Dayang Chem (Hangzhou) Co.,Ltd.
Dayangchem’s R&D center can offer custom synthesis according to the contract research and development services for the fine chemicals, pharmaceutical, biotechnique and some of the other chemicals. DayangChem can provide different quantiti
Cas:90-05-1
Min.Order:100 Gram
Negotiable
Type:Lab/Research institutions
inquiryXi'an Xszo Chem Co., Ltd.
1. Factory price and high quality must be guaranteed, base on 8 years of production and R&D experience2. Free samples will be provided,ensure specifications and quality are right for customer3. Customers will receive the most professional technical s
Cas:90-05-1
Min.Order:1 Gram
FOB Price: $0.1
Type:Manufacturers
inquiryKono Chem Co.,Ltd
Guaiacol Specification Items Specifications Appearance Colorless to light yellow oily liquid or fused solid UN No. 2
Hebei yanxi chemical co.,LTD.
Hebei yanxi chemical co., LTD is a professional research, development and production of lead acetate benzene acetamide enterprise backbone members by local well-known entrepreneurs and professional senior engineers in the party's "low carb
Wuhan Fortuna Chemical Co.,Ltd
Guaiacol CAS 90-05-1 Company profile Wuhan Fortuna Chemical Co.,Ltd established in 2006, is a big integrative chemical enterprise being engaged in Pharmaceutical & its intermediates, Food/Feed additives, Fine chemicals in distributing the pr
Jinan Finer Chemical Co., Ltd
Product Description Product Name Guaiacol CAS No. 90-05-1 Assay ≥99%
Henan Allgreen Chemical Co.,Ltd
high quality Appearance:White or off-white Solid Storage:Sealed, dry, microtherm , avoid light and smell. Package:According to the demand of customer Application:Organic synthesis Transportation:by air or by sea Port:shanghai
EAST CHEMSOURCES LIMITED
Guaiacol Product Name: Guaiacol Molecular Weight: 124.13 CAS NO: 90-05-1 EC NO: 201-964-7 Molecular Formula: C7H8O2 Specifi
Ality Chemical Corporation
The above product is Ality Chemical's strong item with best price, good quality and fast supply. Ality Chemical has been focusing on the research and production of this field for over 14 years. At the same time, we are always committed to providi
Shanghai Seasonsgreen Chemical Co.,Ltd
Shanghai Seasonsgreen Chemical is a high-tech research and development, production, sale and custom synthesis set in one high-tech chemical products enterprises. Our sales and marketing division is located in Shanghai, serving international pharmaceu
Cas:90-05-1
Min.Order:1 Kilogram
FOB Price: $1.0
Type:Manufacturers
inquiryCOLORCOM LTD.
Colorcom is a global leader in industrial chemical manufacturing and is continuously innovating and transforming to exceed client expectations and industry standards. Colorcom prides itself on superior customer and technical focus, while focusing on
Chemwill Asia Co., Ltd.
Guaiacol Manufacturer Factory CAS 90-05-1 Guaiacol Manufacturer High quality Best price In stock factory CAS 90-05-1 Guaiacol COA TDS price MSDS highly quality and immedaite delivery Guaiacol Chemical Properties Melting point 26-29 &
Cas:90-05-1
Min.Order:5 Kiloliter
FOB Price: $3.5 / 5.0
Type:Manufacturers
inquiryLIDE PHARMACEUTICALS LIMITED
LIDE PHARMACEUTICALS LIMITED is a professional chemicals and APIs leading manufacturer in China. Our core business line covers APIs, Intermediates, Herb extract, etc.
Hebei Nengqian Chemical Import and Export Co., LTD
With our good experience, we offer detailed technical support and advice to assist customers. We communicate closely with customers to establish their quality requirements. Consistent Quality Our plant has strict quality control in each manufacturin
Cas:90-05-1
Min.Order:1 Kilogram
FOB Price: $1.0 / 10.0
Type:Trading Company
inquiryHenan Tianfu Chemical Co., Ltd.
Our company engages in Sodium Tripolyphosphate (STPP) and Sodium Hexametabphosphate (SHMP) production; development of noble metal catalysts, synthesis of electronic chemical materials and general chemicals Imp&Exp trading business. The company
Cas:90-05-1
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryZhuozhou Wenxi import and Export Co., Ltd
WITH US,YOUR MONEY IN SAFE,YOUR BUSINESS IN SAFE 1)Quick Response Within 12 hours; 2)Quality Guarantee: All products are strictly tested by our QC, confirmed by QA and approved by third party lab in China, USA, Canada, Germany, UK, Italy, France et
Cas:90-05-1
Min.Order:1 Kilogram
