Hangzhou Dingyan Chem Co., Ltd
Items Standard Result Appearance Colorless,transparent liquid Complies Content% ≥99.90
Cas:141-78-6
Min.Order:1 Kilogram
FOB Price: $3.0 / 10.0
Type:Manufacturers
inquiryHenan Allgreen Chemical Co.,Ltd
T he company has advanced technology, as well as a large number of excellent R & D team, to provide customers from the grams to one hundred kilograms and tons of high-quality products, competitive prices and quality service Appearance:White or
Nanjing Yuance Industry&Trade Co., Ltd.
Purity 99% with competitive price Fast delivery Prompt shipment Appearance:Colorless to pale yellow liquid with fruity ethereal odo Storage:Store in cool dry place Package:170 kg/drum Application:Widely used in the preparation of cherry, peach
Cas:141-78-6
Min.Order:1 Metric Ton
Negotiable
Type:Manufacturers
inquiryEAST CHEMSOURCES LIMITED
ethyl acetate Measuring items Technical Index Appearance Clear liquid, free of any suspended impurities Acetic ester, w/%≥ 99.7 Ethanol, w/%≤ 0.10
Cas:141-78-6
Min.Order:1 Kilogram
FOB Price: $2.0 / 3.0
Type:Manufacturers
inquiryHebei yanxi chemical co.,LTD.
Hebei yanxi chemical co. LTD. has expanded a compositive entity from initially only as a small manufacturer. The company dedicated to the development, production and marketing of chemicals. After many years of efforts, we have established stable
Cas:141-78-6
Min.Order:1 Metric Ton
FOB Price: $1.0 / 2.0
Type:Trading Company
inquiryAlity Chemical Corporation
1.Specification Purity: ≥ 99% Spec.Gravity @ 25 ° C: 0.889-0.909 Ref. Index @ 20 ° C: 1.369-1.377 Boiling Point: 76-77 ° C Flash point:
Henan Tianfu Chemical Co., Ltd.
Our advantage:Our company was built in 2009 with an ISO certificate.In the past 5 years, we have grown up as a famous fine chemicals supplier in China and we had established stable business relationships with Samsung,LG,Merck,Thermo Fisher Scient
Cas:141-78-6
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryJinan Finer Chemical Co., Ltd
Product Description Product website: http://www.finerchem.com Product Name Ethyl acetate CAS No. 141-78-6 A
Cas:141-78-6
Min.Order:1 Metric Ton
FOB Price: $12.0
Type:Lab/Research institutions
inquiryHenan Sinotech Import&Export Corporation
Ethyl acetate Basic information Product Name: Ethyl acetate Synonyms: ALCOHOL, REAGENT, DENATURED;ALCOHOL;ALCOHOL, DENATURED; ALCOHOL C2;ABSOLUTE ALCOHOL;ACETIC ESTER;ACETIC E
Cas:141-78-6
Min.Order:5 Metric Ton
FOB Price: $1.0 / 2.0
Type:Other
inquiryBaoji Guokang Healthchem co.,ltd
Our company has been in existence for 10 years since its establishment. We have our own unique team. The company integrates independent research and development, production and sales. We have established famous brands at home and abroad. At prese
Cas:141-78-6
Min.Order:1 Kilogram
FOB Price: $9.0 / 10.0
Type:Trading Company
inquiryWuhan Zenuo Biological Medicine Technology Co Ltd
Product Name: Ethyl acetate Synonyms: Ethyl Acetate Biosynthesis;(Benzoylthio)?;Ethyl acetate Manufacturer;Ethyl acetate( 99.8%, HyDry, Water≤50 ppm (by K.F.));Ethyl acetate( 99.8%, HyDry, with molecular sieves, Water≤50 ppm (by K.F.));Dich
Cas:141-78-6
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryLonwin Chemical Group Limited
Ethyl acetate CAS:141-78-6 Specification Product Name Ethyl acetate CAS No. 141-78-6 Appearance Colorless transparent liquid Assay ≥99% Capacity 100mt/year Min.packin
Cas:141-78-6
Min.Order:1 Kilogram
FOB Price: $100.0 / 150.0
Type:Other
inquiryTriumph International Development Limilted
Appearance:White or off-white Solid Storage:Store in a cool,dry place and keep away from direct strong light Package:As customer request Application:Organic synthesis Transportation:Common products:Sea/Air/Courier Dangerous Che
Cas:141-78-6
Min.Order:100 Metric Ton
Negotiable
Type:Lab/Research institutions
inquiryDB BIOTECH CO., LTD
Q1: Are you a manufacturer?Answer: Yes, we are factory founded on 2002. Q2: How to contact with us?Click "contact supplier" And then send us message the product you interest in, you will get reply within 12 hours. Q3:Which kind of payment terms do yo
Afine Chemicals Limited
