Hebei yanxi chemical co.,LTD.
Hebei yanxi chemical co. LTD. has expanded a compositive entity from initially only as a small manufacturer. The company dedicated to the development, production and marketing of chemicals. After many years of efforts, we have established stable
Ality Chemical Corporation
The above product is Ality Chemical's strong item with best price, good quality and fast supply. Ality Chemical has been focusing on the research and production of this field for over 14 years. At the same time, we are always committed to providi
Henan Allgreen Chemical Co.,Ltd
high quality Storage:Sealed, dry, microtherm , avoid light and smell. Package:According to the demand of customer Application:Organic synthesis Transportation:by air or by sea Port:shanghai
Chemwill Asia Co., Ltd.
Our main production base is located in Xuzhou industry park. We are certified both to the ISO 9001 and ISO 14001 Standards, have a safety management system in place.Our R&D team masters core technology for process-design of target building block
Changchun Artel lmport and Export trade company
Superiority We can customize and synthesize products that other suppliers may not be able to provide. Advantage cof, mof ligand manufacturer Product Detail
Cas:74-85-1
Min.Order:100 Kilogram
Negotiable
Type:Trading Company
inquiryHangzhou Keyingchem Co.,Ltd
Hangzhou KeyingChem Co., Ltd. exported this product to many countries and regions at best price. If you are looking for the material’s manufacturer or supplier in China, KeyingChem is your best choice. Pls contact with us freely for getting det
Zibo Hangyu Biotechnology Development Co., Ltd
Zibo Hangyu Biotechnology Development Co., Ltd is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality pharmaceuticals, intermediates, special chemicals and OLED intermediates and other fine chemi
HANGZHOU YUNUO CHEMICAL CO.,LTD
Superior quality, moderate price & quick delivery. Appearance:Colourless gas Storage:Stored in cool, dry and ventilation place; Away from fire and heat Package:1kg/bag, 1kg/drum or 25kg/drum or as per your request. Application:A catalyst for pl
Henan Tianfu Chemical Co., Ltd.
ETHYLENE Chemical Properties Melting point −169 °C(lit.) Boiling point −104 °C(lit.) density 0.00126 vapor density 0.97 (vs air) vapor pressure 35.04 atm ( 20 °C) refractive in
Cas:74-85-1
Min.Order:1 Metric Ton
FOB Price: $20.0
Type:Lab/Research institutions
inquiryZibo Dorne chemical technology co. LTD
Product Details Grade: pharmaceutical grade Purity:99%+ ProductionCapacity: 1000 Kilogram/Month Scope of use: For scientific research only(The product must be used legally) Our Advantage 1. Best quality with competitive price. 2. Quick shipping,
Cas:74-85-1
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryHangzhou Dingyan Chem Co., Ltd
R & D enterprises have their own stock in stock Package:1kg Application:pharmaceutical intermediates
Zhuozhou Wenxi import and Export Co., Ltd
Product Description Description & Specification Category Pharmaceutical Raw Materials, Fine Chemicals, Bulk drug Standard Medical standard
Cas:74-85-1
Min.Order:1 Kilogram
FOB Price: $112.0
Type:Trading Company
inquiryAntimex Chemical Limied
Ansciep Chemical is a professional enterprise manufacturing and distributing fine chemicals and speciality chemicals. We have been dedicated to heterocycle compounds and phenyl rings for tens of years. This is our mature product for export. Our quali
Hunan Longxianng Runhui Trading Co.,Ltd
74-85-1 ETHYLENEAppearance:powder Storage:room tempurature Package:As required Application:medical Transportation:By express (Door to door) such as FEDEX, DHL, EMS for small amount. By air(airport to airport) or by sea LCL/FCL for large amount.
Qingdao Sigma Chemical Co., Ltd.
Qingdao Sigma Chemical Ltd is is a global chemical industry manufacturers and suppliers of pharmaceuticals and intermediates, peptide,Nootropis etc API, food and feed additives, herbal extracts, agrochemicals and fine chemicals etc. Our Labo
HENAN SUNLAKE ENTERPRISE CORPORATION
ETHYLENE Basic information Product Name: ETHYLENE Synonyms: ETHYLENE;Acetene;Athylen;bicarburettedhydrogen;Bicarburretted hydrogen;bicarburrettedhydrogen;C2H4;E
HuBei Ipure Biotech CO.,ltd
HuBei ipure is a diversified product production and operation enterprise, with API, pharma intermediates, and other fine chemicals as well as R&D and pigments development and sales as one of the large enterprises, with more than 130 acres of pl
Cas:74-85-1
Min.Order:1 Gram
FOB Price: $1.0
Type:Trading Company
inquiryHenan Fine Chemicals Co., Ltd
Best quality & Attractive price & Professional service; Trial & Pilot & CommercialHenan Fine Chemicals Co., LTD. is a diversified technology - oriented integrated company, which mainly concentrates on fine chemicals. Our company is committed to becom
Chungking Joyinchem Co., Ltd.
Joyinchem have been committed to chemical supply for several years and have built good cooperation records with multinational chemical corporations and importers from all over the world. Our services include:-Spot goods-Contract manufacturing-Custom
Shanghai AngewChem Co., Ltd.
Shanghai AngewChemCo., Ltd. is an innovative enterprise on fine chemicals and pharmaceuticals. Based on Shanghai R&D center and Hunan chemical manufacturing plant, we offer chemical research, process development, and large-scale production. Complete
Hebei mingxing biotech co.,ltd
Known for its best quality and competitve price, this chemicals we offered is widely appreciated by our customers. Prompt reaction, good quality and best service make us reliable and outstanding in this industry.Appearance:yellow to white solid or po
Hebei Minshang Biotechnology Co., Ltd
74-85-1 ETHYLENE Application:medical
Nanjing Raymon Biotech Co., Ltd.
Ethylene Storage:Store in dry and cool condition Package:25kg or according to cutomer's demand Application:Chemical research/Pharmaceutical intermediates Transportation:By Sea,by Air,By courier like DHL or Fedx. Port:Shanghai/Shenzhen
Shochem(Shanghai) Co., Ltd
good quality and best price Application:intermediates
Wuxi Morality Chemical Co., Ltd
Do best quality products, erect the morality model Application:Please email us, thanks
Synova( Tianjin ) Chemical Technology Co,.Ltd
The company have effective management team, professional technical R & D personnel, the service spirit of customer oriented. We have long-term cooperation with famous domestic manufacturer, and excellent customer resources overseas. We are skilled in
Shanghai Yanchu chemicals Co., Ltd
low price and good quality Application:It is an important raw material and intermediate used in Organic Synthesis, Pharmaceuticals, Agrochemicals and D
Synthetic route

