potassium hydroxide In water; dimethyl sulfoxide; ethyl acetate | 89% |

-
-
26171-23-3
1-methyl-5-(p-toluoyl)pyrrole-2-acetic acid

| Conditions | Yield |
|---|---|
| With sodium hydroxide at 60℃; for 2h; | 75% |

| Conditions | Yield |
|---|---|
| With potassium hydroxide In water; acetonitrile | 70% |


-
-
75-08-1
ethanethiol

-
-
26171-23-3
1-methyl-5-(p-toluoyl)pyrrole-2-acetic acid

| Conditions | Yield |
|---|---|
| With potassium hydroxide In water; dimethyl sulfoxide; ethyl acetate | 70% |


-
B
-
26171-23-3
1-methyl-5-(p-toluoyl)pyrrole-2-acetic acid

| Conditions | Yield |
|---|---|
| With phosphate buffer at 37℃; for 6h; pH=7.4; Kinetics; Further Variations:; Reagents; Hydrolysis; |
-
-
96-54-8
N-Methylpyrrole

-
-
26171-23-3
1-methyl-5-(p-toluoyl)pyrrole-2-acetic acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 5 steps 1: 1) POCl3 / 1) RT, 4 h, 2) 1,2-dichloroethane, 40 h 2: 50 percent / 1) POCl3 / 1) 1,2-dichloroethane, reflux, 30 min, 2) 50-60 deg C, 2 h 3: 60 percent / triton B / tetrahydrofuran / 4 h / Heating 4: 35 percent / HCl / ethanol / 70 h / Ambient temperature 5: 95.2 percent / 10percent aq. NaOH / 0.5 h / Heating View Scheme | |
| Multi-step reaction with 5 steps 1.1: dichloromethane / 1 h / -10 - 0 °C / Inert atmosphere 1.2: 0.5 h / 0 °C / pH 10 / Inert atmosphere 1.3: 0.5 h / Inert atmosphere 2.1: hydrazine hydrate; potassium hydroxide / water / 8 h / 100 °C / Inert atmosphere 2.2: 20 °C / pH 2 / Inert atmosphere 3.1: toluene-4-sulfonic acid / 8 h / 70 °C / Inert atmosphere 4.1: DBN / toluene / 4 h / 115 °C / Inert atmosphere 5.1: water; sodium hydroxide / 48 h / 20 °C / Inert atmosphere 5.2: 20 °C / Inert atmosphere View Scheme | |
| Multi-step reaction with 5 steps 1.1: dichloromethane / 1 h / -10 - 0 °C 1.2: 0.5 h / -10 - 0 °C / pH 10 2.1: hydrazine hydrate; potassium hydroxide / water / 5 h / 100 °C 3.1: potassium carbonate / dichloromethane / 3 h / 30 °C 4.1: o-xylene / 24 h / 145 °C 5.1: sodium hydroxide / methanol; water / 2 h / 30 °C View Scheme |
-
-
75820-72-3
1-metil-5-p-metilbenzoil-1H-pirrolo-2-carbossialdeide

-
-
26171-23-3
1-methyl-5-(p-toluoyl)pyrrole-2-acetic acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: 60 percent / triton B / tetrahydrofuran / 4 h / Heating 2: 35 percent / HCl / ethanol / 70 h / Ambient temperature 3: 95.2 percent / 10percent aq. NaOH / 0.5 h / Heating View Scheme |
-
-
85380-92-3
1-(methylsulfinyl)-1-methylthio-2-(1-methyl-5-p-toluoyl-2-pyrrolyl)ethylene

-
-
26171-23-3
1-methyl-5-(p-toluoyl)pyrrole-2-acetic acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 35 percent / HCl / ethanol / 70 h / Ambient temperature 2: 95.2 percent / 10percent aq. NaOH / 0.5 h / Heating View Scheme |
-
-
62128-31-8
(1-methyl-1H-pyrrol-2-yl)-p-tolylmethanone