Our Services 1. New Molecules R&D 2. Own test center HPLC NMR GC LC-MS 3. API and Intermediates from China reputed manufacturers 4. Documents support COA MOA MSDS DMF open part Our advantages 1. Government awarded company. Top 100 enter
Cas:141-78-6
Min.Order:1 Gram
Negotiable
Type:Lab/Research institutions
inquiryHangzhou Keyingchem Co.,Ltd
Hangzhou KeyingChem Co., Ltd. exported this product to many countries and regions at best price. If you are looking for the material’s manufacturer or supplier in China, KeyingChem is your best choice. Pls contact with us freely for getting det
Hangzhou J&H Chemical Co., Ltd.
J&H CHEM R&D center can offer custom synthesis according to the contract research and development services for the fine chemicals, pharmaceutical, biotechnique and some of the other chemicals. J&H CHEM has some Manufacturing base in Jia
Zibo Hangyu Biotechnology Development Co., Ltd
Zibo Hangyu Biotechnology Development Co., Ltd is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality pharmaceuticals, intermediates, special chemicals and OLED intermediates and other fine chemi
Cas:141-78-6
Min.Order:10 Gram
FOB Price: $100.0
Type:Lab/Research institutions
inquiryHANGZHOU YUNUO CHEMICAL CO.,LTD
Superior quality, moderate price & quick delivery. Appearance:colorless liquid Storage:Stored in cool, dry and ventilation place; Away from fire and heat Package:100g/bottle,1kg/bottle,25kg/drum or as per your request Application:Bis(2-hydr
Siwei Development Group Ltd.
Product name: Ethyl Acetate CAS No.: 141-78-6 Molecule Formula:C4H8O2 Molecule Weight:88.11 Purity: 99.0% Package: 190kg/drum Description:Colorless transparent liquid Manufacture Standards:Enterprise Standard TESTI
Henan Wentao Chemical Product Co., Ltd.
We are leading fine chemicals supplier in China and we had established stable business relationships with Samsung,LG,Merck,Thermo Fisher Scientific and so on.Our main business covers the fields below: 1.Noble Metal Catalysts (Pt.Pd...)
Cas:141-78-6
Min.Order:1 Kilogram
FOB Price: $5.0
Type:Lab/Research institutions
inquiryHangzhou Huarong Pharm Co., Ltd.
Hangzhou Huarong Pharm Co., Ltd.established since 2006 , has been actively developing specialty products for Finished Dosages, APIs, Intermediates, and Fine chemicals markets in North America, Europe, Korea, Japan, Mid-East and all over the World. Hu
Hangzhou Zhongqi chem Co.,Ltd.
Located in Hangzhou National Hi-Tech Industrial Development Zone, zhongqichem is a technical company mainly focus on the Custom synthesis, manufacturing, sales of chemicals to various industries. Benefiting from the outstanding customer service and h
Hunan chemfish Pharmaceutical co.,Ltd
Appearance:95%+ Package:R&D,Pilot run Transportation:per client require Port:Express ,Air, Sea
Cas:141-78-6
Min.Order:0
Negotiable
Type:Manufacturers
inquiryResearch Peptide Biotechnology Co., Ltd.
High purity, high success rate, short cycle and moderate priceAppearance:White powder solid Storage:Negative 20 degrees Celsius Package:5mg, 10mg 100mg, 1gram Application:Applied to various scientific research
Kono Chem Co.,Ltd
high purity lowest priceAppearance:solid or liquid Storage:in sealed air resistant place Package:drum and bag Application:for pharma use Transportation:by sea or air Port:Beijing or Guangzhou
JINHUA HUAYI CHEMICAL CO., LTD.
Jinhua huayi chemical co., ltd. is dedicated to the development, production and marketing of chemicals. On the basis of equality and mutual benefit, and under the principle of customer first, credit first, quality first, we are ready to join hands
Aecochem Corp.
Our clients, like BASF,CHEMO,Brenntag,ASR,Evonik,Merck and etc.Appearance:COA Storage:in stock Application:MSDS/TDS
Wuxi TAA Chemical Industry Co.,LTD.
1.A strong technical force and advanced processing equipments. The quality of the products has been strictly inspected and all kinds of index have reached or exceeded domestic and international standards.2. Now we have established long-term stable re
Antimex Chemical Limied
Ansciep Chemical is a professional enterprise manufacturing and distributing fine chemicals and speciality chemicals. We have been dedicated to heterocycle compounds and phenyl rings for tens of years. This is our mature product for export. Our quali
Synthetic route

