Enke Pharma-tech Co.,Ltd. (Cangzhou, China )
Cangzhou Enke Pharma Tech Co.,ltd. is located in Cangzhou City, Hebei province ,where is a famous petroleum chemical industry city in China. Enke Pharma a high-tech enterprise ,and we are dedicated to developing and manufacturing new ap
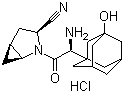
Cas:709031-78-7
Min.Order:1 Kilogram
FOB Price: $5.0 / 10.0
Type:Manufacturers
inquiryChemwill Asia Co., Ltd.
Our main production base is located in Xuzhou industry park. We are certified both to the ISO 9001 and ISO 14001 Standards, have a safety management system in place.Our R&D team masters core technology for process-design of target building block
Cas:709031-78-7
Min.Order:5 Kiloliter
FOB Price: $1.2 / 5.0
Type:Manufacturers
inquiryHebei Nengqian Chemical Import and Export Co., LTD
Our advantages: 1. All inquiries will be replied within 12 hours. 2. Dedication to quality, supply & service. 3. Strictly on selecting raw materials. 4. Reasonable & competitive price, fast lead time. 5. Sample is available for your eva
Cas:709031-78-7
Min.Order:1 Kilogram
FOB Price: $9.0 / 99.0
Type:Trading Company
inquiryHebei Sankai Chemical Technology Co., Ltd
1. Product advantages High purity, all above 98.5%, no impurities after dissolution We will test each batch to ensure quality OEM and private brand services designed for free Various cap colors available We can also provide MT1 peptide powd
Cas:709031-78-7
Min.Order:1 Kilogram
FOB Price: $8.0 / 20.0
Type:Trading Company
inquiryHubei Langyou International Trading Co., Ltd
Advantages: Hubei XinRunde Chemical Co., Ltd is a renowned pharmaceutical manufacturer. We can offer high quality products at competitive price in quick delivery with 100% custom pass guaranteed. Never stop striving to offer our best serv
Cas:709031-78-7
Min.Order:10 Gram
Negotiable
Type:Other
inquiryDayang Chem (Hangzhou) Co.,Ltd.
As a leading manufacturer and supplier of chemicals in China, DayangChem not only supply popular chemicals, but also DayangChem’s R&D center offer custom synthesis according to the contract research and development services for the fine chemicals, ph
Cas:709031-78-7
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryQingdao Beluga Import and Export Co., LTD
Saxagliptin hydrochloride CAS:709031-78-7 Qingdao Belugas Import and Export Co., Ltd. is a scientific and technological company integrating research and development, production and trade of chemical intermediates, specializing in high quality organ
Cas:709031-78-7
Min.Order:1 Gram
Negotiable
Type:Lab/Research institutions
inquiryWuhan Wonda Pharm Limited
1.high quality: quality is life. quality is the most important element for all goods. we have a lab doing research in wuhan china. hplc and nmr is available if needed. 2.reasonable price: we provide high quality products wi
Cas:709031-78-7
Min.Order:10 Gram
Negotiable
Type:Lab/Research institutions
inquiryHenan Wentao Chemical Product Co., Ltd.
Henan Wentao Chemical Product Co.,Ltd is Located in Zhengzhou High-tech Development Zone with import and export license, We passed ISO 9001:2008 as well, Henan Wentao has developed more than 1000 compounds, which are widely used in the fields of prod
Cas:709031-78-7
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryXiamen Hisunny Chemical Co.,Ltd
Best quality & Attractive price & Professional service; Trial & Pilot & Commercial Hisunny Chemical is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality intermediates, specia
Triumph International Development Limilted
Appearance:white or light yellow crystalline powder Storage:Store in a cool,dry place and keep away from direct strong light Package:As customer request Application:Used for research and industrial manufacture. Transportation:As
Cas:709031-78-7
Min.Order:100 Gram
Negotiable
Type:Lab/Research institutions
inquiryTaizhou Crene Biotechnology co.ltd
Our company provides one-stop services of research - development - production for a variety of special prouducts. Not only do we make effective use of our strong technological strength, but also establish of cooperative relations with several well-
Cas:709031-78-7
Min.Order:100 Milligram
Negotiable
Type:Lab/Research institutions
inquiryAfine Chemicals Limited
Our Services 1. New Molecules R&D 2. Own test center HPLC NMR GC LC-MS 3. API and Intermediates from China reputed manufacturers 4. Documents support COA MOA MSDS DMF open part Our advantages 1. Government awarded company. Top 100 enter
Zibo Hangyu Biotechnology Development Co., Ltd
Zibo Hangyu Biotechnology Development Co., Ltd is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality pharmaceuticals, intermediates, special chemicals and OLED intermediates and other fine chemi
Cas:709031-78-7
Min.Order:10 Gram
Negotiable
Type:Lab/Research institutions
inquiryHangzhou Lingrui Chemical Co.,Ltd.
