Hunan Russell Chemicals Technology Co.,Ltd
high purity lowest priceAppearance:solid or liquid Storage:in sealed air resistant place Package:Foil bag; Drum; Plastic bottle Application:Pharma;Industry;Agricultural Transportation:by sea or air Port:any port in China
QINGDAO ON-BILLION INDUSTRAIL CO.,LTD
EINECSNO.202-626-1 FORMULAC5H6O2 MOLWT.98.10 H.S.CODE2932.13 TOXICITY Oral rat LD50: 177 mg/kg SYNONYMS 2-Furylmeth… Appearance:colorless to pale yellow liquid Storage:2-8°C Package:iso tank or 240kg drums Application:Furan polymers, in maki
Pure Chemistry Scientific Inc.
98-01-1 Application:Furfural
Synthetic route

| Conditions | Yield |
|---|---|
| With bis(2,2'-bipyridyl) copper(II) permanganate In acetone for 0.1h; Ambient temperature; | 100% |
| With 4 A molecular sieve; tetrabutylammonium perchlorate; Ru-Cu-Al-hydrotalcite In toluene at 60℃; for 3h; | 100% |
| With potassium carbonate In toluene at 70℃; for 2.5h; Reagent/catalyst; | 100% |

| Conditions | Yield |
|---|---|
| In diethyl ether for 1h; Product distribution; Irradiation; photochemical debromination was investigated; | 100% |

-
-
98-01-1
furfural

| Conditions | Yield |
|---|---|
| With hydrogenchloride for 0.0416667h; Product distribution; Ambient temperature; pH = 4-6, regeneration of aldehyde; | 100% |

| Conditions | Yield |
|---|---|
| With sulphated zirconia In acetonitrile at 60℃; for 2h; Microwave irradiation; | 100% |
| With Montmorillonite K10 In dichloromethane for 0.333333h; Heating; | 98% |
| With water; Sulfate; titanium(IV) oxide In dichloromethane for 0.0833333h; Deacetylation; Heating; | 97% |
-
-
67421-75-4
2-(1,3-dithian-2-yl)furan

-
-
98-01-1
furfural

| Conditions | Yield |
|---|---|
| With dihydrogen peroxide; iodine; sodium dodecyl-sulfate In water at 20℃; for 1h; Micellar solution; | 100% |
| With indium(III) trifluoride; water In acetonitrile for 3.5h; Reflux; chemoselective reaction; | 93% |
| With 2,4,4,6-Tetrabromo-2,5-cyclohexadien-1-one; dihydrogen peroxide In water; acetonitrile at 20℃; for 0.75h; | 90% |
| With eosin y In water; acetonitrile at 20℃; for 4h; Irradiation; | 82% |
| With ammonium iodide; dihydrogen peroxide; sodium dodecyl-sulfate In water at 20℃; for 0.333333h; micellar medium; |
-
-
18708-18-4
furfural tosylhydrazone

-
-
98-01-1
furfural

| Conditions | Yield |
|---|---|
| With Cr-MCM-41 zeolite on silica gel for 0.1h; microwave irradiation; | 98% |
| With benzeneseleninic anhydride In tetrahydrofuran at 40 - 50℃; for 2h; | 88% |
| With 2,3-dicyano-5,6-dichloro-p-benzoquinone In dichloromethane; water at 20℃; for 2h; Oxidation; oxidative cleavage; | 80% |

| Conditions | Yield |
|---|---|
| With sulfonated graphitic carbon nitride In water at 100℃; for 0.5h; Solvent; Temperature; | 96% |
| With Sulfonated graphene at 150℃; for 0.666667h; Temperature; Sealed tube; | 96% |
| With hydrogenchloride; 5-methyl-dihydro-furan-2-one In water at 224.84℃; under 28443.9 Torr; for 0.0375h; Kinetics; Temperature; Reagent/catalyst; Concentration; Flow reactor; | 93% |

| Conditions | Yield |
|---|---|
| With Montmorillonite K 10; water In acetone for 0.5h; Heating; | 96% |
| With aluminum oxide; Oxone for 0.03h; Hydrolysis; Microwave irradiation; | 93% |
| With iron(III) chloride hexahydrate; acetaldehyde In dichloromethane at 20℃; for 0.25h; | 90% |

| Conditions | Yield |
|---|---|
| With water; Dess-Martin periodane In dichloromethane at 5℃; for 0.333333h; | 95% |
| With silica gel; iron(III) chloride for 0.0133333h; microwave irradiation; | 92% |
| With bis(pyridine)silver(I) permanganate In dichloromethane for 0.0833333h; Ambient temperature; | 90% |
-
-
117680-17-8
2-(Iodomethyl)tetrahydrofuran

