Xi'an Xszo Chem Co., Ltd.
1. Factory price and high quality must be guaranteed, base on 8 years of production and R&D experience2. Free samples will be provided,ensure specifications and quality are right for customer3. Customers will receive the most professional technical s
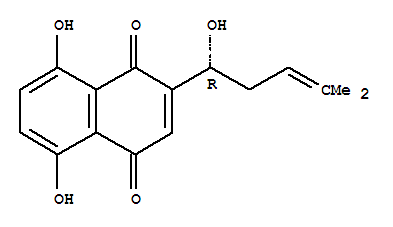
Cas:517-89-5
Min.Order:1 Gram
FOB Price: $0.1
Type:Manufacturers
inquiryGreenutra Resource Inc
Gromwell Root Extract Powder Latin name: Lithospermum erythrorhizon Part used: Root Active ingredient: Shikonin Specification: 10%~98% tested by HPLC Cas No.: 517-89-5 Mol
Cas:517-89-5
Min.Order:1 Kilogram
FOB Price: $12.0
Type:Trading Company
inquiryDayang Chem (Hangzhou) Co.,Ltd.
As a leading manufacturer and supplier of chemicals in China, DayangChem not only supply popular chemicals, but also DayangChem's R&D center offer custom synthesis services. DayangChem can provide different quantities of custom synthesis ch
Cas:517-89-5
Min.Order:1 Kilogram
FOB Price: $3.0
Type:Lab/Research institutions
inquiryKono Chem Co.,Ltd
Specifications Type: Gromwell Root Extract Form: Powder Part: Root Extraction Type: Solvent Extraction Packaging: Bottle, Can, Drum,
Hebei Nengqian Chemical Import and Export Co., LTD
With our good experience, we offer detailed technical support and advice to assist customers. We communicate closely with customers to establish their quality requirements. Consistent Quality Our plant has strict quality control in each manufactu
Cas:517-89-5
Min.Order:1 Kilogram
FOB Price: $1.0 / 10.0
Type:Trading Company
inquiryHenan Tianfu Chemical Co., Ltd.
Henan Tianfu Chemical Co., Ltd. is located in Zhengzhou High-tech Development Zone with import and export license. We passed ISO 9001:2008 in 2009, and won "High-tech Enterprise" by provincial government in 2013.The objective of the com
Cas:517-89-5
Min.Order:1 Gram
FOB Price: $1000.0
Type:Lab/Research institutions
inquiryZhuozhou Wenxi import and Export Co., Ltd
WITH US,YOUR MONEY IN SAFE,YOUR BUSINESS IN SAFE 1)Quick Response Within 12 hours; 2)Quality Guarantee: All products are strictly tested by our QC, confirmed by QA and approved by third party lab in China, USA, Canada, Germany, UK, Italy, France et
Cas:517-89-5
Min.Order:1 Kilogram
FOB Price: $139.0 / 210.0
Type:Trading Company
inquiryHenan Sinotech Import&Export Corporation
Name: Shikonin / ALKANNIN Cas No:517-89-5 Molecular Formula:C16H16O5 Purity:95%min(HPLC) 1% glycerol acetate solution 0.5% aqueous solution Molecular Weight:288.3 Appearance: Dark purple fine Powder Storage:Store in cool and dry place, away from
Shanghai Upbio Tech Co.,Ltd
1.No Less 8 years exporting experience. Clients can 100% received goods 2.Lower Price with higher quality 3,Free sample 4,We are sincerely responsible for the "product quality" and "After Service" Upbio is Specialized
Cas:517-89-5
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryXi'an Faithful Biotech Co., Ltd.
We are the manufacturers and suppliers of API in China, and warehouse in Germany and USA of California, which can quickly and safely deliver to your address 1.High quality and competitive price. 2.Free sample for your evaluation. 3.Promptly delivery
Cas:517-89-5
Min.Order:10 Gram
FOB Price: $8.8
Type:Trading Company
inquiryBaoji Guokang Healthchem co.,ltd
Our company has been in existence for 10 years since its establishment. We have our own unique team. The company integrates independent research and development, production and sales. We have established famous brands at home and abroad. At pres
Cas:517-89-5
Min.Order:100 Gram
FOB Price: $62.0 / 78.9
Type:Trading Company
inquiryQingdao Beluga Import and Export Co., LTD
5,8-Dihydroxy-2-[(1R)-1-hydroxy-4-methyl-pent-3-enyl]naphthalene-1,4-dione CAS:517-89-5 Qingdao Belugas Import and Export Co., Ltd. is a scientific and technological company integrating research and development, production and trade of chemical inte
Cas:517-89-5
Min.Order:1 Gram
Negotiable
Type:Lab/Research institutions
inquiryShandong Hanjiang Chemical Co., Ltd.
Hello, dear friend! I'm Hansen and Allen from China. Welcome to my lookchem mall! The following is a brief introduction of our company's products and services. If you are interested in our products, please contact us by emai
Cas:517-89-5
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryLonwin Chemical Group Limited
5,8-Dihydroxy-2-[(1R)-1-hydroxy-4-methyl-pent-3-enyl]naphthalene-1,4-dione CAS: 517-89-5 Specification product name: gromwell root extract powder appearance: brown yellow fine powder
Cas:517-89-5
Min.Order:1 Kilogram
Negotiable
Type:Other
inquiryHANGZHOU TIANYE CHEMICALS CO., LTD.
We product this chemical more than 10 years . We are very experience to export it to many countries, Our superior & stable quality , competitive price gain warm reception from our customers. Specification : Tokyo Violet;(R)-5,8-Dihydroxy-2-
Cas:517-89-5
Min.Order:1 Gram
Negotiable
Type:Trading Company
inquiryHangzhou Keyingchem Co.,Ltd
Hangzhou KeyingChem Co., Ltd. exported this product to many countries and regions at best price. If you are looking for the material’s manufacturer or supplier in China, KeyingChem is your best choice. Pls contact with us freely for getting det
Cas:517-89-5
Min.Order:0 Metric Ton
Negotiable
Type:Lab/Research institutions
inquiryHangzhou J&H Chemical Co., Ltd.
J&H CHEM R&D center can offer custom synthesis according to the contract research and development services for the fine chemicals, pharmaceutical, biotechnique and some of the other chemicals. J&H CHEM has some Manufacturing base in Jia
SHANGHAI T&W PHARMACEUTICAL CO., LTD.
A substitute for perfluorooctanoic acid, mainly used as a surfactant, dispersant, additive, etc Appearance:White solid or Colorless liquid Purity:99.3 % We will ship the goods in a timely manner as required We can provide relevant documents acc
Ality Chemical Corporation
The above product is Ality Chemical's strong item with best price, good quality and fast supply. Ality Chemical has been focusing on the research and production of this field for over 14 years. At the same time, we are always committed to providi
Shaanxi Cuicheng Biomedical Technology Co., Ltd.
Why Choose Us: 1. Factory direct sales, so we can provide the competitive price and high quality product base on 8 years of production and R&D experience. 2. It is available in stock for quick shipment.Products could be packaged according to cu
Cas:517-89-5
Min.Order:1 Gram
Negotiable
Type:Other
inquiryShanghai Massive Chemical Technology Co., Ltd.
Massive Chemical is certified with ISO9001 and ISO14001 manufacturer for this product. We will offer all documents as requirement for the materials which includes, Certificate of Analysis, Material Safety Data Sheet, and Method of Analysis and
Cas:517-89-5
Min.Order:1 Gram
FOB Price: $1.0
Type:Lab/Research institutions
inquiryZibo Hangyu Biotechnology Development Co., Ltd
Zibo Hangyu Biotechnology Development Co., Ltd is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality pharmaceuticals, intermediates, special chemicals and OLED intermediates and other fine chemi
Cas:517-89-5
Min.Order:10 Gram
FOB Price: $100.0
Type:Lab/Research institutions
inquiryHenan Wentao Chemical Product Co., Ltd.
We are leading fine chemicals supplier in China with ISO certificate, Our main business covers the fields below: 1.Noble Metal Catalysts (Pt.Pd...) 2.Organic Phosphine Ligands (Tert-butyl-phosphine.Cyclohexyl-phosphine...) 3.OLED
Cas:517-89-5
Min.Order:1 Kilogram
FOB Price: $3.0
Type:Lab/Research institutions
inquiryJiangsu Qianyu Molecular Technology Co., LTD.
Our Advantages A. International Top level TechnologyOur company owned biomedicine experts are famous at home and abroad with rich experience in research and development in the field of efficient chiral functional molecules research and development an
Cas:517-89-5
Min.Order:0
Negotiable
Type:Trading Company
inquiryXiamen Jenny Chemical Technology Co., Ltd.
GMP standard, high purity, competitive price, in stock 1. Quick Response: within 6 hours after receiving your email. 2. Quality Guarantee: All products are strictly tested by our QC, confirmed by QA, and approved by a third-party lab in China, USA,
EAST CHEMSOURCES LIMITED
factory?direct?saleAppearance:White Powder Storage:Store In Dry, Cool And Ventilated Place Package:25kg/drum, also according to the clients requirement Application:It is widely used as a thickener, emulsifier and stabilizer Transportation:By Sea Or B
Cas:517-89-5
Min.Order:1 Kilogram
FOB Price: $18.0 / 20.0
Type:Trading Company
inquiryHangzhou Huarong Pharm Co., Ltd.
Hangzhou Huarong Pharm Co., Ltd.established since 2006 , has been actively developing specialty products for Finished Dosages, APIs, Intermediates, and Fine chemicals markets in North America, Europe, Korea, Japan, Mid-East and all over the World. Hu
TAIZHOU ZHENYU BIOTECHNOLOGY CO., LTD
Zhenyu biotech exported this product to many countries and regions at best price. if you are looking for the material's manufacturer or supplier in china, zhenyu biotech is your best choice. pls contact with us freely for getting detailed
Cas:517-89-5
Min.Order:1 Kilogram
FOB Price: $2.0
Type:Lab/Research institutions
inquiryShandong Mopai Biotechnology Co., LTD
Shandong Mopai Biotechnology Co., LTD is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality pharmaceuticals, intermediates, special chemicals and OLED intermediates and other fine chemicals. W
Cas:517-89-5
Min.Order:1 Gram
Negotiable
Type:Lab/Research institutions
inquiryKAISA GROUP INC
1.Applied in food field.it can improve the immune system and prolong life. 2.Appliedin cosmetic field.it can improve the skin care. 3.Applied in pharmaceutical field.it can treat various dieases. 4.Our product quality assurance will make our customer
Cas:517-89-5
Min.Order:1 Metric Ton
FOB Price: $1.5
Type:Trading Company
inquirySynthetic route
-
-
197573-96-9
(1R)-4-methyl-1-(1,4,5,8-tetramethoxynaphthalen-2-yl)pent-3-en-1-ol