-
-
26171-23-3
1-methyl-5-(p-toluoyl)pyrrole-2-acetic acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: 50 percent / 1) POCl3 / 1) 1,2-dichloroethane, reflux, 30 min, 2) 50-60 deg C, 2 h 2: 60 percent / triton B / tetrahydrofuran / 4 h / Heating 3: 35 percent / HCl / ethanol / 70 h / Ambient temperature 4: 95.2 percent / 10percent aq. NaOH / 0.5 h / Heating View Scheme | |
| Multi-step reaction with 2 steps 1: triphenylphosphine; sodium periodate; sodium acetate; acetic acid; manganese (II) acetate tetrahydrate / 24 h / 70 °C / Inert atmosphere 2: water / 24 h / 70 °C / Alkaline conditions View Scheme |
-
-
63833-44-3
morpholino(p-tolyl)methanone

-
-
26171-23-3
1-methyl-5-(p-toluoyl)pyrrole-2-acetic acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 5 steps 1: 1) POCl3 / 1) RT, 4 h, 2) 1,2-dichloroethane, 40 h 2: 50 percent / 1) POCl3 / 1) 1,2-dichloroethane, reflux, 30 min, 2) 50-60 deg C, 2 h 3: 60 percent / triton B / tetrahydrofuran / 4 h / Heating 4: 35 percent / HCl / ethanol / 70 h / Ambient temperature 5: 95.2 percent / 10percent aq. NaOH / 0.5 h / Heating View Scheme |
-
-
21898-59-9
1-methylpyrrole-2-acetic acid

-
-
26171-23-3
1-methyl-5-(p-toluoyl)pyrrole-2-acetic acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1.1: toluene-4-sulfonic acid / 8 h / 70 °C / Inert atmosphere 2.1: DBN / toluene / 4 h / 115 °C / Inert atmosphere 3.1: water; sodium hydroxide / 48 h / 20 °C / Inert atmosphere 3.2: 20 °C / Inert atmosphere View Scheme | |
| Multi-step reaction with 3 steps 1: potassium carbonate / dichloromethane / 3 h / 30 °C 2: o-xylene / 24 h / 145 °C 3: sodium hydroxide / methanol; water / 2 h / 30 °C View Scheme |
-
-
21898-43-1
2-(1-methyl-1H-pyrrol-2-yl)-2-oxoacetic acid

-
-
26171-23-3
1-methyl-5-(p-toluoyl)pyrrole-2-acetic acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1.1: hydrazine hydrate; potassium hydroxide / water / 8 h / 100 °C / Inert atmosphere 1.2: 20 °C / pH 2 / Inert atmosphere 2.1: toluene-4-sulfonic acid / 8 h / 70 °C / Inert atmosphere 3.1: DBN / toluene / 4 h / 115 °C / Inert atmosphere 4.1: water; sodium hydroxide / 48 h / 20 °C / Inert atmosphere 4.2: 20 °C / Inert atmosphere View Scheme | |
| Multi-step reaction with 4 steps 1: hydrazine hydrate; potassium hydroxide / water / 5 h / 100 °C 2: potassium carbonate / dichloromethane / 3 h / 30 °C 3: o-xylene / 24 h / 145 °C 4: sodium hydroxide / methanol; water / 2 h / 30 °C View Scheme |
-
-
51856-79-2
N-methyl-2-(methoxycarbonylmethyl)pyrrole

