Dayang Chem (Hangzhou) Co.,Ltd.
DayangChem exported this product to many countries and regions at best price. If you are looking for the material's manufacturer or supplier in China, DayangChem is your best choice. Pls contact with us freely for getting detailed product spe
Cas:867-13-0
Min.Order:1 Kilogram
FOB Price: $3.0
Type:Lab/Research institutions
inquiryHangzhou Dingyan Chem Co., Ltd
Items Standard Result Appearance Colorless oily liquid Conforms Assay ≥99%
Cas:867-13-0
Min.Order:1 Kilogram
FOB Price: $20.0 / 50.0
Type:Manufacturers
inquirySimagchem Corporation
Welcome to Simagchem, your partner in China as a premier supply of bulk specialty chemicals for industry and life science. We introduce experienced quality product and exceptional JIT service with instant market intelligence in China to benefit our
Cas:867-13-0
Min.Order:1 Gram
Negotiable
Type:Manufacturers
inquiryWuhan Fortuna Chemical Co.,Ltd
1. Guaranteed purity; 2. Large quantity in stock; 3. Largest manufacturer; 4. Best service after shipment with email; 5. High quality & competitive price; ...... Appearance:Colorless to light yellow liquid Storage:Refrigerator Pac
Cas:867-13-0
Min.Order:25 Kilogram
FOB Price: $100.0
Type:Trading Company
inquiryHebei yanxi chemical co.,LTD.
Hebei yanxi chemical co. LTD. has expanded a compositive entity from initially only as a small manufacturer. The company dedicated to the development, production and marketing of chemicals. After many years of efforts, we have established stable
Cas:867-13-0
Min.Order:1 Metric Ton
FOB Price: $1.0 / 2.0
Type:Manufacturers
inquiryHenan Allgreen Chemical Co.,Ltd
The company has a professional R & D team, mature technology, very competitive prices and stable high quality products for customers to order high quality and low price product efforts! Appearance:Clear Liquid Storage:Sealed, dry, microtherm ,
Ality Chemical Corporation
The above product is Ality Chemical's strong item with best price, good quality and fast supply. Ality Chemical has been focusing on the research and production of this field for over 14 years. At the same time, we are always committed to providi
Chemwill Asia Co., Ltd.
Our main production base is located in Xuzhou industry park. We are certified both to the ISO 9001 and ISO 14001 Standards, have a safety management system in place.Our R&D team masters core technology for process-design of target building block
Cas:867-13-0
Min.Order:5 Kiloliter
FOB Price: $1.2 / 5.0
Type:Manufacturers
inquiryHenan Tianfu Chemical Co., Ltd.
Our company was built in 2009 with an ISO certificate.In the past 5 years, we have grown up as a famous fine chemicals supplier in China and we had established stable business relationships with Samsung,LG,Merck,Thermo Fisher Scientific and so on.O
Cas:867-13-0
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryHebei Nengqian Chemical Import and Export Co., LTD
With our good experience, we offer detailed technical support and advice to assist customers. We communicate closely with customers to establish their quality requirements. Consistent Quality Our plant has strict quality control in each manufacturin
Cas:867-13-0
Min.Order:1 Kilogram
FOB Price: $3.0 / 10.0
Type:Trading Company
inquiryZhuozhou Wenxi import and Export Co., Ltd
WITH US,YOUR MONEY IN SAFE,YOUR BUSINESS IN SAFE 1)Quick Response Within 12 hours; 2)Quality Guarantee: All products are strictly tested by our QC, confirmed by QA and approved by third party lab in China, USA, Canada, Germany, UK, Italy, France et
Cas:867-13-0
Min.Order:1 Kilogram
FOB Price: $139.0 / 210.0
Type:Trading Company
inquiryHebei Sankai Chemical Technology Co., Ltd
1. Product advantages High purity, all above 98.5%, no impurities after dissolution We will test each batch to ensure quality OEM and private brand services designed for free Various cap colors available We can also provide MT1 peptide powd
Cas:867-13-0
Min.Order:1 Kilogram
FOB Price: $5.0 / 10.0
Type:Trading Company
inquiryHubei Langyou International Trading Co., Ltd
1. Guaranteed purity; 2. Large quantity in stock; 3. Largest manufacturer; 4. Best service after shipment with email; 5. High quality & competitive price; Appearance:white crystalline powder Storage:Store in sealed containers
Cas:867-13-0
Min.Order:100 Gram
Negotiable
Type:Other
inquiryShanghai Upbio Tech Co.,Ltd
1.No Less 8 years exporting experience. Clients can 100% received goods 2.Lower Price with higher quality 3,Free sample 4,We are sincerely responsible for the "product quality" and "After Service" Upbio is Specialized
Cas:867-13-0
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryXi'an Faithful Biotech Co., Ltd.
We are the manufacturers and suppliers of API in China, and warehouse in Germany and USA of California, which can quickly and safely deliver to your address 1.High quality and competitive price. 2.Free sample for your evaluation. 3.Promptly delivery
Cas:867-13-0
Min.Order:10 Gram
FOB Price: $2.9
Type:Trading Company
inquiryQingdao Beluga Import and Export Co., LTD
