Dayang Chem (Hangzhou) Co.,Ltd.
Dayangchem’s R&D center can offer custom synthesis according to the contract research and development services for the fine chemicals, pharmaceutical, biotechnique and some of the other chemicals. DayangChem can provide different quantiti
Cas:106-50-3
Min.Order:100 Gram
FOB Price: $5.0
Type:Lab/Research institutions
inquiryAntimex Chemical Limied
Quick Details p-Phenylenediamine manufactuer of Antimex Group,local in shanghai China ProName: p-Phenylenediamine CasNo: 106-50-3 Molecular Formula: C6H8N2 Appearance: Brown flakes to White flakes Application: D
Cas:106-50-3
Min.Order:1 Metric Ton
FOB Price: $2.0
Type:Lab/Research institutions
inquiryHefei TNJ chemical industry co.,ltd
P-phenylenediamine(PPD) Product Name: P-phenylenediamine(PPD) Synonyms: p-Phenylene diamine CAS No.: 106-50-3
Ality Chemical Corporation
p-Phenylenediamine Basic information Description Chemical Properties Uses Production method Acute toxicity Irritation data Hazardous characteristics of explosive Flammability and hazard characteristics Storage c
Simagchem Corporation
Welcome to Simagchem, your partner in China as a premier supply of bulk specialty chemicals for industry and life science. We introduce experienced quality product and exceptional JIT service with instant market intelligence in China to benefit our
Cas:106-50-3
Min.Order:1 Kilogram
Negotiable
Type:Manufacturers
inquiryJinan Finer Chemical Co., Ltd
Product Description Product name p-Phenylenediamine CAS 106-50-3 Assay 99% Appearance White crystal or flake Capacity 500mt/year Min.package 100gram Standard En
Cas:106-50-3
Min.Order:1 Kilogram
FOB Price: $2.0
Type:Lab/Research institutions
inquiryHebei yanxi chemical co.,LTD.
hebei yanxi chemical co., LTD who registered capital of 10 million yuan, nearly to $2 million, we have a pharmaceutical raw materials factory production of pharmaceutical raw materials, and a reagent r&d center, and we do research and developm
Cas:106-50-3
Min.Order:1 Metric Ton
FOB Price: $1.0 / 3.0
Type:Manufacturers
inquiryWuhan Fortuna Chemical Co.,Ltd
1. Guaranteed purity; 2. Large quantity in stock; 3. Largest manufacturer; 4. Best service after shipment with email; 5. High quality & competitive price; ...... Appearance:White powder or flake Storage:2-8°C Package:25KG/drum Applica
Cas:106-50-3
Min.Order:25 Kilogram
FOB Price: $10.0 / 20.0
Type:Trading Company
inquiryHenan Allgreen Chemical Co.,Ltd
T he company has advanced technology, as well as a large number of excellent R & D team, to provide customers from the grams to one hundred kilograms and tons of high-quality products, competitive prices and quality service Appearance:White
EAST CHEMSOURCES LIMITED
p-Phenylenediamine Product Name: p-Phenylenediamine Molecular Weight: 108.14 CAS NO: 106-50-3 EC NO: 203-404-7 Molecular Formula: C6H8N2
Cas:106-50-3
Min.Order:1 Kilogram
FOB Price: $2.0 / 3.0
Type:Manufacturers
inquiryChemwill Asia Co., Ltd.
Our main production base is located in Xuzhou industry park. We are certified both to the ISO 9001 and ISO 14001 Standards, have a safety management system in place.Our R&D team masters core technology for process-design of target building block
Cas:106-50-3
Min.Order:5 Kiloliter
FOB Price: $1.2 / 5.0
Type:Manufacturers
inquiryHenan Tianfu Chemical Co., Ltd.
Product Name: p-Phenylenediamine Synonyms: benzofurd;C.I. 76076;C.I. Developer 13;C.I. Oxidation Base 10;c.i.developer13;c.i.oxidationbase10;C.I.OxidationBase10(76060);cideveloper13 CAS: 106-50-3 MF: C6H8N2 MW: 108.14
Cas:106-50-3
Min.Order:1 Gram
FOB Price: $8900.0
Type:Lab/Research institutions
inquiryHenan Sinotech Import&Export Corporation
Chemical Name: p-Phenylene Diamine Synonyms: 1,4-Diaminobenzene;1,4-benzenediamie; Para Phenylene Diamine CAS No.: 106-50-3 Molecular Formula: C6H8N2 Properties: white transparent crystal, it is easily soluble in ether, ethanol, acetone and
Cas:106-50-3
Min.Order:1 Metric Ton
FOB Price: $1.0
Type:Other
inquiryLIDE PHARMACEUTICALS LIMITED
Advantage : LIDE PHARMACEUTICALS LTD. is a mid-small manufacturing-type enterprise, engaged in pharmaceutical intermediates of R&D, custom-made and production, and also involving trading chemicals for export. We have established the R&
Cas:106-50-3
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryBaoji Guokang Healthchem co.,ltd
Our company has been in existence for 10 years since its establishment. We have our own unique team. The company integrates independent research and development, production and sales. We have established famous brands at home and abroad. At present