| Conditions | Yield |
|---|---|
| With mesoporous silica MCM-4l/Al at 399.84℃; under 21.0021 - 94.5095 Torr; for 50h; Inert N2; | 100% |
| With H-USY zeolite at 299.84℃; under 760.051 Torr; for 1.5h; Catalytic behavior; Reagent/catalyst; Time; Temperature; Inert atmosphere; Flow reactor; | 100% |
| With water at 380℃; under 1500.15 Torr; Reagent/catalyst; | 99% |

| Conditions | Yield |
|---|---|
| In dichloromethane for 1h; Yields of byproduct given; | A n/a B 100% |


| Conditions | Yield |
|---|---|
| In dichloromethane for 1h; Yields of byproduct given; | A n/a B 100% |

-
-
3968-30-7
5-cyclopentylidene-2,2-dimethyl-1,3-dioxane-4,6-dione

-
A
-
2873-50-9
butatriene

-
B
-
74-85-1
ethene

-
C
-
124-38-9
carbon dioxide

-
D
-
1165952-91-9
cyclohexa-1,3-diene

-
E
-
67-64-1
acetone

-
F
-
71-43-2
benzene

| Conditions | Yield |
|---|---|
| With variation of temp. at 550℃; Product distribution; | A 4% B 11.9% C 100% D 39.2% E 101.9 % F 3.3% |


| Conditions | Yield |
|---|---|
| With tungsten In octane; chlorobenzene at 80℃; for 1h; | A 100% B n/a |
| With chloroaryloxide neopentylidene complex of tungsten (1) | A 90% B n/a |
| (2,6-Ph2C6H3O)2W(Cl)=CHC(CH3)3*OEt2 In chlorobenzene at 80℃; for 3h; | A 88% B n/a |

| Conditions | Yield |
|---|---|
| With bis(1,5-cyclooctadiene)nickel (0); triphenylphosphine at 54℃; for 20h; Product distribution; Rate constant; Thermodynamic data; other solvents, reagents, reagents ratio, time, temperature; activation energy, ΔH<*>, ΔS<*>; | A 100% B n/a C 100% |


| Conditions | Yield |
|---|---|
| With trifluoroacetic acid at 120℃; | 100% |

| Conditions | Yield |
|---|---|
| With tungsten In octane; chlorobenzene at 80℃; for 2h; | A 100% B n/a |
| With Grubbs catalyst first generation In dichloromethane |
-
-
83044-85-3
3-allylsulfanyl-2-methyl-propene