Triethyl phosphonoacetate CAS: 867-13-0 Qingdao Belugas Import and Export Co., Ltd. is a scientific and technological company integrating research and development, production and trade of chemical intermediates, specializing in high quality organic
Cas:867-13-0
Min.Order:1 Gram
Negotiable
Type:Lab/Research institutions
inquiryShandong Hanjiang Chemical Co., Ltd.
Hello, dear friend! I'm Hansen and Allen from China. Welcome to my lookchem mall! The following is a brief introduction of our company's products and services. If you are interested in our products, please contact us by emai
Cas:867-13-0
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryHenan Wentao Chemical Product Co., Ltd.
Henan Wentao Chemical Product Co.,Ltd is Located in Zhengzhou High-tech Development Zone with import and export license, We passed ISO 9001:2008 as well, Henan Wentao has developed more than 1000 compounds, which are widely used in the fields of prod
Wuhan Han Sheng New Material Technology Co.,Ltd
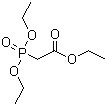
Email Us: admin07@hsnm.com.cn quick details of Triethyl phosphonoacetate raw liquids Product Name Triethyl phosphonoacetate CAS 867-13-0 MF C8H17O5P MW 224.19 EINECS 212-757-6 MOL File 867-13-0.mol Chemical Properties Melting point -24°
SHANGHAI T&W PHARMACEUTICAL CO., LTD.
A substitute for perfluorooctanoic acid, mainly used as a surfactant, dispersant, additive, etc Appearance:White solid or Colorless liquid Purity:99.3 % We will ship the goods in a timely manner as required We can provide relevant documents acc
Cas:867-13-0
Min.Order:4 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryHangzhou Keyingchem Co.,Ltd
Hangzhou KeyingChem Co., Ltd. exported this product to many countries and regions at best price. If you are looking for the material’s manufacturer or supplier in China, KeyingChem is your best choice. Pls contact with us freely for getting det
Cas:867-13-0
Min.Order:0 Metric Ton
Negotiable
Type:Lab/Research institutions
inquiryHangzhou J&H Chemical Co., Ltd.
J&H CHEM R&D center can offer custom synthesis according to the contract research and development services for the fine chemicals, pharmaceutical, biotechnique and some of the other chemicals. J&H CHEM has some Manufacturing base in Jia
Cas:867-13-0
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryShanghai Hope Chem Co., Ltd
1. Product advantages: exquisite appearance and unique functions. 2. Product Advantages: Our products are the best,fast speed ,in large stock 3. High-quality products and thoughtful service are your greatest satisfaction. 4. The product
Cas:867-13-0
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryZibo Hangyu Biotechnology Development Co., Ltd
Zibo Hangyu Biotechnology Development Co., Ltd is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality pharmaceuticals, intermediates, special chemicals and OLED intermediates and other fine chemi
Cas:867-13-0
Min.Order:10 Gram
FOB Price: $100.0
Type:Lab/Research institutions
inquiryHangzhou Lingrui Chemical Co.,Ltd.
our strengths: 1: Fast and guaranteed shipment (TNT;EMS;FEDEX;DHL;UPS;EUB, special line) 2: Various payment items accepted (Btc; MoneyGram; WU) 3: Valued package (Paraffin coating; Double aluminum foil bag; Vacuum packaging) 4: Efficient delivery
Cas:867-13-0
Min.Order:1 Kilogram
Negotiable
Type:Other
inquiryZibo Dorne chemical technology co. LTD
Product Details Grade: pharmaceutical grade Purity:99%+ ProductionCapacity: 1000 Kilogram/Month Scope of use: For scientific research only(The product must be used legally) Our Advantage 1. Best quality with competitive price. 2. Quick shipping,
Cas:867-13-0
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryJiangsu Qianyu Molecular Technology Co., LTD.
Our Advantages A. International Top level TechnologyOur company owned biomedicine experts are famous at home and abroad with rich experience in research and development in the field of efficient chiral functional molecules research and development an
Cas:867-13-0
Min.Order:0
Negotiable
Type:Trading Company
inquirySiwei Development Group Ltd.
Product name: Triethyl Phosphonacetic Acid CAS No.:867-13-0 Molecule Formula:C8H17O5P Molecule Weight:224.19 Purity: 98.0% Package: 200kg/drum Description:Colorless to light yellow liquid Manufacture Standards:Enterprise Standard
Cas:867-13-0
Min.Order:1 Kilogram
Negotiable
Type:Trading Company
inquiryXiamen Jenny Chemical Technology Co., Ltd.
GMP standard, high purity, competitive price, in stock 1. Quick Response: within 6 hours after receiving your email. 2. Quality Guarantee: All products are strictly tested by our QC, confirmed by QA, and approved by a third-party lab in China, USA,
Shanghai Minstar Chemical Co., Ltd
Product Name: Triethyl phosphonoacetate CAS: 867-13-0 MF: C8H17O5P MW: 224.19 EINECS: 212-757-6 Mol File: 867-13-0.mol Triethyl phosphonoacetate Structure Triethyl phosphonoacetate Chemical Properties Melting point -24°C B
Cas:867-13-0
Min.Order:1 Gram
Negotiable
Type:Lab/Research institutions
inquirySynthetic route
-
-
105-36-2
ethyl bromoacetate