FOB Price: $139.0 / 210.0
Type:Trading Company
inquiryWuhan Han Sheng New Material Technology Co.,Ltd
Our Advantage: high quality with competitive price High quality standard: BP/USP/EP Enterprise standard All purity customized Fast and safe delivery We have reliable forwarder who can help us deliver our goods more fast and safe. We
Henan Sinotech Import&Export Corporation
Chemical name: Guaiacol Other name: 2-Mehoxy Phenol Molecular formula: C7H8O2 Relaive molecular weight: 124.14 Physical and chemical properties of 2-Methoxyphenol 99% 90-05-1 : colorless crystal or liquid with special fragrance, sl
Hefei TNJ chemical industry co.,ltd
Focus on vanillin's production, the factory has created a perfect industry chain,and the product line includes vanillin,ethyl vanillin,guaiacol,o-Nitrochlorobenzene,o-Aminoanisole,ortho-Vanillin,glyoxylic acid and others now.With annual sales
Shanghai Upbio Tech Co.,Ltd
1.In No Less SIX years exporting experience. for US clients can 100% received goods 2.Lower Price with higher quality 3,Free sample 4,We are sincerely responsible for the "product quality" and "After Service&quo
Cas:90-05-1
Min.Order:25 Kilogram
FOB Price: $1.0
Type:Lab/Research institutions
inquiryXi'an Faithful Biotech Co., Ltd.
We are the manufacturers and suppliers of API in China, and warehouse in Germany and USA of California, which can quickly and safely deliver to your address 1.High quality and competitive price. 2.Free sample for your evaluation. 3.Promptly delivery
Cas:90-05-1
Min.Order:10 Gram
FOB Price: $3.5
Type:Trading Company
inquiryShanghai Minstar Chemical Co., Ltd
Product Name: Guaiacol CAS: 90-05-1 MF: C7H8O2 MW: 124.14 EINECS: 201-964-7 Mol File: 90-05-1.mol Guaiacol Structure Guaiacol Chemical Properties Melting point 26-29 °C (lit.) Boiling point 205 °C (lit.) density
Leader Biochemical Group
About Our Group Since 1996 when our factory was put into production to year of 2020, our group has successively invested in more than 52 factories with shares and subordinates.We focus on manufacture Pharm & c
Cas:90-05-1
Min.Order:500 Kilogram
FOB Price: $1.0 / 2.0
Type:Lab/Research institutions
inquiryBaoji Guokang Healthchem co.,ltd
Our company has been in existence for 10 years since its establishment. We have our own unique team. The company integrates independent research and development, production and sales. We have established famous brands at home and abroad. At prese
Cas:90-05-1
Min.Order:1 Kilogram
FOB Price: $45.0 / 60.0
Type:Trading Company
inquiryQingdao Beluga Import and Export Co., LTD
Guaiacol CAS:90-05-1 Qingdao Belugas Import and Export Co., Ltd. is a scientific and technological company integrating research and development, production and trade of chemical intermediates, specializing in high quality organic intermediates, ster
Shanghai Terui OP New Material Technology Co., Ltd.
We generally use secure shipping methods that do not require you to do any customs clearance. Free shipping to your address without worrying about any customs issues. We have a professional customs clearance team to 100% guarantee the safe delivery o
Cas:90-05-1
Min.Order:1 Kilogram
Negotiable
Type:Trading Company
inquiryShandong Hanjiang Chemical Co., Ltd.
Hello, dear friend! I'm Hansen and Allen from China. Welcome to my lookchem mall! The following is a brief introduction of our company's products and services. If you are interested in our products, please contact us by emai
Changchun Artel lmport and Export trade company
Minimum Order Qty. 10 Gram Supply Ability 500 Kilograms/Month Storage store in cool, dry, ventilated place 20℃ Delivery Time 3 business days after payment Payment Term TT,western union,Paypal,MoneyGram Package 10g,20g,50g,100g,500g,1KGS,
Cas:90-05-1
Min.Order:10 Gram
Negotiable
Type:Trading Company
inquiryLonwin Chemical Group Limited
Guaiacol CAS: 90-05-1 Specification Item Standard Identification A.H-NMR:Comply with the structure B.LC-MS:Comply with the structure C.The IR spectrum of sample s
Hebei Quanhe Biotechnology Co. LTD
1. Timely and efficient service to ensure communication with customers 2. Produce products of different specifications and sizes according to your requirements. 3. Quality procedures and standards recognized by SGS. Advanced plant equipment ensures s
Synthetic route
-
-
19513-80-5
2-(2-methoxyphenoxy)-1-(4-methoxyphenyl)ethan-1-one