High purity Saxagliptin hydrochloride 709031-78-7 in stock immediately delivery good supplierAppearance:Powder Storage:Dry and ventilated Package:according to customers' requirements Application:APIs Transportation:By air(EMS or EUB or FedEx or TNT e
Cas:709031-78-7
Min.Order:1 Gram
Negotiable
Type:Other
inquiryHANGZHOU YUNUO CHEMICAL CO.,LTD
Superior quality, moderate price & quick delivery. Appearance:white crystalline powder Storage:Stored in cool, dry and ventilation place; Away from fire and heat Package:1kg/bag, 1kg/drum or 25kg/drum or as per your request. Application:Used as
Cas:709031-78-7
Min.Order:1 Kilogram
Negotiable
Type:Trading Company
inquiryZibo Dorne chemical technology co. LTD
Product Details Grade: pharmaceutical grade Purity:99%+ ProductionCapacity: 1000 Kilogram/Month Scope of use: For scientific research only(The product must be used legally) Our Advantage 1. Best quality with competitive price. 2. Quick shipping,
Cas:709031-78-7
Min.Order:1 Gram
Negotiable
Type:Lab/Research institutions
inquiryXiamen Jenny Chemical Technology Co., Ltd.
GMP standard, high purity, competitive price, in stock 1. Quick Response: within 6 hours after receiving your email. 2. Quality Guarantee: All products are strictly tested by our QC, confirmed by QA, and approved by a third-party lab in China, USA,
Cas:709031-78-7
Min.Order:1 Milligram
Negotiable
Type:Trading Company
inquiryHangzhou Huarong Pharm Co., Ltd.
We Huarong Pharm can provide Customized Synthesis & Process R&D & APIs and intermediates Production & Quality Research & Registration Application, especially our GMP validation service which complies with SFDA, FDA, WHO and EU EMPA. O
Shandong Mopai Biotechnology Co., LTD
Shandong Mopai Biotechnology Co., LTD is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality pharmaceuticals, intermediates, special chemicals and OLED intermediates and other fine chemicals. W
Cas:709031-78-7
Min.Order:1 Gram
Negotiable
Type:Lab/Research institutions
inquiryHangzhou Zhongqi chem Co.,Ltd.
Located in Hangzhou National Hi-Tech Industrial Development Zone, zhongqichem is a technical company mainly focus on the Custom synthesis, manufacturing, sales of chemicals to various industries. Benefiting from the outstanding customer service an
Suzhou Health Chemicals Co., Ltd.
High quality,stable supply chain.Appearance:white/off-white or light yellow Storage:Store in cool and dry place, keep away from strong light and heat. Package:aluminum bottle,glass bottle,PTFE bottle,cardboard drum Application:Active Pharmaceutical I
Cas:709031-78-7
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryBluecrystal chem-union
We are a Union of chemistry in China, consists of chemists,engineers, laboratories,factories in China. We organize surplus capacity of R&D and production as well as custom synthesis for chemical products and chemical business project. We are supp
Cas:709031-78-7
Min.Order:0
Negotiable
Type:Trading Company
inquiryGIHI CHEMICALS CO.,LIMITED
Lower price, sample is available,SDS test documents are available,large stock in warehouseAppearance:White powder Storage:Sealed and preserved Package:200/Kilograms Application:Fine chemical intermediates, used as the main raw material for the synthe
Cas:709031-78-7
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryHangzhou Dingyan Chem Co., Ltd
manufacture Application:The refence standard products, for lab use, also we could produce bigger quantity on yr requirement.
Cas:709031-78-7
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryHenan Allgreen Chemical Co.,Ltd
high quality Storage:Sealed, dry, microtherm , avoid light and smell. Package:According to the demand of customer Application:Organic synthesis Transportation:by air or by sea
Hubei Taiho Chemical Co.,LTD
TAIHO Unique Advantages:1.We're factory2.Free samples available3.Commodity inspection can be done4.ISO9001,Kosher certifications5.10 years experiences Storage:Store in cool &dry place Package:aluminium foil bag/fiber can/plastic drum Application:comp
Koning Pharmchem Co., Ltd.
KONING PHARMCHEM CO., LTD. has committed itself to a strategy of providing a unique service for companies involved in the manufacture of pharmaceuticals, healthcare products, food, cosmetics and other fine chemicals. Our products including but not li
BOC Sciences
BOC Sciences provides a wide range of research chemicals and biochemicals including inhibitors, building blocks, GMP Products, impurities and metabolites, APIs for Veterinary, Natural Compounds, ADCs, Stem Cell Molecule and chiral compounds. https:
Hangzhou JINLAN Pharm-Drugs Technology Co., Ltd
Specification:USP/EP Month production ablity:50kg/Month Low price& High quality Manufacturer Appearance:white powder Storage:Room Temperature Application:709031-78-7
Cas:709031-78-7
Min.Order:0 Metric Ton
Negotiable
Type:Trading Company
inquirySynthetic route