-
-
122-52-1
triethyl phosphite

-
-
867-13-0
diethoxyphosphoryl-acetic acid ethyl ester

| Conditions | Yield |
|---|---|
| at 60 - 80℃; for 0.5h; Arbusov reaction; | 100% |
| at 150℃; for 3h; | 99% |
| for 0.0333333h; Michaelis-Arbuzov reaction; microwave irradiation; | 98% |
-
-
105-39-5
chloroacetic acid ethyl ester

-
-
122-52-1
triethyl phosphite

-
-
867-13-0
diethoxyphosphoryl-acetic acid ethyl ester

| Conditions | Yield |
|---|---|
| In toluene at 170℃; under 7500.75 Torr; for 0.333333h; Pressure; Temperature; Arbuzov Reaction; Large scale; | 95% |
| at 120 - 165℃; for 2h; | 93% |
| for 2h; Inert atmosphere; Reflux; | 63% |

| Conditions | Yield |
|---|---|
| With triethyl phosphite | 89.3% |
-
-
78-40-0
triethyl phosphate

-
-
105-36-2
ethyl bromoacetate

-
-
867-13-0
diethoxyphosphoryl-acetic acid ethyl ester

| Conditions | Yield |
|---|---|
| at 140℃; for 3h; | 88% |
-
-
623-73-4
diazoacetic acid ethyl ester

-
-
762-04-9
phosphonic acid diethyl ester

-
-
867-13-0
diethoxyphosphoryl-acetic acid ethyl ester

| Conditions | Yield |
|---|---|
| With copper acetylacetonate In benzene Product distribution; Heating; other catalysts, other diazo compounds, other hydrophosphoryl compounds; | 83% |
| copper acetylacetonate In benzene for 1h; Heating; | 83% |
| With copper acetylacetonate In benzene Heating; | 83% |
| With trifluorormethanesulfonic acid In benzene Heating; | 2% |
-
-
105-39-5
chloroacetic acid ethyl ester

-
-
762-04-9
phosphonic acid diethyl ester

-
-
867-13-0
diethoxyphosphoryl-acetic acid ethyl ester

| Conditions | Yield |
|---|---|
| With potassium carbonate; 18-crown-6 ether at 80℃; for 6h; | 77% |
| With sodium hydroxide; N-benzyl-N,N,N-triethylammonium chloride In dichloromethane at 0 - 5℃; for 0.333333h; | 55% |
| With potassium carbonate In ethanol at 78℃; for 2h; Substitution; Michaelis-Becker reaction; | 33% |
-
-
78-94-4
methyl vinyl ketone

-
-
66700-63-8
diethyl 1-iodo-1-carbethoxy-methylphosphonate

-
A
-
867-13-0
diethoxyphosphoryl-acetic acid ethyl ester

| Conditions | Yield |
|---|---|
| With triethyl borane; tri-n-butyl-tin hydride In hexane; toluene at -78℃; for 6h; Yields of byproduct given; | A n/a B 77% |


| Conditions | Yield |
|---|---|
| With n-butyllithium; lithium diisopropyl amide In tetrahydrofuran; hexane at -60℃; | 74% |
-
-
132646-34-5
2-(5-methyl-2-phenyl-4-oxazolyl)methyl-5-benzofurancarboxaldehyde

-
A
-
867-13-0
diethoxyphosphoryl-acetic acid ethyl ester

-
B
-
159017-57-9
ethyl (E)-3-[2-(5-methyl-2-phenyl-4-oxazolylmethyl)benzofuran-5-yl]acrylate

| Conditions | Yield |
|---|---|
| A n/a B 74% |
-
-
1067-74-9
Methyl diethylphosphonoacetate

-
-
219650-29-0
(-)-methyl (2S,3aS,5R,11bR)-3-benzyl-2,3,3a,4,5,7-hexahydro-3,5-ethano-12-oxo-1H-pyrrolo-<2,3-d>carbazole-6-carboxylate

-
A
-
867-13-0
diethoxyphosphoryl-acetic acid ethyl ester

-
B
-
219650-31-4
(-)-methyl (2S,3aS,5R,11bR)-3-benzyl-2,3,3a,4,5,7-hexahydro-3,5-ethano-12-(E)-<(methoxycarbonyl)methylene>-1H-pyrrolo<2,3-d>carbazole-6-carboxylates

-
C
-
219650-30-3
(-)-methyl (2S,3aS,5R,11bR)-3-benzyl-2,3,3a,4,5,7-hexahydro-3,5-ethano-12-(Z)-<(methoxycarbonyl)methylene>-1H-pyrrolo<2,3-d>carbazole-6-carboxylates

| Conditions | Yield |
|---|---|
| With potassium hexamethyldisilazane In tetrahydrofuran for 0.5h; Ambient temperature; Yield given; | A n/a B 64% C n/a |

-
-
762-04-9, 123-22-8
Diethyl phosphonate

-
-
105-39-5
chloroacetic acid ethyl ester

-
-
867-13-0
diethoxyphosphoryl-acetic acid ethyl ester

| Conditions | Yield |
|---|---|
| With sodium In diethyl ether | 60% |
-
-
159017-37-5
(E)-4-[2-(5-methyl-2-phenyl-4-oxazolyl)ethoxy]cinnamaldehyde

-
A
-
867-13-0
diethoxyphosphoryl-acetic acid ethyl ester

-
B
-
1026936-75-3
ethyl (E)-5-[4-[2-(5-methyl-2-phenyl-4-oxazolyl)ethoxy]phenyl]-2,4-pentadienoate

| Conditions | Yield |
|---|---|
| A n/a B 56% |
-
-
108-05-4
vinyl acetate

-
-
66700-63-8
diethyl 1-iodo-1-carbethoxy-methylphosphonate

-
A
-
867-13-0
diethoxyphosphoryl-acetic acid ethyl ester

| Conditions | Yield |
|---|---|
| With triethyl borane; tri-n-butyl-tin hydride In hexane; toluene at -78℃; for 6h; Yields of byproduct given; | A n/a B 38% |

-
-
7071-12-7
triethyl 2-chloro-2-phosphonoacetate

-
A
-
867-13-0
diethoxyphosphoryl-acetic acid ethyl ester

-
B
-
66051-39-6
(E)-ethyl 2-chloro-3-(4-methoxyphenyl)propenoate

-
C
-
66051-40-9
(E)-methoxyphenyl-3 chloro-2 propenoate d'ethyle

| Conditions | Yield |
|---|---|
| With tetramethylammonium tetrafluoroborate; 4-methoxy-benzaldehyde In N,N-dimethyl-formamide Ambient temperature; controlled potential electrolysis; cathode: Hg; electrode ref.: AgI/Ag; Yields of byproduct given; | A 22% B n/a C n/a |