-
A
-
90-05-1
2-methoxy-phenol

-
B
-
100-06-1
1-(4-methoxyphenyl)ethanone

| Conditions | Yield |
|---|---|
| With formic acid; N-ethyl-N,N-diisopropylamine; Lumogen F Orange 240 In acetonitrile at 25℃; for 8h; Reagent/catalyst; UV-irradiation; Inert atmosphere; | A 99% B 99% |
| With formic acid; (4,4'-di-tert-butyl-2,2'-dipyridyl)-bis-(2-phenylpyridine(-1H))-iridium(III) hexafluorophosphate; N-ethyl-N,N-diisopropylamine In ethanol at 20℃; Irradiation; chemoselective reaction; | A 95% B 72% |
| With formic acid; (4,4'-di-tert-butyl-2,2'-dipyridyl)-bis-(2-phenylpyridine(-1H))-iridium(III) hexafluorophosphate; N-ethyl-N,N-diisopropylamine In acetonitrile at 20℃; for 12h; Reagent/catalyst; Irradiation; chemoselective reaction; | A 89% B 88% |

| Conditions | Yield |
|---|---|
| With glycolic Acid; copper hydroxide; sodium hydroxide In water; dimethyl sulfoxide at 120℃; for 6h; Inert atmosphere; Schlenk technique; | 95% |
| Stage #1: 4-iodoanisol With copper(l) iodide; 1,10-Phenanthroline; potassium hydroxide In water; dimethyl sulfoxide at 20 - 100℃; Inert atmosphere; Stage #2: With hydrogenchloride In water; dimethyl sulfoxide at 20℃; Inert atmosphere; | 94% |
| Stage #1: 4-iodoanisol With copper(l) iodide; 2-methyl-8-quinolinol; tetra(n-butyl)ammonium hydroxide In water; dimethyl sulfoxide at 100℃; for 10h; Stage #2: With hydrogenchloride In water; N,N-dimethyl-formamide at 20℃; | 94% |

| Conditions | Yield |
|---|---|
| With dicyclohexyl-(2′,4′,6′-triisopropyl-3,6-dimethoxy-[1,1′-biphenyl]-2-yl)phosphine; boric acid; palladium diacetate; caesium carbonate In 1-methyl-pyrrolidin-2-one at 80℃; for 24h; Schlenk technique; Inert atmosphere; | 88% |
| Stage #1: 2-bromoanisole With copper(l) iodide; cesium hydroxide; 5-bromo-2-(1H-imidazol-2-yl)pyridine In water; dimethyl sulfoxide; tert-butyl alcohol at 120℃; for 36h; Inert atmosphere; Stage #2: With hydrogenchloride In water; dimethyl sulfoxide; tert-butyl alcohol pH=1 - 2; Inert atmosphere; | 83% |
| With tert.-butylhydroperoxide; n-butyllithium In tetrahydrofuran; hexane at 20℃; for 2h; | 78% |

| Conditions | Yield |
|---|---|
| With sodium tetrahydroborate; palladium on activated charcoal In water; ethyl acetate at 80℃; for 1h; | A 95% B 99% |
| With 5%-palladium/activated carbon; ammonium formate In water at 80℃; for 1h; | A n/a B 98% |
| With C32H25Cl2N6O2Rh2(1+)*Cl(1-); sodium hydroxide In water at 110℃; for 18h; Inert atmosphere; | A 89% B 86% |
-
-
22675-96-3
2-methoxyphenoxy-3',4'-dimethoxyacetophenone

-
A
-
90-05-1
2-methoxy-phenol

-
B
-
93-07-2
Veratric acid

| Conditions | Yield |
|---|---|
| With oxygen; copper(II) nitrate In acetonitrile at 120℃; under 4500.45 Torr; for 10h; | A n/a B 99% |
| With sodium hydroxide for 2h; Reagent/catalyst; Time; Milling; Green chemistry; | A 95 %Chromat. B 80 %Chromat. |
-
-
92409-23-9
1-(4-methoxyphenyl)-3-hydroxy-2-(2-methoxyphenoxy)propan-1-one

-
A
-
90-05-1
2-methoxy-phenol

| Conditions | Yield |
|---|---|
| With formic acid; (4,4'-di-tert-butyl-2,2'-dipyridyl)-bis-(2-phenylpyridine(-1H))-iridium(III) hexafluorophosphate; N-ethyl-N,N-diisopropylamine In acetonitrile at 20℃; for 18h; Irradiation; Flow reactor; chemoselective reaction; | A 94% B 93% |
| With formic acid; N-ethyl-N,N-diisopropylamine In acetonitrile at 20℃; for 24h; Inert atmosphere; Irradiation; | A 82% B 90% |
| With formic acid; (4,4'-di-tert-butyl-2,2'-dipyridyl)-bis-(2-phenylpyridine(-1H))-iridium(III) hexafluorophosphate; N-ethyl-N,N-diisopropylamine In acetonitrile at 20℃; for 15h; Irradiation; chemoselective reaction; | A 88% B 85% |
| With formic acid; (4,4'-di-tert-butyl-2,2'-dipyridyl)-bis-(2-phenylpyridine(-1H))-iridium(III) hexafluorophosphate; N-ethyl-N,N-diisopropylamine In ethanol at 20℃; Irradiation; chemoselective reaction; | A 56% B 34% |
| With formic acid; 10-phenyl-10H-phenothiazine; N-ethyl-N,N-diisopropylamine In acetone at 20℃; for 96h; Inert atmosphere; Irradiation; | A 28 mg B 42 mg |
-
-
10548-77-3
1-(3,4-dimethoxyphenyl)-3-hydroxy-2-(2-methoxyphenoxy)propan-1-one

