Dayang Chem (Hangzhou) Co.,Ltd.
As a leading manufacturer and supplier of chemicals in China, DayangChem not only supply popular chemicals, but also DayangChem's R&D center offer custom synthesis services. DayangChem can provide different quantities of custom synthesis ch
Cas:3796-70-1
Min.Order:1 Kilogram
FOB Price: $3.0
Type:Lab/Research institutions
inquiryShanghai Seasonsgreen Chemical Co.,Ltd
Shanghai Seasonsgreen Chemical is a high-tech research and development, production, sale and custom synthesis set in one high-tech chemical products enterprises. Our sales and marketing division is located in Shanghai, serving international pharmaceu
Cas:3796-70-1
Min.Order:1 Kilogram
FOB Price: $1.0
Type:Manufacturers
inquiryChemwill Asia Co., Ltd.
Our main production base is located in Xuzhou industry park. We are certified both to the ISO 9001 and ISO 14001 Standards, have a safety management system in place.Our R&D team masters core technology for process-design of target building block
Cas:3796-70-1
Min.Order:5 Kiloliter
FOB Price: $1.2 / 5.0
Type:Manufacturers
inquiryZhuozhou Wenxi import and Export Co., Ltd
WITH US,YOUR MONEY IN SAFE,YOUR BUSINESS IN SAFE 1)Quick Response Within 12 hours; 2)Quality Guarantee: All products are strictly tested by our QC, confirmed by QA and approved by third party lab in China, USA, Canada, Germany, UK, Italy, France et
Cas:3796-70-1
Min.Order:1 Kilogram
FOB Price: $139.0 / 210.0
Type:Trading Company
inquiryShanghai Upbio Tech Co.,Ltd
1.No Less 8 years exporting experience. Clients can 100% received goods 2.Lower Price with higher quality 3,Free sample 4,We are sincerely responsible for the "product quality" and "After Service" Upbio is Specialized
Cas:3796-70-1
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryShandong Hanjiang Chemical Co., Ltd.
Hello, dear friend! I'm Hansen and Allen from China. Welcome to my lookchem mall! The following is a brief introduction of our company's products and services. If you are interested in our products, please contact us by emai
Cas:3796-70-1
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryHenan Wentao Chemical Product Co., Ltd.
Henan Wentao Chemical Product Co.,Ltd is Located in Zhengzhou High-tech Development Zone with import and export license, We passed ISO 9001:2008 as well, Henan Wentao has developed more than 1000 compounds, which are widely used in the fields of prod
Changchun Artel lmport and Export trade company
Price, service, company and transport advantage : 1. best service, high quality and reasonable price 2. it's customers' right to choose the package (ems, dhl, fedex, ups); 3. it's customers' r
Wuhan Han Sheng New Material Technology Co.,Ltd
Our Advantage: high quality with competitive price High quality standard: BP/USP/EP Enterprise standard All purity customized Fast and safe delivery We have reliable forwarder who can help us deliver our goods more fast and safe. We
Hangzhou Keyingchem Co.,Ltd
Hangzhou KeyingChem Co., Ltd. exported this product to many countries and regions at best price. If you are looking for the material’s manufacturer or supplier in China, KeyingChem is your best choice. Pls contact with us freely for getting det
Cas:3796-70-1
Min.Order:0 Metric Ton
Negotiable
Type:Lab/Research institutions
inquiryHangzhou J&H Chemical Co., Ltd.
J&H CHEM R&D center can offer custom synthesis according to the contract research and development services for the fine chemicals, pharmaceutical, biotechnique and some of the other chemicals. J&H CHEM has some Manufacturing base in Jia
Cas:3796-70-1
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquirySHANGHAI SYSTEAM BIOCHEM CO., LTD
We are one of a few suppliers that can offer custom synthesis service of this product We are specialized in custom synthesis, chemical/pharmaceutical/ pesticides outsourcing and contract research. We are committed to prov
Cas:3796-70-1
Min.Order:100 Gram
FOB Price: $100.0 / 2000.0
Type:Lab/Research institutions
inquiryAlity Chemical Corporation
The above product is Ality Chemical's strong item with best price, good quality and fast supply. Ality Chemical has been focusing on the research and production of this field for over 14 years. At the same time, we are always committed to providing h
Henan Tianfu Chemical Co., Ltd.
Our company was built in 2009 with an ISO certificate.In the past 5 years, we have grown up as a famous fine chemicals supplier in China and we had established stable business relationships with Samsung, LG, Merck, Thermo Fisher Scientific and so o
Xiamen Jenny Chemical Technology Co., Ltd.