-
-
26171-23-3
1-methyl-5-(p-toluoyl)pyrrole-2-acetic acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1.1: DBN / toluene / 4 h / 115 °C / Inert atmosphere 2.1: water; sodium hydroxide / 48 h / 20 °C / Inert atmosphere 2.2: 20 °C / Inert atmosphere View Scheme | |
| Multi-step reaction with 2 steps 1: o-xylene / 24 h / 145 °C 2: sodium hydroxide / methanol; water / 2 h / 30 °C View Scheme | |
| Multi-step reaction with 4 steps 1.1: 1-amino-3-(dimethylamino)propane / 5,5-dimethyl-1,3-cyclohexadiene / 24 h / Reflux 1.2: 0.5 h / 20 °C 2.1: copper(l) iodide; tetrakis(triphenylphosphine) palladium(0) / 18 h / 70 °C 3.1: palladium 10% on activated carbon; hydrogen / methanol / 3 h 4.1: sodium hydroxide / methanol; tetrahydrofuran; water / 16 h / 20 °C View Scheme | |
| Multi-step reaction with 4 steps 1.1: 1-amino-3-(dimethylamino)propane / 5,5-dimethyl-1,3-cyclohexadiene / 24 h / Reflux 1.2: 0.5 h / 20 °C 2.1: bis(dibenzylideneacetone)-palladium(0); 4,5-bis(diphenylphosphino)-9,9-dimethylxanthene; N,N,N,N,-tetramethylethylenediamine / N,N-dimethyl-formamide / 0.06 h / 160 °C / Sealed tube; Microwave irradiation 3.1: palladium 10% on activated carbon; hydrogen / methanol / 3 h 4.1: sodium hydroxide / methanol; tetrahydrofuran; water / 16 h / 20 °C View Scheme | |
| Multi-step reaction with 2 steps 1: 4-methyl-morpholine / o-xylene / 12 h / Reflux 2: sodium hydroxide; water / 2 h / 60 °C View Scheme |
-
-
1711-02-0
4-iodobenzoic acid chloride

-
-
26171-23-3
1-methyl-5-(p-toluoyl)pyrrole-2-acetic acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1.1: 1-amino-3-(dimethylamino)propane / 5,5-dimethyl-1,3-cyclohexadiene / 24 h / Reflux 1.2: 0.5 h / 20 °C 2.1: copper(l) iodide; tetrakis(triphenylphosphine) palladium(0) / 18 h / 70 °C 3.1: palladium 10% on activated carbon; hydrogen / methanol / 3 h 4.1: sodium hydroxide / methanol; tetrahydrofuran; water / 16 h / 20 °C View Scheme | |
| Multi-step reaction with 4 steps 1.1: 1-amino-3-(dimethylamino)propane / 5,5-dimethyl-1,3-cyclohexadiene / 24 h / Reflux 1.2: 0.5 h / 20 °C 2.1: bis(dibenzylideneacetone)-palladium(0); 4,5-bis(diphenylphosphino)-9,9-dimethylxanthene; N,N,N,N,-tetramethylethylenediamine / N,N-dimethyl-formamide / 0.06 h / 160 °C / Sealed tube; Microwave irradiation 3.1: palladium 10% on activated carbon; hydrogen / methanol / 3 h 4.1: sodium hydroxide / methanol; tetrahydrofuran; water / 16 h / 20 °C View Scheme |

-
-
26171-23-3
1-methyl-5-(p-toluoyl)pyrrole-2-acetic acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: bis(dibenzylideneacetone)-palladium(0); 4,5-bis(diphenylphosphino)-9,9-dimethylxanthene; N,N,N,N,-tetramethylethylenediamine / N,N-dimethyl-formamide / 0.06 h / 160 °C / Sealed tube; Microwave irradiation 2: palladium 10% on activated carbon; hydrogen / methanol / 3 h 3: sodium hydroxide / methanol; tetrahydrofuran; water / 16 h / 20 °C View Scheme | |
| Multi-step reaction with 3 steps 1: copper(l) iodide; tetrakis(triphenylphosphine) palladium(0) / 18 h / 70 °C 2: palladium 10% on activated carbon; hydrogen / methanol / 3 h 3: sodium hydroxide / methanol; tetrahydrofuran; water / 16 h / 20 °C View Scheme |
-
-
617721-38-7
methyl 2-(5-(4-cyanobenzoyl)-1-methyl-1H-pyrrol-2-yl)acetate