-
A
-
104397-78-6
1-(3,4-dimethoxyphenyl)-3-hydroxypropan-1-one

-
B
-
90-05-1
2-methoxy-phenol

| Conditions | Yield |
|---|---|
| With formic acid; N-ethyl-N,N-diisopropylamine; Lumogen F Orange 240 In acetonitrile at 25℃; for 8h; Reagent/catalyst; UV-irradiation; Inert atmosphere; | A 98% B 97% |
| With formic acid; (4,4'-di-tert-butyl-2,2'-dipyridyl)-bis-(2-phenylpyridine(-1H))-iridium(III) hexafluorophosphate; N-ethyl-N,N-diisopropylamine In acetonitrile at 20℃; for 48h; Irradiation; chemoselective reaction; | A 90% B 95% |
| With formic acid; N-ethyl-N,N-diisopropylamine In acetonitrile at 20℃; for 24h; Inert atmosphere; Irradiation; | A 90% B 80% |

| Conditions | Yield |
|---|---|
| With palladium diacetate; sodium hydride In N,N-dimethyl acetamide at 25℃; for 3h; Inert atmosphere; | 96% |
| With palladium diacetate; sodium hydride In N,N-dimethyl acetamide at 50℃; for 5h; Inert atmosphere; | 96% |
| With sulfosuccinic acid functionalized mesoporous silica In ethanol at 100℃; |
-
-
106534-23-0
1-(4-Hydroxyphenyl)-2-(2-methoxyphenoxy)-1-ethanol

-
A
-
90-05-1
2-methoxy-phenol

-
B
-
99-93-4
4-Hydroxyacetophenone

| Conditions | Yield |
|---|---|
| With sodium tetrahydroborate; palladium on activated charcoal In water; ethyl acetate at 80℃; for 4h; | A n/a B 97% |
| With C32H25Cl2N6O2Rh2(1+)*Cl(1-); sodium hydroxide In water at 110℃; for 18h; Inert atmosphere; | A 90% B 76% |
| With C32H25Cl2N6O2Rh2(1+)*Cl(1-); sodium hydroxide In water at 110℃; for 18h; Inert atmosphere; Green chemistry; | A 90% B 76% |
| With C22H17Cl3N3ORh; sodium hydroxide In water for 60h; Inert atmosphere; |
-
-
7382-68-5
1-(4-hydroxy-3-methoxyphenyl)-2-(2-methoxyphenoxy)ethanol

-
A
-
90-05-1
2-methoxy-phenol

-
B
-
498-02-2
1-(3-methoxy-4-hydroxyphenyl)ethanone

| Conditions | Yield |
|---|---|
| With sodium tetrahydroborate; palladium on activated charcoal In water; ethyl acetate at 80℃; for 14h; | A n/a B 96% |
| With C32H25Cl2N6O2Rh2(1+)*Cl(1-); sodium hydroxide In water at 110℃; for 18h; Inert atmosphere; | A 86% B 82% |
| With C32H25Cl2N6O2Rh2(1+)*Cl(1-); sodium hydroxide In water at 110℃; for 18h; Inert atmosphere; Green chemistry; | A 86% B 82% |
| With C22H17Cl3N3ORh; sodium hydroxide In water for 60h; Inert atmosphere; |
-
-
73220-26-5
1-methoxy-2-(methoxymethoxy)benzene

-
-
90-05-1
2-methoxy-phenol

| Conditions | Yield |
|---|---|
| With β‐cyclodextrin In water; acetone at 60℃; for 5.5h; Green chemistry; | 95% |
| With Montmorillonite K 10 In benzene at 50℃; for 1h; | 91% |
| With bismuth(III) chloride In water; acetonitrile at 50℃; for 1h; chemoselective reaction; | 83% |
-
-
10548-77-3
1-(3,4-dimethoxyphenyl)-3-hydroxy-2-(2-methoxyphenoxy)propan-1-one

-
A
-
90-05-1
2-methoxy-phenol

-
B
-
93-07-2
Veratric acid

| Conditions | Yield |
|---|---|
| With dihydrogen peroxide; sodium hydroxide In tetrahydrofuran; methanol; water at 50℃; for 10h; | A 45% B 88% |
| With dihydrogen peroxide; sodium hydroxide In tetrahydrofuran; methanol at 50℃; for 10h; | |
| With sodium hydroxide for 2h; Milling; Green chemistry; | A 92 %Chromat. B 70 %Chromat. |
-
-
89466-08-0
2-hydroxyphenyl boronic acid

-
-
90-05-1
2-methoxy-phenol

| Conditions | Yield |
|---|---|
| With oxygen; triethylamine In 2-methyltetrahydrofuran at 20℃; under 760.051 Torr; for 48h; UV-irradiation; | 69% |
-
-
19513-80-5
2-(2-methoxyphenoxy)-1-(4-methoxyphenyl)ethan-1-one