GMP standard, high purity, competitive price, in stock 1. Quick Response: within 6 hours after receiving your email. 2. Quality Guarantee: All products are strictly tested by our QC, confirmed by QA, and approved by a third-party lab in China, USA,
Cas:3796-70-1
Min.Order:1 Milligram
Negotiable
Type:Trading Company
inquiryShanghai Minstar Chemical Co., Ltd
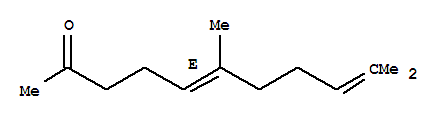
Product Name: Geranylacetone CAS: 3796-70-1 MF: C13H22O MW: 194.31 EINECS: 223-269-8 Product Categories: Mol File: 3796-70-1.mol Geranylacetone Structure Geranylacetone Chemical Properties Boiling point 254-258 °C(lit.)
EAST CHEMSOURCES LIMITED
factory?direct?saleAppearance:White Powder Storage:Store In Dry, Cool And Ventilated Place Package:25kg/drum, also according to the clients requirement Application:It is widely used as a thickener, emulsifier and stabilizer Transportation:By Sea Or B
Cas:3796-70-1
Min.Order:1 Kilogram
FOB Price: $18.0 / 20.0
Type:Trading Company
inquiryShandong Mopai Biotechnology Co., LTD
Shandong Mopai Biotechnology Co., LTD is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality pharmaceuticals, intermediates, special chemicals and OLED intermediates and other fine chemicals. W
Cas:3796-70-1
Min.Order:1 Gram
Negotiable
Type:Lab/Research institutions
inquiryHangzhou Huarong Pharm Co., Ltd.
Hangzhou Huarong Pharm Co., Ltd.established since 2006 , has been actively developing specialty products for Finished Dosages, APIs, Intermediates, and Fine chemicals markets in North America, Europe, Korea, Japan, Mid-East and all over the World. Hu
KAISA GROUP INC
1.Applied in food field.it can improve the immune system and prolong life. 2.Appliedin cosmetic field.it can improve the skin care. 3.Applied in pharmaceutical field.it can treat various dieases. 4.Our product quality assurance will make our customer
Cas:3796-70-1
Min.Order:1 Metric Ton
FOB Price: $1.5
Type:Trading Company
inquiryHunan chemfish Pharmaceutical co.,Ltd
Appearance:95%+ Package:R&D,Pilot run Transportation:per client require Port:Express ,Air, Sea
Bluecrystal chem-union
We are a Union of chemistry in China, consists of chemists,engineers, laboratories,factories in China. We organize surplus capacity of R&D and production as well as custom synthesis for chemical products and chemical business project. We are supp
GIHI CHEMICALS CO.,LIMITED
Lower price, sample is available,SDS test documents are available,large stock in warehouseAppearance:White powder Storage:Sealed and preserved Package:200/Kilograms Application:Fine chemical intermediates, used as the main raw material for the synthe
Kono Chem Co.,Ltd
high purity lowest priceAppearance:solid or liquid Storage:in sealed air resistant place Package:drum and bag Application:for pharma use Transportation:by sea or air Port:Beijing or Guangzhou
JINHUA HUAYI CHEMICAL CO., LTD.
Jinhua huayi chemical co., ltd. is dedicated to the development, production and marketing of chemicals. On the basis of equality and mutual benefit, and under the principle of customer first, credit first, quality first, we are ready to join hands
Shanghai Acmec Biochemical Technology Co., Ltd.
Acmec is a leading manufacturer and supplier of biochemical reagents and life science products. We have over 40,000 items in stock (real-time inventory) and offer discounted prices to registered members of the online store ( www.acmec.com.cn ) Appea
Cas:3796-70-1
Min.Order:1 bottle
Negotiable
Type:Lab/Research institutions
inquiryHenan Allgreen Chemical Co.,Ltd
high quality Storage:Sealed, dry, microtherm , avoid light and smell. Package:According to the demand of customer Application:Organic synthesis Transportation:by air or by sea
Aecochem Corp.
Our clients, like BASF,CHEMO,Brenntag,ASR,Evonik,Merck and etc.Appearance:COA Storage:in stock Application:MSDS/TDS
Antimex Chemical Limied
Ansciep Chemical is a professional enterprise manufacturing and distributing fine chemicals and speciality chemicals. We have been dedicated to heterocycle compounds and phenyl rings for tens of years. This is our mature product for export. Our quali
Cas:3796-70-1
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryQingdao Beluga Import and Export Co., LTD
We are a trading company aiming at providing high-quality services to customers. Established for many years, with good reputation in the industry Storage:Sealed and preserved Application:Fine chemical intermediates, used as the main raw material for
Synthetic route
-
-
19894-78-1
(E)-2-acetyl-5,9-dimethyldeca-4,8-dienoic acid ethyl ester