-
-
26171-23-3
1-methyl-5-(p-toluoyl)pyrrole-2-acetic acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: palladium 10% on activated carbon; hydrogen / methanol / 3 h 2: sodium hydroxide / methanol; tetrahydrofuran; water / 16 h / 20 °C View Scheme |
-
-
24437-41-0
1-methyl-2-pyrroleacetonitrile

-
-
874-60-2
4-methyl-benzoyl chloride

-
-
26171-23-3
1-methyl-5-(p-toluoyl)pyrrole-2-acetic acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: aluminium trichloride / 1,1-dichloroethane; hexane; benzene 2: sodium hydroxide; sodium chloride / ethanol View Scheme |

-
-
26171-23-3
1-methyl-5-(p-toluoyl)pyrrole-2-acetic acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: sodium ethanolate / dimethyl sulfoxide / 3 h / 100 °C / Inert atmosphere 2: sodium hydride / N,N-dimethyl-formamide / 3.5 h / 20 °C 3: triphenylphosphine; sodium periodate; sodium acetate; acetic acid; manganese (II) acetate tetrahydrate / 24 h / 70 °C / Inert atmosphere 4: water / 24 h / 70 °C / Alkaline conditions View Scheme |
-
-
55895-62-0
2-(4-methylbenzoyl)pyrrole

-
-
26171-23-3
1-methyl-5-(p-toluoyl)pyrrole-2-acetic acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: sodium hydride / N,N-dimethyl-formamide / 3.5 h / 20 °C 2: triphenylphosphine; sodium periodate; sodium acetate; acetic acid; manganese (II) acetate tetrahydrate / 24 h / 70 °C / Inert atmosphere 3: water / 24 h / 70 °C / Alkaline conditions View Scheme |
-
-
104-87-0
4-methyl-benzaldehyde

-
-
26171-23-3
1-methyl-5-(p-toluoyl)pyrrole-2-acetic acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 5 steps 1: tert.-butylhydroperoxide; palladium diacetate; Trimethylacetic acid / toluene; decane / 1.5 h / 60 °C / Inert atmosphere; Sealed tube 2: sodium ethanolate / dimethyl sulfoxide / 3 h / 100 °C / Inert atmosphere 3: sodium hydride / N,N-dimethyl-formamide / 3.5 h / 20 °C 4: triphenylphosphine; sodium periodate; sodium acetate; acetic acid; manganese (II) acetate tetrahydrate / 24 h / 70 °C / Inert atmosphere 5: water / 24 h / 70 °C / Alkaline conditions View Scheme |

-
-
26171-23-3
1-methyl-5-(p-toluoyl)pyrrole-2-acetic acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: 2 h / 190 °C 2: 4-methyl-morpholine / o-xylene / 12 h / Reflux 3: sodium hydroxide; water / 2 h / 60 °C View Scheme |

-
-
26171-23-3
1-methyl-5-(p-toluoyl)pyrrole-2-acetic acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: 2 h / 190 °C 2: 4-methyl-morpholine / o-xylene / 12 h / Reflux 3: sodium hydroxide / 2 h / 60 °C View Scheme |

-
-
26171-23-3
1-methyl-5-(p-toluoyl)pyrrole-2-acetic acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 4-methyl-morpholine / o-xylene / 12 h / Reflux 2: sodium hydroxide / 2 h / 60 °C View Scheme |
-
-
83863-74-5
2-(carboxymethyl)-1-methyl-1H-pyrrole-3-carboxylic acid