-
A
-
90-05-1
2-methoxy-phenol

-
B
-
100-09-4
4-methoxybenzoic acid

| Conditions | Yield |
|---|---|
| With oxygen; copper(II) nitrate In acetonitrile at 120℃; under 4500.45 Torr; for 10h; | A n/a B 99% |
| With oxygen; copper diacetate; triethylamine In dimethyl sulfoxide at 20℃; for 12h; Sealed tube; | A 93% B 83% |
| With water; oxalic acid at 100℃; for 24h; Reagent/catalyst; | A 21 mg B 73% |

| Conditions | Yield |
|---|---|
| With Ru/CNT; hydrogen In water; decalin at 200℃; under 7500.75 Torr; for 3h; | |
| With hydrogen In water; decalin at 200℃; under 7500.75 Torr; for 6h; chemoselective reaction; | |
| With formic acid In water at 130℃; for 6h; Reagent/catalyst; Green chemistry; | A 69 %Chromat. B 17 %Chromat. |

| Conditions | Yield |
|---|---|
| With ammonium bicarbonate In water at 20℃; for 2h; Schlenk technique; | 95% |

| Conditions | Yield |
|---|---|
| With cucumber juice at 30 - 35℃; for 48h; Inert atmosphere; Green chemistry; | 80% |
| With copper dichloride In water at 200℃; for 6h; Inert atmosphere; Autoclave; | 55% |
| decarboxylase | |
| decarboxylase Enzymatic reaction; | |
| With hydrogen In water at 170℃; under 7500.75 Torr; for 4h; Autoclave; |

| Conditions | Yield |
|---|---|
| With Ni7Au3; hydrogen In water at 130℃; under 7500.75 Torr; for 0.5h; Reagent/catalyst; | A 44% B 14% |
| With hydrogen In water at 60℃; under 7500.75 Torr; for 1h; Reagent/catalyst; Overall yield = 60 %Chromat.; |

| Conditions | Yield |
|---|---|
| With potassium hydroxide In toluene; tert-butyl alcohol at 100℃; for 0.5h; Inert atmosphere; | 87% |

| Conditions | Yield |
|---|---|
| Stage #1: 2-Chloroanisole With tris-(dibenzylideneacetone)dipalladium(0); cesium hydroxide monohydrate; 2-((di-adamantan-1-yl)phosphaneyl)-1-(2,6-diisopropylphenyl)-1H-imidazole In 1,4-dioxane at 120℃; for 20h; Inert atmosphere; Stage #2: With hydrogenchloride In 1,4-dioxane; water at 20℃; Inert atmosphere; | 90% |
| With 2-di-tertbutylphosphino-3,4,5,6-tetramethyl-2',4',6'-triisopropyl-1,1'-biphenyl; potassium hydroxide; tris-(dibenzylideneacetone)dipalladium(0) In 1,4-dioxane; water at 100℃; for 24h; | 79% |
| With trans-di(μ-acetato)bis[o-(di-o-tolyl-phosphino)benzyl]dipalladium(II); C29H45Pt; potassium carbonate In water; N,N-dimethyl-formamide at 115℃; for 0.5h; Inert atmosphere; Microwave irradiation; | 60% |

-
A
-
3929-47-3
3-(3,4-dimethoxyphenyl)-1-propanol

-
B
-
5888-52-8
4-propyl-1,2-dimethoxybenzene

-
C
-
100699-21-6
Acetic acid 3-(3,4-dimethoxy-phenyl)-propyl ester

-
D
-
90-05-1
2-methoxy-phenol

| Conditions | Yield |
|---|---|
| With palladium on activated charcoal; hydrogen; lanthanum(lll) triflate In 1,2-dichloro-ethane at 140℃; under 31029.7 Torr; for 16h; | A 18% B 32% C 19% D 69% |
| With palladium on activated charcoal; hydrogen; lanthanum(lll) triflate In 1,1,2,2-tetrachloroethane at 140℃; under 31029.7 Torr; for 2h; | A 14% B 7% C 34% D 59% |
| With palladium on activated charcoal; hydrogen; lanthanum(lll) triflate In 1,2-dichloro-ethane at 140℃; under 31029.7 Torr; for 16h; | A 6% B 11% C 19% D 56% |
| With palladium on activated charcoal; hydrogen; lanthanum(lll) triflate In 1,2-dichloro-ethane at 140℃; under 31029.7 Torr; for 16h; | A 9% B 17% C 9% D 43% |
| With palladium on activated charcoal; hydrogen; lanthanum(lll) triflate In 1,2-dichloro-ethane at 140℃; under 31029.7 Torr; for 16h; | A 22% B 12% C 13% D 13% |