-
-
7071-12-7
triethyl 2-chloro-2-phosphonoacetate

-
-
123-11-5
4-methoxy-benzaldehyde

-
A
-
867-13-0
diethoxyphosphoryl-acetic acid ethyl ester

-
B
-
66051-39-6
(E)-ethyl 2-chloro-3-(4-methoxyphenyl)propenoate

-
C
-
66051-40-9
(E)-methoxyphenyl-3 chloro-2 propenoate d'ethyle

| Conditions | Yield |
|---|---|
| With tetramethylammonium tetrafluoroborate In N,N-dimethyl-formamide Ambient temperature; controlled potential electrolysis; cathode: Hg; electrode ref.: AgI/Ag; Yield given; | A 22% B n/a C n/a |

-
-
7071-12-7
triethyl 2-chloro-2-phosphonoacetate

-
-
123-11-5
4-methoxy-benzaldehyde

-
A
-
867-13-0
diethoxyphosphoryl-acetic acid ethyl ester

-
B
-
24393-56-4
ethyl (E)-3-(4-methoxyphenyl)prop-2-enoate

-
C
-
51507-22-3
(Z)-ethyl 3-(4-methoxyphenyl)acrylate

| Conditions | Yield |
|---|---|
| With tetramethylammonium tetrafluoroborate In N,N-dimethyl-formamide Ambient temperature; controlled potential electrolysis; cathode: Hg; electrode ref.: AgI/Ag; Yield given; | A 14% B n/a C n/a |

-
-
60-29-7
diethyl ether

-
-
2303-76-6, 118080-94-7
sodium diethyl phosphite

-
-
105-39-5
chloroacetic acid ethyl ester

-
-
867-13-0
diethoxyphosphoryl-acetic acid ethyl ester

-
-
2303-76-6, 118080-94-7
sodium diethyl phosphite

-
-
105-39-5
chloroacetic acid ethyl ester

-
-
867-13-0
diethoxyphosphoryl-acetic acid ethyl ester

| Conditions | Yield |
|---|---|
| With hexane | |
| With ligroine | |
| With ethanol |
-
-
2303-76-6, 118080-94-7
sodium diethyl phosphite

-
-
105-39-5
chloroacetic acid ethyl ester

-
-
71-43-2
benzene

-
-
867-13-0
diethoxyphosphoryl-acetic acid ethyl ester

-
-
2303-76-6, 118080-94-7
sodium diethyl phosphite

-
-
105-36-2
ethyl bromoacetate

-
-
867-13-0
diethoxyphosphoryl-acetic acid ethyl ester

| Conditions | Yield |
|---|---|
| With benzene |
-
-
64-17-5
ethanol

-
-
34164-06-2
dichlorophosphoryl-acetyl chloride

-
-
867-13-0
diethoxyphosphoryl-acetic acid ethyl ester

| Conditions | Yield |
|---|---|
| With pyridine In diethyl ether |
-
-
34159-39-2
diethyl ester of 2,2-diethoxyethenephosphonic acid

-
-
867-13-0
diethoxyphosphoryl-acetic acid ethyl ester

| Conditions | Yield |
|---|---|
| With sulfuric acid; water |
-
-
74119-48-5
2-Diethoxyphosphoryl-3-dimethylaminoacrylsaeure-ethylester

-
A
-
51-17-2
benzoimidazole

-
B
-
867-13-0
diethoxyphosphoryl-acetic acid ethyl ester

| Conditions | Yield |
|---|---|
| With 1,2-diamino-benzene In ethanol Heating; |
-
-
589-57-1
diethyl phosphorylchloridite

-
-
141-78-6
ethyl acetate

-
-
867-13-0
diethoxyphosphoryl-acetic acid ethyl ester

| Conditions | Yield |
|---|---|
| Yield given. Multistep reaction. Further byproducts given; |
-
-
1117-96-0
Diazoethan

-
-
124-38-9
carbon dioxide

-
-
762-04-9
phosphonic acid diethyl ester

-
-
74-88-4
methyl iodide

-
-
867-13-0
diethoxyphosphoryl-acetic acid ethyl ester

| Conditions | Yield |
|---|---|
| In diethyl ether at 0℃; for 0.5h; |

-
-
74119-48-5
2-Diethoxyphosphoryl-3-dimethylaminoacrylsaeure-ethylester

-
-
95-54-5
1,2-diamino-benzene

-
A
-
51-17-2
benzoimidazole

-
B
-
867-13-0
diethoxyphosphoryl-acetic acid ethyl ester

| Conditions | Yield |
|---|---|
| In ethanol Heating; |

-
-
683-08-9
Diethyl methylphosphonate

-
-
105-58-8
Diethyl carbonate

-
-
867-13-0
diethoxyphosphoryl-acetic acid ethyl ester

| Conditions | Yield |
|---|---|
| With n-butyllithium; diisopropylamine 1.) hexane, THF, -78 deg C, 5 min, 2.) THF, -78 deg C --> 0 deg C; Yield given. Multistep reaction; | |
| With n-butyllithium 1.) THF, hexane, -55 deg C, 20 min, 2.) THF, hexane, -55 deg C, 30 min; Yield given. Multistep reaction; |
-
-
105-36-2
ethyl bromoacetate

-
-
122-52-1
triethyl phosphite

-
A
-
867-13-0
diethoxyphosphoryl-acetic acid ethyl ester

-
B
-
63720-98-9
phosphoric acid 1-ethoxy-vinyl ester diethyl ester

| Conditions | Yield |
|---|---|
| In benzene for 8h; Heating; | |
| In benzene for 8h; Heating; |