-
-
26171-23-3
1-methyl-5-(p-toluoyl)pyrrole-2-acetic acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: toluene-4-sulfonic acid / 1 h / Reflux 2: 2 h / 190 °C 3: 4-methyl-morpholine / o-xylene / 12 h / Reflux 4: sodium hydroxide; water / 2 h / 60 °C View Scheme | |
| Multi-step reaction with 4 steps 1: hydrogenchloride / water / 1 h / Reflux 2: 2 h / 190 °C 3: 4-methyl-morpholine / o-xylene / 12 h / Reflux 4: sodium hydroxide / 2 h / 60 °C View Scheme | |
| Multi-step reaction with 4 steps 1: sulfuric acid / 1 h / Reflux 2: 2 h / 190 °C 3: 4-methyl-morpholine / o-xylene / 12 h / Reflux 4: sodium hydroxide; water / 2 h / 60 °C View Scheme |

-
-
26171-23-3
1-methyl-5-(p-toluoyl)pyrrole-2-acetic acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: 2 h / 190 °C 2: 4-methyl-morpholine / o-xylene / 12 h / Reflux 3: sodium hydroxide; water / 2 h / 60 °C View Scheme |
-
-
26171-23-3
1-methyl-5-(p-toluoyl)pyrrole-2-acetic acid

-
-
85622-93-1
temozolomide

-
-
33369-52-7
methyl 1-Methyl-5-(4-methylbenzoyl)-1H-pyrrole-2-acetate

| Conditions | Yield |
|---|---|
| With sodium carbonate In 1,4-dioxane; water at 60℃; for 6h; Sealed tube; | 81% |
-
-
67-56-1
methanol

-
-
26171-23-3
1-methyl-5-(p-toluoyl)pyrrole-2-acetic acid

-
-
33369-52-7
methyl 1-Methyl-5-(4-methylbenzoyl)-1H-pyrrole-2-acetate

| Conditions | Yield |
|---|---|
| With sulfuric acid for 3h; Reflux; | 77% |
| With sulfuric acid for 3h; Reflux; |

| Conditions | Yield |
|---|---|
| With (4s,6s)-2,4,5,6-tetra(9H-carbazol-9-yl)isophthalonitrile; caesium carbonate In dimethyl sulfoxide at 20℃; under 760.051 Torr; for 0.0833333h; Glovebox; Inert atmosphere; Sealed tube; Irradiation; | 75% |
| Stage #1: 1-methyl-5-(p-toluoyl)pyrrole-2-acetic acid With caesium carbonate In methanol at 20℃; for 1h; Inert atmosphere; Stage #2: carbon dioxide In dimethyl sulfoxide at 150℃; for 0.0833333h; Inert atmosphere; Cooling with liquid nitrogen; | 5.7 mg |
-
-
26171-23-3
1-methyl-5-(p-toluoyl)pyrrole-2-acetic acid

-
-
90-05-1
2-methoxy-phenol

-
-
1207070-76-5
1-methyl-5-p-toluoylpyrrole-2-acetamidoacetic acid isopropyl alcohol ester

| Conditions | Yield |
|---|---|
| With benzotriazol-1-ol; triethylamine; dicyclohexyl-carbodiimide In dichloromethane at 30℃; | 70% |

-
-
26171-23-3
1-methyl-5-(p-toluoyl)pyrrole-2-acetic acid

| Conditions | Yield |
|---|---|
| With trifluorormethanesulfonic acid; bis(4-methoxyphenyl)selenide In chloroform at 20℃; for 16h; Reagent/catalyst; | 70% |
-
-
26171-23-3
1-methyl-5-(p-toluoyl)pyrrole-2-acetic acid

| Conditions | Yield |
|---|---|
| With 2,4,6-trimethyl-pyridine; 2,4,6-Triisopropylthiophenol; water-d2; 9-(2-mesityl)-10-methylacridinium perchlorate In dichloromethane Irradiation; | 69% |
-
-
6265-73-2
N-(2-hydroxyethyl)nicotinamide