-
-
3796-70-1
trans geranyl acetone

| Conditions | Yield |
|---|---|
| With sodium hydroxide In ethanol; water for 3h; Reflux; | 95% |
| With potassium hydroxide In methanol Reflux; | 85% |
| Multi-step reaction with 2 steps 1: aq. NaOH / 1.) rt, 2 h; 60 deg C, overnight 2: benzene / 1 h / Heating View Scheme | |
| With methanol; potassium hydroxide at 80℃; Inert atmosphere; |

| Conditions | Yield |
|---|---|
| With polystyrene-supported(cathecholato)oxoRe cat. act. by iPrOH; dimethyl sulfoxide In toluene for 4h; Heating; Dean-Stark apparatus; | 93% |
| With pyridinium chlorochromate |
-
-
80868-08-2
(E)-6,10-Dimethyl-3-(toluene-4-sulfonyl)-undeca-5,9-dien-2-one

-
-
3796-70-1
trans geranyl acetone

| Conditions | Yield |
|---|---|
| With disodium hydrogenphosphate; sodium amalgam In methanol at 0℃; for 2h; | 90% |
-
-
7733-91-7
(E)-6,10-dimethyl-5,9-undecadien-2-ol

-
-
107-21-1
ethylene glycol

-
A
-
3796-62-1
(5E)-6,10-dimethylundeca-5,9-dien-2-one ethylene acetal

-
B
-
3796-70-1
trans geranyl acetone

| Conditions | Yield |
|---|---|
| With dimethyl sulfoxide; [ReOCl3(PPh3)2] In toluene for 5h; Heating; | A 82% B 6% |

-
-
152526-59-5
C25H32S2

-
-
3796-70-1
trans geranyl acetone

| Conditions | Yield |
|---|---|
| With water; mercury dichloride In acetonitrile for 2h; | 80% |
-
-
51933-45-0
3-methoxycarbonyl-6,10-dimethyl-E,E-5,9-undecadien-2-one

-
-
3796-70-1
trans geranyl acetone

| Conditions | Yield |
|---|---|
| With potassium hydroxide In ethanol; water at 20℃; for 4h; Decarboxylation; | 65% |

| Conditions | Yield |
|---|---|
| Stage #1: trans-geranyl bromide; ethyl acetoacetate With potassium carbonate In acetone at 70℃; Inert atmosphere; Stage #2: With potassium hydroxide In methanol for 3h; Inert atmosphere; Reflux; | 65% |

| Conditions | Yield |
|---|---|
| With sodium diethylmalonate; ethyl-diphenyl-phosphane; bis(dibenzylideneacetone)-palladium(0) In dimethyl sulfoxide at 100℃; for 18h; | 50% |
-
-
10032-00-5
acetoacetic acid-(3,7-dimethyl-octa-2,6-dienyl ester)