-
-
98-01-1
furfural

| Conditions | Yield |
|---|---|
| With oxygen; kieselguhr; copper(l) chloride In hexane for 2h; Oxidation; Heating; | 93% |

-
-
98-01-1
furfural

| Conditions | Yield |
|---|---|
| With aluminium trichloride; 1-benzyl-4-aza-1-azoniabicyclo[2.2.2]octane dichromate at 20℃; for 0.0125h; | 93% |
| With sodium perborate In acetic acid at 40℃; for 1h; Oxidation; |

| Conditions | Yield |
|---|---|
| Stage #1: 2-(1,3-dithiolan-2-yl)furan In ethanol at 20℃; Stage #2: With water In ethanol at 20℃; | 93% |
| With indium(III) trifluoride; water In acetonitrile for 3.5h; Reflux; chemoselective reaction; | 89% |
| With indium (III) iodide; dihydrogen peroxide In water; toluene at 20℃; for 15h; Inert atmosphere; Sealed tube; |
-
-
81932-19-6
2-(furan-2-yl)-1,3-oxathiolane

-
-
98-01-1
furfural

| Conditions | Yield |
|---|---|
| Stage #1: 2-(furan-2-yl)-1,3-oxathiolane In ethanol at 20℃; Stage #2: With water In ethanol at 20℃; | 93% |
| With copper(II) nitrate monohydrate at 90℃; for 0.333333h; | 85% |

| Conditions | Yield |
|---|---|
| With potassium carbonate In chloroform at 20℃; for 24h; Irradiation; Inert atmosphere; | 93% |
-
-
14757-78-9
3-bromofurfural

-
-
98-01-1
furfural

| Conditions | Yield |
|---|---|
| In diethyl ether for 1h; Product distribution; Irradiation; photochemical debromination was investigated; | 92% |
-
-
10323-20-3
D-Arabinose

-
-
98-01-1
furfural

| Conditions | Yield |
|---|---|
| With Dowex 50Wx8-100 ion-exchange resin at 100℃; for 6h; Ionic liquid; Sealed tube; | 92% |
| With silicoaluminophosphate-44 In water; toluene at 170℃; for 8h; | 63% |
| With 1-butyl-3-methylimidazolium tetrachloridoferrate(III) In water; butanone at 160℃; for 3h; | 50.7% |

| Conditions | Yield |
|---|---|
| With nickel-doped graphene carbon nitride nanoparticles; air In ethanol at 25℃; for 8h; Irradiation; Green chemistry; | 92% |
| With oxygen In acetonitrile at 20℃; for 18h; Irradiation; | 95 %Spectr. |
| With tert.-butylhydroperoxide; C29H25Cl2N4Ru(1+)*F6P(1-) In acetonitrile at 60℃; for 3h; Schlenk technique; Inert atmosphere; | 71 %Chromat. |

| Conditions | Yield |
|---|---|
| With 1-methyl-3-(4-sulfobutyl)-1H-imidazol-3-ium hydrogensulfate; 4-methyl-2-pentanone In water at 150℃; under 760.051 Torr; for 0.416667h; Autoclave; | 91.4% |

| Conditions | Yield |
|---|---|
| With D-Xylose In water at 180℃; for 0.666667h; Temperature; Reagent/catalyst; | 91.4% |

| Conditions | Yield |
|---|---|
| With phosphorus and fluorine co-doped amorphous carbon nitride In tetrahydrofuran; water at 130℃; for 5h; | 91% |
| With hydrogen In water at 150℃; under 750.075 Torr; Temperature; | 87.6% |
| With vanadyl pyrophosphate In water; toluene at 170℃; for 6h; | 56% |

| Conditions | Yield |
|---|---|
| With aluminum oxide; potassium permanganate In dichloromethane at 20℃; | 90% |
-
-
1027382-27-9
tert-butyldimethyl(2,2,2-trichloro-1-furan-2-ylethoxy)silane