| Conditions | Yield |
|---|---|
| With [{lμ2-3-1κO-((carboxylatomethyl)amino)-4-chlorobenzoato}(N,N,N-κ3-2,3,5,6-tetrakis(2-pyridyl)pyrazine)zinc(II)]n(dimethylformamide)(water) In dimethyl sulfoxide at 80℃; for 5h; Time; | 85% |

| Conditions | Yield |
|---|---|
| niobium(V) oxide at 200℃; for 1h; Product distribution; examination of niobic acid as an efficient catalyst; var. catalysts, temps;; | 100% |
| With sulfuric acid at 70℃; for 5h; Reagent/catalyst; | 96% |
| With 1-butyl-2-methylbenzimidazolium tetrafluoroborate at 80℃; for 2h; | 95% |

| Conditions | Yield |
|---|---|
| With oxygen; Pd on Zeolite Y at 90 - 110℃; under 760.051 - 1520.1 Torr; Product distribution / selectivity; | A 0.5% B 7.5% C 49.6% |
| With oxygen; Pd on Zeolite Y In water at 70 - 150℃; under 22801.5 Torr; Conversion of starting material; | A 0.1% B 3.6% C 22.5% |
| With oxygen; Pt on Zeolite ZSM-5 at 110℃; under 1520.1 Torr; Product distribution / selectivity; | A 7.4% B 3.9% C 10.2% |


| Conditions | Yield |
|---|---|
| With copper supported on ZnAl2O4 at 399.84℃; Temperature; Flow reactor; | A 90% B n/a |
| With oxygen; Sb2O4-MoO3 (Sb4Mo10O31) at 200 - 350℃; in a flow system; | A n/a B 30% |
| With oxygen; Sb2O4-MoO3 (Sb4Mo10O31) at 200 - 350℃; Product distribution; selectivity and activity of the Sb-Mo-O catalyst in the oxidation of ethanol; | A n/a B 30% |

| Conditions | Yield |
|---|---|
| With lithium hydroxide at 35℃; under 50 Torr; Temperature; Pressure; Reagent/catalyst; | 62% |

| Conditions | Yield |
|---|---|
| With [{(PhN)MeC(Nt-Bu)}AlMe(μ-OMe)]2 at 20℃; for 16h; Tishchenko-Claisen Dismutation; Inert atmosphere; Schlenk technique; Green chemistry; | 95% |
| With Rh(PhBP3)(H)2(NCMe); hydrogen In toluene at 20℃; under 760.051 Torr; for 0.166667h; Tishchenko reaction; | 68% |
| With magnesium oxide; copper at 400℃; |
-
-
74-85-1
ethene

-
-
64-19-7
acetic acid

-
A
-
60-29-7
diethyl ether

-
B
-
64-17-5
ethanol

-
C
-
141-78-6
ethyl acetate

| Conditions | Yield |
|---|---|
| Stage #1: acetic acid With water at 92.4℃; Stage #2: ethene; cesium nitrate; tungstophosphoric acid; water; mixture of, dried, tabletted at 92.4 - 194.4℃; under 6750.68 Torr; Product distribution / selectivity; Gas phase; | A 3.2% B 3.6% C 92.7% |
| With water; cesium nitrate; tungstophosphoric acid; water; mixture of, dried, tabletted at 92.4 - 165℃; under 6750.68 Torr; Product distribution / selectivity; Gas phase; | A 3% B 3.4% C 91.5% |
| With water; lithium nitrate; silica; tungstophosphoric acid; water; mixture of, heated at 150 C at 102.2 - 165℃; under 6750.68 Torr; Product distribution / selectivity; Gas phase; | A 2.2% B 5% C 90.1% |
| With water; lithium nitrate; silica; tungstosilicic acid; water; mixture of, heated at 150 C at 102.2 - 165℃; under 6750.68 Torr; Conversion of starting material; Gas phase; | A 4.7% B 7.6% C 87.7% |

-
-
943406-22-2
4-(3-(3,4-dimethylcyclopenta-1,3-dienone)-4-(cyclohexylamino)benzyl)-2-(3,4-dimethylcyclopenta-1,3-dienone)-N-cyclohexylbenzeneamine