-
-
517-89-5
shikonin

| Conditions | Yield |
|---|---|
| Stage #1: (1R)-4-methyl-1-(1,4,5,8-tetramethoxynaphthalen-2-yl)pent-3-en-1-ol With pyridine; dmap; acetic anhydride In dichloromethane at 0 - 20℃; for 0.333333h; Stage #2: With ammonium cerium (IV) nitrate In dichloromethane; water at 20℃; for 0.25h; Further stages; | 85% |
| Multi-step reaction with 3 steps 1.1: dmap; triethylamine / dichloromethane / 0.33 h / 20 °C 1.2: 0.25 h / 20 °C 2.1: dmap; triethylamine; zinc / 2 h / 20 °C 3.1: ammonium cerium (IV) nitrate / acetonitrile / 0.17 h / 20 °C 3.2: 6 h View Scheme | |
| Multi-step reaction with 3 steps 1.1: dmap; triethylamine / dichloromethane / 0.33 h / 20 °C 1.2: 0.25 h / 20 °C 2.1: dmap; triethylamine; zinc / 2 h / 20 °C 3.1: ammonium cerium (IV) nitrate / acetonitrile / 0.17 h / 20 °C 3.2: 6 h View Scheme |
-
-
135504-96-0
(R)-2-(<(1,1-Dimethylethyl)dimethylsilyloxy>-4-methyl-3-pentenyl)-5,8-dihydroxynaphtho-1,4-quinone

-
-
517-89-5
shikonin

| Conditions | Yield |
|---|---|
| With tetrabutyl ammonium fluoride In tetrahydrofuran for 40h; Ambient temperature; | 51% |
-
-
206996-08-9
(R)-4-Methyl-1-(1,3,6,8-tetraoxa-pyren-4-yl)-pent-3-en-1-ol