-
-
709031-78-7
Saxagliptin hydrochloride

| Conditions | Yield |
|---|---|
| With hydrogenchloride In water; ethyl acetate at 5 - 10℃; | 99% |
| With chloro-trimethyl-silane In acetone for 1h; | |
| Multi-step reaction with 2 steps 1: 20 - 30 °C 2: hydrogenchloride; water / acetone / 1.25 h View Scheme |

-
-
709031-78-7
Saxagliptin hydrochloride

| Conditions | Yield |
|---|---|
| With hydrogenchloride; water In methanol at 25℃; for 3h; | 90% |

-
-
709031-78-7
Saxagliptin hydrochloride

| Conditions | Yield |
|---|---|
| Stage #1: C25H31N3O2 With palladium 10% on activated carbon In methanol at 25 - 30℃; for 2h; Stage #2: With hydrogenchloride In ethyl acetate at 25 - 30℃; for 4h; | 80% |
| Stage #1: C25H31N3O2 With palladium 10% on activated carbon In methanol at 25 - 30℃; for 2h; Stage #2: With hydrogenchloride In water; ethyl acetate at 25 - 30℃; for 4h; | 80% |

-
-
709031-78-7
Saxagliptin hydrochloride

| Conditions | Yield |
|---|---|
| Stage #1: C18H23N5O2 With ammonium chloride; zinc In methanol; water at 25 - 30℃; for 2h; Stage #2: With hydrogenchloride In water at 25 - 30℃; for 4h; | 73% |
| Stage #1: C18H23N5O2 With ammonium chloride; zinc In methanol; water at 25 - 30℃; for 2h; Stage #2: With hydrogenchloride In water; ethyl acetate at 25 - 30℃; for 4h; | 73% |

-
-
709031-78-7
Saxagliptin hydrochloride

| Conditions | Yield |
|---|---|
| In ethanol at 45℃; |
-
-
709031-43-6
3-cyano-(αS)-α-(3-hydroxytricyclo[3.3.1.13,7]dec-1-yl)-β-oxo-(1S,3S,5S)-2-azabicyclo[3.1.0]hexane-2-ethanecarbamic acid 1,1-dimethylethyl ester

-
-
709031-78-7
Saxagliptin hydrochloride

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: hydrogenchloride / isopropyl alcohol; water / 2.08 h / 20 - 65 °C 2: 20 - 30 °C 3: hydrogenchloride; water / acetone / 1.25 h View Scheme | |
| With hydrogenchloride In propan-1-ol; water Product distribution / selectivity; Reflux; | |
| With hydrogenchloride; water In isopropyl alcohol at 20 - 65℃; for 1h; | 50 g |
| With dichloromethane In methanol at 20℃; Inert atmosphere; |
-
-
361442-01-5
tert-butyl [(1S)-2-[(1S,3S,5S)-3-carbamoyl-2-azabicyclo[3.1.0]hex-2-yl]-1-(3-hydroxytricyclo[3.3.1.13'7]dec-1-yl)-2-oxoethyl]carbamate

-
-
709031-78-7
Saxagliptin hydrochloride

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1.1: triethylamine / dichloromethane / 0.5 h / 0 °C 1.2: 0.58 h / 0 - 7 °C 2.1: hydrogenchloride / isopropyl alcohol; water / 2.08 h / 20 - 65 °C 3.1: 20 - 30 °C 4.1: hydrogenchloride; water / acetone / 1.25 h View Scheme | |
| Multi-step reaction with 2 steps 1: triethylamine; trifluoroacetyl chloride / dichloromethane / 0 - 10 °C 2: hydrogenchloride / water; propan-1-ol / Reflux View Scheme | |
| Multi-step reaction with 2 steps 1.1: pyridine; trifluoroacetic anhydride / 0.5 h / Cooling with ice 1.2: 7.55 h / Cooling 2.1: dichloromethane / methanol / 20 °C / Inert atmosphere View Scheme |
-
-
1346445-79-1
(1S,3S,5S)-2-[(2S)-2-propan-2-ylideneamino-2-(3-hydroxy-1-adamantyl)acetyl]-2-azabicyclo[3.1.0]-hexane-3-carbonitrile

-
-
709031-78-7
Saxagliptin hydrochloride

| Conditions | Yield |
|---|---|
| With hydrogenchloride; water In acetone for 1.25h; | |
| With ammonium chloride at 20℃; pH=4.53; |
-
-
361442-05-9
(S)-3-hydroxyadamantylglycine-L-cis-4,5-methanoprolinenitrile TFA salt

-
-
709031-78-7
Saxagliptin hydrochloride

| Conditions | Yield |
|---|---|
| Stage #1: (S)-3-hydroxyadamantylglycine-L-cis-4,5-methanoprolinenitrile TFA salt With sodium hydroxide In water Stage #2: With hydrogenchloride In dichloromethane; water |

-
-
709031-78-7
Saxagliptin hydrochloride

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1.1: trifluoroacetic anhydride; 3-pyridinecarboxylic acid ethyl ester / ethyl acetate / 6 h / 0 - 5 °C 1.2: 2 h / 25 °C 2.1: 5%-palladium/activated carbon; hydrogen / acetonitrile / 4 h / 20 - 25 °C 3.1: hydrogenchloride / water; ethyl acetate / 5 - 10 °C View Scheme |