Cas:106-50-3
Min.Order:1 Kilogram
FOB Price: $30.0 / 35.0
Type:Trading Company
inquiryChangchun Artel lmport and Export trade company
Product Detail Minimum Order Qty. 10 Gram
Cas:106-50-3
Min.Order:20 Metric Ton
Negotiable
Type:Trading Company
inquiryLonwin Chemical Group Limited
p-Phenylenediamine CAS: 106-50-3 Specification mp 139 °c bp 267 °c(lit.) vapor density 3.7 (vs air) vapor pressure 1.08 mm hg ( 100 °c) fp 156 °c storage temp.
Wuhan Zenuo Biological Medicine Technology Co Ltd
Product Name: p-Phenylenediamine Synonyms: P-Phenylenediamine(P.P.D.)WHITE;cioxidationbase10;Developer 13;Developer PF;developer13;developerpf;Durafur Black R;durafurblackr CAS: 106-50-3 MF: C6H8N2 MW: 108.14 EINECS: 203-404-7 Product C
Cas:106-50-3
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryAfine Chemicals Limited
Our Services 1. New Molecules R&D 2. Own test center HPLC NMR GC LC-MS 3. API and Intermediates from China reputed manufacturers 4. Documents support COA MOA MSDS DMF open part Our advantages 1. Government awarded company. Top 100 enter
Cas:106-50-3
Min.Order:1 Kilogram
FOB Price: $1.0 / 100000.0
Type:Lab/Research institutions
inquiryHangzhou Keyingchem Co.,Ltd
Hangzhou KeyingChem Co., Ltd. exported this product to many countries and regions at best price. If you are looking for the material’s manufacturer or supplier in China, KeyingChem is your best choice. Pls contact with us freely for getting det
Cas:106-50-3
Min.Order:0 Metric Ton
Negotiable
Type:Lab/Research institutions
inquiryHangzhou J&H Chemical Co., Ltd.
J&H CHEM R&D center can offer custom synthesis according to the contract research and development services for the fine chemicals, pharmaceutical, biotechnique and some of the other chemicals. J&H CHEM has some Manufacturing base in Jia
SHANGHAI T&W PHARMACEUTICAL CO., LTD.
A substitute for perfluorooctanoic acid, mainly used as a surfactant, dispersant, additive, etc Appearance:White solid or Colorless liquid Purity:99.3 % We will ship the goods in a timely manner as required We can provide relevant documents acc
Cas:106-50-3
Min.Order:4 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryZibo Hangyu Biotechnology Development Co., Ltd
Zibo Hangyu Biotechnology Development Co., Ltd is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality pharmaceuticals, intermediates, special chemicals and OLED intermediates and other fine chemi
HANGZHOU YUNUO CHEMICAL CO.,LTD
Superior quality, moderate price & quick delivery. Appearance:Off-white flakes Storage:Stored in cool, dry and ventilation place; Away from fire and heat Package:as per your request Application:Used as Pharmaceutical Intermediates, etc. Tra
Zibo Dorne chemical technology co. LTD
Product Details Grade: pharmaceutical grade Purity:99%+ ProductionCapacity: 1000 Kilogram/Month Scope of use: For scientific research only(The product must be used legally) Our Advantage 1. Best quality with competitive price. 2. Quick shipping,
Cas:106-50-3
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryHenan Wentao Chemical Product Co., Ltd.
1)quick response within 12 hours; 2)quality guarantee: all products are strictly tested by our qc, confirmed by qa and approved by third party lab in china, usa, canada, germany, uk, italy, france etc. 3) oem/odm available; 4) rea
Cas:106-50-3
Min.Order:1 Kilogram
FOB Price: $4.0
Type:Lab/Research institutions
inquirySiwei Development Group Ltd.
Product name:P-Phenylenediamine CAS No.:106-50-3 Molecule Formula:C6H8N2 Molecule Weight:108.14 Purity: 99.0% Package: 25kg/drum Description: White flakes Manufacture Standards:Enterprise Standard TESTING ITEMS
Cas:106-50-3
Min.Order:1 Kilogram
Negotiable
Type:Trading Company
inquiryHangzhou Zhongqi chem Co.,Ltd.
Located in Hangzhou National Hi-Tech Industrial Development Zone, zhongqichem is a technical company mainly focus on the Custom synthesis, manufacturing, sales of chemicals to various industries. Benefiting from the outstanding customer service an
Kono Chem Co.,Ltd
high purity lowest priceAppearance:solid or liquid Storage:in sealed air resistant place Package:drum and bag Application:for pharma use Transportation:by sea or air Port:Beijing or Guangzhou
Hangzhou Dingyan Chem Co., Ltd
R & D enterprises have their own stock in stockAppearance:To be subject to the object Package:Customized Application:pharmaceutical intermediates Transportation:Air Port:Shanghai;Guangzhou
Synthetic route