-
-
5823-12-1
ethyl 2,2-dichloro-2-diethoxyphosphorylacetate

-
-
867-13-0
diethoxyphosphoryl-acetic acid ethyl ester

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 7 percent / p-methoxybenzaldehyde, Me4NBF4 / dimethylformamide / Ambient temperature; controlled potential electrolysis; cathode: Hg; electrode ref.: AgI/Ag 2: 22 percent / Me4NBF4, p-methoxybenzaldehyde / dimethylformamide / Ambient temperature; controlled potential electrolysis; cathode: Hg; electrode ref.: AgI/Ag View Scheme | |
| Multi-step reaction with 2 steps 1: 7 percent / p-methoxybenzaldehyde, Me4NBF4 / dimethylformamide / Ambient temperature; controlled potential electrolysis; cathode: Hg; electrode ref.: AgI/Ag 2: 22 percent / Me4NBF4 / dimethylformamide / Ambient temperature; controlled potential electrolysis; cathode: Hg; electrode ref.: AgI/Ag View Scheme |
-
-
50-00-0
formaldehyd

-
-
867-13-0
diethoxyphosphoryl-acetic acid ethyl ester

-
-
20345-61-3
ethyl 2-diethoxyphosphinoylacrylate

| Conditions | Yield |
|---|---|
| Stage #1: formaldehyd; diethoxyphosphoryl-acetic acid ethyl ester With piperidine In methanol at 80℃; for 36h; Stage #2: With toluene-4-sulfonic acid In toluene for 16h; Dean-Stark; Reflux; | 100% |
| Stage #1: formaldehyd With piperidine In methanol for 0.5h; Heating; Stage #2: diethoxyphosphoryl-acetic acid ethyl ester In methanol for 70h; Heating; | 73% |
| piperidine In methanol Heating; | 60% |
-
-
867-13-0
diethoxyphosphoryl-acetic acid ethyl ester

-
-
108-94-1
cyclohexanone

-
-
1552-92-7
ethyl cyclohexylideneacetate

| Conditions | Yield |
|---|---|
| Stage #1: diethoxyphosphoryl-acetic acid ethyl ester With sodium hydride In tetrahydrofuran; mineral oil for 1h; Stage #2: cyclohexanone In tetrahydrofuran; mineral oil for 18h; | 100% |
| With sodium hydride In tetrahydrofuran for 8h; | 98% |
| Stage #1: diethoxyphosphoryl-acetic acid ethyl ester With 15-crown-5; sodium hydride In tetrahydrofuran; mineral oil at 0℃; Stage #2: cyclohexanone In tetrahydrofuran; mineral oil at -40 - 20℃; | 97% |

| Conditions | Yield |
|---|---|
| With triethylamine; magnesium bromide In tetrahydrofuran at 25℃; for 12h; | 100% |
| (i) NaH, Et2O, (ii) /BRN= 1560236/; Multistep reaction; |
-
-
867-13-0
diethoxyphosphoryl-acetic acid ethyl ester

-
-
123-19-3
4-heptanone

-
-
10297-63-9
ethyl 3-propylhex-2-enoate

| Conditions | Yield |
|---|---|
| Stage #1: diethoxyphosphoryl-acetic acid ethyl ester With sodium hydride In tetrahydrofuran at 20℃; Stage #2: 4-heptanone at 20℃; for 24h; Reflux; | 100% |
| Stage #1: diethoxyphosphoryl-acetic acid ethyl ester With sodium hydride In tetrahydrofuran; mineral oil at 0 - 20℃; for 0.5h; Schlenk technique; Inert atmosphere; Stage #2: 4-heptanone In tetrahydrofuran; mineral oil at 20℃; Schlenk technique; Inert atmosphere; | 88% |
| Stage #1: diethoxyphosphoryl-acetic acid ethyl ester; 4-heptanone With sodium hydride In tetrahydrofuran; mineral oil at 20℃; for 0.333333h; Inert atmosphere; Schlenk technique; Stage #2: In tetrahydrofuran; mineral oil for 20h; Reflux; | 86% |

| Conditions | Yield |
|---|---|
| With potassium hydroxide In ethanol; water at 20℃; | 100% |
| With water; sodium hydroxide In ethanol at 20℃; Inert atmosphere; | 97% |
| With hydrogenchloride; potassium hydroxide 1.) aq. EtOH, 20 h; | 95% |
-
-
867-13-0
diethoxyphosphoryl-acetic acid ethyl ester

-
-
630-19-3
pivalaldehyde

-
-
87995-20-8
3-tert-butyl-acrylic acid ethyl ester

| Conditions | Yield |
|---|---|
| Stage #1: diethoxyphosphoryl-acetic acid ethyl ester With sodium hydride In tetrahydrofuran Cooling with ice; Stage #2: pivalaldehyde In tetrahydrofuran at 20℃; for 48h; Stage #3: With ammonium chloride In tetrahydrofuran; water | 100% |
| (i) NaH, Et2O, (ii) /BRN= 506060/; Multistep reaction; | |
| Stage #1: diethoxyphosphoryl-acetic acid ethyl ester With sodium hydride In tetrahydrofuran at 0℃; Inert atmosphere; Stage #2: pivalaldehyde In tetrahydrofuran at 0 - 20℃; Inert atmosphere; | |
| With sodium tert-pentoxide at 0 - 20℃; for 1h; Horner-Wadsworth-Emmons Olefination; Green chemistry; | |
| With sodium hydride In tetrahydrofuran at 0℃; Inert atmosphere; |
-
-
98-01-1
furfural

-
-
867-13-0
diethoxyphosphoryl-acetic acid ethyl ester

-
-
623-20-1
ethyl (E)-3-(2-furyl)prop-2-enoate

| Conditions | Yield |
|---|---|
| With potassium carbonate In neat (no solvent) Mechanism; var. other bases, effect of water; | 100% |
| With water; barium dihydroxide In 1,4-dioxane at 70℃; for 0.416667h; | 100% |
| With water; barium dihydroxide In 1,4-dioxane at 70℃; for 0.416667h; Product distribution; other catalyst, other solvents, influence of water; | 100% |
-
-
498-62-4
3-thiophene carboxaldehyde