-
-
517-89-5
shikonin

| Conditions | Yield |
|---|---|
| With lithium perchlorate In water; acetonitrile at 25℃; anodic oxidation, graphite electrodes, 3V; Yield given; |
-
-
443686-76-8
(R,S)-acetic acid 4-acetoxy-3-(1-acetoxy-4-methyl-pent-3-enyl)-5,8-dimethoxynaphthalen-1-yl ester

-
A
-
517-89-5
shikonin

-
B
-
517-88-4, 517-89-5, 11031-58-6, 54952-43-1, 85921-41-1
(S)-5,8-dihydroxy-2-(1-hydroxy-4-methylpent-3-enyl)naphthalene-1,4-dione

| Conditions | Yield |
|---|---|
| Stage #1: (R,S)-acetic acid 4-acetoxy-3-(1-acetoxy-4-methyl-pent-3-enyl)-5,8-dimethoxynaphthalen-1-yl ester With ammonium cerium(IV) nitrate In acetonitrile at 25℃; for 0.25h; Stage #2: With sodium hydroxide at 25℃; for 1h; Title compound not separated from byproducts; |
-
-
24502-78-1, 34232-27-4, 38222-13-8, 54984-93-9, 106295-33-4
acetylshikonin

-
-
517-89-5
shikonin

| Conditions | Yield |
|---|---|
| With sodium hydroxide In methanol |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 6 steps 1.1: 85 percent / CBr4; PPh3; pyridine / CH2Cl2 2.1: tetrahydrofuran / 0.33 h / -20 °C 3.1: n-BuLi; t-BuOH / tetrahydrofuran; hexane / 0.33 h / -78 °C 3.2: 8 percent / tetrahydrofuran; hexane / 3 h 4.1: NaBH4 / methanol / 0.33 h / 0 °C 5.1: 254.8 mg / Et3N; DMAP / CH2Cl2 / 5 h / 25 °C 6.1: aq. CAN / acetonitrile / 0.25 h / 25 °C 6.2: aq. NaOH / 1 h / 25 °C View Scheme | |
| Multi-step reaction with 6 steps 1.1: 85 percent / CBr4; PPh3; pyridine / CH2Cl2 2.1: tetrahydrofuran / 0.33 h / -20 °C 3.1: LDA / tetrahydrofuran / 0.5 h / -78 °C 3.2: 60 percent / tetrahydrofuran / 0.08 h / -78 °C 4.1: NaBH4 / methanol / 0.33 h / 0 °C 5.1: 254.8 mg / Et3N; DMAP / CH2Cl2 / 5 h / 25 °C 6.1: aq. CAN / acetonitrile / 0.25 h / 25 °C 6.2: aq. NaOH / 1 h / 25 °C View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 7 steps 1.1: 90 percent / aq. NaOH / methanol / 8 h / Heating 2.1: 85 percent / CBr4; PPh3; pyridine / CH2Cl2 3.1: tetrahydrofuran / 0.33 h / -20 °C 4.1: n-BuLi; t-BuOH / tetrahydrofuran; hexane / 0.33 h / -78 °C 4.2: 8 percent / tetrahydrofuran; hexane / 3 h 5.1: NaBH4 / methanol / 0.33 h / 0 °C 6.1: 254.8 mg / Et3N; DMAP / CH2Cl2 / 5 h / 25 °C 7.1: aq. CAN / acetonitrile / 0.25 h / 25 °C 7.2: aq. NaOH / 1 h / 25 °C View Scheme | |
| Multi-step reaction with 7 steps 1.1: 90 percent / aq. NaOH / methanol / 8 h / Heating 2.1: 85 percent / CBr4; PPh3; pyridine / CH2Cl2 3.1: tetrahydrofuran / 0.33 h / -20 °C 4.1: LDA / tetrahydrofuran / 0.5 h / -78 °C 4.2: 60 percent / tetrahydrofuran / 0.08 h / -78 °C 5.1: NaBH4 / methanol / 0.33 h / 0 °C 6.1: 254.8 mg / Et3N; DMAP / CH2Cl2 / 5 h / 25 °C 7.1: aq. CAN / acetonitrile / 0.25 h / 25 °C 7.2: aq. NaOH / 1 h / 25 °C View Scheme |
-
-
33698-69-0
6-methyl-hepta-1,5-dien-3-one

-
-
517-89-5
shikonin

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1.1: n-BuLi; t-BuOH / tetrahydrofuran; hexane / 0.33 h / -78 °C 1.2: 8 percent / tetrahydrofuran; hexane / 3 h 2.1: NaBH4 / methanol / 0.33 h / 0 °C 3.1: 254.8 mg / Et3N; DMAP / CH2Cl2 / 5 h / 25 °C 4.1: aq. CAN / acetonitrile / 0.25 h / 25 °C 4.2: aq. NaOH / 1 h / 25 °C View Scheme | |
| Multi-step reaction with 4 steps 1.1: LDA / tetrahydrofuran / 0.5 h / -78 °C 1.2: 60 percent / tetrahydrofuran / 0.08 h / -78 °C 2.1: NaBH4 / methanol / 0.33 h / 0 °C 3.1: 254.8 mg / Et3N; DMAP / CH2Cl2 / 5 h / 25 °C 4.1: aq. CAN / acetonitrile / 0.25 h / 25 °C 4.2: aq. NaOH / 1 h / 25 °C View Scheme |
-
-
145668-25-3
2-(1'-hydroxy-4'-methylpent-3'-en-1'-yl)-5,8-dimethoxy-1,4-naphthaquinone