-
A
-
42855-50-5
3-methyl-2,5-dihydro-thiophene

-
B
-
74-85-1
ethene

| Conditions | Yield |
|---|---|
| With tungsten In octane; chlorobenzene at 80℃; for 1h; | A 100% B n/a |
-
-
60-12-8
2-phenylethanol

-
-
754-05-2
ethenyltrimethylsilane

-
A
-
74-85-1
ethene

-
B
-
14629-58-4
trimethyl(phenethyloxy)silane

| Conditions | Yield |
|---|---|
| hydrogenchloride; chlorobis(ethylene)rhodium(I) dimer In 1,4-dioxane; chloroform at 20℃; for 2h; Product distribution / selectivity; | A n/a B 100% |
| chlorobis(cyclooctene)rhodium(I) dimer In toluene at 70℃; for 3h; Product distribution / selectivity; | A n/a B 100% |
| hydrogenchloride; chlorobis(cyclooctene)rhodium(I) dimer In 1,4-dioxane; chloroform at 20℃; for 2h; Product distribution / selectivity; | A n/a B 96% |

-
-
64-17-5
ethanol

-
-
1125-26-4
dimethylphenylvinylsilane

-
A
-
74-85-1
ethene

-
B
-
1825-58-7
dimethyl(ethoxy)phenylsilane

| Conditions | Yield |
|---|---|
| hydrogenchloride; chlorobis(cyclooctene)rhodium(I) dimer In 1,4-dioxane; chloroform at 20℃; for 18h; Kinetics; | A n/a B 100% |

-
-
67-63-0
isopropyl alcohol

-
-
1125-26-4
dimethylphenylvinylsilane

-
A
-
74-85-1
ethene

-
B
-
17988-21-5
dimethyl(isopropoxy)phenylsilane

| Conditions | Yield |
|---|---|
| hydrogenchloride; chlorobis(cyclooctene)rhodium(I) dimer In 1,4-dioxane; chloroform at 20℃; for 24h; Kinetics; | A n/a B 100% |


| Conditions | Yield |
|---|---|
| With oxygen In acetonitrile byproducts: {Co(2,2'-bipyridine)2}(2+); one-electron oxidn. of cis-Co complex by (Fe(phen)3(3+) in presence of O2 at 298 K; monitored by (1)H-NMR; | A <1 B <1 C 100% |
| In acetonitrile byproducts: {Co(2,2'-bipyridine)2}(2+); one-electron oxidn. of cis-Co complex by (Fe(phen)3(3+) at 298 K; monitored by (1)H-NMR; | A <1 B <1 C 98% |


| Conditions | Yield |
|---|---|
| In acetonitrile byproducts: {Co(2,2'-bipyridine)2}(2+); one-electron oxidn. of cis-Co complex by (Fe(bpy)3(3+) at 298 K; monitored by (1)H-NMR; | A <1 B <1 C 100% |

-
-
75847-39-1, 76189-28-1
diethylbis(triethylphosphine)platinum(II)

-
A
-
76136-93-1
(ethylene)bis(triethylphosphine)platinum(0)

-
B
-
74-85-1
ethene

| Conditions | Yield |
|---|---|
| In cyclohexane Pt-complex evacuated to 0.1 torr, flushed with Ar, dry degassed cyclohexane added, cooled (liq. N2), evacuated (0.1 torr), sealed, warmed to room temp., heated at 118°C (oil bath) for 30 min; | A 100% B 0% |
-
-
1295-35-8
bis(1,5-cyclooctadiene)nickel (0)

-
-
15133-82-1
tetrakis(triphenylphosphine)nickel(0)

-
B
-
15376-83-7
(triphenylphosphine)3(CO)nickelk

-
C
-
74-85-1
ethene

-
D
-
108-95-2
phenol

| Conditions | Yield |
|---|---|
| With triphenylphosphine In neat (no solvent) (N2 or Ar or vac.), EtCOOPh added to Ni(cod)2 and Ni(PPh3)4, mixture stirred at 54°C for 20 h; GLC; | A 73% B n/a C 100% D 91% |