-
B
-
141-78-6
ethyl acetate

| Conditions | Yield |
|---|---|
| With methyllithium; CeCl3 In tetrahydrofuran; diethyl ether | A 46% B n/a |

| Conditions | Yield |
|---|---|
| With silver trifluoromethanesulfonate at 60℃; for 0.0666667h; neat (no solvent); | 97% |
| With tetrakis(triphenylphosphineoxide)dioxouranium(VI) perchlorate In dichloromethane-d2 at 22℃; for 2h; Mechanism; Reagent/catalyst; Solvent; | 97% |
| With vanadyl sulfate trihydrate at 20℃; for 8h; | 93% |

| Conditions | Yield |
|---|---|
| With water; potassium iodide at 20℃; for 3h; Electrochemical reaction; Green chemistry; | 95.2% |
| With HCl(CO)Ru(Phen-(tBu)PNN); potassium tert-butylate In neat (no solvent) for 14h; Schlenk technique; Inert atmosphere; | 59% |
| With iodine; hexachloroplatinate(II) In water | 47% |

| Conditions | Yield |
|---|---|
| With 0.08%-SO3H functionalized-benzylated Al-SBA-15 nanoporous catalyst at 79.84℃; for 2h; | 90% |
| With Ru4H4(CO)8(PBu3)4; hydrogen under 98800 Torr; for 48h; Heating; | 40.7% |
-
-
74-85-1
ethene

-
-
64-19-7
acetic acid

-
C
-
60-29-7
diethyl ether

-
D
-
64-17-5
ethanol

-
E
-
141-78-6
ethyl acetate

| Conditions | Yield |
|---|---|
| With water; silicotungstic acid / SiO2 at 175℃; under 9000.9 Torr; Gas phase; Industry scale; | |
| With water; silicotungstic acid / SiO2 at 175℃; under 9000.9 Torr; Gas phase; Industry scale; |

-
-
64-17-5
ethanol

-
A
-
34557-54-5
methane

-
B
-
124-38-9
carbon dioxide

-
C
-
75-07-0
acetaldehyde

-
D
-
64-19-7
acetic acid

-
E
-
141-78-6
ethyl acetate

| Conditions | Yield |
|---|---|
| With oxygen at 140℃; for 15h; Reagent/catalyst; Temperature; Inert atmosphere; Autoclave; | A n/a B n/a C n/a D 71% E n/a |


| Conditions | Yield |
|---|---|
| With 1-hydroxytetraphenylcyclopentadienyl(tetraphenyl-2,4-cyclopentadien-1-one)-μ-hydrotetracarbonyldiruthenium(II) In toluene at 80℃; under 11103.3 Torr; for 1h; Temperature; Inert atmosphere; Autoclave; |

-
-
78-83-1
2-methyl-propan-1-ol

-
-
75-07-0
acetaldehyde

-
A
-
1540-94-9
N-isobutylacetamide

-
B
-
141-78-6
ethyl acetate

| Conditions | Yield |
|---|---|
| With 1-hydroxytetraphenylcyclopentadienyl(tetraphenyl-2,4-cyclopentadien-1-one)-μ-hydrotetracarbonyldiruthenium(II) In toluene at 80℃; under 11103.3 Torr; for 1h; Inert atmosphere; Autoclave; |


| Conditions | Yield |
|---|---|
| With water at 179.84℃; under 7500.75 Torr; Inert atmosphere; Gas phase; | 67% |
| With sulfuric acid | |
| With pumice stone; water at 450℃; under 14710.2 Torr; |

| Conditions | Yield |
|---|---|
| With hydrogen at 270℃; under 19502 Torr; for 1h; Catalytic behavior; Reagent/catalyst; | A 90% B 9% |
| With hydrogen Reagent/catalyst; | A 75% B n/a |
| With hydrogen Reagent/catalyst; | A 71% B n/a |


-
B
-
141-78-6
ethyl acetate

| Conditions | Yield |
|---|---|
| With hydrogenchloride; sodium borohydrid In methanol; water | A 66.4% B n/a |

| Conditions | Yield |
|---|---|
| With hydrogen at 240 - 280℃; under 15751.6 Torr; Reagent/catalyst; Flow reactor; |


| Conditions | Yield |
|---|---|
| With 5Cu/ZrO2 at 224.84℃; under 760.051 Torr; Reagent/catalyst; |