-
-
517-89-5
shikonin

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1.1: aq. Na2S2O4 / diethyl ether / 0.33 h 2.1: 254.8 mg / Et3N; DMAP / CH2Cl2 / 5 h / 25 °C 3.1: aq. CAN / acetonitrile / 0.25 h / 25 °C 3.2: aq. NaOH / 1 h / 25 °C View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 8 steps 1.1: 86 percent / dimethylformamide / 48 h / 25 °C 2.1: 90 percent / aq. NaOH / methanol / 8 h / Heating 3.1: 85 percent / CBr4; PPh3; pyridine / CH2Cl2 4.1: tetrahydrofuran / 0.33 h / -20 °C 5.1: n-BuLi; t-BuOH / tetrahydrofuran; hexane / 0.33 h / -78 °C 5.2: 8 percent / tetrahydrofuran; hexane / 3 h 6.1: NaBH4 / methanol / 0.33 h / 0 °C 7.1: 254.8 mg / Et3N; DMAP / CH2Cl2 / 5 h / 25 °C 8.1: aq. CAN / acetonitrile / 0.25 h / 25 °C 8.2: aq. NaOH / 1 h / 25 °C View Scheme | |
| Multi-step reaction with 8 steps 1.1: 86 percent / dimethylformamide / 48 h / 25 °C 2.1: 90 percent / aq. NaOH / methanol / 8 h / Heating 3.1: 85 percent / CBr4; PPh3; pyridine / CH2Cl2 4.1: tetrahydrofuran / 0.33 h / -20 °C 5.1: LDA / tetrahydrofuran / 0.5 h / -78 °C 5.2: 60 percent / tetrahydrofuran / 0.08 h / -78 °C 6.1: NaBH4 / methanol / 0.33 h / 0 °C 7.1: 254.8 mg / Et3N; DMAP / CH2Cl2 / 5 h / 25 °C 8.1: aq. CAN / acetonitrile / 0.25 h / 25 °C 8.2: aq. NaOH / 1 h / 25 °C View Scheme |
-
-
206996-05-6
N-methoxy-N,4-dimethylpent-3-enamide

-
-
517-89-5
shikonin

| Conditions | Yield |
|---|---|
| Multi-step reaction with 5 steps 1.1: tetrahydrofuran / 0.33 h / -20 °C 2.1: n-BuLi; t-BuOH / tetrahydrofuran; hexane / 0.33 h / -78 °C 2.2: 8 percent / tetrahydrofuran; hexane / 3 h 3.1: NaBH4 / methanol / 0.33 h / 0 °C 4.1: 254.8 mg / Et3N; DMAP / CH2Cl2 / 5 h / 25 °C 5.1: aq. CAN / acetonitrile / 0.25 h / 25 °C 5.2: aq. NaOH / 1 h / 25 °C View Scheme | |
| Multi-step reaction with 5 steps 1.1: tetrahydrofuran / 0.33 h / -20 °C 2.1: LDA / tetrahydrofuran / 0.5 h / -78 °C 2.2: 60 percent / tetrahydrofuran / 0.08 h / -78 °C 3.1: NaBH4 / methanol / 0.33 h / 0 °C 4.1: 254.8 mg / Et3N; DMAP / CH2Cl2 / 5 h / 25 °C 5.1: aq. CAN / acetonitrile / 0.25 h / 25 °C 5.2: aq. NaOH / 1 h / 25 °C View Scheme |

-
-
517-89-5
shikonin

| Conditions | Yield |
|---|---|
| Multi-step reaction with 6 steps 1.1: (Me3Si)2NNa / tetrahydrofuran / 2 h / -10 °C 1.2: 764 mg / tetrahydrofuran / 2 h / 25 °C 2.1: Amberlyst 15; H2O / tetrahydrofuran / 6 h / 25 °C 3.1: n-BuLi; t-BuOH / tetrahydrofuran; hexane / 0.33 h / -78 °C 3.2: 8 percent / tetrahydrofuran; hexane / 3 h 4.1: NaBH4 / methanol / 0.33 h / 0 °C 5.1: 254.8 mg / Et3N; DMAP / CH2Cl2 / 5 h / 25 °C 6.1: aq. CAN / acetonitrile / 0.25 h / 25 °C 6.2: aq. NaOH / 1 h / 25 °C View Scheme | |
| Multi-step reaction with 6 steps 1.1: (Me3Si)2NNa / tetrahydrofuran / 2 h / -10 °C 1.2: 764 mg / tetrahydrofuran / 2 h / 25 °C 2.1: Amberlyst 15; H2O / tetrahydrofuran / 6 h / 25 °C 3.1: LDA / tetrahydrofuran / 0.5 h / -78 °C 3.2: 60 percent / tetrahydrofuran / 0.08 h / -78 °C 4.1: NaBH4 / methanol / 0.33 h / 0 °C 5.1: 254.8 mg / Et3N; DMAP / CH2Cl2 / 5 h / 25 °C 6.1: aq. CAN / acetonitrile / 0.25 h / 25 °C 6.2: aq. NaOH / 1 h / 25 °C View Scheme |
-
-
443686-72-4
3-(3-methyl-buten-2-yl)-2-vinyl-[1,3]-dioxolane

-
-
517-89-5
shikonin

| Conditions | Yield |
|---|---|
| Multi-step reaction with 5 steps 1.1: Amberlyst 15; H2O / tetrahydrofuran / 6 h / 25 °C 2.1: n-BuLi; t-BuOH / tetrahydrofuran; hexane / 0.33 h / -78 °C 2.2: 8 percent / tetrahydrofuran; hexane / 3 h 3.1: NaBH4 / methanol / 0.33 h / 0 °C 4.1: 254.8 mg / Et3N; DMAP / CH2Cl2 / 5 h / 25 °C 5.1: aq. CAN / acetonitrile / 0.25 h / 25 °C 5.2: aq. NaOH / 1 h / 25 °C View Scheme | |
| Multi-step reaction with 5 steps 1.1: Amberlyst 15; H2O / tetrahydrofuran / 6 h / 25 °C 2.1: LDA / tetrahydrofuran / 0.5 h / -78 °C 2.2: 60 percent / tetrahydrofuran / 0.08 h / -78 °C 3.1: NaBH4 / methanol / 0.33 h / 0 °C 4.1: 254.8 mg / Et3N; DMAP / CH2Cl2 / 5 h / 25 °C 5.1: aq. CAN / acetonitrile / 0.25 h / 25 °C 5.2: aq. NaOH / 1 h / 25 °C View Scheme |
-
-
443686-71-3
[2-(3-methyl-buten-2-yl)-[1,3]-dioxolan-2-yl]-methanol