-
-
98-01-1
furfural

| Conditions | Yield |
|---|---|
| With tetrabutyl ammonium fluoride In tetrahydrofuran; N,N-dimethyl-formamide at 50℃; | 90% |

| Conditions | Yield |
|---|---|
| With water; 4-acetylamino-2,2,6,6-tetramethylpiperidine-1-oxoammonium tetrafluoroborate In acetonitrile at 20℃; for 8h; | 90% |
-
-
71778-41-1
2-(bis(phenylthio)methyl)furan

-
-
98-01-1
furfural

| Conditions | Yield |
|---|---|
| With indium(III) trifluoride; water In acetonitrile for 3h; Reflux; chemoselective reaction; | 90% |

| Conditions | Yield |
|---|---|
| With Dowex 50Wx8-200 ion-exchange resin at 100℃; for 3h; Ionic liquid; Sealed tube; | 90% |
| With 3-butyl-1-methyl-1H-imidazol-3-ium hexafluorophosphate; copper dichloride at 120℃; for 0.3h; Reagent/catalyst; Ionic liquid; | 67 %Chromat. |

| Conditions | Yield |
|---|---|
| With indium(III) trifluoride; water In acetonitrile for 2.5h; Reflux; chemoselective reaction; | 89% |

| Conditions | Yield |
|---|---|
| With pentacoordinated hydrogenosilane 1 | 87% |
| With bis(triphenylphosphine)copper(I) tetrahydroborate; triphenylphosphine In acetone at 25℃; for 1h; | 78% |
| With tert-butyl isocyanide; CpRu(PiPr3)(CH3CN)2PF6; Dimethylphenylsilane In [(2)H6]acetone at 20℃; for 24h; chemoselective reaction; | 100 %Spectr. |
| With tert-butyl isocyanide; CpRu(PiPr3)(CH3CN)2PF6; Dimethylphenylsilane In [(2)H6]acetone at 20℃; for 24h; chemoselective reaction; | Ca. 100 %Spectr. |

| Conditions | Yield |
|---|---|
| With indium(III) trifluoride; water In acetonitrile for 2h; Reflux; chemoselective reaction; | 87% |
| indium(III) chloride In methanol; water for 2h; Heating; | 85% |

| Conditions | Yield |
|---|---|
| With 2,2,6,6-Tetramethyl-1-piperidinyloxy free radical; [bis(acetoxy)iodo]benzene In dichloromethane at 0 - 20℃; for 0.333333h; Inert atmosphere; Green chemistry; | 87% |
| With pyridoxal 5'-phosphate; sodium pyruvate In aq. phosphate buffer at 30℃; pH=7.5; Reagent/catalyst; Enzymatic reaction; |

| Conditions | Yield |
|---|---|
| With manganese triacetate In benzene for 1h; Heating; | 86% |
| With antimonypentachloride In dichloromethane at 20℃; for 2.3h; | 75% |

| Conditions | Yield |
|---|---|
| With sodium hydroxide In ethanol for 0.025h; microwave irradiation; | 100% |
| With sodium hydroxide In ethanol; water at 20℃; for 24h; Inert atmosphere; Green chemistry; | 99% |
| aluminum oxide for 0.0416667h; microwave irradiation; | 98% |

| Conditions | Yield |
|---|---|
| In methanol at 20℃; for 24h; | 100% |
| In methanol at 20℃; for 24h; | 100% |
| In methanol at 20℃; | 85% |

| Conditions | Yield |
|---|---|
| With sodium sulfate In benzene for 0.5h; Ambient temperature; | 100% |
| In methanol at 20℃; for 24h; | 100% |
| In methanol at 20℃; for 24h; | 100% |

| Conditions | Yield |
|---|---|
| With copper(II) bis(trifluoromethanesulfonate) In water at 20℃; for 0.0166667h; | 100% |
| With aluminum oxide for 5h; Milling; | 100% |
| sodium hydrogen sulfate; silica gel at 56 - 58℃; for 0.0244444h; microwave irradiation; | 98% |
-
-
98-01-1
furfural