-
-
105-54-4
butanoic acid ethyl ester

-
A
-
64-17-5
ethanol

-
B
-
109-21-7
butyl butyrate

-
C
-
141-78-6
ethyl acetate

-
D
-
71-36-3
butan-1-ol

| Conditions | Yield |
|---|---|
| With C21H35BrMnN2O2P; hydrogen; potassium hydride In toluene at 100℃; under 15001.5 Torr; for 22h; | A 35 %Spectr. B 12 %Spectr. C 11 %Spectr. D 32 %Spectr. |


| Conditions | Yield |
|---|---|
| With (K(1+))2<(Ph3P)3(Ph2P)Ru2H4>(2-)*2C6H14O3; hydrogen In toluene under 4650.4 Torr; 1) 23 deg C, 0.5 h; 2) 90 deg C, 20 h; | |
| With hydrogen; <(Ph3P)(Ph2P)RuH2-K+*diglyme>2 In toluene at 90℃; under 4650.4 Torr; for 20h; Title compound not separated from byproducts; | |
| With (K(1+))2<(Ph3P)3(Ph2P)Ru2H4>(2-)*2C6H14O3; hydrogen In toluene under 4650.4 Torr; Product distribution; further carboxylic acid esters; other reagents; 1) 23 deg C, 0.5 h; 2) 90 deg C, 20 h;; |

-
-
201230-82-2
carbon monoxide

-
-
64-19-7
acetic acid

-
A
-
67-56-1
methanol

-
B
-
64-17-5
ethanol

-
C
-
79-20-9
acetic acid methyl ester

-
D
-
141-78-6
ethyl acetate

| Conditions | Yield |
|---|---|
| With hydrogen; Cu/ZnO/Al2O3 catalyst (MK-121) at 260℃; under 34375.6 Torr; |

-
-
64-19-7
acetic acid

-
A
-
60-29-7
diethyl ether

-
B
-
64-17-5
ethanol

-
C
-
75-07-0
acetaldehyde

-
D
-
141-78-6
ethyl acetate

| Conditions | Yield |
|---|---|
| With hydrogen at 240 - 280℃; under 15751.6 Torr; Reagent/catalyst; Flow reactor; |


| Conditions | Yield |
|---|---|
| With styrene macroporous cation exchange resin In toluene at 50℃; for 3h; | |
| With styrene-based macroporous cation exchange resin In toluene at 50℃; for 3h; Green chemistry; |
-
-
108-05-4
vinyl acetate

-
-
201230-82-2
carbon monoxide

-
A
-
22094-23-1
2-acetoxypropanal

-
B
-
18545-28-3
3-acetoxypropanal

-
C
-
64-19-7
acetic acid

-
D
-
141-78-6
ethyl acetate

| Conditions | Yield |
|---|---|
| With hydrogen; dicobalt octacarbonyl In toluene at 120℃; under 72402.6 - 77574.3 Torr; for 1.83333 - 2h; Product distribution / selectivity; | |
| With hydrogen; dicobalt octacarbonyl In dichloromethane at 120℃; under 77574.3 Torr; for 1h; Product distribution / selectivity; | |
| With hydrogen; dicobalt octacarbonyl In pyridine at 120℃; under 31029.7 Torr; for 1h; Product distribution / selectivity; | |
| With hydrogen; dicobalt octacarbonyl In dichlorobenzene, 1,2- at 120℃; under 77574.3 Torr; for 1h; Product distribution / selectivity; | |
| Stage #1: carbon monoxide With hydrogen; cobalt(II) acetate In toluene at 200℃; under 51716.2 - 103432 Torr; for 0.75h; Stage #2: vinyl acetate In toluene at 120℃; under 93089.1 Torr; Product distribution / selectivity; |

-
-
108-05-4
vinyl acetate

-
A
-
64-17-5
ethanol

-
B
-
123-86-4
acetic acid butyl ester

-
C
-
141-78-6
ethyl acetate

-
D
-
67-64-1
acetone

| Conditions | Yield |
|---|---|
| With hydrogen; p-benzoquinone; Grace Raney 4310 w/ Mo In Isopropyl acetate at 50 - 89℃; under 6190.25 - 11310.2 Torr; for 0.1 - 1.68333h; Product distribution / selectivity; | |
| With hydrogen; p-benzoquinone; Grace Raney 4310 w/ Mo at 44.1 - 51℃; under 5983.38 - 10999.9 Torr; for 0.433333 - 1.5h; Product distribution / selectivity; | |
| With hydrogen; p-benzoquinone; 5%-palladium/activated carbon In Isopropyl acetate at 36 - 50℃; under 2311.54 - 6138.53 Torr; for 0.0666667h; Product distribution / selectivity; | |
| With hydrogen; p-benzoquinone; 5%-palladium/activated carbon at 54.25 - 59℃; under 6086.82 - 8827.77 Torr; for 0.266667h; Product distribution / selectivity; |


| Conditions | Yield |
|---|---|
| With hydrogenchloride; n-butyllithium In tetrahydrofuran; hexane | 41% |
-
-
67-56-1
methanol