| Conditions | Yield |
|---|---|
| With sodium tetrahydroborate In water at 50℃; for 0.0833333h; Green chemistry; | 100% |
| With hydrogen In tetrahydrofuran at 20℃; under 760.051 Torr; for 3h; chemoselective reaction; | 100% |
| With sodium hypophosphite monohydrate; 5%-palladium/activated carbon; hypophosphorous acid In 2-methyltetrahydrofuran; water for 3h; Sonication; chemoselective reaction; | 99% |

| Conditions | Yield |
|---|---|
| Stage #1: 4-nitrophenyl azide With hydrazine hydrate at 135℃; for 3h; Stage #2: With potassium hydroxide at 135℃; for 24h; | 100% |
| With hydrogen In ethanol at 20℃; under 760.051 Torr; for 5h; | 94% |
| With hydrogenchloride; tin |

| Conditions | Yield |
|---|---|
| With palladium diacetate; carbon monoxide; triphenylphosphine In water; acetic acid at 56℃; under 532 Torr; for 11h; | 100% |
| With triethylsilane; palladium dichloride In ethanol at 20℃; for 1h; Inert atmosphere; | 100% |
| With 10% palladium on activated charcoal; hydrogen for 2h; | 100% |

| Conditions | Yield |
|---|---|
| With graphitic carbon nitride; hydrazine hydrate In water at 70℃; for 24h; Irradiation; Sealed tube; Green chemistry; chemoselective reaction; | 100% |

| Conditions | Yield |
|---|---|
| Stage #1: nitrobenzene With Oxalamide; tetramethyl ammoniumhydroxide; ammonia In dimethyl sulfoxide at 100℃; under 18751.9 Torr; for 7h; Autoclave; Stage #2: With 5%-palladium/activated carbon; hydrogen In methanol at 55℃; under 9000.9 Torr; for 4h; Reagent/catalyst; Autoclave; | 99.1% |
| Multi-step reaction with 2 steps 1: urea, monosodium salt; urea / dimethyl sulfoxide / 2 h / 90 °C 2: 5%-palladium/activated carbon; hydrogen / ethanol / 80 °C / 15001.5 - 22502.3 Torr View Scheme |

| Conditions | Yield |
|---|---|
| With ammonium bromide; ethylenediamine at 70℃; for 5h; Microwave irradiation; Inert atmosphere; neat (no solvent); | 99% |
| With ammonium iodide; hydrazine at 50℃; for 12h; | 99% |
| With ammonium iodide; hydrazine hydrate at 50℃; for 12h; Inert atmosphere; Sealed tube; | 99% |
-
-
14860-64-1
4-azidoaniline