-
-
762-04-9
phosphonic acid diethyl ester

-
-
20627-09-2
diethyl (hydroxy(furan-2-yl)methyl)phosphonate

| Conditions | Yield |
|---|---|
| With triethylamine In neat (no solvent) at 20℃; Pudovik Reaction; Inert atmosphere; | 100% |
| With triethylamine at 50℃; Pudovik Reaction; Inert atmosphere; Sealed tube; | 100% |
| With 2-tert-butylimino-2-diethylamino-1,3-dimethylperhydro-1,3,2-diazaphosphorine supported on polystyrene In neat (no solvent) at 30℃; for 3h; Pudovik Reaction; Green chemistry; | 99% |
-
-
98-01-1
furfural

-
-
105-56-6
ethyl 2-cyanoacetate

-
-
67449-75-6, 23973-22-0
ethyl (E)-2-cyano-3-(2-furyl)-2-propenoate

| Conditions | Yield |
|---|---|
| With L-proline for 0.0333333h; Knoevenagel condensation; microwave irradiation; | 100% |
| ytterbium(III) perfluorooctanesulfonate In toluene at 80℃; for 3h; Knoevenagel condensation; | 99% |
| With polyacrylonitrile fiber modified with triethylenetetramine In water at 50℃; for 1.5h; Knoevenagel condensation; | 99% |
-
-
98-01-1
furfural

-
-
91-59-8
naphthalen-2-ylamine

-
-
6233-18-7
(E)-1-(furan-2-yl)-N-(naphthalen-2-yl)methanimine

| Conditions | Yield |
|---|---|
| In diethyl ether at 20℃; Inert atmosphere; Molecular sieve; Darkness; | 100% |

| Conditions | Yield |
|---|---|
| With piperidine; acetic acid In dichloromethane at 0 - 20℃; Michael Addition; Molecular sieve; | 100% |
| With L-Lysine hydrochloride; triethylamine In N,N-dimethyl-formamide at 20℃; for 3h; Knoevenagel Condensation; | 98.3% |
| With cross-linked polystyrene-titanium tetrachloride complex In neat (no solvent) at 60℃; for 2h; Knoevenagel Condensation; | 98% |

| Conditions | Yield |
|---|---|
| With copper(II) bis(trifluoromethanesulfonate) In water at 20℃; for 0.0166667h; | 100% |
| In water at 20℃; for 2h; | 93% |
| In dichloromethane Inert atmosphere; Molecular sieve; | 81% |

| Conditions | Yield |
|---|---|
| With 1-butyl-1,4-diazabicyclo[2.2.2]octanylium hydrotetrafluoroborate In water at 20℃; for 0.0166667h; Knoevenagel condensation; | 100% |
| With 1,4-diaza-bicyclo[2.2.2]octane In water at 20℃; for 0.0166667h; Knoevenagel Condensation; Green chemistry; | 100% |
| With hydroquinone; p-benzoquinone In water at 20℃; for 3h; Reagent/catalyst; Knoevenagel Condensation; Inert atmosphere; Sealed tube; | 100% |

| Conditions | Yield |
|---|---|
| With Yb(III)-coordinated adamantane-based porous polymer In methanol at 20℃; for 12h; Catalytic behavior; Reagent/catalyst; | 100% |
| indium(III) triflate In dichloromethane at 20℃; for 0.0833333h; | 99% |
| With cerium triflate In methanol at 20℃; for 0.0333333h; | 99% |

| Conditions | Yield |
|---|---|
| With hydrogen In ethanol at 60℃; under 15001.5 Torr; for 4h; Catalytic behavior; Reagent/catalyst; Temperature; Solvent; Pressure; Autoclave; Green chemistry; | 100% |
| With hydrogen In butan-1-ol at 80℃; under 30003 Torr; for 5h; Catalytic behavior; Temperature; Reagent/catalyst; | 99% |
| With hydrogen In isopropyl alcohol at 179.84℃; under 22502.3 Torr; for 1.25h; Reagent/catalyst; | 99% |