-
-
1295-35-8
bis(1,5-cyclooctadiene)nickel (0)

-
-
637-27-4
phenyl propionate

-
A
-
15376-83-7
(triphenylphosphine)3(CO)nickelk

-
B
-
74-85-1
ethene

-
C
-
108-95-2
phenol

| Conditions | Yield |
|---|---|
| With triphenylphosphine In neat (no solvent) Kinetics; byproducts: diethyl ketone, 1,3-cyclooctadiene, 1,4-cyclooctadiene; further byproducts: 1,5-cyclooctadiene, bicyclooctene-1, bicyclooctene-2, (N2 or Ar or vac.), EtCOOPh added to Ni(cod)2 and PPh3 (PPh3/Ni = 10), mixture stirred at 54°C for 21 h; gas chromy., volatile removed in vac., recrystd. from THF-hexane; | A 80% B 100% C 100% |
| With triphenylphosphine In neat (no solvent) Kinetics; byproducts: diethyl ketone, 1,3-cyclooctadiene, 1,4-cyclooctadiene; further byproducts: 1,5-cyclooctadiene, bicyclooctene-1, bicyclooctene-2, (N2 or Ar or vac.), EtCOOPh added to Ni(cod)2 and PPh3 (PPh3/Ni = 4),mixture stirred at 54°C for 20 h; gas chromy., volatile removed in vac., recrystd. from THF-hexane; | A 60% B 100% C 100% |
| With triphenylphosphine In neat (no solvent) Kinetics; byproducts: diethyl ketone, 1,3-cyclooctadiene, 1,4-cyclooctadiene; further byproducts: 1,5-cyclooctadiene, bicyclooctene-1, bicyclooctene-2, (N2 or Ar or vac.), EtCOOPh added to Ni(cod)2 and PPh3 (PPh3/Ni = 3),mixture stirred at 54°C for 20 h; gas chromy., volatile removed in vac., recrystd. from THF-hexane; | A 60% B 90% C 90% |
| With triphenylphosphine In neat (no solvent) Kinetics; byproducts: diethyl ketone, 1,3-cyclooctadiene, 1,4-cyclooctadiene; further byproducts: 1,5-cyclooctadiene, bicyclooctene-1, bicyclooctene-2, (N2 or Ar or vac.), EtCOOPh added to Ni(cod)2 and PPh3 (PPh3/Ni = 2),mixture stirred at 54°C for 12 h; gas chromy., volatile removed in vac., recrystd. from THF-hexane; | A 30% B 60% C 60% |
| With triphenylphosphine In neat (no solvent) Kinetics; byproducts: diethyl ketone, 1,3-cyclooctadiene, 1,4-cyclooctadiene; further byproducts: 1,5-cyclooctadiene, bicyclooctene-1, bicyclooctene-2, (N2 or Ar or vac.), EtCOOPh added to Ni(cod)2 and PPh3 (PPh3/Ni = 1),mixture stirred at 54°C for 12 h; gas chromy., volatile removed in vac., recrystd. from THF-hexane; | A n/a B 40% C 50% |

-
-
1295-35-8
bis(1,5-cyclooctadiene)nickel (0)

-
-
637-27-4
phenyl propionate

-
-
1038-95-5
Tri(p-tolyl)phosphine

-
A
-
74-85-1
ethene

-
B
-
74887-07-3
Ni(CO)(P(C6H4CH3)3)3

-
C
-
108-95-2
phenol

| Conditions | Yield |
|---|---|
| In further solvent(s) Kinetics; (N2 or Ar or vac.), EtCOOPh added to Ni(cod)2 and P(C6H4CH3)3 (P(Ph-CH3)3/Ni = 3) in acetophenone, mixture stirred at 65°C for 50 h; | A 80% B 85% C 100% |

-
-
50487-72-4
N,N-Diallyltosylamide

-
A
-
74-85-1
ethene

-
B
-
16851-72-2
1-[(4-methylphenyl)sulfonyl]-2,5-dihydro-1H-pyrrole

| Conditions | Yield |
|---|---|
| With Hoveyda-Grubbs catalyst second generation In dichloromethane at 40℃; for 1h; Concentration; Solvent; Temperature; Grubbs Olefin Metathesis; Flow reactor; | A n/a B 100% |
| With Hoveyda-Grubbs catalyst second generation In (2)H8-toluene at 29.84℃; Reagent/catalyst; |
-
-
822-38-8
1,3-dithiolane-2-thione

