Antimex Chemical Limied
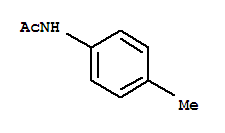
ProName: p-Toluidine Manufacturer/High quality/... CasNo: 106-49-0 Molecular Formula: C7H9N Appearance: Powder Application: It is an important raw material and in... DeliveryTime: prompt PackAge: according to the clients
Cas:106-49-0
Min.Order:1 Metric Ton
FOB Price: $5.0
Type:Lab/Research institutions
inquiryDayang Chem (Hangzhou) Co.,Ltd.
Dayangchem's R&D center can offer custom synthesis according to the contract research and development services for the fine chemicals, pharmaceutical, biotechnique and some of the other chemicals. DayangChem can provide different quantities
Cas:106-49-0
Min.Order:1 Kilogram
FOB Price: $3.0
Type:Lab/Research institutions
inquirySimagchem Corporation
Welcome to Simagchem, your partner in China as a premier supply of bulk specialty chemicals for industry and life science. We introduce experienced quality product and exceptional JIT service with instant market intelligence in China to benefit our
Cas:106-49-0
Min.Order:0 Metric Ton
Negotiable
Type:Manufacturers
inquiryChemwill Asia Co., Ltd.
Our main production base is located in Xuzhou industry park. We are certified both to the ISO 9001 and ISO 14001 Standards, have a safety management system in place.Our R&D team masters core technology for process-design of target building block
Cas:106-49-0
Min.Order:5 Kiloliter
FOB Price: $1.2 / 5.0
Type:Manufacturers
inquiryAlity Chemical Corporation
Products: p-Toluidine Synonyms: 4-Methylaniline C.A.S: 106-49-0 Assay: 98% Molecular: C7H9N Pack: Stru
Henan Tianfu Chemical Co., Ltd.
p-Toluidine Basic information Product Name: p-Toluidine Synonyms: P-TOLUIDINE;P-TOLYLAMINE;P-AMINOTOLUENE;PARA-TOLUIDINE;P-METHYLANILINE;1-Amino-4-methylbenzene;4-Aminotoluen;4-aminotolu
Cas:106-49-0
Min.Order:1 Gram
Negotiable
Type:Lab/Research institutions
inquiryBaoji Guokang Healthchem co.,ltd
Our company has been in existence for 10 years since its establishment. We have our own unique team. The company integrates independent research and development, production and sales. We have established famous brands at home and abroad. At present
Cas:106-49-0
Min.Order:1 Kilogram
FOB Price: $23.0 / 33.0
Type:Trading Company
inquirySHANGHAI T&W PHARMACEUTICAL CO., LTD.
A substitute for perfluorooctanoic acid, mainly used as a surfactant, dispersant, additive, etc Appearance:White solid or Colorless liquid Purity:99.3 % We will ship the goods in a timely manner as required We can provide relevant documents acc
Hangzhou Keyingchem Co.,Ltd
Hangzhou KeyingChem Co., Ltd. exported this product to many countries and regions at best price. If you are looking for the material’s manufacturer or supplier in China, KeyingChem is your best choice. Pls contact with us freely for getting det
Hangzhou J&H Chemical Co., Ltd.
J&H CHEM R&D center can offer custom synthesis according to the contract research and development services for the fine chemicals, pharmaceutical, biotechnique and some of the other chemicals. J&H CHEM has some Manufacturing base in Jia
Zibo Hangyu Biotechnology Development Co., Ltd
Zibo Hangyu Biotechnology Development Co., Ltd is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality pharmaceuticals, intermediates, special chemicals and OLED intermediates and other fine chemi
Cas:106-49-0
Min.Order:10 Gram
FOB Price: $100.0
Type:Lab/Research institutions
inquiryZibo Dorne chemical technology co. LTD
Product Details Grade: pharmaceutical grade Purity:99%+ ProductionCapacity: 1000 Kilogram/Month Scope of use: For scientific research only(The product must be used legally) Our Advantage 1. Best quality with competitive price. 2. Quick shipping,
Cas:106-49-0