-
-
709031-78-7
Saxagliptin hydrochloride

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 5%-palladium/activated carbon; hydrogen / acetonitrile / 4 h / 20 - 25 °C 2: hydrogenchloride / water; ethyl acetate / 5 - 10 °C View Scheme |

-
-
709031-78-7
Saxagliptin hydrochloride

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: N-(3-dimethylaminopropyl)-N-ethylcarbodiimide; benzotriazol-1-ol; N-ethyl-N,N-diisopropylamine / ethyl acetate; acetonitrile / 8.25 h / 20 °C 2: 5%-palladium/activated carbon; hydrogen / acetonitrile / 4 h / 20 - 25 °C 3: hydrogenchloride / water; ethyl acetate / 5 - 10 °C View Scheme | |
| Multi-step reaction with 4 steps 1.1: N-(3-dimethylaminopropyl)-N-ethylcarbodiimide; benzotriazol-1-ol; N-ethyl-N,N-diisopropylamine / N,N-dimethyl-formamide / 4.25 h / 20 °C 2.1: trifluoroacetic anhydride; 3-pyridinecarboxylic acid ethyl ester / ethyl acetate / 6 h / 0 - 5 °C 2.2: 2 h / 25 °C 3.1: 5%-palladium/activated carbon; hydrogen / acetonitrile / 4 h / 20 - 25 °C 4.1: hydrogenchloride / water; ethyl acetate / 5 - 10 °C View Scheme |
-
-
865999-64-0
(1S)-α-amino-3-hydroxyadamantane-acetic acid hydrochloride

-
-
709031-78-7
Saxagliptin hydrochloride

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: sodium hydroxide / water / 4 h / 25 - 30 °C / pH 8.5 - 9.5 2: N-(3-dimethylaminopropyl)-N-ethylcarbodiimide; benzotriazol-1-ol; N-ethyl-N,N-diisopropylamine / ethyl acetate; acetonitrile / 8.25 h / 20 °C 3: 5%-palladium/activated carbon; hydrogen / acetonitrile / 4 h / 20 - 25 °C 4: hydrogenchloride / water; ethyl acetate / 5 - 10 °C View Scheme | |
| Multi-step reaction with 5 steps 1.1: sodium hydroxide / water / 4 h / 25 - 30 °C / pH 8.5 - 9.5 2.1: N-(3-dimethylaminopropyl)-N-ethylcarbodiimide; benzotriazol-1-ol; N-ethyl-N,N-diisopropylamine / N,N-dimethyl-formamide / 4.25 h / 20 °C 3.1: trifluoroacetic anhydride; 3-pyridinecarboxylic acid ethyl ester / ethyl acetate / 6 h / 0 - 5 °C 3.2: 2 h / 25 °C 4.1: 5%-palladium/activated carbon; hydrogen / acetonitrile / 4 h / 20 - 25 °C 5.1: hydrogenchloride / water; ethyl acetate / 5 - 10 °C View Scheme | |
| Multi-step reaction with 4 steps 1.1: triethylamine / dichloromethane / 4 h / 15 - 35 °C 2.1: benzotriazol-1-ol; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride; triethylamine / dichloromethane / 12 h / 5 - 35 °C 3.1: pyridine; trifluoroacetic anhydride / tetrahydrofuran / 3.5 h / 0 - 10 °C 3.2: 3.5 h / 25 °C / pH 10 - 11 4.1: water; hydrogenchloride / methanol / 3 h / 25 °C View Scheme |
-
-
929106-57-0
(1S,3S,5S)-2-azabicyclo[3.1.0]hexane-3-carbonitrile 4-methylbenzenesulfonate

-
-
709031-78-7
Saxagliptin hydrochloride

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: N-(3-dimethylaminopropyl)-N-ethylcarbodiimide; benzotriazol-1-ol; N-ethyl-N,N-diisopropylamine / ethyl acetate; acetonitrile / 8.25 h / 20 °C 2: 5%-palladium/activated carbon; hydrogen / acetonitrile / 4 h / 20 - 25 °C 3: hydrogenchloride / water; ethyl acetate / 5 - 10 °C View Scheme |

-
-
709031-78-7
Saxagliptin hydrochloride

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1.1: N-(3-dimethylaminopropyl)-N-ethylcarbodiimide; benzotriazol-1-ol; N-ethyl-N,N-diisopropylamine / N,N-dimethyl-formamide / 4.25 h / 20 °C 2.1: trifluoroacetic anhydride; 3-pyridinecarboxylic acid ethyl ester / ethyl acetate / 6 h / 0 - 5 °C 2.2: 2 h / 25 °C 3.1: 5%-palladium/activated carbon; hydrogen / acetonitrile / 4 h / 20 - 25 °C 4.1: hydrogenchloride / water; ethyl acetate / 5 - 10 °C View Scheme |