-
-
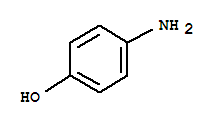
106-50-3
1,4-phenylenediamine

| Conditions | Yield |
|---|---|
| With iron(III) oxide; hydrazine hydrate In water at 120℃; for 2h; Concentration; Temperature; Time; Inert atmosphere; | 99% |
| With zinc In methanol at 20℃; for 5h; | 95% |
| With hydrazine hydrate In ethanol at 20℃; chemoselective reaction; | 90% |

| Conditions | Yield |
|---|---|
| With C24H12Cu2F9N4O7; tetrabutylammomium bromide; ammonia; caesium carbonate In water at 110 - 140℃; for 16h; | 99% |
| With ammonia; C16H16N2O2*0.8Ni*0.2Cu at 85℃; under 1500.15 Torr; for 8.5h; Autoclave; Inert atmosphere; | 92% |
| Multi-step reaction with 2 steps 1: copper(ll) sulfate pentahydrate; sodium L-ascorbate; potassium carbonate; ammonium hydroxide / dimethyl sulfoxide; glycerol / 8 h / 100 °C 2: copper(ll) sulfate pentahydrate; sodium L-ascorbate; potassium carbonate; ammonium hydroxide / dimethyl sulfoxide; glycerol / 8 h / 100 °C View Scheme |

| Conditions | Yield |
|---|---|
| With C24H12Cu2F9N4O7; tetrabutylammomium bromide; ammonia; caesium carbonate In water at 110 - 140℃; for 16h; | 99% |
| With ammonia at 20℃; Reagent/catalyst; Temperature; | 97% |
| With [10%-Pd/Al2O3]; ammonia at 250℃; Catalytic behavior; Temperature; Reagent/catalyst; Inert atmosphere; |

| Conditions | Yield |
|---|---|
| With hydrogen In ethanol at 109.84℃; under 18751.9 Torr; for 7.5h; Autoclave; Green chemistry; chemoselective reaction; | 99% |

| Conditions | Yield |
|---|---|
| With [Cu2(2,7-bis(pyridin-2-yl)-l,8-naphthyridine)(OH)(CF3COO)3]; tetrabutylammomium bromide; ammonia; caesium carbonate In water at 120℃; for 16h; Sealed tube; chemoselective reaction; | 98% |
| Multi-step reaction with 2 steps 1: chlorotris(triphenylphosphine)cobalt(I); bis[2-(diphenylphosphino)phenyl] ether / toluene / 12 h / 100 °C 2: hydrogenchloride; water / methanol / 1 h / 20 °C View Scheme |

| Conditions | Yield |
|---|---|
| With copper(l) iodide; tetra(n-butyl)ammonium hydroxide; ammonia In water at 25℃; for 24h; Inert atmosphere; Sealed tube; chemoselective reaction; | 97% |
| With acetamidine hydrochloride; caesium carbonate; L-proline; copper(l) iodide In N,N-dimethyl-formamide at 120℃; for 10h; | 94% |
| With copper(l) iodide; ammonia; potassium carbonate In water; dimethyl sulfoxide at 100℃; for 12h; | 93% |
-
-
71026-66-9
N-(tert-butoxycarbonyl)-1,4-phenylenediamine

-
-
106-50-3
1,4-phenylenediamine

| Conditions | Yield |
|---|---|
| In various solvent(s) at 150℃; for 0.5h; microwave irradiation; | 97% |
| With 1,1,1,3',3',3'-hexafluoro-propanol at 150℃; for 0.5h; Product distribution / selectivity; Microwave irradiation; | 97% |
| With 1,1,1,3',3',3'-hexafluoro-propanol at 150℃; for 0.5h; Product distribution / selectivity; Microwave irradiation; | 97% |
| With 2,2,2-trifluoroethanol at 150℃; for 3h; Product distribution / selectivity; Microwave irradiation; | 93% |

| Conditions | Yield |
|---|---|
| With chloro-trimethyl-silane; sodium iodide In acetonitrile for 0.166667h; Ambient temperature; | 96% |
| With water for 5h; Inert atmosphere; UV-irradiation; Sealed tube; chemoselective reaction; | 92% |
-
-
105-11-3, 6133-83-1, 6421-98-3
1,4-Benzochinondioxim