-
-
3796-70-1
trans geranyl acetone

| Conditions | Yield |
|---|---|
| With palladium diacetate; sodium hydride; triphenylphosphine In tert-butyl alcohol at 50℃; for 4h; | 44% |
| With palladium diacetate; triphenylphosphine In tert-butyl alcohol at 50℃; for 4h; | 44% |
-
-
674-82-8
4-methyleneoxetan-2-one

-
-
78-70-6
3,7-dimethylocta-1,6-dien-3-ol

-
A
-
3879-26-3
(Z)-nerylacetone

-
B
-
3796-70-1
trans geranyl acetone

-
-
4490-10-2
geranyl chloride

-
-
1007476-32-5
sodium ethyl acetylacetate enolate

-
-
3796-70-1
trans geranyl acetone

| Conditions | Yield |
|---|---|
| With ethanol und Erhitzen des Reaktionsprodukts mit wss.-aethanol.Natronlauge; |
-
-
6138-90-5
geranyl bromide

-
-
1007476-32-5
sodium ethyl acetylacetate enolate

-
-
3796-70-1
trans geranyl acetone

| Conditions | Yield |
|---|---|
| With ethanol und Erhitzen des Reaktionsprodukts mit wss.-aethanol.Natronlauge; |
-
-
78-70-6
3,7-dimethylocta-1,6-dien-3-ol

-
-
141-97-9
ethyl acetoacetate

-
A
-
3879-26-3
(Z)-nerylacetone

-
B
-
3796-70-1
trans geranyl acetone

| Conditions | Yield |
|---|---|
| With aluminum isopropoxide | |
| With aluminum tri-tert-butoxide |

-
-
25456-03-5
3,7-Dimethylocta-1,6-dien-3-yl acetoacetate

-
A
-
3879-26-3
(Z)-nerylacetone

-
B
-
3796-70-1
trans geranyl acetone

| Conditions | Yield |
|---|---|
| With palladium diacetate; sodium hydride; triphenylphosphine In tert-butyl alcohol at 50℃; for 2h; Yield given. Yields of byproduct given; | |
| In diphenylether at 190 - 220℃; for 18h; Yield given. Yields of byproduct given; |

| Conditions | Yield |
|---|---|
| With sulfur dioxide; water at 50℃; for 5h; Yield given; |

| Conditions | Yield |
|---|---|
| With silica gel In benzene for 20h; Heating; dark; Yield given. Yields of byproduct given. Title compound not separated from byproducts; |
-
-
111-02-4
2,6,10,15,19,23-hexamethyltetracosa-2,6,10,14,18,22-hexaene

-
A
-
3796-70-1
trans geranyl acetone

-
B
-
110-93-0
6-Methyl-hept-5-en-2-on

-
C
-
67858-78-0
(4E, 8E)-5,9,13-trimethyl-4,8,12-tetradecatrienal

-
D
-
56882-09-8
(4E,8E,12E)-4,9,13,17-Tetramethyloctadeca-4,8,12,16-tetraen-1-al

| Conditions | Yield |
|---|---|
| With periodic acid; 3-chloro-benzenecarboperoxoic acid 1.) CH2Cl2, 0 deg C, 30 min, 2.) ether, 15 min; Yield given. Multistep reaction; |

-
-
68754-01-8
(E)-4-methyl-8-oxo-non-4-enal

-
-
1530-33-2
isopropyltriphenylphosphonium bromide

-
-
3796-70-1
trans geranyl acetone

| Conditions | Yield |
|---|---|
| With n-butyllithium 1) THF, hexane, 25 deg C for 1 h, then r.t. 1.5 h, 2) THF, hexane, 25 deg C, 1.5 h, then r.t. 1 h; Yield given. Multistep reaction; |

| Conditions | Yield |
|---|---|
| In tetrachloromethane at 77℃; for 1h; Yields of byproduct given; | |
| In benzene for 1h; Heating; Yield given. Yields of byproduct given; | |
| In tetrachloromethane at 77℃; for 1h; Yield given. Yields of byproduct given; |