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryHenan Wentao Chemical Product Co., Ltd.
We are leading fine chemicals supplier in China with ISO certificate, Our main business covers the fields below: 1.Noble Metal Catalysts (Pt.Pd...) 2.Organic Phosphine Ligands (Tert-butyl-phosphine.Cyclohexyl-phosphine...) 3.OLED
Cas:106-49-0
Min.Order:1 Gram
FOB Price: $3.0
Type:Lab/Research institutions
inquiryHangzhou Huarong Pharm Co., Ltd.
Hangzhou Huarong Pharm Co., Ltd.established since 2006 , has been actively developing specialty products for Finished Dosages, APIs, Intermediates, and Fine chemicals markets in North America, Europe, Korea, Japan, Mid-East and all over the World. Hu
Hangzhou Fandachem Co.,Ltd
Hangzhou Fandachem Co.,Ltd, a China-based chemical company, specialize in exporting P-Toluidine 99.5%, CAS:106-49-0 Please contact us by email freely. We are leading exporter in China. If you really need this cargo, please do not hes
Cas:106-49-0
Min.Order:1 Kilogram
Negotiable
Type:Other
inquiryHangzhou Zhongqi chem Co.,Ltd.
Located in Hangzhou National Hi-Tech Industrial Development Zone, zhongqichem is a technical company mainly focus on the Custom synthesis, manufacturing, sales of chemicals to various industries. Benefiting from the outstanding customer service and h
Hunan chemfish Pharmaceutical co.,Ltd
Appearance:95%+ Package:R&D,Pilot run Transportation:per client require Port:Express ,Air, Sea
HANGZHOU YUNUO CHEMICAL CO.,LTD
Superior quality, moderate price & quick delivery. Appearance:Colorless or light yellow liquid Storage:stored in a cool, dry and ventilated place to provent sun and rain Package:25kg/drum, or as per your request. Application:Used as Pharmac
Kono Chem Co.,Ltd
high purity lowest priceAppearance:solid or liquid Storage:in sealed air resistant place Package:drum and bag Application:for pharma use Transportation:by sea or air Port:Beijing or Guangzhou
GIHI CHEMICALS CO.,LIMITED
high purity,in stock Package:25kg/drum,or as per customers'demand Application:API,Pharmaceutical intermediates Transportation:air,sea,courier
Henan Allgreen Chemical Co.,Ltd
high quality Storage:Sealed, dry, microtherm , avoid light and smell. Package:According to the demand of customer Application:Organic synthesis Transportation:by air or by sea
Aecochem Corp.
Our clients, like BASF,CHEMO,Brenntag,ASR,Evonik,Merck and etc.Appearance:COA Storage:in stock Application:MSDS/TDS
Jiangsu Glory Chemical Co., Ltd.
Para Toluidine (PT) M. F. :C7H9N M. W. :107Appearance:White to yellow brown crystal Package:In 200KGS net weight iron drum; 16MT per 20’FCL
Zhuozhou Wenxi import and Export Co., Ltd
Product Description Description & Specification Category Pharmaceutical Raw Materials, Fine Chemicals, Bulk drug Standard Medical standard
Cas:106-49-0
Min.Order:1 Kilogram
FOB Price: $112.0
Type:Trading Company
inquiryXiamen AmoyChem Co.,Ltd
Amoychem is committed to providing the top-quality chemical products and services Internationally. We offer our customers with friendly, professional service and reliable, high performance products that have been manufactured according to the accredi
Hangzhou ZeErRui Chemical Co., Ltd.
Hangzhou ZeErRui Chemical Co., Ltd. located in Lingang industrial areas, our plant covers an area of 6000 square meters.ZeErRui dedicated to the development, production and marketing of chemicals. We have earned ourselves a good reputation at home an
Cas:106-49-0
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryHunan Russell Chemicals Technology Co.,Ltd
high purity lowest priceAppearance:solid or liquid Storage:in sealed air resistant place Package:Foil bag; Drum; Plastic bottle Application:Pharma;Industry;Agricultural Transportation:by sea or air Port:any port in China
Synthetic route