-
-
517-89-5
shikonin

| Conditions | Yield |
|---|---|
| Multi-step reaction with 7 steps 1.1: (COCl)2; DMSO; Et3N / CH2Cl2 / 1.5 h / -78 - 25 °C 2.1: (Me3Si)2NNa / tetrahydrofuran / 2 h / -10 °C 2.2: 764 mg / tetrahydrofuran / 2 h / 25 °C 3.1: Amberlyst 15; H2O / tetrahydrofuran / 6 h / 25 °C 4.1: n-BuLi; t-BuOH / tetrahydrofuran; hexane / 0.33 h / -78 °C 4.2: 8 percent / tetrahydrofuran; hexane / 3 h 5.1: NaBH4 / methanol / 0.33 h / 0 °C 6.1: 254.8 mg / Et3N; DMAP / CH2Cl2 / 5 h / 25 °C 7.1: aq. CAN / acetonitrile / 0.25 h / 25 °C 7.2: aq. NaOH / 1 h / 25 °C View Scheme | |
| Multi-step reaction with 7 steps 1.1: (COCl)2; DMSO; Et3N / CH2Cl2 / 1.5 h / -78 - 25 °C 2.1: (Me3Si)2NNa / tetrahydrofuran / 2 h / -10 °C 2.2: 764 mg / tetrahydrofuran / 2 h / 25 °C 3.1: Amberlyst 15; H2O / tetrahydrofuran / 6 h / 25 °C 4.1: LDA / tetrahydrofuran / 0.5 h / -78 °C 4.2: 60 percent / tetrahydrofuran / 0.08 h / -78 °C 5.1: NaBH4 / methanol / 0.33 h / 0 °C 6.1: 254.8 mg / Et3N; DMAP / CH2Cl2 / 5 h / 25 °C 7.1: aq. CAN / acetonitrile / 0.25 h / 25 °C 7.2: aq. NaOH / 1 h / 25 °C View Scheme |
-
-
443686-50-8
1-(1,4-dihydroxy-5,8-dimethoxy-naphthalen-2-yl)-4-methyl-pent-3-en-1-one

-
-
517-89-5
shikonin

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1.1: NaBH4 / methanol / 0.33 h / 0 °C 2.1: 254.8 mg / Et3N; DMAP / CH2Cl2 / 5 h / 25 °C 3.1: aq. CAN / acetonitrile / 0.25 h / 25 °C 3.2: aq. NaOH / 1 h / 25 °C View Scheme |
-
-
443686-67-7
2-(1-hydroxy-4-methyl-pent-3-enyl)-5,8-dimethoxy-naphthalene-1,4-diol

-
-
517-89-5
shikonin

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1.1: 254.8 mg / Et3N; DMAP / CH2Cl2 / 5 h / 25 °C 2.1: aq. CAN / acetonitrile / 0.25 h / 25 °C 2.2: aq. NaOH / 1 h / 25 °C View Scheme |
-
-
88051-30-3
2-bromo-1,8:4,5-bis(methylenedioxy)naphthalene

-
-
517-89-5
shikonin

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: 63 percent / t-BuLi / tetrahydrofuran / 1.) -78 deg C, 1 h, 2.) -78 deg C, 1.5 h 2: 1.) (-)-DIP-Cl, 2.) CH3CHO / 1.) THF, -40 to -25 deg C, 3 h, 2.) THF, 0 deg C, 12 h 3: LiClO4 / acetonitrile; H2O / 25 °C / anodic oxidation, graphite electrodes, 3V View Scheme |
-
-
88051-28-9
naphtho[1,8-de:4,5-d'e']bis([1,3]dioxine)

-
-
517-89-5
shikonin

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: 70 percent / NBS / CHCl3 / 12 h / 25 °C 2: 63 percent / t-BuLi / tetrahydrofuran / 1.) -78 deg C, 1 h, 2.) -78 deg C, 1.5 h 3: 1.) (-)-DIP-Cl, 2.) CH3CHO / 1.) THF, -40 to -25 deg C, 3 h, 2.) THF, 0 deg C, 12 h 4: LiClO4 / acetonitrile; H2O / 25 °C / anodic oxidation, graphite electrodes, 3V View Scheme |
-
-
206996-06-7
2-(4-methyl-3-pentene-1-one)-1,8:4,5-bis(methylenedioxy)naphthalene

-
-
517-89-5
shikonin

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 1.) (-)-DIP-Cl, 2.) CH3CHO / 1.) THF, -40 to -25 deg C, 3 h, 2.) THF, 0 deg C, 12 h 2: LiClO4 / acetonitrile; H2O / 25 °C / anodic oxidation, graphite electrodes, 3V View Scheme |
-
-
135504-90-4
(R)-3-Hydroxy-3-(1,4,5,8-tetramethoxy-2-naphthyl)propanoic Acid

-
-
517-89-5
shikonin

| Conditions | Yield |
|---|---|
| Multi-step reaction with 7 steps 1: p-toluenesulfonic acid / 48 h / Ambient temperature 2: 4-(dimethylamino)pyridine, imdazole / dimethylformamide / 72 h / Ambient temperature 3: diisobutylaluminum hydride / CH2Cl2 / 29 h / -78 °C 4: 1) 1.6M n-BuLi / 1) Et2O, 0 deg C, 1.5 h, 2) Et2O, r.t., 24 h 5: 54 percent / 4)2(NO3)6> / acetonitrile; CHCl3; H2O / 1 h / 0 °C 6: 29 percent / AgO, 6 N HNO3 / dioxane / 0.75 h / Ambient temperature 7: 51 percent / (n-C4H9)4NF*3H2O / tetrahydrofuran / 40 h / Ambient temperature View Scheme |
-
-
135523-23-8
Ethyl (R)-3-Hydroxy-3-(1,4,5,8-tetramethoxy-2-naphthyl)propanoate

-
-
517-89-5
shikonin

| Conditions | Yield |
|---|---|
| Multi-step reaction with 6 steps 1: 4-(dimethylamino)pyridine, imdazole / dimethylformamide / 72 h / Ambient temperature 2: diisobutylaluminum hydride / CH2Cl2 / 29 h / -78 °C 3: 1) 1.6M n-BuLi / 1) Et2O, 0 deg C, 1.5 h, 2) Et2O, r.t., 24 h 4: 54 percent / 4)2(NO3)6> / acetonitrile; CHCl3; H2O / 1 h / 0 °C 5: 29 percent / AgO, 6 N HNO3 / dioxane / 0.75 h / Ambient temperature 6: 51 percent / (n-C4H9)4NF*3H2O / tetrahydrofuran / 40 h / Ambient temperature View Scheme |
-
-
135504-95-9
(R)-2-(1-<(1,1-Dimethylethyl)dimethylsilyloxy>-4-methyl-3-pentenyl)-5,8-dimethoxynaphtho-1,4-quinone