-
-
106-50-3
1,4-phenylenediamine

| Conditions | Yield |
|---|---|
| With sodium tetrahydroborate at 20℃; for 0.0333333h; neat (no solvent, solid phase); | 96% |
| With iron oxide; zirconium(IV) chloride; sodium cyanoborohydride In neat (no solvent) at 20℃; for 0.0833333h; | 94% |
-
-
105-11-3, 6133-83-1, 6421-98-3
p-benzoquinone dioxime

-
-
106-50-3
1,4-phenylenediamine

| Conditions | Yield |
|---|---|
| With (pyridine)(tetrahydroborato)zinc In tetrahydrofuran for 1h; Heating; | 95% |
| With fermenting. yeast | |
| With hydrogenchloride; tin(ll) chloride |

| Conditions | Yield |
|---|---|
| With formic acid; zinc In methanol at 20℃; for 0.0833333h; | A 95% B 93% |
| With hydrazine hydrate; aluminium In ethanol Heating; | A 94% B 93% |
| With ammonium acetate; zinc In methanol at 20℃; for 0.05h; | A n/a B 93% |

-
-
106-50-3
1,4-phenylenediamine

| Conditions | Yield |
|---|---|
| With acetic acid; zinc In water at 0℃; for 0.5h; | 95% |

| Conditions | Yield |
|---|---|
| With [Zn(BH4)2(py)] In tetrahydrofuran for 3.1h; Heating; | 94% |
| With mineral acid durch elektrolytische Reduktion; | |
| With hydrogenchloride; tin |
-
-
25811-68-1
N,N,N',N'-tetrakis(trimethylsilyl)-1,4-phenylenediamine

-
-
106-50-3
1,4-phenylenediamine

| Conditions | Yield |
|---|---|
| With hydrogenchloride; water In methanol at 20℃; for 1h; | 94% |
| With hydrogenchloride In diethyl ether |

| Conditions | Yield |
|---|---|
| With ammonium formate; nickel In methanol at 20℃; for 0.0833333h; | 93% |
| With hydrazine hydrate In ethanol Heating; | 93% |
| With water; zinc |

| Conditions | Yield |
|---|---|
| With nickel; hydrazinium monoformate In methanol for 0.0833333h; Heating; | 93% |

| Conditions | Yield |
|---|---|
| With ammonium hydroxide; trans-bis(triphenylphosphine)palladium dichloride; sodium carbonate In neat (no solvent) at 80℃; for 7h; | 93% |
| With ammonium hydroxide; (2-methylacetatobenzyl)triphenylphosphonium hexabromodipalladate(II) at 60℃; for 5h; | 92% |
| With ammonium hydroxide; bis[(2-methylacetatobenzyl)tri(p-tolyl)phosphonium] hexabromodipalladate(II) In neat (no solvent) at 60℃; for 5h; | 90% |

| Conditions | Yield |
|---|---|
| With water; chlorine; sodium hydroxide at 5 - 80℃; for 4.5h; Reagent/catalyst; Hofmann Rearrangement; | 93% |
-
-
65052-89-3
N,N-dibenzyl-4-nitroaniline

-
-
106-50-3
1,4-phenylenediamine

| Conditions | Yield |
|---|---|
| With formic acid; potassium hydroxide In ethanol at 70℃; for 1h; | 93% |
-
-
53821-12-8
benzyl 4-(nitro)phenylcarbamate

-
-
106-50-3
1,4-phenylenediamine

| Conditions | Yield |
|---|---|
| With hydrogen In ethanol at 20℃; under 760.051 Torr; for 5h; | 92% |
| With methanol; sodium tetrahydroborate; nickel(II) chloride hexahydrate at 20℃; for 0.25h; chemoselective reaction; | 86% |

| Conditions | Yield |
|---|---|
| With ammonium hydroxide; copper(II) ferrite at 150℃; under 31503.2 - 36003.6 Torr; for 10h; Catalytic behavior; Pressure; Temperature; Autoclave; | 91.7% |
| With copper(l) iodide; 2,2-[μ-(N,N'-piperazindiyl)dimethyl]-bis(4,6-di-tert-butyl-phenol); ammonia at 150℃; under 31503.2 Torr; for 10h; Reagent/catalyst; Temperature; Pressure; Autoclave; | 90.6% |
| With ammonium hydroxide; 1,10-Phenanthroline; copper(I) bromide at 150℃; under 45004.5 Torr; for 0.00555556h; Reagent/catalyst; Temperature; Pressure; | 90.6% |