-
-
762-42-5
dimethyl acetylenedicarboxylate

-
A
-
7396-41-0
4,5-bis(methoxycarbonyl)-1,3-dithiole-2-thione

-
B
-
74-85-1
ethene

| Conditions | Yield |
|---|---|
| at 120 - 140℃; | A 99.6% B n/a |


| Conditions | Yield |
|---|---|
| With triethylamine In water at 20℃; for 2h; Inert atmosphere; Irradiation; | 99% |
| With vanadocene In hexane Product distribution; vanadocene monobromide, vanadocene monochloride; other temperature and reaction time.; | 62% |
| Electrolysis; |

| Conditions | Yield |
|---|---|
| In chlorobenzene for 24h; Heating; | A 99% B n/a |
-
-
85597-87-1
7-Aza-bicyclo[2.2.1]hept-2-ene-1,2,3,4-tetracarboxylic acid diisopropyl ester dimethyl ester

-
A
-
74-85-1
ethene

-
B
-
85597-95-1
1H-Pyrrole-2,3,4,5-tetracarboxylic acid diisopropyl ester dimethyl ester

| Conditions | Yield |
|---|---|
| at 90 - 120℃; for 3h; | A n/a B 99% |

| Conditions | Yield |
|---|---|
| With alumina at 449.84℃; Catalytic behavior; Reagent/catalyst; Temperature; Inert atmosphere; Overall yield = 100 %; | A 0.1% B 98.9% |
| C2I2O2Rh(1-)*C8H20N(1+); tetraethylammonium iodide; hydrogen iodide In water at 110℃; Product distribution / selectivity; Inert atmosphere; Autoclave; | A 10% B 50% |
| 1-methyl-3-(propyl-3-sulfonyl)imidazolium trifluoromethanesulfonate; CF3O3S(1-)*CHF3O3S*C7H13N2O3S(1+) at 240 - 260℃; for 4h; Product distribution / selectivity; | A n/a B 12% |
-
-
34557-54-5
methane

-
-
74-85-1
ethene

| Conditions | Yield |
|---|---|
| under 760.051 Torr; Recess waveguide; Gas phase; | 98.53% |
| With hydrogen under 760.051 Torr; Product distribution / selectivity; Microwave irradiation; | 96.4% |
| Stage #1: methane Stage #2: With alumina at 125℃; Reagent/catalyst; Temperature; | 55% |

| Conditions | Yield |
|---|---|
| With oxygen; V-Mo-Nb-Te oxide | 98% |
| at 800℃; under 760.051 Torr; for 1h; Catalytic behavior; Gas phase; Flow reactor; | 84% |
| With disulfur; iron(II,III) oxide at 940℃; Reagent/catalyst; Temperature; Flow reactor; | 75.9% |

| Conditions | Yield |
|---|---|
| With iron sulfide at 800 - 900℃; other metal sulfides; | A 2% B 98% |
| With SAPO-34/HZSM-5 nanostructure In water at 370℃; for 20h; Reagent/catalyst; Inert atmosphere; |
-
-
85597-84-8
7-Aza-bicyclo[2.2.1]hept-2-ene-1,2,3,4-tetracarboxylic acid tetramethyl ester

-
A
-
74-85-1
ethene

-
B
-
2703-15-3
tetramethyl 1H-pyrrole-2,3,4,5-tetracarboxylate

| Conditions | Yield |
|---|---|
| at 90 - 120℃; for 3h; | A n/a B 98% |
-
-
85597-90-6
7-Aza-bicyclo[2.2.1]hept-2-ene-1,2,3,4-tetracarboxylic acid di-tert-butyl ester dimethyl ester

-
A
-
74-85-1
ethene

-
B
-
85597-98-4
1H-Pyrrole-2,3,4,5-tetracarboxylic acid di-tert-butyl ester dimethyl ester

| Conditions | Yield |
|---|---|
| at 90 - 120℃; for 3h; | A n/a B 98% |
-
-
85597-92-8
7-Aza-bicyclo[2.2.1]hept-2-ene-1,2,3,4-tetracarboxylic acid dicyclohexyl ester dimethyl ester