-
-
3796-70-1
trans geranyl acetone

| Conditions | Yield |
|---|---|
| In benzene for 1h; Heating; Yield given; |
-
-
78-70-6
3,7-dimethylocta-1,6-dien-3-ol

-
-
77-76-9
2,2-dimethoxy-propane

-
A
-
3879-26-3
(Z)-nerylacetone

-
B
-
3796-70-1
trans geranyl acetone

| Conditions | Yield |
|---|---|
| phosphoric acid; acetic acid at 150℃; for 18h; Yield given. Yields of byproduct given. Title compound not separated from byproducts; |


| Conditions | Yield |
|---|---|
| With water; mercury dichloride In acetonitrile for 2h; Yield given. Yields of byproduct given. Title compound not separated from byproducts; |
-
-
79221-63-9
4-methyl-4(E)-nonene-1,8-diol

-
-
3796-70-1
trans geranyl acetone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 1)oxalyl chloride, DMSO, 2)triethylamine / 1) CH2Cl2, -60 deg C, 15 min, 2) CH2Cl2, -60 deg C, 5 min, then to r.t. 2: 1) butyllithium / 1) THF, hexane, 25 deg C for 1 h, then r.t. 1.5 h, 2) THF, hexane, 25 deg C, 1.5 h, then r.t. 1 h View Scheme |
-
-
87791-09-1
bis(trimethylsilyl) ether of 4-methyl-4E-nonene-1,8-diol

-
-
3796-70-1
trans geranyl acetone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: 98 percent / aq. HCl / ethanol / 2 h / Ambient temperature 2: 1)oxalyl chloride, DMSO, 2)triethylamine / 1) CH2Cl2, -60 deg C, 15 min, 2) CH2Cl2, -60 deg C, 5 min, then to r.t. 3: 1) butyllithium / 1) THF, hexane, 25 deg C for 1 h, then r.t. 1.5 h, 2) THF, hexane, 25 deg C, 1.5 h, then r.t. 1 h View Scheme |
-
-
79235-13-5
diacetate of 4-methyl-4E-nonene-1,8-diol

-
-
3796-70-1
trans geranyl acetone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: 97 percent / NaOH / aq. ethanol / 5 h / Ambient temperature 2: 1)oxalyl chloride, DMSO, 2)triethylamine / 1) CH2Cl2, -60 deg C, 15 min, 2) CH2Cl2, -60 deg C, 5 min, then to r.t. 3: 1) butyllithium / 1) THF, hexane, 25 deg C for 1 h, then r.t. 1.5 h, 2) THF, hexane, 25 deg C, 1.5 h, then r.t. 1 h View Scheme |
-
-
118723-77-6
linalyl phenylcarbamate

-
-
3796-70-1
trans geranyl acetone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 86 percent / Pd2(dba)3*CHCl3; Ph3P / tetrahydrofuran / 12 h / 40 °C 2: 65 percent / KOH / ethanol; H2O / 4 h / 20 °C View Scheme |
-
-
77532-60-6
3-Carbomethoxy-6,10-dimethyl-5,9-undecadien-2-on

-
A
-
3796-70-1
trans geranyl acetone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 10percent aq. NaOH / methanol / Ambient temperature 2: benzene / 1 h / Heating View Scheme |
-
-
85217-73-8
(Z)-3,7-dimethylocta-2,6-dien-1-yl methyl carbonate

-
A
-
3796-70-1
trans geranyl acetone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: 1.) Pd2(dba)3*CHCl3, dppe / 2.) THF, 50 deg C, 3 h 2: 10percent aq. NaOH / methanol / Ambient temperature 3: benzene / 1 h / Heating View Scheme |
-
-
85217-72-7
geranyl methyl carbonate

-
A
-
3796-70-1
trans geranyl acetone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: 1.) Pd2(dba)3*CHCl3, dppe / 2.) THF, 50 deg C, 3 h 2: 10percent aq. NaOH / methanol / Ambient temperature 3: benzene / 1 h / Heating View Scheme |
-
-
3796-70-1
trans geranyl acetone