| Conditions | Yield |
|---|---|
| With hydrogen under 2250.23 Torr; for 15h; | 100% |
| With hydrogen at 200℃; under 760.051 Torr; | 95.5% |
| With hydrogen at 120℃; under 760.051 Torr; for 24h; Catalytic behavior; Reagent/catalyst; Temperature; | 94.5% |

| Conditions | Yield |
|---|---|
| With Pt(3)Co(3)/C; hydrogen In water at 35℃; under 750.075 Torr; for 10h; Reagent/catalyst; Pressure; Temperature; Solvent; Concentration; | 100% |
| With HRO/TiO2; hydrogen In water at 150℃; under 15001.5 Torr; for 3h; | 100% |
| With hydrogen In isopropyl alcohol at 179.84℃; under 22502.3 Torr; for 1.25h; Reagent/catalyst; | 100% |
-
-
98-01-1
furfural

-
-
14032-66-7
5-hydroxy-2-(5H)-furanone

| Conditions | Yield |
|---|---|
| With methanol; oxygen Irradiation; | 100% |
| With oxygen; methylene blue In methanol at -78 - 20℃; for 23h; Irradiation; | 98% |
| With oxygen; Rose Bengal lactone In methanol at 20℃; for 24h; Photolysis; | 93% |

| Conditions | Yield |
|---|---|
| With ammonia; hydrogen In tetrahydrofuran at 80℃; for 2h; Autoclave; | 100% |
| With ammonia; hydrogen In methanol at 30℃; for 24h; Autoclave; | 91% |
| With ammonium hydroxide; Ni6AlO(z); hydrogen at 100℃; under 3000.3 Torr; for 5h; Autoclave; | 90% |

| Conditions | Yield |
|---|---|
| With pyridine; hydroxylamine hydrochloride In ethanol | 100% |
| With hydroxylamine hydrochloride In ethanol; water for 0.166667h; Microwave irradiation; | 89% |
| With N-hydroxyphthalimide In water at 90℃; for 3h; Sealed tube; | 89% |
-
-
98-01-1
furfural

-
-
4464-77-1, 69314-24-5, 69314-25-6, 116204-42-3
1,2-di-furan-2-yl-ethane-1,2-diol

| Conditions | Yield |
|---|---|
| With tris(2,2’-bipyridine)ruthenium(II); ascorbate In water for 3h; pH=12.7; Irradiation; | 100% |
| With ammonium chloride; magnesium Ambient temperature; | 92% |
| With triethylammonium formate; magnesium In methanol; water at 25℃; for 0.333333h; | 74% |

| Conditions | Yield |
|---|---|
| With lithium tetrafluoroborate at 0℃; for 5h; | 100% |
| With amberlyst-15 In acetonitrile for 1h; | 99.92% |
| With dimethylbromosulphonium bromide at 20℃; for 0.0833333h; | 98% |
-
-
98-01-1
furfural

-
-
867-13-0
diethoxyphosphoryl-acetic acid ethyl ester

-
-
623-20-1
ethyl (E)-3-(2-furyl)prop-2-enoate

| Conditions | Yield |
|---|---|
| With potassium carbonate In neat (no solvent) Mechanism; var. other bases, effect of water; | 100% |
| With water; barium dihydroxide In 1,4-dioxane at 70℃; for 0.416667h; | 100% |
| With water; barium dihydroxide In 1,4-dioxane at 70℃; for 0.416667h; Product distribution; other catalyst, other solvents, influence of water; | 100% |

| Conditions | Yield |
|---|---|
| With (R)-oxynitrilase (almond meal) In di-isopropyl ether at 4℃; for 48h; | 100% |
| With almond meal ((R)-oxynitrilase) In di-isopropyl ether at 4 - 30℃; | 100% |
| With almond meal In di-isopropyl ether at 15℃; | 100% |
-
-
98-01-1
furfural