| Conditions | Yield |
|---|---|
| With triethylsilane; palladium dichloride In ethanol at 20℃; for 0.5h; Inert atmosphere; | 100% |
| With hydrazine hydrate In ethanol; water at 80℃; for 1h; chemoselective reaction; | 100% |
| With sodium tetrahydroborate In ethanol; water at 39.84℃; for 2h; Reagent/catalyst; | 100% |

| Conditions | Yield |
|---|---|
| With triethylsilane; palladium dichloride In ethanol at 20℃; for 0.5h; Inert atmosphere; | 100% |
| With hydrogen In ethanol at 80℃; under 2250.23 Torr; for 5h; Catalytic behavior; Inert atmosphere; | 95% |
| With [IrCl(CO)(PPh3)2]; hydrazine hydrate; potassium hydroxide In methanol at 160℃; for 3h; Wolff-Kishner Reduction; Sealed tube; | 51% |

| Conditions | Yield |
|---|---|
| With triethylsilane; palladium dichloride In ethanol at 20℃; for 0.75h; Inert atmosphere; | 100% |
| With palladium 10% on activated carbon; ammonium formate In ethanol at 100℃; for 24h; Reagent/catalyst; | 90% |
| Stage #1: 4-nitrobenzaldehdye With hydrazine hydrate at 135℃; for 3h; Stage #2: With potassium hydroxide at 135℃; for 24h; | 63% |
| With hydrogen In ethanol; water at 25℃; under 750.075 Torr; for 4h; Schlenk technique; |
-
-
14618-59-8
N-(tert-butoxycarbonyl)-4-methylaniline

-
-
106-49-0
p-toluidine

| Conditions | Yield |
|---|---|
| With water at 100℃; for 10h; Inert atmosphere; | 99% |
| With 3-butyl-l-methyl-1H-imidazol-3-iumtrifloroacetate In 1,4-dioxane; water at 70 - 72℃; for 1h; | 98% |
| With Montmorillonite K10 In dichloromethane for 2h; deacylation; Heating; | 97% |
| With water at 150℃; for 4h; Subcritical conditions; | 88% |
| With zinc(II) iodide In toluene at 120℃; for 24h; Reagent/catalyst; Schlenk technique; Inert atmosphere; Glovebox; |

-
-
106-49-0
p-toluidine

| Conditions | Yield |
|---|---|
| With potassium phosphate; tris(2,2-bipyridine)ruthenium(II) hexafluorophosphate; ascorbic acid In water; acetonitrile at 20℃; for 3h; Sealed tube; Irradiation; Inert atmosphere; | 99% |

| Conditions | Yield |
|---|---|
| With butyltriphenylphosphonium tetrahydroborate In dichloromethane at 20℃; for 0.266667h; | 98% |
| With iron(III) chloride; sodium iodide In acetonitrile at 20℃; for 0.166667h; | 97% |
| With chloro-trimethyl-silane; sodium iodide In acetonitrile for 0.0833333h; Ambient temperature; | 97% |

| Conditions | Yield |
|---|---|
| With copper(I) oxide; ammonium hydroxide In 1-methyl-pyrrolidin-2-one at 80℃; for 15h; | 98% |
| Stage #1: para-bromotoluene With bis(bis(trimethylsilyl)amido)zinc(II); tri-tert-butyl phosphine; lithium chloride; bis(dibenzylideneacetone)-palladium(0) In tetrahydrofuran at 50℃; for 9h; Stage #2: With hydrogenchloride In tetrahydrofuran; diethyl ether | 93% |
| With ammonium hydroxide In neat (no solvent) at 60℃; for 8h; | 93% |

| Conditions | Yield |
|---|---|
| With water; sodium hydroxide In ethanol at 65 - 70℃; for 1h; | 98% |
| With sodium hydroxide; water In 1,4-dioxane at 70℃; Kinetics; Thermodynamic data; isotope effect (D2O); ΔH(excit.), ΔS(excit.); | |
| With sodium hydroxide In ethanol; water at 40℃; Kinetics; |