-
-
107-21-1
ethylene glycol

-
-
3796-62-1
(5E)-6,10-dimethylundeca-5,9-dien-2-one ethylene acetal

| Conditions | Yield |
|---|---|
| With pyridine; toluene-4-sulfonic acid In benzene Heating; | 100% |
| With toluene-4-sulfonic acid | 93% |
| With toluene-4-sulfonic acid |
-
-
3796-70-1
trans geranyl acetone

-
-
412042-43-4
4-{3-[2-(3,3-dimethyl-oxiranyl)-ethyl]-3-methyl-oxiranyl}-butan-2-one

| Conditions | Yield |
|---|---|
| With 3-chloro-benzenecarboperoxoic acid In dichloromethane | 100% |
| With Oxone; Na2B4O7 - Na2(EDTA) buffer; 1,2,4,5-di-O-isopropylidene-D-erythro-2,3-hexodiuro-2,6-pyranose In water; acetonitrile at 0℃; pH=10.5; |
-
-
3796-70-1
trans geranyl acetone

-
-
4294-57-9
para-methylphenylmagnesium bromide

-
-
70026-33-4
E-6,10-dimethyl-2-hydroxy-2-(p-tolyl)-5,9-undecadiene

| Conditions | Yield |
|---|---|
| In diethyl ether at 20℃; | 100% |
-
-
3796-70-1
trans geranyl acetone

-
-
2356-16-3
ethyl(diethylphosphono)(fluoro)acetate

-
-
1994-91-8
cis,trans-2-Fluor-3,7,11-trimethyl-Δ2,6,10-dodecatriensaeureethylester

| Conditions | Yield |
|---|---|
| With sodium hydride In tetrahydrofuran | 99% |
-
-
3796-70-1
trans geranyl acetone

| Conditions | Yield |
|---|---|
| With borane N,N-diethylaniline complex; (3aR)-1-methyl-3,3-diphenyl-tetrahydro-pyrrolo[1,2-c][1,3,2]oxazaborole In toluene Product distribution / selectivity; | 99% |
-
-
3796-70-1
trans geranyl acetone

-
-
17508-17-7
O-(2,4-dinitrophenyl)hydroxylamine

| Conditions | Yield |
|---|---|
| With hydrogenchloride In ethanol at 20℃; | 99% |

| Conditions | Yield |
|---|---|
| With sodium tetrahydroborate In methanol at 0℃; for 0.5h; | 98% |
| With lithium aluminium tetrahydride In diethyl ether for 0.166667h; Heating; | 97% |
| With lithium aluminium tetrahydride In diethyl ether at 0℃; for 0.5h; | 96% |
-
-
3796-70-1
trans geranyl acetone

-
-
76248-60-7
(4aS,8aS)-2,5,5,8a-tetramethyl-4a,5,6,7,8,8a-hexahydro-4H-chromene

| Conditions | Yield |
|---|---|
| With class II squalene-hopene cyclase from Alicyclobacillus acidocaldarius variant G600R In aq. acetate buffer at 30℃; for 24h; pH=6; Enzymatic reaction; | 98% |
| With squalene–hopene cyclase from Zymomonas mobilis for 20h; Concentration; Reagent/catalyst; Enzymatic reaction; |
-
-
3796-70-1
trans geranyl acetone

| Conditions | Yield |
|---|---|
| With potassium osmate(VI) dihydrate; methanesulfonamide; C70H63N5O3; potassium carbonate; potassium hexacyanoferrate(III) In water; tert-butyl alcohol at 24℃; for 10h; Reagent/catalyst; Temperature; Sharpless Dihydroxylation; Cooling with ice; enantioselective reaction; | 97% |
| With methanesulfonamide; AD-mix-β In water; tert-butyl alcohol at 0℃; for 192h; Sharpless Dihydroxylation; | 7% |
-
-
3796-70-1
trans geranyl acetone