-
-
867-13-0
diethoxyphosphoryl-acetic acid ethyl ester

-
-
50266-60-9
(E)-3-thiophen-3-yl-acrylic acid ethyl ester

| Conditions | Yield |
|---|---|
| Stage #1: diethoxyphosphoryl-acetic acid ethyl ester With sodium hydride In tetrahydrofuran; mineral oil at 0 - 20℃; for 0.5h; Stage #2: 3-thiophene carboxaldehyde In tetrahydrofuran; mineral oil at 20℃; for 1h; | 100% |
| Stage #1: diethoxyphosphoryl-acetic acid ethyl ester With sodium hydride In tetrahydrofuran; mineral oil at 0℃; for 0.5h; Stage #2: 3-thiophene carboxaldehyde In tetrahydrofuran; mineral oil at 0 - 20℃; for 3.25h; | 100% |
| Stage #1: diethoxyphosphoryl-acetic acid ethyl ester With potassium tert-butylate In tetrahydrofuran at 20℃; Stage #2: 3-thiophene carboxaldehyde In tetrahydrofuran at 90℃; for 0.5h; | 90% |
-
-
29943-42-8
Tetrahydro-4H-pyran-4-one

-
-
867-13-0
diethoxyphosphoryl-acetic acid ethyl ester

-
-
130312-00-4
ethyl tetrahydro-4H-pyran-4-ylideneacetate

| Conditions | Yield |
|---|---|
| With sodium hydride In mineral oil; benzene at 0 - 20℃; for 2.33333h; Inert atmosphere; | 100% |
| Stage #1: diethoxyphosphoryl-acetic acid ethyl ester With sodium hydride In tetrahydrofuran; mineral oil at 0 - 20℃; for 0.5h; Schlenk technique; Inert atmosphere; Stage #2: Tetrahydro-4H-pyran-4-one In tetrahydrofuran; mineral oil at 20℃; Schlenk technique; Inert atmosphere; | 100% |
| With potassium carbonate In N,N-dimethyl-formamide at 80℃; | 96% |
-
-
4746-97-8
cyclohexanedione monoethylene ketal

-
-
867-13-0
diethoxyphosphoryl-acetic acid ethyl ester

-
-
51656-91-8
ethyl 1,4-dioxaspiro[4,5]dec-8-ylideneacetate

| Conditions | Yield |
|---|---|
| With sodium hydride In 1,2-dimethoxyethane for 11.5h; Ambient temperature; | 100% |
| With sodium hydride In tetrahydrofuran at 0℃; for 0.666667h; Horner-Wadsworth-Emmons reaction; | 100% |
| Stage #1: cyclohexanedione monoethylene ketal With sodium hydride In tetrahydrofuran; mineral oil at 0℃; for 1h; Stage #2: diethoxyphosphoryl-acetic acid ethyl ester In tetrahydrofuran; mineral oil at -20 - 20℃; for 3h; | 100% |
-
-
75-77-4
chloro-trimethyl-silane

-
-
867-13-0
diethoxyphosphoryl-acetic acid ethyl ester

-
-
65868-40-8
ethyl P,P-bis(trimethylsilyl) phosphonoacetate

| Conditions | Yield |
|---|---|
| at 100℃; for 168h; Inert atmosphere; | 100% |
| With potassium iodide In acetonitrile at 25 - 35℃; for 0.5h; | |
| In chlorobenzene at 140℃; for 18h; |

| Conditions | Yield |
|---|---|
| With n-butyllithium In 1,2-dimethoxyethane; hexane 1.) 0 deg C, 10 min, 2.) 30 min; | 100% |
| Stage #1: diethoxyphosphoryl-acetic acid ethyl ester With sodium hydride In diethyl ether at 0℃; for 0.0833333h; Stage #2: Octanal In diethyl ether for 0.25h; | 96% |
| Stage #1: diethoxyphosphoryl-acetic acid ethyl ester With sodium hydride In diethyl ether; mineral oil at 0℃; Stage #2: Octanal In diethyl ether; mineral oil at 0℃; | 96% |
-
-
867-13-0
diethoxyphosphoryl-acetic acid ethyl ester

-
-
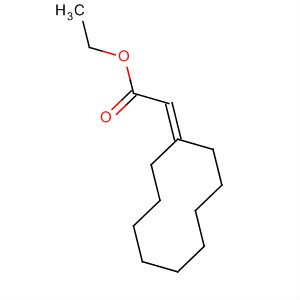
91200-48-5
6-(6-ethoxycarbonyl-4,4-dimethylhex-5-enyl)cyclohex-2-enone

| Conditions | Yield |
|---|---|
| With sodium hydride In 1,2-dimethoxyethane for 1h; Ambient temperature; | 100% |
-
-
867-13-0
diethoxyphosphoryl-acetic acid ethyl ester

-
-
57711-44-1
(3S,10S,13S)-3-((tert-butyldimethylsilyl)oxy)-10,13-dimethyl-1,2,3,4,7,8,9,10,11,12,13,14,15,16-tetradecahydro-17H-cyclopenta-[a]phenanthren-17-one

-
-
115019-45-9
(E)-ethyl 3β-(tert-butyldimethylsiloxy)-5α-pregn-17-(20)-en-21-oate

| Conditions | Yield |
|---|---|
| With sodium ethanolate In tetrahydrofuran; ethanol at 85℃; for 15h; | 100% |
| With sodium ethanolate In tetrahydrofuran; ethanol for 10h; Heating; | 98.6% |
-
-
867-13-0
diethoxyphosphoryl-acetic acid ethyl ester