| Conditions | Yield |
|---|---|
| With 40% potassium fluoride/alumina at 85℃; for 0.0666667h; Microwave irradiation; Neat (no solvent); | 98% |
| With sodium hydroxide In methanol at 80℃; | 92% |
| With pepsin immobilized on terephthalaldehyde functionalized chitosan magnetic nanoparticle In acetonitrile at 20℃; for 48h; pH=2; | 83% |

| Conditions | Yield |
|---|---|
| With potassium carbonate In dimethyl sulfoxide at 90℃; for 2h; Lossen rearrangement; | 98% |
| With palladium diacetate; triethylamine In acetonitrile at 90℃; for 24h; Solvent; Lossen Rearrangement; Sealed tube; | 98% |
| With potassium carbonate In dimethyl sulfoxide at 90℃; for 2h; Reagent/catalyst; Solvent; Lossen Rearrangement; | 98% |
-
-
63534-35-0
3-azidohomoadamantane

-
-
108-88-3
toluene

-
A
-
76429-91-9
1-(adamantan-1-ylmethyl)-4-methylbenzene

-
B
-
106-49-0
p-toluidine

| Conditions | Yield |
|---|---|
| With aluminium trichloride at 80℃; for 1.5h; Yields of byproduct given; | A 97% B n/a |

-
-
29540-83-8
p-tolyl triflate

-
-
106-49-0
p-toluidine

| Conditions | Yield |
|---|---|
| Stage #1: p-tolyl triflate With bis(bis(trimethylsilyl)amido)zinc(II); tri-tert-butyl phosphine; tetrabutylammomium bromide; bis(dibenzylideneacetone)-palladium(0) In tetrahydrofuran at 50℃; for 6h; Stage #2: With hydrogenchloride In tetrahydrofuran; diethyl ether | 97% |
| Multi-step reaction with 2 steps 1: Trimethyl borate; water; trifluoroacetic acid; acetone / 15 °C / UV-irradiation; Inert atmosphere 2: sodium hydroxide / methanol / 80 °C View Scheme |
-
-
74552-28-6
N-(p-Tolyl)carbamidsaeure-β-chlorethylester

-
-
106-49-0
p-toluidine

| Conditions | Yield |
|---|---|
| With ammonium chloride; zinc; hydroxycobalamine In ethanol; water at 20℃; for 15h; | 96% |

| Conditions | Yield |
|---|---|
| With sodium tetrahydroborate; titanium tetrachloride In 1,2-dimethoxyethane for 14h; Ambient temperature; | 96% |
-
-
31935-68-9
1,1,1-Trimethyl-N-(4-methylphenyl)-N-(trimethylsilyl)silanamine

-
-
106-49-0
p-toluidine

| Conditions | Yield |
|---|---|
| With hydrogenchloride; water In methanol at 20℃; for 1h; | 96% |
| With hydrogenchloride In diethyl ether | |
| With hydrogenchloride In chloroform; water for 1h; | 23.6 mg |

| Conditions | Yield |
|---|---|
| With copper(I) oxide; ammonium hydroxide; C17H14N2O3; potassium hydroxide In ethanol at 60℃; for 24h; Schlenk technique; Inert atmosphere; | 96% |
| With ammonia; triethylamine In water at 20℃; for 3.5h; | 96% |
| With copper(l) iodide; ascorbic acid In ammonia at 25℃; for 18h; Kinetics; liquid NH3; | 95% |
-
-
635-46-1
1,2,3,4-tetrahydroisoquinoline

-
-
99-99-0
1-methyl-4-nitrobenzene

-
A
-
91-22-5
quinoline

-
B
-
106-49-0
p-toluidine

| Conditions | Yield |
|---|---|
| With nickel-nitrogen-doped carbon framework In water at 145℃; for 18h; Inert atmosphere; Sealed tube; Green chemistry; | A 94% B 96% |


| Conditions | Yield |
|---|---|
| at 230℃; under 10 Torr; for 0.0833333h; Product distribution; pyrolysis without solvent; | A 95% B n/a |
-
-
76-05-1
trifluoroacetic acid