-
-
29389-04-6
2-(2,6-dimethoxyphenoxy)-1-(3,4-dimethoxyphenyl)ethan-1-one

-
A
-
37803-48-8
β-oxo-β-(3,4-dimethoxyphenyl)-ethanol

-
B
-
90-05-1
2-methoxy-phenol

| Conditions | Yield |
|---|---|
| With 1,10-Phenanthroline; oxygen; copper diacetate In methanol at 80℃; under 3000.3 Torr; for 3h; | A 93% B 21% |
-
-
14385-48-9
2-(2-methoxyphenoxy)-acetophenone

-
A
-
582-24-1
1-phenyl-2-hydroxyethanone

-
B
-
90-05-1
2-methoxy-phenol

| Conditions | Yield |
|---|---|
| With 1,10-Phenanthroline; oxygen; copper diacetate In methanol at 80℃; under 3000.3 Torr; for 3h; | A 95% B 27% |
-
-
19513-80-5
2-(2-methoxyphenoxy)-1-(4-methoxyphenyl)ethan-1-one

-
A
-
4136-21-4
p-methoxybenzoylmethanol

-
B
-
90-05-1
2-methoxy-phenol

| Conditions | Yield |
|---|---|
| With 1,10-Phenanthroline; oxygen; copper diacetate In methanol at 80℃; under 3000.3 Torr; for 3h; | A 85% B 10% |

| Conditions | Yield |
|---|---|
| With C32H25Cl2N6O2Rh2(1+)*Cl(1-); sodium hydroxide In water at 110℃; for 48h; Inert atmosphere; | A 90% B 85% |
-
-
14385-48-9
2-(2-methoxyphenoxy)-acetophenone

-
-
142-84-7
di-n-propylamine

-
A
-
14657-86-4
N,N-dipropylbenzamide

-
B
-
90-05-1
2-methoxy-phenol

| Conditions | Yield |
|---|---|
| With copper diacetate In dimethyl sulfoxide at 20℃; for 10h; Sealed tube; | A 81% B 91% |
| With copper(II) acetate monohydrate In dimethyl sulfoxide at 25℃; under 760.051 Torr; for 12h; | A 46 %Chromat. B 94 %Chromat. |

-
-
19513-80-5
2-(2-methoxyphenoxy)-1-(4-methoxyphenyl)ethan-1-one

-
-
124-40-3
dimethyl amine

-
A
-
7291-00-1
N,N-dimethyl-4-methoxybenzamide

-
B
-
90-05-1
2-methoxy-phenol

| Conditions | Yield |
|---|---|
| With copper diacetate In water; dimethyl sulfoxide at 20℃; for 12h; Sealed tube; | A 78% B 87% |
| With copper(II) acetate monohydrate In dimethyl sulfoxide at 25℃; under 760.051 Torr; for 9h; | A 67 %Chromat. B 86 %Chromat. |

-
-
22675-96-3
2-methoxyphenoxy-3',4'-dimethoxyacetophenone

-
-
124-40-3
dimethyl amine

-
A
-
6967-45-9
3,4-dimethoxy-N,N-dimethylbenzamide

-
B
-
90-05-1
2-methoxy-phenol

| Conditions | Yield |
|---|---|
| With copper diacetate In water; dimethyl sulfoxide at 20℃; for 12h; Sealed tube; | A 80% B 76% |
| With copper(II) acetate monohydrate In dimethyl sulfoxide at 25℃; under 760.051 Torr; for 9h; | A 80 %Chromat. B 75 %Chromat. |


| Conditions | Yield |
|---|---|
| Stage #1: 4-bromoguaiacol With nickel(II) iodide; 1,4-bis(dicyclohexylphosphino)butane; sodium carbonate; cesium iodide In tetrahydrofuran for 0.25h; Schlenk technique; Glovebox; Stage #2: In tetrahydrofuran at 35℃; for 72h; Irradiation; | 76% |

| Conditions | Yield |
|---|---|
| With hydrazine hydrate; caesium carbonate at 80℃; for 8h; | 99% |
| With water; dihydrogen peroxide at 20℃; for 0.166667h; Green chemistry; | 97% |
| With water; caesium carbonate; hydrazine hydrate at 80℃; for 24h; | 97% |

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetone at 20℃; Reflux; | 100% |
| With potassium carbonate; potassium iodide In acetone for 18h; Reflux; | 100% |
| With potassium carbonate | 99% |

| Conditions | Yield |
|---|---|
| With benzyltriphenylphosphonium peroxodisulfate; potassium bromide In acetonitrile for 4h; Heating; | 100% |
| With bromine In dichloromethane at -78 - -5℃; for 1h; Inert atmosphere; | 100% |
| With bromine In dichloromethane | 97% |

| Conditions | Yield |
|---|---|
| With triethylamine In dichloromethane at 0℃; for 2h; Inert atmosphere; | 100% |
| With triethylamine In ethyl acetate at 0 - 20℃; for 0.166667h; Green chemistry; | 97% |
| With triethylamine In dichloromethane at 20℃; for 1h; Inert atmosphere; | 97% |
-
-
107-30-2
chloromethyl methyl ether