-
-
709031-78-7
Saxagliptin hydrochloride

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1.1: benzotriazol-1-ol / ethyl acetate; acetonitrile / 0.17 h / 25 - 30 °C / Inert atmosphere 1.2: 0.17 h / 25 - 30 °C 1.3: 0 - 30 °C / Inert atmosphere 2.1: pyridine; trifluoroacetic anhydride / ethyl acetate / 0 - 5 °C 2.2: 3 h / 25 - 45 °C 3.1: zinc; ammonium chloride / water; methanol / 2 h / 25 - 30 °C 3.2: 4 h / 25 - 30 °C View Scheme | |
| Multi-step reaction with 4 steps 1.1: (S)-1-phenyl-ethylamine / acetone / 7 h / 25 - 65 °C 2.1: benzotriazol-1-ol; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride / ethyl acetate; acetonitrile / 0.25 h / 20 °C 2.2: 4 h / 0 - 20 °C 3.1: pyridine; trifluoroacetic anhydride / ethyl acetate / 0 - 5 °C 3.2: 3 h / 25 - 45 °C 4.1: zinc; ammonium chloride / water; methanol / 2 h / 25 - 30 °C 4.2: 4 h / 25 - 30 °C View Scheme | |
| Multi-step reaction with 4 steps 1.1: (S)-1-phenyl-ethylamine / acetone / 7 h / 25 - 65 °C 1.2: 2 h / 25 - 30 °C 2.1: 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride; benzotriazol-1-ol; N-ethyl-N,N-diisopropylamine / acetonitrile; ethyl acetate / 4 h / 0 - 20 °C 3.1: pyridine; trifluoroacetic anhydride / ethyl acetate / 0 - 5 °C 3.2: 3 h / 25 - 45 °C 4.1: zinc; ammonium chloride / water; methanol / 2 h / 25 - 30 °C 4.2: 4 h / 25 - 30 °C View Scheme |

-
-
709031-78-7
Saxagliptin hydrochloride

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1.1: benzotriazol-1-ol; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride / ethyl acetate; acetonitrile / 0.25 h / 20 °C 1.2: 4 h / 0 - 20 °C 2.1: pyridine; trifluoroacetic anhydride / ethyl acetate / 0 - 5 °C 2.2: 3 h / 25 - 45 °C 3.1: zinc; ammonium chloride / water; methanol / 2 h / 25 - 30 °C 3.2: 4 h / 25 - 30 °C View Scheme | |
| Multi-step reaction with 3 steps 1.1: 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride; benzotriazol-1-ol; N-ethyl-N,N-diisopropylamine / acetonitrile; ethyl acetate / 4 h / 0 - 20 °C 2.1: pyridine; trifluoroacetic anhydride / ethyl acetate / 0 - 5 °C 2.2: 3 h / 25 - 45 °C 3.1: zinc; ammonium chloride / water; methanol / 2 h / 25 - 30 °C 3.2: 4 h / 25 - 30 °C View Scheme |

-
-
709031-78-7
Saxagliptin hydrochloride

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1.1: pyridine; trifluoroacetic anhydride / ethyl acetate / 0 - 5 °C 1.2: 3 h / 25 - 45 °C 2.1: zinc; ammonium chloride / water; methanol / 2 h / 25 - 30 °C 2.2: 4 h / 25 - 30 °C View Scheme | |
| Multi-step reaction with 2 steps 1.1: pyridine; trifluoroacetic anhydride / ethyl acetate / 0 - 5 °C 1.2: 3 h / 25 - 45 °C 2.1: zinc; ammonium chloride / water; methanol / 2 h / 25 - 30 °C 2.2: 4 h / 25 - 30 °C View Scheme |

-
-
709031-78-7
Saxagliptin hydrochloride

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1.1: (S)-1-phenyl-ethylamine / methanol / 7 h / 25 - 65 °C 2.1: benzotriazol-1-ol; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride / ethyl acetate; acetonitrile / 0.25 h / 20 °C 2.2: 4 h / 0 - 20 °C 3.1: pyridine; trifluoroacetic anhydride / ethyl acetate / 0 - 5 °C 3.2: 3 h / 25 - 45 °C 4.1: palladium 10% on activated carbon / methanol / 2 h / 25 - 30 °C 4.2: 4 h / 25 - 30 °C View Scheme | |
| Multi-step reaction with 4 steps 1.1: (S)-1-phenyl-ethylamine / methanol / 7 h / 25 - 65 °C 1.2: 1 h / 25 - 30 °C 2.1: 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride; benzotriazol-1-ol; N-ethyl-N,N-diisopropylamine / acetonitrile; ethyl acetate / 4 h / 0 - 20 °C 3.1: pyridine; trifluoroacetic anhydride / ethyl acetate / 0 - 5 °C 3.2: 3 h / 25 - 45 °C 4.1: palladium 10% on activated carbon / methanol / 2 h / 25 - 30 °C 4.2: 4 h / 25 - 30 °C View Scheme |