-
-
26171-23-3
1-methyl-5-(p-toluoyl)pyrrole-2-acetic acid

-
-
761415-92-3
[1-methyl-5-(4-methyl-benzoyl)-1H-pyrrol-2-yl]-acetic acid 2-[(pyridine-3-carbonyl)-amino]-ethyl ester

| Conditions | Yield |
|---|---|
| With dmap; dicyclohexyl-carbodiimide In acetonitrile | 68% |
-
-
753-90-2
trifluoroethylamine

-
-
26171-23-3
1-methyl-5-(p-toluoyl)pyrrole-2-acetic acid

-
-
75-05-8
acetonitrile

| Conditions | Yield |
|---|---|
| With copper(l) iodide; tert.-butylnitrite at 65℃; for 12h; | 67% |

-
-
75368-99-9
amino acetic acid guacil ester hydrochloride

-
-
26171-23-3
1-methyl-5-(p-toluoyl)pyrrole-2-acetic acid

| Conditions | Yield |
|---|---|
| With potassium carbonate; benzotriazol-1-ol; dicyclohexyl-carbodiimide In acetone at -5 - 30℃; for 5h; | 60% |
-
-
887144-97-0
Togni's reagent

-
-
26171-23-3
1-methyl-5-(p-toluoyl)pyrrole-2-acetic acid

| Conditions | Yield |
|---|---|
| With 2,2'-dipyridyl ketone; [4,4′-bis(1,1-dimethylethyl)-2,2′-bipyridine-N1,N1′]bis{3,5-difluoro-2-[5-(trifluoromethyl)-2-pyridinyl-κN]phenyl-κC}iridium(III) hexafluorophosphate; copper dichloride; N,N,N',N'-tetramethylguanidine In ethyl acetate at 20℃; for 12h; Inert atmosphere; Irradiation; Sealed tube; chemoselective reaction; | 43% |
-
-
26171-23-3
1-methyl-5-(p-toluoyl)pyrrole-2-acetic acid

-
-
62128-36-3
1,5-dimethyl-2-(4-methylbenzoyl)pyrrole

| Conditions | Yield |
|---|---|
| In water Rate constant; Mechanism; Irradiation; |
-
-
26171-23-3
1-methyl-5-(p-toluoyl)pyrrole-2-acetic acid

-
A
-
62128-36-3
1,5-dimethyl-2-(4-methylbenzoyl)pyrrole

| Conditions | Yield |
|---|---|
| With copper(II) ion In water pH=7.4; Product distribution; Kinetics; Further Variations:; Reagents; various Cu(2+) concentrations; also in the absence of Cu(2+); Decarboxylation; photodecarboxylation; Irradiation; |
-
-
26171-23-3
1-methyl-5-(p-toluoyl)pyrrole-2-acetic acid

| Conditions | Yield |
|---|---|
| With tritium; Crabtree's catalyst In dichloromethane | |
| With tritium; Crabtree's catalyst regioselective reaction; |
-
-
26171-23-3
1-methyl-5-(p-toluoyl)pyrrole-2-acetic acid

| Conditions | Yield |
|---|---|
| With tritium; polystyrene-bound PPh3-[(COD)Ir(PCy3)(Py)]PF6 complex In dichloromethane at 20℃; for 3h; |
-
-
26171-23-3
1-methyl-5-(p-toluoyl)pyrrole-2-acetic acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: 68 percent / dicyclohexylcarbodiimide; 4-dimethylaminopyridine / acetonitrile 2: 79 percent / CH2Cl2; acetonitrile / 144 h / 20 °C 3: 100 percent / NaHCO3; Na2S2O4 / H2O; ethyl acetate / 0.25 h / 0 °C / pH 7 View Scheme |
-
-
26171-23-3
1-methyl-5-(p-toluoyl)pyrrole-2-acetic acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 68 percent / dicyclohexylcarbodiimide; 4-dimethylaminopyridine / acetonitrile 2: 79 percent / CH2Cl2; acetonitrile / 144 h / 20 °C View Scheme |
Related products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View