-
-
106-50-3
1,4-phenylenediamine

| Conditions | Yield |
|---|---|
| With tris-(trimethylsilyl)silane; 2-hydroxyethanethiol In water at 100℃; for 4h; | 90% |

| Conditions | Yield |
|---|---|
| With Saccharomyces cerevisiae BY In methanol at 20℃; pH=7.0; aq. buffer; Enzymatic reaction; | 90% |

| Conditions | Yield |
|---|---|
| With aluminum oxide for 5h; Milling; | 100% |
| In chloroform at 20℃; Inert atmosphere; | 82% |
| With sodium hydrogen sulfate; silica gel for 0.0666667h; microwave irradiation; | 78% |
-
-
90-02-8
salicylaldehyde

-
-
106-50-3
1,4-phenylenediamine

-
-
119643-02-6
2,2'-{(1E,1'E)-[1,4-phenylenebis(azanylylidene)]bis(methanylylidene)}diphenol

| Conditions | Yield |
|---|---|
| In methanol for 0.5h; Reflux; | 100% |
| In ethanol for 2h; Heating; | 88% |
| With tin(II) chloride dihdyrate In dichloromethane at 20℃; for 6h; | 87.7% |
-
-
98-59-9
p-toluenesulfonyl chloride

-
-
106-50-3
1,4-phenylenediamine

-
-
41595-29-3
N,N'-ditosyl-p-phenylenediamine

| Conditions | Yield |
|---|---|
| With pyridine for 2h; Reflux; | 100% |
| With triethylamine In dichloromethane at 4℃; | 100% |
| With sodium hydroxide In diethyl ether at 0 - 20℃; | 90% |
| With pyridine In tetrahydrofuran at 40℃; for 2h; | 74% |
-
-
24424-99-5
di-tert-butyl dicarbonate

-
-
106-50-3
1,4-phenylenediamine

-
-
71026-66-9
N-(tert-butoxycarbonyl)-1,4-phenylenediamine

| Conditions | Yield |
|---|---|
| With potassium carbonate In tetrahydrofuran at 20℃; | 100% |
| With glycerol at 20℃; for 0.333333h; Green chemistry; chemoselective reaction; | 97% |
| In dichloromethane at 20℃; for 18h; Cooling with ice; | 97% |

-
-
137819-03-5
2,4-diamino-(n-hexadecyloxy)benzene

-
-
106-50-3
1,4-phenylenediamine

- soluble polyimide, inherent viscosity: 0.78 dl/g; monomer(s): 5-(2,5-dioxotetrahydrofuryl)-3-methyl-3-cyclohexene-1,2-dicarboxylic anhydride; p-phenylenediamine; 2,4-diamino-(n-hexadecyloxy)benzene
-
soluble polyimide, inherent viscosity: 0.78 dl/g; monomer(s): 5-(2,5-dioxotetrahydrofuryl)-3-methyl-3-cyclohexene-1,2-dicarboxylic anhydride; p-phenylenediamine; 2,4-diamino-(n-hexadecyloxy)benzene

| Conditions | Yield |
|---|---|
| With isoquinoline In various solvent(s) at 70 - 200℃; | 100% |


-
-
137819-03-5
2,4-diamino-(n-hexadecyloxy)benzene

-
-
106-50-3
1,4-phenylenediamine

- soluble polyimide, inherent viscosity: 0.67 dl/g; monomer(s): 5-(2,5-dioxotetrahydrofuryl)-3-methyl-3-cyclohexene-1,2-dicarboxylic anhydride; p-phenylenediamine; 2,4-diamino-(n-hexadecyloxy)benzene
-
soluble polyimide, inherent viscosity: 0.67 dl/g; monomer(s): 5-(2,5-dioxotetrahydrofuryl)-3-methyl-3-cyclohexene-1,2-dicarboxylic anhydride; p-phenylenediamine; 2,4-diamino-(n-hexadecyloxy)benzene

| Conditions | Yield |
|---|---|
| With isoquinoline In various solvent(s) at 70 - 200℃; | 100% |