-
-
709031-78-7
Saxagliptin hydrochloride

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1.1: benzotriazol-1-ol; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride / ethyl acetate; acetonitrile / 0.25 h / 20 °C 1.2: 4 h / 0 - 20 °C 2.1: pyridine; trifluoroacetic anhydride / ethyl acetate / 0 - 5 °C 2.2: 3 h / 25 - 45 °C 3.1: palladium 10% on activated carbon / methanol / 2 h / 25 - 30 °C 3.2: 4 h / 25 - 30 °C View Scheme | |
| Multi-step reaction with 3 steps 1.1: 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride; benzotriazol-1-ol; N-ethyl-N,N-diisopropylamine / acetonitrile; ethyl acetate / 4 h / 0 - 20 °C 2.1: pyridine; trifluoroacetic anhydride / ethyl acetate / 0 - 5 °C 2.2: 3 h / 25 - 45 °C 3.1: palladium 10% on activated carbon / methanol / 2 h / 25 - 30 °C 3.2: 4 h / 25 - 30 °C View Scheme |

-
-
709031-78-7
Saxagliptin hydrochloride

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1.1: pyridine; trifluoroacetic anhydride / ethyl acetate / 0 - 5 °C 1.2: 3 h / 25 - 45 °C 2.1: palladium 10% on activated carbon / methanol / 2 h / 25 - 30 °C 2.2: 4 h / 25 - 30 °C View Scheme |
-
-
4942-47-6
(adamant-1-yl)-acetic acid

-
-
709031-78-7
Saxagliptin hydrochloride

| Conditions | Yield |
|---|---|
| Multi-step reaction with 6 steps 1.1: thionyl chloride / 1.5 h / 20 °C / Inert atmosphere 1.2: 3 h / 25 - 75 °C 2.1: sulfuric acid; nitric acid / water / 0 - 10 °C 3.1: tetrabutylammomium bromide; sodium azide / water; toluene / 25 - 80 °C 4.1: benzotriazol-1-ol / ethyl acetate; acetonitrile / 0.17 h / 25 - 30 °C / Inert atmosphere 4.2: 0.17 h / 25 - 30 °C 4.3: 0 - 30 °C / Inert atmosphere 5.1: pyridine; trifluoroacetic anhydride / ethyl acetate / 0 - 5 °C 5.2: 3 h / 25 - 45 °C 6.1: zinc; ammonium chloride / water; methanol / 2 h / 25 - 30 °C 6.2: 4 h / 25 - 30 °C View Scheme | |
| Multi-step reaction with 7 steps 1.1: thionyl chloride / 1.5 h / 20 °C / Inert atmosphere 1.2: 3 h / 25 - 75 °C 2.1: sulfuric acid; nitric acid / water / 0 - 10 °C 3.1: tetrabutylammomium bromide; sodium azide / water; toluene / 25 - 80 °C 4.1: (S)-1-phenyl-ethylamine / acetone / 7 h / 25 - 65 °C 5.1: benzotriazol-1-ol; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride / ethyl acetate; acetonitrile / 0.25 h / 20 °C 5.2: 4 h / 0 - 20 °C 6.1: pyridine; trifluoroacetic anhydride / ethyl acetate / 0 - 5 °C 6.2: 3 h / 25 - 45 °C 7.1: zinc; ammonium chloride / water; methanol / 2 h / 25 - 30 °C 7.2: 4 h / 25 - 30 °C View Scheme | |
| Multi-step reaction with 7 steps 1.1: thionyl chloride / 1.5 h / 20 °C / Inert atmosphere 1.2: 3 h / 25 - 75 °C 2.1: sulfuric acid; nitric acid / water / 0 - 10 °C 3.1: 3 h / 110 °C 4.1: (S)-1-phenyl-ethylamine / methanol / 7 h / 25 - 65 °C 5.1: benzotriazol-1-ol; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride / ethyl acetate; acetonitrile / 0.25 h / 20 °C 5.2: 4 h / 0 - 20 °C 6.1: pyridine; trifluoroacetic anhydride / ethyl acetate / 0 - 5 °C 6.2: 3 h / 25 - 45 °C 7.1: palladium 10% on activated carbon / methanol / 2 h / 25 - 30 °C 7.2: 4 h / 25 - 30 °C View Scheme | |
| Multi-step reaction with 7 steps 1.1: thionyl chloride / 1.5 h / 20 °C / Inert atmosphere 1.2: 3 h / 25 - 75 °C 1.3: 3 h / 0 - 75 °C 2.1: sulfuric acid; nitric acid / 0 - 10 °C 2.2: 2 h / 0 - 10 °C 3.1: 3 h / 110 °C 4.1: (S)-1-phenyl-ethylamine / methanol / 7 h / 25 - 65 °C 4.2: 1 h / 25 - 30 °C 5.1: 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride; benzotriazol-1-ol; N-ethyl-N,N-diisopropylamine / acetonitrile; ethyl acetate / 4 h / 0 - 20 °C 6.1: pyridine; trifluoroacetic anhydride / ethyl acetate / 0 - 5 °C 6.2: 3 h / 25 - 45 °C 7.1: palladium 10% on activated carbon / methanol / 2 h / 25 - 30 °C 7.2: 4 h / 25 - 30 °C View Scheme | |
| Multi-step reaction with 7 steps 1.1: thionyl chloride / 1.5 h / 20 °C / Inert atmosphere 1.2: 3 h / 25 - 75 °C 1.3: 3 h / 0 - 75 °C 2.1: sulfuric acid; nitric acid / 0 - 10 °C 2.2: 2 h / 0 - 10 °C 3.1: tetrabutylammomium bromide; sodium azide / toluene; water / 25 - 80 °C 4.1: (S)-1-phenyl-ethylamine / acetone / 7 h / 25 - 65 °C 4.2: 2 h / 25 - 30 °C 5.1: 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride; benzotriazol-1-ol; N-ethyl-N,N-diisopropylamine / acetonitrile; ethyl acetate / 4 h / 0 - 20 °C 6.1: pyridine; trifluoroacetic anhydride / ethyl acetate / 0 - 5 °C 6.2: 3 h / 25 - 45 °C 7.1: zinc; ammonium chloride / water; methanol / 2 h / 25 - 30 °C 7.2: 4 h / 25 - 30 °C View Scheme |
-
-
59768-70-6
2-(1-adamantyl)-2-bromoacetic acid