-
-
201230-82-2
carbon monoxide

-
-
74-88-4
methyl iodide

-
A
-
534-15-6
1,1-dimethoxyethane

-
B
-
64-17-5
ethanol

-
C
-
79-20-9
acetic acid methyl ester

-
D
-
75-07-0
acetaldehyde

-
E
-
141-78-6
ethyl acetate

| Conditions | Yield |
|---|---|
| With (acetylacetonato)dicarbonylrhodium (l); hydrogen In toluene at 170℃; under 16276.6 - 25375 Torr; for 4h; Autoclave; | |
| With (acetylacetonato)dicarbonylrhodium (l); rhodium(III) chloride hydrate; 1,3-bis-(diphenylphosphino)propane; hydrogen In toluene at 170℃; under 31015.6 - 47307.2 Torr; for 4h; Concentration; Autoclave; |

-
-
67-56-1
methanol

-
-
201230-82-2
carbon monoxide

-
-
74-88-4
methyl iodide

-
A
-
534-15-6
1,1-dimethoxyethane

-
B
-
79-20-9
acetic acid methyl ester

-
C
-
75-07-0
acetaldehyde

-
D
-
141-78-6
ethyl acetate

| Conditions | Yield |
|---|---|
| With (acetylacetonato)dicarbonylrhodium (l); 1,3-bis-(diphenylphosphino)propane; hydrogen In toluene at 170℃; under 16276.6 - 25375 Torr; for 4h; Autoclave; |


| Conditions | Yield |
|---|---|
| Stage #1: phenylacetylene With n-butyllithium In tetrahydrofuran; hexane at -78℃; for 1h; Stage #2: ethyl acetate With boron trifluoride diethyl etherate In tetrahydrofuran; hexane at -78℃; | 100% |
| Stage #1: phenylacetylene With n-butyllithium In tetrahydrofuran; hexane at 0℃; for 0.5h; Inert atmosphere; Stage #2: ethyl acetate With boron trifluoride diethyl etherate In tetrahydrofuran; hexane at -78℃; for 0.5h; Inert atmosphere; Stage #3: With water; ammonium chloride In tetrahydrofuran; hexane at -78 - 20℃; | 96% |
| Stage #1: phenylacetylene With n-butyllithium In tetrahydrofuran; hexane at -78℃; for 0.5h; Schlenk technique; Stage #2: ethyl acetate With boron trifluoride diethyl etherate In tetrahydrofuran at -78 - 20℃; for 1h; | 89% |

| Conditions | Yield |
|---|---|
| Stage #1: ethyl acetate With lithium hexamethyldisilazane In tetrahydrofuran at -78℃; for 1h; Stage #2: acetone In tetrahydrofuran at -78℃; for 0.5h; | 100% |
| Stage #1: ethyl acetate With lithium hexamethyldisilazane In tetrahydrofuran at 20℃; for 1h; Stage #2: acetone In tetrahydrofuran for 0.5h; | 100% |
| Stage #1: ethyl acetate With lithium hexamethyldisilazane In tetrahydrofuran at -78℃; for 0.5h; Inert atmosphere; Stage #2: acetone In tetrahydrofuran for 0.166667h; | 88% |

| Conditions | Yield |
|---|---|
| With dilithium tetra(tert-butyl)zincate at 0℃; for 1h; Temperature; Inert atmosphere; | 100% |
| K2CO3 + 5percent Carbowax 6000 at 170℃; Product distribution; various catalysts, various amounts of catalysts; | 54 % Chromat. |
| With trans-5,15-bis(2-hydroxy-1-naphthyl)octaethylporphyrine; silver perchlorate In benzene at 50℃; without AgClO4, other catalysts; |

| Conditions | Yield |
|---|---|
| zirconium(IV) oxide at 200℃; in vapor-phase; | 100% |
| With K5 for 2h; Heating; | 90% |
| 18-crown-6 ether; potassium carbonate at 170℃; Product distribution; various catalysts; | 61 % Chromat. |
| 18-crown-6 ether; potassium carbonate at 170℃; | 61 % Chromat. |
| With Mycobacterium smegmatis acyl transferase In aq. phosphate buffer at 21℃; for 1h; pH=7.5; Inert atmosphere; Enzymatic reaction; |
-
-
141-78-6
ethyl acetate