-
A
-
350-96-9
2,2,2-trifluoro-N-(4-methylphenyl)acetamide

-
B
-
106-49-0
p-toluidine

-
C
-
67-68-5
dimethyl sulfoxide

| Conditions | Yield |
|---|---|
| at 50℃; for 5h; Elimination; acylation; retro-Pummerer reaction; | A 3% B 95% C 67% |


| Conditions | Yield |
|---|---|
| With ammonium formate; zinc In ethylene glycol for 0.05h; microwave irradiation; | 95% |
-
-
2415-85-2
3-oxo-N-(p-tolyl)butanamide

-
-
95-54-5
1,2-diamino-benzene

-
A
-
615-15-6
2-Methyl-1H-benzimidazole

-
B
-
106-49-0
p-toluidine

| Conditions | Yield |
|---|---|
| With 1-n-butyl-3-methylimidazolim bromide In neat (no solvent) at 120℃; for 2h; | A 95% B 90% |

-
-
14933-93-8
4'-methylbenzenesulphenanilide

-
A
-
3236-92-8
2,7-dimethylphenazine

-
B
-
106-49-0
p-toluidine

-
C
-
882-33-7
diphenyldisulfane

| Conditions | Yield |
|---|---|
| With trifluoroacetic acid In benzene at 25℃; | A 35% B 55% C 94% |

-
-
58960-03-5
7-methyl-1,2,3,4-tetrahydroquinoline

-
-
99-99-0
1-methyl-4-nitrobenzene

-
A
-
612-60-2
7-methylquinoline

-
B
-
106-49-0
p-toluidine

| Conditions | Yield |
|---|---|
| With nickel-nitrogen-doped carbon framework In water at 145℃; for 18h; Inert atmosphere; Sealed tube; Green chemistry; | A 92% B 94% |


| Conditions | Yield |
|---|---|
| With dimethylsulfide borane complex In chlorobenzene 1) 15 min, r.t. 2) 5 h, reflux; | 93% |
-
-
501-60-0, 21650-54-4, 30926-02-4
1,2-di(p-tolyl)diazene

-
-
106-49-0
p-toluidine

| Conditions | Yield |
|---|---|
| With potassium hydroxide; nickel-incorporated hexagonal mesoporous aluminophosphate In isopropyl alcohol at 82.84℃; for 1.5h; | 93% |
| With perchloric acid In isopropyl alcohol; acetonitrile at 25℃; for 0.75h; pH=2; Inert atmosphere; Irradiation; | 92% |
| With ethanol; iron; calcium chloride at 60℃; for 0.5h; | 92% |

| Conditions | Yield |
|---|---|
| With copper(I) oxide; ammonium hydroxide; air In methanol at 20℃; for 12h; | 93% |
| With sodium hydroxide; hydroxylamine-O-sulfonic acid In acetonitrile at 20℃; for 16h; | 90% |
| With N-Bromosuccinimide; CYANAMID; bis-[(trifluoroacetoxy)iodo]benzene In acetonitrile at 20℃; for 1h; chemoselective reaction; | 90% |
-
-
126629-91-2
N-{(Z)-1,3-Diphenyl-3-[(Z)-p-tolylimino]-propenyl}-methanesulfonamide

-
A
-
96355-37-2
3,5-Diphenyl-2H-1,2-thiazin-1,1-dioxid

-
B
-
106-49-0
p-toluidine

| Conditions | Yield |
|---|---|
| With lithium diisopropyl amide In tetrahydrofuran at -78 - 25℃; for 12h; | A 92% B n/a |
-
-
126629-93-4
N-{(Z)-3-Phenyl-1-p-tolyl-3-[(Z)-p-tolylimino]-propenyl}-methanesulfonamide