-
-
123718-34-3
(E)-4-<(E)-1-methyl-3-(2,6,6-trimethyl-1-cyclohexenyl)-2-propenylidene>-2-cyclohexen-1-one

| Conditions | Yield |
|---|---|
| With sodium hydride In tetrahydrofuran for 2.5h; Ambient temperature; | 100% |
-
-
867-13-0
diethoxyphosphoryl-acetic acid ethyl ester

-
-
144148-22-1
7-(4-tert-butylbenzyl)-2-(4-fluorophenyl)-4,5,6,7-tetrahydro-2H-indazole-3-carboxaldehyde

-
-
144148-78-7
ethyl (E)-3-<7-(4-tert-butylbenzyl)-2-(4-fluorophenyl)-4,5,6,7-tetrahydro-2H-indazol-3-yl>-2-propenoate

| Conditions | Yield |
|---|---|
| With sodium hydride In tetrahydrofuran Ambient temperature; | 100% |
-
-
867-13-0
diethoxyphosphoryl-acetic acid ethyl ester

-
-
157330-99-9
rac-(E)-ethyl 7-(methoxycarbonyl)amino-8-(trityloxy)-2-octenoate

| Conditions | Yield |
|---|---|
| With sodium hydride In tetrahydrofuran for 0.5h; Ambient temperature; | 100% |
-
-
867-13-0
diethoxyphosphoryl-acetic acid ethyl ester

-
-
99-61-6
3-nitro-benzaldehyde

-
-
621-19-2
(E)-ethyl 3-(3-nitrophenyl)acrylate

| Conditions | Yield |
|---|---|
| With water; barium dihydroxide In 1,4-dioxane at 70℃; for 0.416667h; | 100% |
| Stage #1: diethoxyphosphoryl-acetic acid ethyl ester; 3-nitro-benzaldehyde With lithium chloride In acetonitrile at 0℃; for 0.0833333h; Horner-Wadsworth-Emmons reaction; Stage #2: With 1,8-diazabicyclo[5.4.0]undec-7-ene In acetonitrile at 0 - 20℃; | 96% |
| Stage #1: diethoxyphosphoryl-acetic acid ethyl ester With sodium hydride In tetrahydrofuran; mineral oil at 0℃; for 0.5h; Stage #2: 3-nitro-benzaldehyde In tetrahydrofuran; mineral oil at 0 - 20℃; for 1h; | 93% |
-
-
867-13-0
diethoxyphosphoryl-acetic acid ethyl ester

-
-
86-81-7
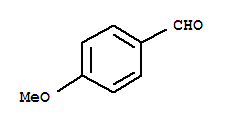
3,4,5-trimethoxy-benzaldehyde

-
-
1878-29-1, 30273-65-5, 31892-98-5
ethyl (E)-3-(3,4,5-trimethoxyphenyl)acrylate

| Conditions | Yield |
|---|---|
| Stage #1: diethoxyphosphoryl-acetic acid ethyl ester With lithium chloride In tetrahydrofuran at 20℃; for 0.25h; Horner-Wadsworth-Emmons Olefination; Inert atmosphere; Stage #2: 3,4,5-trimethoxy-benzaldehyde With 1,8-diazabicyclo[5.4.0]undec-7-ene In tetrahydrofuran Horner-Wadsworth-Emmons Olefination; Inert atmosphere; | 100% |
| With potassium carbonate In water at 70℃; for 4h; | 90% |
| With potassium carbonate In water at 70℃; for 4h; Wittig Olefination; stereoselective reaction; | 90% |

| Conditions | Yield |
|---|---|
| With sodium hydride In tetrahydrofuran for 1h; | 100% |
| With potassium carbonate; 1,8-diazabicyclo[5.4.0]undec-7-ene at 20℃; for 2h; Horner-Wadsworth-Emmons reaction; neat (no solvent); optical yield given as %de; stereoselective reaction; | 100% |
| With n-butyllithium In tetrahydrofuran; hexane 1.) 0 deg C, 10 min, 2.) 30 min; | 99% |
-
-
867-13-0
diethoxyphosphoryl-acetic acid ethyl ester

-
-
587-04-2
3-Chlorobenzaldehyde

-
-
2373-88-8, 118315-76-7, 62174-98-5
ethyl (E)-3-(3-chlorophenyl)prop-2-enoate

| Conditions | Yield |
|---|---|
| Stage #1: diethoxyphosphoryl-acetic acid ethyl ester With sodium hydride In tetrahydrofuran at 23℃; for 1h; Stage #2: m-Chlorobenzaldehyde In tetrahydrofuran for 1h; | 100% |
| With sodium hydride In N,N-dimethyl-formamide at -60℃; Horner-Wadsworth-Emmons Olefination; | 95% |
| With tetrabutyl ammonium fluoride In tetrahydrofuran at 30℃; for 4h; Glovebox; | 84% |
-
-
867-13-0
diethoxyphosphoryl-acetic acid ethyl ester

-
-
591-31-1
3-methoxy-benzaldehyde

-
-
33877-04-2, 24393-55-3
ethyl (E)-3-(3-methoxyphenyl)acrylate

| Conditions | Yield |
|---|---|
| Stage #1: diethoxyphosphoryl-acetic acid ethyl ester With lithium chloride In tetrahydrofuran at 20℃; for 0.25h; Horner-Wadsworth-Emmons Olefination; Inert atmosphere; Stage #2: 3-methoxy-benzaldehyde With 1,8-diazabicyclo[5.4.0]undec-7-ene In tetrahydrofuran Horner-Wadsworth-Emmons Olefination; Inert atmosphere; | 100% |
| With sodium hydride In tetrahydrofuran; mineral oil at 0 - 20℃; for 1h; | 100% |
| With tetrabutyl ammonium fluoride In tetrahydrofuran at 30℃; for 4h; Glovebox; | 98% |
-
-
867-13-0
diethoxyphosphoryl-acetic acid ethyl ester