-
A
-
74-85-1
ethene

-
B
-
85598-00-1
1H-Pyrrole-2,3,4,5-tetracarboxylic acid dicyclohexyl ester dimethyl ester

| Conditions | Yield |
|---|---|
| at 90 - 120℃; for 3h; | A n/a B 98% |

| Conditions | Yield |
|---|---|
| With trimethylamine-N-oxide; [N(4-C6H4Br)3][B(C6F5)4]; triethylaluminum; [Cr(CO)4(2-C6H4(MeO))2PN(Me)P(2-C6H4(MeO))2] In toluene at 60℃; under 30002.4 Torr; for 1h; | 100% |
| In chlorobenzene at 45℃; under 36201.3 Torr; for 4h; Product distribution / selectivity; | 99% |
| In chlorobenzene at 60℃; under 36201.3 Torr; for 4h; Product distribution / selectivity; | 99% |

| Conditions | Yield |
|---|---|
| With hydrogen; [Ru2(μ-O2C-C6H4-CO2)2] at 20℃; for 5.4h; Kinetics; Product distribution; Further Variations:; Catalysts; reaction times; | 100% |
| With [1,1-(1,3-dimethylimidazol-2-ylidene)(PPh3)-3-(Py)-1,2-RhSB9H8]; hydrogen In dichloromethane-d2 under 3750.38 Torr; for 12h; Catalytic behavior; Time; Inert atmosphere; | 73% |
| With hydrogen; palladium | 25% |
-
-
74-85-1
ethene

-
-
106471-36-7
bis(2-chloroethyl)selenium dichloride

| Conditions | Yield |
|---|---|
| With selenium tetrachloride In benzene | 100% |
| With tetrachlorosilane; chloroform | |
| With diselenium dichloride; benzene |

| Conditions | Yield |
|---|---|
| With bis(benzonitrile)palladium(II) dichloride; py.Co(N,N'-bis(alicylidene-o-phenylene)diamino).NO2 In tetrahydrofuran at 50℃; under 760 Torr; for 0.916667h; | 100% |
| With bis(benzonitrile)palladium(II) dichloride; py.Co(N,N'-bis(alicylidene-o-phenylene)diamino).NO2 In tetrahydrofuran at 50℃; under 760 Torr; for 0.916667h; Product distribution; influence of Pd/Co ratio, solvent, further olefins; | 100% |
| With aluminum(III) sulfate; water at 350 - 360℃; |

| Conditions | Yield |
|---|---|
| With silver trifluoromethanesulfonate; triphenylphosphine; bi(allylnickel bromide) In dichloromethane at -55℃; under 760 Torr; for 2h; | 100% |
| With bi(allylnickel bromide); silver trifluoromethanesulfonate; triphenylphosphine In dichloromethane at -55℃; under 760 Torr; for 2h; Addition; Hydrovinylation; | 95% |
| With 2; diethylaluminium chloride; triphenylphosphine In dichloromethane; toluene under 7500.6 Torr; for 0.5h; | 90% |

| Conditions | Yield |
|---|---|
| Irradiation (UV/VIS); 44 h; | 100% |
| Irradiation (UV/VIS); 44 h; | 100% |
| Irradiation; |
-
-
74-85-1
ethene

-
-
106726-64-1
1,4-Dithia-7-azanorbornylium hexafluoroarsenate

| Conditions | Yield |
|---|---|
| With dithionitronium hexafluoroarsenate In liquid sulphur dioxide Ambient temperature; | 100% |
| With dithionitronium hexafluoroarsenate In liquid sulphur dioxide for 1h; Ambient temperature; | 100% |

| Conditions | Yield |
|---|---|
| di(rhodium)tetracarbonyl dichloride In ethanol at 115℃; under 37503 Torr; for 1.5h; | 100% |

-
-
74-85-1
ethene

-
-
10368-35-1
4-ethoxyphenylacetic acid chloride

-
-
69788-78-9
6-ethoxy-3,4-dihydro-2(1H)-naphthalenone

| Conditions | Yield |
|---|---|
| With aluminium trichloride In dichloromethane for 4h; Ambient temperature; | 100% |
-
-
74-85-1
ethene

| Conditions | Yield |
|---|---|
| With triphenylphosphine; bis(1,5-cyclooctadiene)nickel (0) In toluene at 25℃; under 22502.3 Torr; for 70h; | 100% |
| With tetramethyldialuminoxane; N,N'-(1,1-Me2-ethylene)bis(salicylideneaminato)Zr(IV)Cl2*THF In toluene at 25℃; under 7500.6 Torr; for 24h; Polymerization; | |
| Pd-2 at 22℃; under 4137.18 Torr; for 12h; Polymerization; |
-
-
74-85-1
ethene