-
-
517-89-5
shikonin

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 29 percent / AgO, 6 N HNO3 / dioxane / 0.75 h / Ambient temperature 2: 51 percent / (n-C4H9)4NF*3H2O / tetrahydrofuran / 40 h / Ambient temperature View Scheme |
-
-
135504-92-6
(R)-3-(tert-butyldimethylsilyloxy)-3-(1,4,5,8-tetramethoxynaphthalen-2-yl)propanal

-
-
517-89-5
shikonin

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: 1) 1.6M n-BuLi / 1) Et2O, 0 deg C, 1.5 h, 2) Et2O, r.t., 24 h 2: 54 percent / 4)2(NO3)6> / acetonitrile; CHCl3; H2O / 1 h / 0 °C 3: 29 percent / AgO, 6 N HNO3 / dioxane / 0.75 h / Ambient temperature 4: 51 percent / (n-C4H9)4NF*3H2O / tetrahydrofuran / 40 h / Ambient temperature View Scheme | |
| Multi-step reaction with 3 steps 1.1: n-butyllithium / diethyl ether / 0.5 h / 0 °C / Inert atmosphere 1.2: 2 h / 20 °C 2.1: tetrabutyl ammonium fluoride / tetrahydrofuran / 3 h / 20 °C 3.1: pyridine; dmap; acetic anhydride / dichloromethane / 0.33 h / 0 - 20 °C 3.2: 0.25 h / 20 °C View Scheme |
-
-
135504-93-7
(R)-tert-butyldimethyl-(4-methyl-1-(1,4,5,8-tetramethoxynaphthalen-2-yl)pent-3-enyloxy)silane

-
-
517-89-5
shikonin

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: 54 percent / 4)2(NO3)6> / acetonitrile; CHCl3; H2O / 1 h / 0 °C 2: 29 percent / AgO, 6 N HNO3 / dioxane / 0.75 h / Ambient temperature 3: 51 percent / (n-C4H9)4NF*3H2O / tetrahydrofuran / 40 h / Ambient temperature View Scheme | |
| Multi-step reaction with 2 steps 1.1: tetrabutyl ammonium fluoride / tetrahydrofuran / 3 h / 20 °C 2.1: pyridine; dmap; acetic anhydride / dichloromethane / 0.33 h / 0 - 20 °C 2.2: 0.25 h / 20 °C View Scheme |
-
-
135504-91-5
Ethyl (R)-3-<(1,1-Dimethylethyl)dimethylsilyloxy>-3-(1,4,5,8-tetramethoxy-2-naphthyl)propanoate

-
-
517-89-5
shikonin

| Conditions | Yield |
|---|---|
| Multi-step reaction with 5 steps 1: diisobutylaluminum hydride / CH2Cl2 / 29 h / -78 °C 2: 1) 1.6M n-BuLi / 1) Et2O, 0 deg C, 1.5 h, 2) Et2O, r.t., 24 h 3: 54 percent / 4)2(NO3)6> / acetonitrile; CHCl3; H2O / 1 h / 0 °C 4: 29 percent / AgO, 6 N HNO3 / dioxane / 0.75 h / Ambient temperature 5: 51 percent / (n-C4H9)4NF*3H2O / tetrahydrofuran / 40 h / Ambient temperature View Scheme |
-
-
135504-88-0, 135504-89-1
(1'R)-3-Hydroxy-3-(1,4,5,8-tetramethoxynaphthyl)propanoic Acid 2'-Hydroxy-1',2',2'-triphenylethyl Ester

-
-
517-89-5
shikonin

| Conditions | Yield |
|---|---|
| Multi-step reaction with 8 steps 1: 90 percent / KOH / methanol; H2O / 2.5 h / Heating 2: p-toluenesulfonic acid / 48 h / Ambient temperature 3: 4-(dimethylamino)pyridine, imdazole / dimethylformamide / 72 h / Ambient temperature 4: diisobutylaluminum hydride / CH2Cl2 / 29 h / -78 °C 5: 1) 1.6M n-BuLi / 1) Et2O, 0 deg C, 1.5 h, 2) Et2O, r.t., 24 h 6: 54 percent / 4)2(NO3)6> / acetonitrile; CHCl3; H2O / 1 h / 0 °C 7: 29 percent / AgO, 6 N HNO3 / dioxane / 0.75 h / Ambient temperature 8: 51 percent / (n-C4H9)4NF*3H2O / tetrahydrofuran / 40 h / Ambient temperature View Scheme |

-
A
-
517-89-5
shikonin

-
B
-
517-88-4, 517-89-5, 11031-58-6, 54952-43-1, 85921-41-1
(S)-5,8-dihydroxy-2-(1-hydroxy-4-methylpent-3-enyl)naphthalene-1,4-dione

| Conditions | Yield |
|---|---|
| With methanol; sodium hydroxide at 20℃; for 12h; |
-
-
438036-54-5
ethyl 3-hydroxy-3-(1,4,5,8-tetramethoxynaphthalen-2-yl)propanoate

-
-
517-89-5
shikonin

| Conditions | Yield |
|---|---|
| Multi-step reaction with 10 steps 1.1: sodium hydroxide / methanol; water / 2 h / 20 °C 2.1: dmap; bis(2-oxo-3-oxazolidinyl)phosphane; triethylamine / N,N-dimethyl-formamide / 3 h / 20 °C 3.1: 1H-imidazole; dmap / dichloromethane / 24 h / 0 - 20 °C 4.1: pyridine; phosphorus pentachloride / dichloromethane / 8 h / 0 °C 5.1: dichloromethane / 1 h / -30 - -20 °C 6.1: water / dichloromethane / 12 h / 20 °C 7.1: diisobutylaluminium hydride / dichloromethane; toluene / 12 h / -78 °C 8.1: n-butyllithium / diethyl ether / 0.5 h / 0 °C / Inert atmosphere 8.2: 2 h / 20 °C 9.1: tetrabutyl ammonium fluoride / tetrahydrofuran / 3 h / 20 °C 10.1: pyridine; dmap; acetic anhydride / dichloromethane / 0.33 h / 0 - 20 °C 10.2: 0.25 h / 20 °C View Scheme |