-
-
4301-14-8
acetylenemagnesium bromide

-
-
59905-15-6, 115460-14-5
dehydronerolidol

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at 0 - 20℃; | 96% |
| In tetrahydrofuran; dichloromethane for 1h; Ambient temperature; | 90% |
| In tetrahydrofuran; diethyl ether at 20℃; for 4h; | 70% |
| Stage #1: trans geranyl acetone; acetylenemagnesium bromide In tetrahydrofuran at 23℃; Inert atmosphere; Stage #2: With water In tetrahydrofuran |
-
-
3796-70-1
trans geranyl acetone

-
-
142669-95-2
(5E)-9-chloro-6,10-dimethylundeca-5,10-dien-2-one

| Conditions | Yield |
|---|---|
| With N-chloro-succinimide; selenenyl bromide In dichloromethane at 20℃; for 3.5h; | 96% |
| With N-chloro-succinimide; Phenylselenyl chloride In dichloromethane at 20℃; for 1.16667h; Inert atmosphere; | 95% |
| With sulfuryl dichloride; Cl2CuI; sodium carbonate In various solvent(s) at -5 - 0℃; for 1h; | 172 mg |

| Conditions | Yield |
|---|---|
| With palladium on activated charcoal; hydrogen In methanol | 95% |
| With hydrogen; [(C6Me6)2Ru2(PPh2)H2][BF4] In ethanol at 60℃; under 37503 Torr; for 24h; | 85% |
| With hydrogen; C16AlF36O4(1-)*C32H44IrNO2P(1+) In dichloromethane at 20℃; under 37503.8 Torr; for 2h; Product distribution / selectivity; | 24% |

| Conditions | Yield |
|---|---|
| With potassium 2-methylbutan-2-olate In diethyl ether | 94% |
| With potassium hydroxide In diethyl ether | |
| With potassium hydroxide |
-
-
3796-70-1
trans geranyl acetone

-
-
38622-91-2, 36635-61-7
[(p-methylphenyl)sulfonylmethyl]isonitrile

-
-
57963-93-6
(E)-2,6,10-trimethylundeca-5,9-dienenitrile

| Conditions | Yield |
|---|---|
| Stage #1: trans geranyl acetone; [(p-methylphenyl)sulfonylmethyl]isonitrile With potassium tert-butylate In tetrahydrofuran; diethyl ether at 0℃; Inert atmosphere; Stage #2: With ethanol In tetrahydrofuran; diethyl ether at 0 - 20℃; for 4h; Inert atmosphere; | 94% |
| With potassium tert-butylate | 94% |

| Conditions | Yield |
|---|---|
| Stage #1: cyclopropyltriphenylphosphonium bromide With potassium tert-butylate In tetrahydrofuran at 0 - 5℃; for 2h; Wittig Olefination; Inert atmosphere; Stage #2: trans geranyl acetone In tetrahydrofuran at -2 - 35℃; for 24h; Wittig Olefination; Inert atmosphere; | 93% |
-
-
3796-70-1
trans geranyl acetone

-
-
7677-24-9
trimethylsilyl cyanide

-
-
87509-03-3
(E)-2,6,10-Trimethyl-2-trimethylsilanyloxy-undeca-5,9-dienenitrile

| Conditions | Yield |
|---|---|
| With mercury(II) iodide | 90% |
| With zinc(II) iodide In nitromethane |
-
-
3796-70-1
trans geranyl acetone

-
-
18444-76-3
(E)-6,10-dimethyl-undeca-5,9-dien-2-one oxime

| Conditions | Yield |
|---|---|
| With pyridine; hydroxylamine hydrochloride In ethanol for 0.5h; Heating; | 90% |
-
-
3796-70-1
trans geranyl acetone

-
-
616-38-6
carbonic acid dimethyl ester

-
-
56523-17-2
methyl 3-oxo-7,11-dimethyl-dodeca-6,10-dienoate

| Conditions | Yield |
|---|---|
| With sodium hydride In diethyl ether for 8h; Heating; | 88% |
| With sodium hydride In diethyl ether for 2h; Heating; | 82% |
-
-
3796-70-1
trans geranyl acetone