-
-
590-86-3
isovaleraldehyde

-
-
34993-63-0
ethyl (E)-5-methylhex-2-enoate

| Conditions | Yield |
|---|---|
| Stage #1: diethoxyphosphoryl-acetic acid ethyl ester With sodium hydride In 1,2-dimethoxyethane at 0℃; for 0.416667h; Stage #2: isovaleraldehyde In 1,2-dimethoxyethane for 16h; Horner-Emmons homologation; Heating; | 100% |
| With n-butyllithium In tetrahydrofuran; hexane at 0℃; for 6h; Wadsworth-Honer-Emmons olefination; | 95% |
| With sodium hydride In tetrahydrofuran | 90% |
-
-
867-13-0
diethoxyphosphoryl-acetic acid ethyl ester

-
-
1123-56-4
2,6-dimethylbenzaldehyde

-
-
527759-23-5
ethyl (E)-3-(2,6-dimethylphenyl)-2-propenoate

| Conditions | Yield |
|---|---|
| Stage #1: diethoxyphosphoryl-acetic acid ethyl ester With sodium hydride In tetrahydrofuran at 20℃; for 0.5h; Stage #2: 2,6-dimethylbenzaldehyde In tetrahydrofuran at 20℃; for 6h; Horner-Wadsworth-Emmons reaction; | 100% |
| With tetrabutyl ammonium fluoride In tetrahydrofuran at 30℃; for 4h; Glovebox; | 97% |
| With potassium tert-butylate Horner-Wadsworth-Emmons olefination; | 89% |
| With potassium tert-butylate In tetrahydrofuran Ambient temperature; |
-
-
867-13-0
diethoxyphosphoryl-acetic acid ethyl ester

-
-
78-84-2
isobutyraldehyde

-
-
173724-80-6
(E)-2-(Diethoxy-phosphoryl)-4-methyl-pent-2-enoic acid ethyl ester

| Conditions | Yield |
|---|---|
| With piperidine; acetic acid In benzene for 48h; Heating; | 100% |
| With piperidine; acetic acid for 62h; Condensation; Knoevenagel condensation; | 80% |
| Stage #1: diethoxyphosphoryl-acetic acid ethyl ester With triethylamine; titanium tetrachloride In dichloromethane at 0 - 5℃; Stage #2: isobutyraldehyde In dichloromethane at 0 - 5℃; Horner-Wadsworth-Emmos reaction; |
-
-
5447-02-9
3,4-dibenzyloxybenzaldehyde

-
-
867-13-0
diethoxyphosphoryl-acetic acid ethyl ester

-
-
203571-40-8
3-(3,4-bis-benzyloxy-phenyl)-acrylic acid ethyl ester

| Conditions | Yield |
|---|---|
| Stage #1: diethoxyphosphoryl-acetic acid ethyl ester With sodium hydride In tetrahydrofuran Stage #2: 3,4-dibenzyloxybenzaldehyde In tetrahydrofuran for 0.166667h; Further stages.; | 100% |
| With sodium hydride In 1,2-dimethoxyethane for 0.333333h; | 99.5% |
| With sodium In ethanol Ambient temperature; | 76% |
-
-
867-13-0
diethoxyphosphoryl-acetic acid ethyl ester

-
-
185996-42-3
(S)-3-[2-((S)-2,2-Dimethyl-[1,3]dioxolan-4-ylmethyl)-thiazol-4-yl]-butyraldehyde

-
-
185996-43-4
(E)-(S)-5-[2-((S)-2,2-Dimethyl-[1,3]dioxolan-4-ylmethyl)-thiazol-4-yl]-hex-2-enoic acid ethyl ester

| Conditions | Yield |
|---|---|
| With 1,8-diazabicyclo[5.4.0]undec-7-ene; lithium chloride In acetonitrile for 0.25h; Ambient temperature; | 100% |
-
-
867-13-0
diethoxyphosphoryl-acetic acid ethyl ester

-
-
198336-00-4
(1R,5R,6R)-2-[6-(tert-butyldimethylsilanyloxymethyl)-5-isopropyl-2-methylcyclohex-2-enyl]acetaldehyde

-
-
198336-01-5
(E)-4-[(1R,5R,6R)-6-(tert-Butyl-dimethyl-silanyloxymethyl)-5-isopropyl-2-methyl-cyclohex-2-enyl]-but-2-enoic acid ethyl ester

| Conditions | Yield |
|---|---|
| With sodium hydride In tetrahydrofuran at 0 - 25℃; | 100% |
| With sodium hydride 1.) THF, 25 deg C, 30 min, 2.) THF, from 0 to 25 deg C, 5 h; Yield given. Multistep reaction; |
-
-
867-13-0
diethoxyphosphoryl-acetic acid ethyl ester

-
-
181646-38-8
(1,1-dimethyl-3-oxo-propyl)-carbamic acid tert-butyl ester

-
-
181646-40-2
ethyl (2E)-5-(tert-butoxycarbonylamino)-5-methylhex-2-enoate

| Conditions | Yield |
|---|---|
| Stage #1: diethoxyphosphoryl-acetic acid ethyl ester With potassium tert-butylate In toluene at 60℃; for 0.5h; Wittig-Horner reaction; Stage #2: (1,1-dimethyl-3-oxo-propyl)-carbamic acid tert-butyl ester In toluene at 60℃; for 0.166667h; Wittig-Horner reaction; | 100% |
| With potassium tert-butylate 1.) THF, RT, 40 min, 2.) THF, RT, 75 min; Yield given. Multistep reaction; | |
| With potassium tert-butylate; sodium hydrogencarbonate In tetrahydrofuran; n-heptane |
Related products
Raw Materials
Downstream Products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View














 N,
N, Xi,
Xi, Xn
Xn