-
-
137819-03-5
2,4-diamino-(n-hexadecyloxy)benzene

-
-
106-50-3
1,4-phenylenediamine

- soluble polyimide, inherent viscosity: 0.69 dl/g; monomer(s): 5-(2,5-dioxotetrahydrofuryl)-3-methyl-3-cyclohexene-1,2-dicarboxylic anhydride; p-phenylenediamine; 2,4-diamino-(n-hexadecyloxy)benzene
-
soluble polyimide, inherent viscosity: 0.69 dl/g; monomer(s): 5-(2,5-dioxotetrahydrofuryl)-3-methyl-3-cyclohexene-1,2-dicarboxylic anhydride; p-phenylenediamine; 2,4-diamino-(n-hexadecyloxy)benzene

| Conditions | Yield |
|---|---|
| With isoquinoline In various solvent(s) at 70 - 200℃; | 100% |


-
-
137819-03-5
2,4-diamino-(n-hexadecyloxy)benzene

-
-
106-50-3
1,4-phenylenediamine

- soluble polyimide, inherent viscosity: 0.65 dl/g; monomer(s): 5-(2,5-dioxotetrahydrofuryl)-3-methyl-3-cyclohexene-1,2-dicarboxylic anhydride; p-phenylenediamine; 2,4-diamino-(n-hexadecyloxy)benzene
-
soluble polyimide, inherent viscosity: 0.65 dl/g; monomer(s): 5-(2,5-dioxotetrahydrofuryl)-3-methyl-3-cyclohexene-1,2-dicarboxylic anhydride; p-phenylenediamine; 2,4-diamino-(n-hexadecyloxy)benzene

| Conditions | Yield |
|---|---|
| With isoquinoline In various solvent(s) at 70 - 200℃; | 100% |


-
-
137819-03-5
2,4-diamino-(n-hexadecyloxy)benzene

-
-
106-50-3
1,4-phenylenediamine

- soluble polyimide, inherent viscosity: 0.44 dl/g; monomer(s): 5-(2,5-dioxotetrahydrofuryl)-3-methyl-3-cyclohexene-1,2-dicarboxylic anhydride; p-phenylenediamine; 2,4-diamino-(n-hexadecyloxy)benzene
-
soluble polyimide, inherent viscosity: 0.44 dl/g; monomer(s): 5-(2,5-dioxotetrahydrofuryl)-3-methyl-3-cyclohexene-1,2-dicarboxylic anhydride; p-phenylenediamine; 2,4-diamino-(n-hexadecyloxy)benzene

| Conditions | Yield |
|---|---|
| With isoquinoline In various solvent(s) at 70 - 200℃; | 100% |


-
-
137819-03-5
2,4-diamino-(n-hexadecyloxy)benzene

-
-
106-50-3
1,4-phenylenediamine

- soluble polyimide, inherent viscosity: 0.27 dl/g; monomer(s): 5-(2,5-dioxotetrahydrofuryl)-3-methyl-3-cyclohexene-1,2-dicarboxylic anhydride; p-phenylenediamine; 2,4-diamino-(n-hexadecyloxy)benzene
-
soluble polyimide, inherent viscosity: 0.27 dl/g; monomer(s): 5-(2,5-dioxotetrahydrofuryl)-3-methyl-3-cyclohexene-1,2-dicarboxylic anhydride; p-phenylenediamine; 2,4-diamino-(n-hexadecyloxy)benzene

| Conditions | Yield |
|---|---|
| With isoquinoline In various solvent(s) at 70 - 200℃; | 100% |


-
-
106-50-3
1,4-phenylenediamine

- soluble polyimide, inherent viscosity: 1.42 dl/g; monomer(s): 5-(2,5-dioxotetrahydrofuryl)-3-methyl-3-cyclohexene-1,2-dicarboxylic anhydride; p-phenylenediamine
-
soluble polyimide, inherent viscosity: 1.42 dl/g; monomer(s): 5-(2,5-dioxotetrahydrofuryl)-3-methyl-3-cyclohexene-1,2-dicarboxylic anhydride; p-phenylenediamine

| Conditions | Yield |
|---|---|
| With isoquinoline In various solvent(s) at 70 - 200℃; | 100% |
-
-
50606-95-6
4-hexylbenzoyl chloride