-
A
-
126629-99-0
5-Phenyl-3-p-tolyl-2H-[1,2]thiazine 1,1-dioxide

-
B
-
106-49-0
p-toluidine

| Conditions | Yield |
|---|---|
| With lithium diisopropyl amide In tetrahydrofuran at -78 - 25℃; for 12h; | A 92% B n/a |

| Conditions | Yield |
|---|---|
| With copper(ll) sulfate pentahydrate; ammonium hydroxide In PEG1000-DIL; methyl cyclohexane at 60℃; for 8h; | 92% |
| Stage #1: para-chlorotoluene With bis(bis(trimethylsilyl)amido)zinc(II); tri-tert-butyl phosphine; lithium chloride; bis(dibenzylideneacetone)-palladium(0) In tetrahydrofuran at 90℃; for 12h; Stage #2: With hydrogenchloride In tetrahydrofuran; diethyl ether | 90% |
| With copper(I) oxide; ammonium hydroxide In 1-methyl-pyrrolidin-2-one at 110℃; for 12h; Microwave irradiation; | 89% |

| Conditions | Yield |
|---|---|
| In methanol at 20℃; for 24h; | 100% |
| In methanol at 20℃; for 24h; | 100% |
| In methanol at 20℃; | 85% |

| Conditions | Yield |
|---|---|
| for 1h; Solid phase reaction; condensation; | 100% |
| With 3-butyl-1-methyl-1H-imidazol-3-ium hexafluorophosphate at 80℃; for 8h; | 97% |
| In decaethylene glycol at 120℃; for 3h; | 97% |

| Conditions | Yield |
|---|---|
| In dichloromethane at 20℃; Inert atmosphere; | 100% |
| With Methylenediphosphonic acid at 20℃; for 1h; neat (no solvent); | 99% |
| With pyridine; aluminum oxide at 125 - 127℃; for 2h; microwave irradiation; | 98% |
-
-
102-92-1
cinnamoyl chloride

-
-
106-49-0
p-toluidine

-
-
6876-68-2
N-(4-methylphenyl)-3-phenyl-2-propenamide

| Conditions | Yield |
|---|---|
| With triethylamine In dichloromethane at 20℃; Inert atmosphere; | 100% |
| With triethylamine In ethyl acetate at 0 - 20℃; | 88% |
| With potassium hydroxide |
-
-
99-61-6
3-nitro-benzaldehyde

-
-
106-49-0
p-toluidine

- N-(3-nitrobenzylidene)-4-methylbenzenamine
-
17064-95-8
N-(3-nitrobenzylidene)-4-methylbenzenamine

| Conditions | Yield |
|---|---|
| 100% | |
| With chitosan In ethanol; water at 20℃; for 0.333333h; | 85% |
| With ethanol |
-
-
555-16-8
4-nitrobenzaldehdye

-
-
106-49-0
p-toluidine

-
-
730-39-2, 33442-37-4, 149742-47-2
N-(4-nitrobenzylidene)-4-methylaniline

| Conditions | Yield |
|---|---|
| for 24h; Ambient temperature; | 100% |
| 100% | |
| With montmorillonite at 20℃; for 0.0666667h; Neat (no solvent); | 98% |
-
-
83-72-7
2-Hydroxy-1,4-naphthoquinone

-
-
106-49-0
p-toluidine

-
-
57182-49-7
2-(p-toluidino)-1,4-naphthoquinone

| Conditions | Yield |
|---|---|
| In neat (no solvent) at 100℃; under 750.075 Torr; for 0.05h; Microwave irradiation; | 100% |
| With acetic acid | |
| With ethanol |

| Conditions | Yield |
|---|---|
| for 6h; Ambient temperature; | 100% |
| 100% | |
| In ethyl 2-hydroxypropionate at 20℃; for 0.0333333h; | 90% |

| Conditions | Yield |
|---|---|
| at 20℃; for 0.5h; Solid phase reaction; condensation; | 100% |
| With fibrous nano silica sulfuric acid In neat (no solvent) at 80℃; for 0.0333333h; Green chemistry; | 97% |
| With silica sulfuric acid In acetonitrile for 0.0194444h; Microwave irradiation; | 96% |
-
-
106-49-0
p-toluidine