-
-
99520-55-5
3-acetoxy-3-(4-methoxyphenyl)prop-1-yne

| Conditions | Yield |
|---|---|
| With RuCl2(P(C6H11)3)(1,3-dimesityl-4,5-dihydroimidazol-2-ylidene)(=CHC6H5) In toluene at 80℃; under 760 Torr; for 0.5h; | 100% |
| With tricyclohexylphosphine[1,3-bis(2,4,6-trimethylphenyl)-4,5-dihydroimidazol-2-ylidine][benzylidene]ruthenium(II) dichloride In toluene at 80℃; under 760.051 Torr; for 0.5h; | 100% |
-
-
74-85-1
ethene

| Conditions | Yield |
|---|---|
| With RuCl2(P(C6H11)3)(1,3-dimesityl-4,5-dihydroimidazol-2-ylidene)(=CHC6H5) In toluene at 80℃; under 760 Torr; for 0.5h; | 100% |
| With tricyclohexylphosphine[1,3-bis(2,4,6-trimethylphenyl)-4,5-dihydroimidazol-2-ylidine][benzylidene]ruthenium(II) dichloride In toluene at 80℃; under 760.051 Torr; for 2h; | 94% |
-
-
74-85-1
ethene

-
-
441741-15-7
(1S,4R,5S,6R)-5,6-Bis-prop-2-ynyloxy-bicyclo[2.2.1]hept-2-ene

-
-
441741-16-8
(4aS,4bR,8aS,9aR)-2,7-Divinyl-4a,4b,6,8a,9,9a-hexahydro-3H-4,5-dioxa-fluorene

| Conditions | Yield |
|---|---|
| Grubbs catalyst first generation In dichloromethane at 20℃; for 4h; | 100% |
| Grubbs catalyst first generation In dichloromethane at 20℃; for 4h; | 53% |
-
-
74-85-1
ethene

-
-
477977-14-3
N-(cyclopentenylmethyl)-4-methyl-N-(prop-2-ynyl)benzenesulfonamide

| Conditions | Yield |
|---|---|
| Cl2(PCy3)(N,N'-(Mes)2-imidazolidin-2-yl)Ru=CHC6H5 In dichloromethane under 760.051 Torr; for 26h; Heating; | 100% |
-
-
74-85-1
ethene

-
-
780755-05-7
8,8-dimethyl-4-phenyl-6,10-dioxa-spiro[4.5]deca-1,3-diene-1,2-dicarboxylic acid dimethyl ester

| Conditions | Yield |
|---|---|
| In toluene at 100℃; for 192h; Diels-Alder reaction; ambient pressure; | 100% |

| Conditions | Yield |
|---|---|
| With Vazo64 | 100% |
| With triethyl phosphite | 71% |
| With hydrazine hydrate; copper (I) acetate In isopropyl alcohol at 30℃; under 22800 Torr; for 4h; | 69% |
| copper at 80℃; under 6000.6 - 7500.75 Torr; for 1.33333h; Product distribution / selectivity; | |
| With VAZO 64 at 65 - 81℃; under 4897.34 - 8931.21 Torr; for 3h; Industry scale; Autoclave; | 97.3 %Chromat. |

-
-
74-85-1
ethene

| Conditions | Yield |
|---|---|
| tricyclohexylphosphine[1,3-bis(2,4,6-trimethylphenyl)-4,5-dihydroimidazol-2-ylidine][benzylidene]ruthenium(II) dichloride In toluene at 80℃; | 100% |

-
-
74-85-1
ethene

| Conditions | Yield |
|---|---|
| tricyclohexylphosphine[1,3-bis(2,4,6-trimethylphenyl)-4,5-dihydroimidazol-2-ylidine][benzylidene]ruthenium(II) dichloride In toluene at 80℃; | 100% |

-
-
74-85-1
ethene

| Conditions | Yield |
|---|---|
| tricyclohexylphosphine[1,3-bis(2,4,6-trimethylphenyl)-4,5-dihydroimidazol-2-ylidine][benzylidene]ruthenium(II) dichloride In toluene at 80℃; | 100% |
Related products
Raw Materials
Downstream Products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View




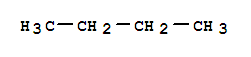


 F+
F+