-
-
517-89-5
shikonin

| Conditions | Yield |
|---|---|
| Multi-step reaction with 6 steps 1.1: dichloromethane / 1 h / -30 - -20 °C 2.1: water / dichloromethane / 12 h / 20 °C 3.1: diisobutylaluminium hydride / dichloromethane; toluene / 12 h / -78 °C 4.1: n-butyllithium / diethyl ether / 0.5 h / 0 °C / Inert atmosphere 4.2: 2 h / 20 °C 5.1: tetrabutyl ammonium fluoride / tetrahydrofuran / 3 h / 20 °C 6.1: pyridine; dmap; acetic anhydride / dichloromethane / 0.33 h / 0 - 20 °C 6.2: 0.25 h / 20 °C View Scheme |

-
-
517-89-5
shikonin

| Conditions | Yield |
|---|---|
| Multi-step reaction with 5 steps 1.1: water / dichloromethane / 12 h / 20 °C 2.1: diisobutylaluminium hydride / dichloromethane; toluene / 12 h / -78 °C 3.1: n-butyllithium / diethyl ether / 0.5 h / 0 °C / Inert atmosphere 3.2: 2 h / 20 °C 4.1: tetrabutyl ammonium fluoride / tetrahydrofuran / 3 h / 20 °C 5.1: pyridine; dmap; acetic anhydride / dichloromethane / 0.33 h / 0 - 20 °C 5.2: 0.25 h / 20 °C View Scheme |
-
-
517-89-5
shikonin

-
-
3588-64-5, 5123-55-7, 32150-90-6, 38229-08-2
(R)-2-(1,3-dioxo-1,3-dihydroisoindol-2-yl)-3-phenylpropionic acid

| Conditions | Yield |
|---|---|
| Stage #1: (R)-2-(1,3-dioxo-1,3-dihydroisoindol-2-yl)-3-phenylpropionic acid With dicyclohexyl-carbodiimide In dichloromethane for 0.25h; Inert atmosphere; Cooling with ice; Stage #2: shikonin With dmap In dichloromethane for 12h; Cooling with ice; | 98% |

| Conditions | Yield |
|---|---|
| Stage #1: N-phthaloylphenylalanine With dicyclohexyl-carbodiimide In dichloromethane for 0.25h; Inert atmosphere; Cooling with ice; Stage #2: shikonin With dmap In dichloromethane for 12h; Cooling with ice; | 98% |

| Conditions | Yield |
|---|---|
| Stage #1: Nα-phthaloyl-L-phenylalanine With dicyclohexyl-carbodiimide In dichloromethane for 0.25h; Inert atmosphere; Cooling with ice; Stage #2: shikonin With dmap In dichloromethane for 12h; Cooling with ice; | 97% |

-
-
517-89-5
shikonin

| Conditions | Yield |
|---|---|
| Stage #1: 2-(1,3-dioxooctahydroisoindol-2-yl)propionic acid With dicyclohexyl-carbodiimide In dichloromethane for 0.25h; Inert atmosphere; Cooling with ice; Stage #2: shikonin With dmap In dichloromethane for 12h; Cooling with ice; | 93% |

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide at 60℃; for 6h; | 92.3% |
| Stage #1: shikonin With potassium carbonate; potassium iodide In N,N-dimethyl-formamide at 60℃; for 0.5h; Stage #2: methyl iodide In N,N-dimethyl-formamide for 5h; | 76.6% |
-
-
517-89-5
shikonin

-
-
77-78-1
dimethyl sulfate

-
-
197573-96-9
(1R)-4-methyl-1-(1,4,5,8-tetramethoxynaphthalen-2-yl)pent-3-en-1-ol

| Conditions | Yield |
|---|---|
| With sodium dithionite; tetrabutylammomium bromide; sodium hydroxide In tetrahydrofuran; water at 20℃; for 3h; Inert atmosphere; | 90% |
-
-
517-89-5
shikonin

| Conditions | Yield |
|---|---|
| Stage #1: C11H15NO4 With dicyclohexyl-carbodiimide In dichloromethane for 0.25h; Inert atmosphere; Cooling with ice; Stage #2: shikonin With dmap In dichloromethane for 12h; Cooling with ice; | 90% |

| Conditions | Yield |
|---|---|
| With dmap; dicyclohexyl-carbodiimide In dichloromethane at 20℃; for 1h; Inert atmosphere; | 87.5% |
-
-
517-89-5
shikonin

| Conditions | Yield |
|---|---|
| Stage #1: C11H15NO4 With dicyclohexyl-carbodiimide In dichloromethane for 0.25h; Inert atmosphere; Cooling with ice; Stage #2: shikonin With dmap In dichloromethane for 12h; Cooling with ice; | 87% |
-
-
1196463-67-8
(R)-2-(4-bromophenyl)-4,5-dihydrothiazole-4-carboxylic acid

-
-
517-89-5
shikonin

| Conditions | Yield |
|---|---|
| With dmap; dicyclohexyl-carbodiimide In dichloromethane for 12h; Cooling with ice; | 87% |

| Conditions | Yield |
|---|---|
| Stage #1: 4-Chlorophenoxyacetic acid With dmap; dicyclohexyl-carbodiimide In dichloromethane at 0℃; for 0.25h; Stage #2: shikonin In dichloromethane at 0 - 20℃; for 6h; | 85.2% |
-
-
517-89-5
shikonin

-
-
107-30-2
chloromethyl methyl ether

-
-
1253934-34-7
2-(1-hydroxy-4-methylpent-3-enyl)-5,8-bis-(methoxymethoxy)-naphthalene-1,4-dione

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetone at 20℃; for 3h; | 84.5% |
| Stage #1: shikonin With potassium carbonate In N,N-dimethyl-formamide at 40℃; for 0.5h; Inert atmosphere; Stage #2: chloromethyl methyl ether In N,N-dimethyl-formamide at 40℃; for 1h; Inert atmosphere; | 77.1% |
-
-
517-89-5
shikonin

-
-
64-19-7
acetic acid

-
-
24502-78-1, 34232-27-4, 38222-13-8, 54984-93-9, 106295-33-4
acetylshikonin

| Conditions | Yield |
|---|---|
| With dmap; dicyclohexyl-carbodiimide In dichloromethane at -10 - 0℃; Inert atmosphere; | 83% |
| With dmap; dicyclohexyl-carbodiimide In dichloromethane at 0 - 20℃; for 4h; | |
| With dmap; dicyclohexyl-carbodiimide In dichloromethane for 3h; ice cooling; |
-
-
517-89-5
shikonin

| Conditions | Yield |
|---|---|
| With dmap; dicyclohexyl-carbodiimide In dichloromethane for 12h; Cooling with ice; | 83% |
-
-
517-89-5
shikonin