-
-
120-14-9
3,4-dimethoxy-benzaldehyde

-
-
67101-90-0
1,2-dimethoxy-4-<<(4-methylphenyl)imino>methyl>benzene

| Conditions | Yield |
|---|---|
| In toluene Heating; | 100% |
-
-
106-49-0
p-toluidine

-
-
98-59-9
p-toluenesulfonyl chloride

-
-
599-86-0
4-methyl-N-(4-methylphenyl)benzenesulfonamide

| Conditions | Yield |
|---|---|
| With pyridine In dichloromethane at 20℃; for 16h; | 100% |
| With pyridine at 0 - 25℃; | 100% |
| In water at 110℃; for 0.0833333h; Microwave irradiation; Green chemistry; chemoselective reaction; | 98% |
-
-
106-49-0
p-toluidine

-
-
625-36-5
2-chloropropionyl chloride

-
-
19342-88-2
3-chloro-N-(4-methylphenyl)propionamide

| Conditions | Yield |
|---|---|
| In acetone for 1h; Acylation; Heating; | 100% |
| In water; acetone for 6h; Reflux; | 100% |
| With potassium carbonate In acetone at 0℃; for 1h; Condensation; | 95% |

| Conditions | Yield |
|---|---|
| 100% | |
| In ethanol for 3h; Reflux; | 77.2% |
| In ethanol for 3h; Reflux; | 77.2% |

| Conditions | Yield |
|---|---|
| With triethylamine In tetrahydrofuran for 24h; Ambient temperature; | 100% |
| With toluene |
-
-
106-49-0
p-toluidine

-
-
762-42-5
dimethyl acetylenedicarboxylate

-
-
24559-80-6
dimethyl (Z)-N-(4-methylphenyl)aminofumarate

| Conditions | Yield |
|---|---|
| at 20℃; for 0.133333h; Michael addition; | 100% |
| In benzene at 5 - 10℃; | 70% |
| In diethyl ether |

| Conditions | Yield |
|---|---|
| In N,N-dimethyl-formamide at 110℃; for 5h; | 100% |
| In diethyl ether at 20℃; for 2h; | 98% |
| In chloroform for 7h; | 97% |

| Conditions | Yield |
|---|---|
| With sodium hydrogencarbonate In N,N,N,N,N,N-hexamethylphosphoric triamide for 0.5h; Heating; 3 equivalents of BzBr; | 100% |
| With montmorillonite-KSF clay supported CuO nanoparticles In neat (no solvent) at 20℃; for 0.75h; Sonication; Green chemistry; chemoselective reaction; | 78% |
| With sodium dodecyl-sulfate; sodium hydrogencarbonate In water at 80℃; for 1h; Inert atmosphere; | 71% |
| With sodium dodecyl-sulfate; sodium hydrogencarbonate In water at 80℃; for 1h; | 70% |

| Conditions | Yield |
|---|---|
| for 6h; Ambient temperature; | 100% |
| In ethyl 2-hydroxypropionate at 20℃; for 0.0333333h; | 96% |
| With montmorillonite at 20℃; for 0.1h; Neat (no solvent); | 96% |
-
-
924-44-7
glyoxylic acid ethyl ester

-
-
106-49-0
p-toluidine

-
-
121641-60-9
ethyl 2-(4-methylphenylimino)acetate

| Conditions | Yield |
|---|---|
| With sodium sulfate In dichloromethane for 0.5h; | 100% |
| With magnesium sulfate In toluene at 25℃; for 0.5h; | 100% |
| for 1h; Green chemistry; | 75% |
-
-
40657-29-2
1,1,1-trifluoro-4,4-diethoxy-3-buten-2-one

-
-
106-49-0
p-toluidine

-
-
128648-64-6
(E)-4-Ethoxy-1,1,1-trifluoro-4-p-tolylamino-but-3-en-2-one

| Conditions | Yield |
|---|---|
| In acetonitrile for 18h; Ambient temperature; | 100% |
-
-
128346-30-5
(2-formylphenyl)butyl tellurium dibromide