-
-
709031-78-7
Saxagliptin hydrochloride

| Conditions | Yield |
|---|---|
| Multi-step reaction with 5 steps 1.1: sulfuric acid; nitric acid / water / 0 - 10 °C 2.1: tetrabutylammomium bromide; sodium azide / water; toluene / 25 - 80 °C 3.1: benzotriazol-1-ol / ethyl acetate; acetonitrile / 0.17 h / 25 - 30 °C / Inert atmosphere 3.2: 0.17 h / 25 - 30 °C 3.3: 0 - 30 °C / Inert atmosphere 4.1: pyridine; trifluoroacetic anhydride / ethyl acetate / 0 - 5 °C 4.2: 3 h / 25 - 45 °C 5.1: zinc; ammonium chloride / water; methanol / 2 h / 25 - 30 °C 5.2: 4 h / 25 - 30 °C View Scheme | |
| Multi-step reaction with 6 steps 1.1: sulfuric acid; nitric acid / water / 0 - 10 °C 2.1: tetrabutylammomium bromide; sodium azide / water; toluene / 25 - 80 °C 3.1: (S)-1-phenyl-ethylamine / acetone / 7 h / 25 - 65 °C 4.1: benzotriazol-1-ol; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride / ethyl acetate; acetonitrile / 0.25 h / 20 °C 4.2: 4 h / 0 - 20 °C 5.1: pyridine; trifluoroacetic anhydride / ethyl acetate / 0 - 5 °C 5.2: 3 h / 25 - 45 °C 6.1: zinc; ammonium chloride / water; methanol / 2 h / 25 - 30 °C 6.2: 4 h / 25 - 30 °C View Scheme | |
| Multi-step reaction with 6 steps 1.1: sulfuric acid; nitric acid / water / 0 - 10 °C 2.1: 3 h / 110 °C 3.1: (S)-1-phenyl-ethylamine / methanol / 7 h / 25 - 65 °C 4.1: benzotriazol-1-ol; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride / ethyl acetate; acetonitrile / 0.25 h / 20 °C 4.2: 4 h / 0 - 20 °C 5.1: pyridine; trifluoroacetic anhydride / ethyl acetate / 0 - 5 °C 5.2: 3 h / 25 - 45 °C 6.1: palladium 10% on activated carbon / methanol / 2 h / 25 - 30 °C 6.2: 4 h / 25 - 30 °C View Scheme | |
| Multi-step reaction with 6 steps 1.1: sulfuric acid; nitric acid / 0 - 10 °C 1.2: 2 h / 0 - 10 °C 2.1: 3 h / 110 °C 3.1: (S)-1-phenyl-ethylamine / methanol / 7 h / 25 - 65 °C 3.2: 1 h / 25 - 30 °C 4.1: 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride; benzotriazol-1-ol; N-ethyl-N,N-diisopropylamine / acetonitrile; ethyl acetate / 4 h / 0 - 20 °C 5.1: pyridine; trifluoroacetic anhydride / ethyl acetate / 0 - 5 °C 5.2: 3 h / 25 - 45 °C 6.1: palladium 10% on activated carbon / methanol / 2 h / 25 - 30 °C 6.2: 4 h / 25 - 30 °C View Scheme | |
| Multi-step reaction with 6 steps 1.1: sulfuric acid; nitric acid / 0 - 10 °C 1.2: 2 h / 0 - 10 °C 2.1: tetrabutylammomium bromide; sodium azide / toluene; water / 25 - 80 °C 3.1: (S)-1-phenyl-ethylamine / acetone / 7 h / 25 - 65 °C 3.2: 2 h / 25 - 30 °C 4.1: 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride; benzotriazol-1-ol; N-ethyl-N,N-diisopropylamine / acetonitrile; ethyl acetate / 4 h / 0 - 20 °C 5.1: pyridine; trifluoroacetic anhydride / ethyl acetate / 0 - 5 °C 5.2: 3 h / 25 - 45 °C 6.1: zinc; ammonium chloride / water; methanol / 2 h / 25 - 30 °C 6.2: 4 h / 25 - 30 °C View Scheme |