-
-
802294-64-0
propionic acid

| Conditions | Yield |
|---|---|
| With dmap; dicyclohexyl-carbodiimide In dichloromethane at -10 - 0℃; Inert atmosphere; | 81% |
| With dmap; N-(3-dimethylaminopropyl)-N-ethylcarbodiimide In dichloromethane | 67% |
| With dmap; dicyclohexyl-carbodiimide In dichloromethane at 0 - 20℃; for 4h; |

| Conditions | Yield |
|---|---|
| With dmap; dicyclohexyl-carbodiimide In dichloromethane at -10 - 0℃; Inert atmosphere; | 81% |
| With dmap; N-(3-dimethylaminopropyl)-N-ethylcarbodiimide In dichloromethane | 59% |
| With dmap; dicyclohexyl-carbodiimide In dichloromethane at 0 - 20℃; for 4h; |
-
-
517-89-5
shikonin

| Conditions | Yield |
|---|---|
| With dmap; dicyclohexyl-carbodiimide In dichloromethane for 12h; Cooling with ice; | 80% |

| Conditions | Yield |
|---|---|
| Stage #1: shikonin With dicyclohexyl-carbodiimide In dichloromethane for 0.25h; Inert atmosphere; Cooling with ice; Stage #2: With dmap In dichloromethane for 0.25h; Inert atmosphere; Cooling with ice; Stage #3: (E)-3-phenylacrylic acid In dichloromethane for 12h; Cooling with ice; Inert atmosphere; | 79% |
-
-
517-89-5
shikonin

| Conditions | Yield |
|---|---|
| With dmap; dicyclohexyl-carbodiimide In dichloromethane for 12h; Cooling with ice; | 79% |

| Conditions | Yield |
|---|---|
| Stage #1: shikonin With dicyclohexyl-carbodiimide In dichloromethane for 0.25h; Inert atmosphere; Cooling with ice; Stage #2: With dmap In dichloromethane for 0.25h; Inert atmosphere; Cooling with ice; Stage #3: (E)-3-phenylacrylic acid In dichloromethane for 12h; Cooling with ice; Inert atmosphere; | 76% |

-
-
517-89-5
shikonin

| Conditions | Yield |
|---|---|
| Stage #1: C17H17NO5 With dicyclohexyl-carbodiimide In dichloromethane for 0.25h; Inert atmosphere; Cooling with ice; Stage #2: shikonin With dmap In dichloromethane for 12h; Cooling with ice; | 76% |
-
-
517-89-5
shikonin

-
-
19983-15-4, 116179-34-1, 62096-93-9
(4R)-2-phenyl-4,5-dihydro-1,3-thiazole-4-carboxylic acid

| Conditions | Yield |
|---|---|
| With dmap; dicyclohexyl-carbodiimide In dichloromethane for 12h; Cooling with ice; | 75% |
-
-
406-93-9
4,4,4-trifluorobutyric acid

-
-
517-89-5
shikonin

-
-
1380298-17-8
2-[1-(4,4,4-trifluorobutyryloxy)-4-methyl-3-pentenyl]naphthazarin

| Conditions | Yield |
|---|---|
| With dmap; dicyclohexyl-carbodiimide In dichloromethane at -10 - 0℃; Inert atmosphere; | 74% |
-
-
4653-11-6
2-thiophenebutyric acid

-
-
517-89-5
shikonin

-
-
1397292-84-0
(R)-1-(5,8-dihydroxy-1,4-dioxo-1,4-dihydronaphthalen-2-yl)-4-methylpent-3-enyl 4-(thiophen-2-yl)butanoate

| Conditions | Yield |
|---|---|
| With dmap; dicyclohexyl-carbodiimide In dichloromethane at -10 - 0℃; for 12h; Inert atmosphere; | 74% |
-
-
15687-27-1
ibuprofen

-
-
517-89-5
shikonin

| Conditions | Yield |
|---|---|
| Stage #1: shikonin With dicyclohexyl-carbodiimide In dichloromethane for 0.333333h; Inert atmosphere; Cooling with ice; Stage #2: With dmap In dichloromethane for 0.25h; Cooling with ice; Inert atmosphere; Stage #3: ibuprofen In dichloromethane for 14h; Cooling with ice; Inert atmosphere; | 74% |

| Conditions | Yield |
|---|---|
| Stage #1: shikonin With dicyclohexyl-carbodiimide In dichloromethane for 0.25h; Inert atmosphere; Cooling with ice; Stage #2: With dmap In dichloromethane for 0.25h; Inert atmosphere; Cooling with ice; Stage #3: (E)-3-phenylacrylic acid In dichloromethane for 12h; Cooling with ice; Inert atmosphere; | 73% |
-
-
381-73-7
Difluoroacetic acid

-
-
517-89-5
shikonin

-
-
1380298-10-1
2-[1-(2,2-difluoroacetoxy)-4-methyl-3-pentenyl]naphthazarin

| Conditions | Yield |
|---|---|
| With dmap; dicyclohexyl-carbodiimide In dichloromethane at -10 - 0℃; Inert atmosphere; | 73% |
-
-
527-72-0
2-thiophenylcarboxylic acid

-
-
517-89-5
shikonin

-
-
1397292-79-3
(R)-1-(5,8-dihydroxy-1,4-dioxo-1,4-dihydronaphthalen-2-yl)-4-methylpent-3-enyl thiophene-2-carboxylate

| Conditions | Yield |
|---|---|
| With dmap; dicyclohexyl-carbodiimide In dichloromethane at -10 - 0℃; for 12h; Inert atmosphere; | 73% |
-
-
517-89-5
shikonin

-
-
15254-22-5
(E)-4-(2,5-dimethylphenyl)-4-oxo-2-butenoic acid

| Conditions | Yield |
|---|---|
| Stage #1: (E)-4-(2,5-dimethylphenyl)-4-oxo-2-butenoic acid With dmap; dicyclohexyl-carbodiimide In dichloromethane at 20℃; for 0.333333h; Stage #2: shikonin In dichloromethane | 73% |
Related products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View