-
-
106-49-0
p-toluidine

-
-
130191-28-5
2-(4'-methylphenyliminomethinyl)phenyltellurenyl bromide

| Conditions | Yield |
|---|---|
| With trifluoroacetic acid In benzene | 100% |
-
-
94507-26-3
2-<(3-Chloropropyl)amino>-4H-3,1-benzoxazin-4-one

-
-
106-49-0
p-toluidine

-
-
94507-35-4
N-(4-Methylphenyl)-2-<3-(3-chloropropyl)ureido>benzamide

| Conditions | Yield |
|---|---|
| for 0.05h; Heating; | 100% |
-
-
84410-20-8
4-[4a-Methoxy-7-(4-methoxy-3,6-dioxo-cyclohexa-1,4-dienyl)-5,8-dioxo-4-phenyl-4a,5,8,8a-tetrahydro-4H-quinolin-1-yl]-benzoic acid

-
-
106-49-0
p-toluidine

-
-
84423-43-8
4-(9-Hydroxy-4a,8-dimethoxy-5,11-dioxo-4-phenyl-6-p-tolyl-4,4a,5,6,11,11a-hexahydro-pyrido[3,2-b]carbazol-1-yl)-benzoic acid

| Conditions | Yield |
|---|---|
| With sodium hydrogencarbonate In ethanol; water for 4h; Heating; | 100% |

| Conditions | Yield |
|---|---|
| for 2h; Ambient temperature; | 100% |
| In ethanol for 0.333333h; Reflux; | 95.51% |
| sodium hydrogen sulfate; silica gel at 54 - 56℃; for 0.0236111h; microwave irradiation; | 91% |
-
-
106-49-0
p-toluidine

-
-
15486-96-1
3-Bromopropionyl chloride

-
-
21437-82-1
3-bomo-N-(4-methylphenyl)propanamide

| Conditions | Yield |
|---|---|
| With potassium carbonate In 1,2-dichloro-ethane at 20℃; for 1h; Time; Inert atmosphere; | 100% |
| With dmap In tetrahydrofuran at 0 - 20℃; for 1h; | 75% |
| With triethylamine In benzene | 70% |

| Conditions | Yield |
|---|---|
| With hydrogen; AV-17-8-Pd In ethanol at 45℃; under 750.06 Torr; Rate constant; Thermodynamic data; E(a); | 100% |
| With hydrogen; AV-17-8-Pd In ethanol at 45℃; under 750.06 Torr; | 100% |
| With Pd/Al2O3; hydrogen In ethanol at 80℃; under 750.075 Torr; for 12h; | 94% |
-
-
106-49-0
p-toluidine

-
-
102-92-1
Cinnamoyl chloride

-
-
134430-88-9
(2E)-N-(4-methylphenyl)-3-phenylprop-2-enamide

| Conditions | Yield |
|---|---|
| With potassium carbonate In water; acetone at 0℃; for 2h; | 100% |
| Stage #1: Cinnamoyl chloride With pyridine; dmap In dichloromethane at 0℃; for 0.416667h; Inert atmosphere; Stage #2: p-toluidine In dichloromethane at 0 - 20℃; | 86% |
| With dmap; sodium hydrogencarbonate In dichloromethane at 20℃; | 84% |

| Conditions | Yield |
|---|---|
| With zirconium(IV) chloride at 20℃; for 0.25h; | 100% |
| With [{(CH3)5Cp}2Zr(CH3CN)2(H2O)][OSO2C6F5]2·CH3CN In neat (no solvent) at 20℃; for 0.166667h; diastereospecific reaction; | 99% |
| With zinc(II) perchlorate hexahydrate at 20℃; for 0.5h; | 98% |

| Conditions | Yield |
|---|---|
| With trimethylamine Ambient temperature; | 100% |
| In diethyl ether; tetrahydrofyran at 0℃; for 4h; | 95% |
| With triethylamine In diethyl ether at 0℃; | 83% |
Related products
Raw Materials
Downstream Products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View















 T,
T, N
N