-
-
90-05-1
2-methoxy-phenol

-
-
73220-26-5
1-methoxy-2-(methoxymethoxy)benzene

| Conditions | Yield |
|---|---|
| Stage #1: 2-methoxy-phenol With potassium tert-butylate In tetrahydrofuran; N,N-dimethyl-formamide at 20℃; for 2h; Stage #2: chloromethyl methyl ether In tetrahydrofuran; N,N-dimethyl-formamide at 20℃; for 5h; | 100% |
| With N-ethyl-N,N-diisopropylamine In dichloromethane at 0 - 20℃; | 99% |
| With N-ethyl-N,N-diisopropylamine at 0 - 20℃; | 99% |

| Conditions | Yield |
|---|---|
| With Trimethylacetic acid In toluene for 20h; Heating; | 100% |

| Conditions | Yield |
|---|---|
| With [bis(acetoxy)iodo]benzene at 20℃; | 100% |
| With iodobenzene; 3-chloro-benzenecarboperoxoic acid at 25℃; for 0.166667h; Reagent/catalyst; Diels-Alder Cycloaddition; | 95% |
| With [bis(acetoxy)iodo]benzene at 0 - 20℃; for 1.5h; | 94% |

| Conditions | Yield |
|---|---|
| With triethylamine In tetrahydrofuran | 100% |
-
-
90-05-1
2-methoxy-phenol

-
-
160257-85-2
5-iodo-2-methoxyphenol

| Conditions | Yield |
|---|---|
| With iodine; n-butyltriphenylphosphonium peroxodisulfate In acetonitrile for 2h; Heating; | 100% |
| Multi-step reaction with 3 steps 1: 99 percent / pyridine / CH2Cl2 / 24 h / 0 - 18 °C 2: 73 percent / silver trifluoroacetate; iodine / CHCl3 / 5 h / 0 - 18 °C 3: 99 percent / potassium carbonate / methanol / 16 h / 18 °C View Scheme | |
| Multi-step reaction with 3 steps 1: 97 percent / DMAP; pyridine / CH2Cl2 / 1 h / 0 °C 2: ICl / CH2Cl2 / 168 h / 25 °C 3: 17.14 g / LiOH*H2O / methanol; tetrahydrofuran; H2O / 120 h / 25 °C View Scheme |
-
-
90-05-1
2-methoxy-phenol

-
-
350-46-9
4-Fluoronitrobenzene

-
-
32795-85-0
2-methoxyphenyl 4'-nitrophenyl ether

| Conditions | Yield |
|---|---|
| 100% | |
| With potassium carbonate In acetonitrile Heating; | |
| With potassium carbonate In N,N-dimethyl-formamide for 8h; Inert atmosphere; Heating; | |
| With potassium carbonate In N,N-dimethyl-formamide at 120℃; |
-
-
358-23-6
trifluoromethylsulfonic anhydride

-
-
90-05-1
2-methoxy-phenol

-
-
59099-58-0
2-methoxyphenyl triflate

| Conditions | Yield |
|---|---|
| With pyridine at 0 - 20℃; for 2h; | 100% |
| With pyridine In dichloromethane at 0 - 20℃; for 1h; Inert atmosphere; | 98% |
| With pyridine In dichloromethane at 20℃; for 0.5h; | 96% |
-
-
934421-88-2
(S,S)-1-chloro-2,5-trans-diphenylphospholane

-
-
90-05-1
2-methoxy-phenol

| Conditions | Yield |
|---|---|
| With lithium In diethyl ether at 20℃; for 3h; | 100% |
-
-
40400-13-3
2-Iodobenzyl bromide

-
-
90-05-1
2-methoxy-phenol

-
-
1332326-21-2
2-(2-iodobenzyloxy)-1-methoxybenzene

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetone at 50℃; | 100% |
-
-
96-32-2
bromoacetic acid methyl ester

-
-
90-05-1
2-methoxy-phenol

-
-
38768-62-6
methyl (2-methoxyphenoxy) ethanoate

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetone at 20℃; for 16h; | 100% |
| With potassium carbonate In acetone at 70℃; for 2h; | 34% |
| With potassium carbonate In tetrahydrofuran at 20 - 80℃; |
-
-
90-05-1
2-methoxy-phenol

-
-
1493-27-2
ortho-nitrofluorobenzene

-
-
74865-12-6
1-methoxy-2-(2-nitrophenoxy)benzene

| Conditions | Yield |
|---|---|
| With potassium carbonate In dimethyl sulfoxide at 95℃; for 20h; | 100% |
| With potassium carbonate In dimethyl sulfoxide at 95℃; |
-
-
350-30-1
3-chloro-4-fluoronitrobenzene