-
-
77405-59-5
(11E,13E)-(4R,5S,6S,7R,9R,16R)-4,6-Dihydroxy-7-(2-hydroxy-ethyl)-5-methoxy-9,16-dimethyl-oxacyclohexadeca-11,13-diene-2,10-dione

-
-
63838-05-1
Acetic acid (7E,9E)-(3aR,5R,12R,16R,17S,17aS)-17-methoxy-5,12-dimethyl-2,6,14-trioxo-2,3,3a,5,6,11,12,14,15,16,17,17a-dodecahydro-4H-1,13-dioxa-cyclopentacyclohexadecen-16-yl ester

| Conditions | Yield |
|---|---|
| With oxygen; platinum at 25℃; | 100% |
-
-
141-78-6
ethyl acetate

| Conditions | Yield |
|---|---|
| With boron trichloride at -78 - 0℃; | 100% |
-
-
141-78-6
ethyl acetate

| Conditions | Yield |
|---|---|
| Stage #1: ethyl acetate; methyl 1-methyl-2-oxo-1,2-dihydroquinoline-3-carboxylate With sodium methylate for 7h; Heating; Stage #2: With hydrogenchloride In ethyl acetate for 3h; Heating; | 100% |

| Conditions | Yield |
|---|---|
| With dilithium tetra(tert-butyl)zincate at 0℃; for 1h; Inert atmosphere; | 100% |
| With 1,3-dicyclohexylimidazol-2-ylidene at 25℃; for 0.5h; | 68% |
| phosphotungstic acid at 65 - 70℃; for 2h; | 24% |
-
-
141-78-6
ethyl acetate

-
-
157764-10-8
2‐[3‐chloro‐5‐(trifluoromethyl)pyridin‐2‐yl]acetonitrile

-
-
744209-92-5
2-(3-chloro-5-trifluoromethyl-pyridin-2-yl)-3-oxo-butyronitrile

| Conditions | Yield |
|---|---|
| With sodium hydride In tetrahydrofuran | 100% |
-
-
58313-23-8
3-iodobenzoic acid methyl ester

-
-
141-78-6
ethyl acetate

-
-
68332-33-2
3-(3-iodophenyl)-3-oxopropionic acid ethyl ester

| Conditions | Yield |
|---|---|
| With potassium tert-butylate In tetrahydrofuran at 0 - 20℃; for 2.25h; | 100% |
-
-
233745-94-3
[3R,4S]-1-(t-Butyloxycarbonyl)-3-ethenyl-4-piperidine Acetic Acid

-
-
141-78-6
ethyl acetate

| Conditions | Yield |
|---|---|
| With thionyl chloride In methanol | 100% |
-
-
52341-13-6, 12264-20-9
trans-dichloro(ethylene)(2,4,6-trimethylpyridine)platinum

-
-
141-78-6
ethyl acetate

-
-
91068-20-1
trans-dichloro(ethyl acetate)(2,4,6-trimethylpyridine)platinum(II)

| Conditions | Yield |
|---|---|
| In ethyl acetate byproducts: ethylene; Irradiation (UV/VIS); | 100% |
| In ethyl acetate Irradiation (UV/VIS); the Pt-complex dissolved in ethyl acetate was introduced into a muffshaped Schlenk tube surrounding a 125-W medium-pressure mercury lamp, Philips HPK 125, irradn. for 15 min at room temp., λ<310 nm was eliminated by Pyrex filter; the solvent was removed under reduced pressure at -30°C, the solid was recrystd. at -30°C in pentane-CH2Cl2; | 85% |

| Conditions | Yield |
|---|---|
| In diethyl ether; N,N-dimethyl acetamide; ethyl acetate (Ar); vapor diffusion of Et2O into a (CH3)2NCOCH3/EtOAc soln. of Co complex; | 100% |
| In N,N-dimethyl acetamide; ethyl acetate (Ar); stirred for 2 h at room temp.; |
-
-
838855-60-0
1,1-dimethylethyl 4-({[4-(cyanomethyl)-2-(methyloxy)phenyl]oxy}methyl)piperidine-1-carboxylate