-
-
7677-24-9
trimethylsilyl cyanide

-
-
40861-56-1
2-(2-furyl)-2-(trimethylsilyloxy)acetonitrile

| Conditions | Yield |
|---|---|
| With Eu2(benzene-1,2,3,4,5,6-hexacarboxylate)(H2O)3 In acetonitrile at 20 - 100℃; for 1h; | 100% |
| With 1-methoxy-2-methyl-1-(trimethylsiloxy)propene at 19℃; for 10h; | 99% |
| With potassium phtalimide at 20℃; for 1.16667h; solvent-free; | 99% |

| Conditions | Yield |
|---|---|
| With water; barium dihydroxide In 1,4-dioxane at 70℃; for 0.666667h; | 100% |
| Stage #1: diethyl 1-cyanomethylphosphonate With sodium hydride In tetrahydrofuran at 20℃; for 1h; Inert atmosphere; Stage #2: furfural In tetrahydrofuran at 28℃; | |
| Stage #1: diethyl 1-cyanomethylphosphonate With sodium hydride In tetrahydrofuran; mineral oil at 0℃; for 0.5h; Stage #2: furfural In tetrahydrofuran; mineral oil at 0 - 25℃; for 2h; |
-
-
98-01-1
furfural

-
-
45732-93-2
1-(Hydroxyaminomethyl)-1-cyclohexanol

-
-
84966-16-5
N-Furfuryliden-(1-hydroxycyclohexyl)methanamin-N-oxid

| Conditions | Yield |
|---|---|
| In ethanol Ambient temperature; | 100% |
-
-
98-01-1
furfural

-
-
56613-80-0
(R)-Phenylglycinol

-
-
139437-47-1
(R)-2-{[1-Furan-2-yl-meth-(E)-ylidene]-amino}-2-phenyl-ethanol

| Conditions | Yield |
|---|---|
| With magnesium sulfate In dichloromethane at 20℃; for 12h; | 100% |
| In benzene Heating; | 88% |
| In benzene Heating; | 85% |
| With magnesium sulfate In dichloromethane | |
| In toluene Condensation; Heating; |

| Conditions | Yield |
|---|---|
| With potassium hydroxide In neat (no solvent) Mechanism; var. other bases, effect of water; | 100% |
| With potassium hydroxide In water; acetonitrile | 98% |
| With potassium hydroxide; water In acetonitrile at 40℃; for 0.833333h; Product distribution; | 93% |

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at 0℃; for 1h; Inert atmosphere; | 100% |
| In tetrahydrofuran at 0 - 20℃; Inert atmosphere; | 98% |
| In tetrahydrofuran at 0 - 20℃; Inert atmosphere; | 92% |
-
-
98-01-1
furfural

-
-
143-33-9
sodium cyanide

-
-
541-41-3
chloroformic acid ethyl ester

-
-
20893-23-6
carbonic acid, cyano(2-furyl)methyl ethyl ester

| Conditions | Yield |
|---|---|
| With tetrabutyl-ammonium chloride In dichloromethane; water Heating; | 100% |


| Conditions | Yield |
|---|---|
| In methanol at 20℃; for 24h; | 100% |
| for 2h; Yield given; | |
| With magnesium sulfate In ethanol at 20℃; |
-
-
98-01-1
furfural

-
-
34164-50-6
4-penten-1-ylmagnesium bromide

-
-
106549-86-4, 84735-65-9
(+/-)-1-(furan-2-yl)hex-5-en-1-ol

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at 0℃; | 100% |
| In tetrahydrofuran at 0 - 20℃; for 2h; Inert atmosphere; | 86% |
| In tetrahydrofuran at 0℃; | 73% |
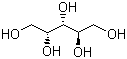
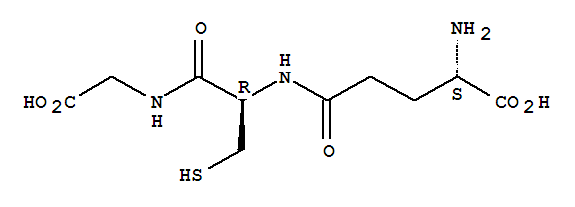
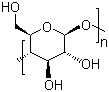
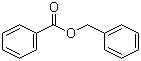
Related products
Downstream Products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View











 T,
T, Xi
Xi