-
-
90-05-1
2-methoxy-phenol

-
-
1097189-03-1
2-chloro-1-(2-methoxyphenoxy)-4-nitrobenzene

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide at 20℃; for 17h; | 100% |

| Conditions | Yield |
|---|---|
| With perchloric acid; d(4)-methanol at 75℃; for 72h; Inert atmosphere; | 100% |

| Conditions | Yield |
|---|---|
| With hydrogen In dodecane at 200℃; under 15001.5 Torr; for 2h; | 99.6% |
| With rhodium contaminated with carbon; N-methyldiethanolamine trifluoromethanesulfonate; hydrogen at 120℃; under 30003 Torr; for 6h; Autoclave; | 93.5% |
| With Ni-doped silica; hydrogen In decalin at 140℃; under 22502.3 Torr; for 5h; Reagent/catalyst; Autoclave; | 91.7% |

| Conditions | Yield |
|---|---|
| In dichloromethane at 0 - 20℃; for 1h; Inert atmosphere; | 99% |
| With pyridine |

| Conditions | Yield |
|---|---|
| With magnesium(II) perchlorate at 20℃; for 1h; | 99% |
| With iodine at 25℃; for 0.0166667h; | 99% |
| With Methylenediphosphonic acid at 20℃; for 2h; neat (no solvent); | 99% |
-
-
88-10-8
N,N-diethylcarbamyl chloride

-
-
90-05-1
2-methoxy-phenol

-
-
85630-19-9
N,N-diethyl-1-carbamyloxy-2-methoxybenzene

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetonitrile for 14h; Reflux; | 99% |
| With pyridine for 6h; Heating; | 96% |
| With sodium hydride In diethyl ether | 93% |
-
-
1835-02-5
2-Bromo-1-(3,4-dimethoxyphenyl)ethanone

-
-
90-05-1
2-methoxy-phenol

-
-
22675-96-3
2-methoxyphenoxy-3',4'-dimethoxyacetophenone

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetone at 20℃; for 4h; Reflux; | 99% |
| With potassium carbonate In acetone | 97% |
| Stage #1: 2-methoxy-phenol With potassium carbonate In acetone at 20℃; for 0.5h; Stage #2: 2-Bromo-1-(3,4-dimethoxyphenyl)ethanone In acetone Reflux; | 95% |

| Conditions | Yield |
|---|---|
| With sodium hydroxide In tetrahydrofuran; water at 0 - 20℃; for 2h; Reagent/catalyst; Green chemistry; | 99% |
| Stage #1: 2-methoxy-phenol With potassium hydroxide In tetrahydrofuran at 20℃; for 0.0833333h; Inert atmosphere; Stage #2: p-toluenesulfonyl chloride In tetrahydrofuran at 60℃; Inert atmosphere; | 99% |
| With triethylamine In dichloromethane at 20℃; | 99% |

| Conditions | Yield |
|---|---|
| With 3 weight% Pd/C; hydrogen In Hexadecane at 249.84℃; under 22502.3 Torr; Reagent/catalyst; Autoclave; | 99% |
| With hydrogen In water at 80℃; under 15001.5 Torr; for 6.5h; | 99% |
| With Ni-doped silica; hydrogen In decalin at 140℃; under 22502.3 Torr; for 5h; Reagent/catalyst; Autoclave; | 95.5% |

| Conditions | Yield |
|---|---|
| Stage #1: 2-methoxy-phenol With pyridine; iodine; aluminium In acetonitrile for 18h; Reflux; Stage #2: With hydrogenchloride In water; acetonitrile at 20℃; | 99% |
| With hydrogenchloride In water at 250℃; under 37503.8 Torr; for 3h; Reagent/catalyst; Autoclave; Inert atmosphere; Green chemistry; | 97% |
| With aluminium(III) iodide; calcium oxide In acetonitrile at 80℃; for 18h; Reagent/catalyst; | 94% |

| Conditions | Yield |
|---|---|
| With hydrogen In water at 160℃; for 10h; Temperature; Concentration; Reagent/catalyst; | 99% |
| With potassium hydroxide; Raney Ni-Al; water at 90℃; for 16h; | 90.8% |
| With acetic acid; platinum Hydrogenation; |

| Conditions | Yield |
|---|---|
| With copper(I) oxide; caesium carbonate; imidazole-4-carboxylic acid In acetonitrile at 80℃; for 24h; | 99% |
| With copper(l) iodide; 1-(dimethylamino-1-yl-pyridin-2-yl-methyl)naphthalen-2-ol; potassium carbonate In dimethyl sulfoxide at 120℃; for 24h; Ullmann Condensation; Green chemistry; | 99% |
| With potassium hydroxide In dimethyl sulfoxide at 110℃; for 15h; Inert atmosphere; | 96% |

| Conditions | Yield |
|---|---|
| 99% | |
| With pyridine In dichloromethane at 0 - 18℃; for 24h; | 99% |
| With pyridine In dichloromethane at 0 - 20℃; for 18h; | 99% |
Related products
Raw Materials
Downstream Products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View











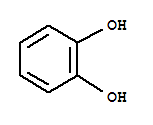



 Xn,
Xn, T,
T, Xi
Xi