-
-
141-78-6
ethyl acetate

| Conditions | Yield |
|---|---|
| With sodium hydride In tetrahydrofuran at 60℃; for 15h; | 100% |

| Conditions | Yield |
|---|---|
| Stage #1: ethyl acetate With lithium diisopropyl amide In tetrahydrofuran; hexane at -78℃; for 0.333333h; Stage #2: (Z)-4-heptenal In tetrahydrofuran; hexane at -78℃; for 2h; | 100% |

| Conditions | Yield |
|---|---|
| Stage #1: ethyl acetate With lithium diisopropyl amide In tetrahydrofuran at -78℃; for 1h; Inert atmosphere; Stage #2: ethyl 2-fluoroacetate In tetrahydrofuran at -78 - 20℃; Inert atmosphere; | 100% |
| Stage #1: ethyl acetate With lithium diisopropyl amide In tetrahydrofuran; diethyl ether; cyclohexane; ethylbenzene at -78℃; for 2h; Cooling with acetone-dry ice; Stage #2: ethyl 2-fluoroacetate In tetrahydrofuran; diethyl ether; cyclohexane; ethylbenzene at -78 - 20℃; | 86% |
| Stage #1: ethyl acetate With sodium ethanolate at 0 - 5℃; for 1h; Stage #2: ethyl 2-fluoroacetate at 0 - 65℃; for 25h; | 65.6% |
| Stage #1: ethyl acetate With lithium diisopropyl amide In diethyl ether at -78℃; for 0.75h; Stage #2: ethyl 2-fluoroacetate In diethyl ether at -78 - 20℃; for 16h; | |
| Stage #1: ethyl acetate With lithium diisopropyl amide In tetrahydrofuran; n-heptane; ethylbenzene at -78℃; for 0.5h; Inert atmosphere; Stage #2: ethyl 2-fluoroacetate In tetrahydrofuran; n-heptane; ethylbenzene at -78 - 20℃; Inert atmosphere; |
-
-
15529-49-4, 41756-81-4
tris(triphenylphosphine)ruthenium(II) chloride

-
-
141-78-6
ethyl acetate

| Conditions | Yield |
|---|---|
| In toluene under Ar or N2; soln. of Ru compd. (0.156 mmol) and Ni compd. (0.303 mmol) in toluene heated at reflux for 4 h; cooling; addn. of small amt. of silica; evapn. under vac.; column chromy. (silica, CH2Cl2/light petroleum (1/4 and 3/7)); recrystn. from ethyl acetate and hydrocarbons (pentane, hexane, heptane); elem. anal.; | 100% |

-
-
1049734-37-3
3,4,5-trimethoxyphenylbiguanide hydrochloride

-
-
141-78-6
ethyl acetate

-
-
1232144-64-7
2-amino-4-(3',4',5'-trimethoxyphenyl)amino-6-methyl-1,3,5-triazine

| Conditions | Yield |
|---|---|
| With sodium methylate In tetrahydrofuran at 70℃; for 0.333333h; Microwave irradiation; | 100% |
-
-
120743-99-9
p-[(tert-butyldimethylsilyl)oxy]benzaldehyde

-
-
141-78-6
ethyl acetate

-
-
221079-70-5
ethyl 3-(4-{[tert-butyl(dimethyl)silyl]oxy}phenyl)-3-hydroxypropionate

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at -78℃; for 1.5h; Inert atmosphere; | 100% |
| Stage #1: ethyl acetate With lithium hexamethyldisilazane In tetrahydrofuran; hexane at -78℃; for 0.333333h; Inert atmosphere; Stage #2: p-[(tert-butyldimethylsilyl)oxy]benzaldehyde In tetrahydrofuran; hexane at -78℃; for 0.5h; Inert atmosphere; | 100% |
-
-
141-78-6
ethyl acetate

-
-
274-07-7
benzo[1,3,2]dioxaborole

-
-
72035-40-6
2-ethoxy-o-phenylene-1,3,2-dioxaborolane

| Conditions | Yield |
|---|---|
| [Mo(H)(Cl)(P(CH3)3)3(N(C6H3(CH3CHCH3)2))] In benzene-d6 react. of HBCat with 0.4 M soln. of MeC(O)OEt (2:1 molar ratio) in C6D6 at temp. of 22°C for 24 h in presence of 5 mol% of Mo complex as catalyst; detd. by NMR; | 100% |
| With tetramethylsilane; (2,6-diisopropylphenyl-N=)Mo(H)(Cl)(PMe3)3 In benzene-d6 at 22℃; for 24h; Inert atmosphere; | |
| With (2,6-diisopropylphenyl-N=)Mo(H)(Cl)(PMe3)3 In benzene-d6 at 22℃; for 2h; Catalytic behavior; Reagent/catalyst; Time; Inert atmosphere; | 100 %Spectr. |
Related products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View

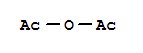

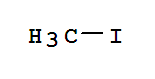












 F,
F, Xi
Xi