-
-
106-50-3
1,4-phenylenediamine

-
-
1055298-70-8
N-(4-aminophenyl)-4-hexylbenzamide

| Conditions | Yield |
|---|---|
| In dichloromethane at 20℃; for 17h; | 100% |
-
-
24424-99-5
di-tert-butyl dicarbonate

-
-
106-50-3
1,4-phenylenediamine

-
-
121680-23-7
di(tert‐butyl) 1,4‐phenylenedicarbamate

| Conditions | Yield |
|---|---|
| With 1,4-disulfopiperazine-1,4-diium chloride In neat (no solvent) at 20℃; for 0.0833333h; Green chemistry; chemoselective reaction; | 100% |
| With guanidine hydrochloride In ethanol at 35 - 40℃; for 0.333333h; | 98% |
| Stage #1: 1,4-phenylenediamine With caesium carbonate In tetrahydrofuran at 20℃; for 0.5h; Schlenk technique; Stage #2: di-tert-butyl dicarbonate In tetrahydrofuran at 70℃; Schlenk technique; | 90% |
| With triethylamine In dichloromethane at 20℃; | 51% |
| With guanidine hydrochloride In ethanol at 40℃; for 8h; |

| Conditions | Yield |
|---|---|
| Stage #1: C50H30N4NiO4 With 1,2,4-Trichlorobenzene for 1h; Molecular sieve; Reflux; Stage #2: 1,4-phenylenediamine In pyridine at 220℃; for 16h; | 100% |
-
-
77716-16-6
4-tert-butoxycarbonylamino-1-methyl-1H-pyrrole-2-carboxylic acid benzotriazol-1yl ester

-
-
106-50-3
1,4-phenylenediamine

| Conditions | Yield |
|---|---|
| In N,N-dimethyl-formamide at 60℃; for 2h; | 100% |

| Conditions | Yield |
|---|---|
| With 1H-imidazole In N,N-dimethyl-formamide; toluene at 150℃; Inert atmosphere; | 100% |

| Conditions | Yield |
|---|---|
| Stage #1: Pyromellitic dianhydride In water at 80℃; for 1h; Inert atmosphere; Stage #2: 1,4-phenylenediamine In water at 80℃; for 4h; Inert atmosphere; | 100% |

| Conditions | Yield |
|---|---|
| With hydrogenchloride In 1,4-dioxane; methanol for 0.166667h; | 100% |
-
-
106-50-3
1,4-phenylenediamine

| Conditions | Yield |
|---|---|
| With hydrogenchloride; tetrabutylammomium bromide In N,N-dimethyl-formamide at 10℃; for 2.5h; Temperature; Reagent/catalyst; | 99.5% |

| Conditions | Yield |
|---|---|
| With [((5-Me)PyNPPh2)IrACHTUNGTRENUNG(cod)]; potassium tert-butylate In diethylene glycol dimethyl ether at 70℃; for 48h; Inert atmosphere; | 99% |
| With C41H36AsClN3OPRuS; potassium hydroxide In toluene at 100℃; for 12h; | 91% |
| at 120℃; for 48h; Molecular sieve; Green chemistry; | 78% |

| Conditions | Yield |
|---|---|
| In dichloromethane for 1h; | 99% |

| Conditions | Yield |
|---|---|
| With C80H124Cl2N8Nd2O2 In tetrahydrofuran at 60℃; for 0.5h; Reagent/catalyst; Temperature; Time; Schlenk technique; Inert atmosphere; Glovebox; | 99% |
| With [Li(THF)(DME)]3La[μ-η2η1(iPrN)2C(NC6H4p-Cl)]3 at 25℃; for 2h; Inert atmosphere; | 98% |
| [(Me3Si)2N]3Yb(μ-Cl)Li(THF)3 In tetrahydrofuran at 60℃; for 4h; | 97% |
-
-
85-44-9
phthalic anhydride

-
-
106-50-3
1,4-phenylenediamine

-
-
935-79-5
cis-1,2,3,6-tetrahydrophthalic anhydride

| Conditions | Yield |
|---|---|
| In acetone at 20℃; for 12h; | 99% |

Related products
Downstream Products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View














 T,
T, N,
N, T+,
T+, Xn
Xn