-
-
1826-67-1
vinyl magnesium bromide

-
-
40716-66-3
(E)-3,7,11-trimethyl-1,6,10-dodecatrien-3-ol

| Conditions | Yield |
|---|---|
| In dichloromethane 1.) 0 deg C, 15 min, 2.) room temperature, 3 h; | 87% |
| In tetrahydrofuran at 0 - 20℃; for 72h; Inert atmosphere; | 27% |
| With tetrahydrofuran | |
| In tetrahydrofuran at 0 - 20℃; Inert atmosphere; |
-
-
3796-70-1
trans geranyl acetone

-
-
149-73-5
trimethyl orthoformate

-
-
149075-51-4
(E)-10,10-dimethoxy-2,6-dimethylundeca-2,6-diene

| Conditions | Yield |
|---|---|
| With sulfuric acid In methanol at 65℃; for 3h; | 87% |
| With sulfuric acid In methanol at 65℃; for 3h; Inert atmosphere; | 87% |
| With sulfuric acid In methanol at 65℃; for 3h; | 87% |
| With sulfuric acid In methanol at 5℃; for 3.08333h; Reflux; | 87% |
-
-
3796-70-1
trans geranyl acetone

-
-
123499-01-4
(5E)-[1,1,1,3,3-2H5]-6,10-dimethyl-undeca-5,9-dien-2-one

| Conditions | Yield |
|---|---|
| With water-d2; potassium carbonate at 70℃; Inert atmosphere; | 86% |
| With water-d2; potassium carbonate at 70℃; Inert atmosphere; | 86% |
| With deuteriated sodium hydroxide; deuteromethanol; water-d2 |

| Conditions | Yield |
|---|---|
| With trimethylsilyl trifluoromethanesulfonate In tetrahydrofuran at -30℃; for 19.5h; Inert atmosphere; | 85.7% |
-
-
867-13-0
diethoxyphosphoryl-acetic acid ethyl ester

-
-
3796-70-1
trans geranyl acetone

-
-
19954-66-6
ethyl farnesoate

| Conditions | Yield |
|---|---|
| With sodium hydride In tetrahydrofuran; mineral oil at 20℃; Wittig-Horner Reaction; | 85% |
| Stage #1: diethoxyphosphoryl-acetic acid ethyl ester With sodium hydride In tetrahydrofuran at 0℃; for 2h; Inert atmosphere; Schlenk technique; Stage #2: trans geranyl acetone In tetrahydrofuran at 0 - 20℃; for 3h; Inert atmosphere; Schlenk technique; | 72% |
| With sodium hydride In benzene | 65% |
-
-
3796-70-1
trans geranyl acetone

-
-
17665-58-6
benzenesulfinyl-acetonitrile

-
-
93040-86-9
3,7,11-trimethyl-4-hydroxy-2,6,10-trienenitrile

| Conditions | Yield |
|---|---|
| With piperidine In methanol for 17h; Ambient temperature; | 85% |
-
-
3796-70-1
trans geranyl acetone

-
-
24163-89-1
rac-(E)-8-(3,3-dimethyloxiran-2-yl)-6-methyloct-5-en-2-one

| Conditions | Yield |
|---|---|
| With 3-chloro-benzenecarboperoxoic acid In dichloromethane at 0℃; for 2h; | 85% |
| With disodium hydrogenphosphate; 3-chloro-benzenecarboperoxoic acid In dichloromethane at 0 - 20℃; | 71% |
| In water at 27℃; for 24h; biotransformation with Mucor griseocyanus DSM 1173, culture-medium: glucose, malt extract, yeast extract, peptone, pH 6.0; | 17.2% |
-
-
3796-70-1
trans geranyl acetone

-
-
62026-31-7
[3-(2-methyl-[1,3]dioxolan-2-yl)-propyl]-magnesium chloride

-
-
1117-52-8
6,10,14-trimethyl-5,9,13-pentadecatriene-2-on

| Conditions | Yield |
|---|---|
| In tert-butyl methyl ether at 0℃; for 6h; Inert atmosphere; | 85% |
Related products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View
 Xi
Xi