-
-
709031-78-7
Saxagliptin hydrochloride

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1.1: tetrabutylammomium bromide; sodium azide / water; toluene / 25 - 80 °C 2.1: benzotriazol-1-ol / ethyl acetate; acetonitrile / 0.17 h / 25 - 30 °C / Inert atmosphere 2.2: 0.17 h / 25 - 30 °C 2.3: 0 - 30 °C / Inert atmosphere 3.1: pyridine; trifluoroacetic anhydride / ethyl acetate / 0 - 5 °C 3.2: 3 h / 25 - 45 °C 4.1: zinc; ammonium chloride / water; methanol / 2 h / 25 - 30 °C 4.2: 4 h / 25 - 30 °C View Scheme | |
| Multi-step reaction with 5 steps 1.1: tetrabutylammomium bromide; sodium azide / water; toluene / 25 - 80 °C 2.1: (S)-1-phenyl-ethylamine / acetone / 7 h / 25 - 65 °C 3.1: benzotriazol-1-ol; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride / ethyl acetate; acetonitrile / 0.25 h / 20 °C 3.2: 4 h / 0 - 20 °C 4.1: pyridine; trifluoroacetic anhydride / ethyl acetate / 0 - 5 °C 4.2: 3 h / 25 - 45 °C 5.1: zinc; ammonium chloride / water; methanol / 2 h / 25 - 30 °C 5.2: 4 h / 25 - 30 °C View Scheme | |
| Multi-step reaction with 5 steps 1.1: 3 h / 110 °C 2.1: (S)-1-phenyl-ethylamine / methanol / 7 h / 25 - 65 °C 3.1: benzotriazol-1-ol; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride / ethyl acetate; acetonitrile / 0.25 h / 20 °C 3.2: 4 h / 0 - 20 °C 4.1: pyridine; trifluoroacetic anhydride / ethyl acetate / 0 - 5 °C 4.2: 3 h / 25 - 45 °C 5.1: palladium 10% on activated carbon / methanol / 2 h / 25 - 30 °C 5.2: 4 h / 25 - 30 °C View Scheme | |
| Multi-step reaction with 5 steps 1.1: tetrabutylammomium bromide; sodium azide / toluene; water / 25 - 80 °C 2.1: (S)-1-phenyl-ethylamine / acetone / 7 h / 25 - 65 °C 2.2: 2 h / 25 - 30 °C 3.1: 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride; benzotriazol-1-ol; N-ethyl-N,N-diisopropylamine / acetonitrile; ethyl acetate / 4 h / 0 - 20 °C 4.1: pyridine; trifluoroacetic anhydride / ethyl acetate / 0 - 5 °C 4.2: 3 h / 25 - 45 °C 5.1: zinc; ammonium chloride / water; methanol / 2 h / 25 - 30 °C 5.2: 4 h / 25 - 30 °C View Scheme | |
| Multi-step reaction with 5 steps 1.1: 3 h / 110 °C 2.1: (S)-1-phenyl-ethylamine / methanol / 7 h / 25 - 65 °C 2.2: 1 h / 25 - 30 °C 3.1: 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride; benzotriazol-1-ol; N-ethyl-N,N-diisopropylamine / acetonitrile; ethyl acetate / 4 h / 0 - 20 °C 4.1: pyridine; trifluoroacetic anhydride / ethyl acetate / 0 - 5 °C 4.2: 3 h / 25 - 45 °C 5.1: palladium 10% on activated carbon / methanol / 2 h / 25 - 30 °C 5.2: 4 h / 25 - 30 °C View Scheme |
-
-
361442-00-4
(αS)-α-[[(1,1-dimethylethoxy)carbonyl]-amino]-3-hydroxytricyclo[3.3.1.13,7]decane-1-acetic acid

-
-
709031-78-7
Saxagliptin hydrochloride

| Conditions | Yield |
|---|---|
| Multi-step reaction with 5 steps 1.1: hydrogenchloride / isopropyl alcohol; dichloromethane / 8 h / 10 - 35 °C 2.1: triethylamine / dichloromethane / 4 h / 15 - 35 °C 3.1: benzotriazol-1-ol; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride; triethylamine / dichloromethane / 12 h / 5 - 35 °C 4.1: pyridine; trifluoroacetic anhydride / tetrahydrofuran / 3.5 h / 0 - 10 °C 4.2: 3.5 h / 25 °C / pH 10 - 11 5.1: water; hydrogenchloride / methanol / 3 h / 25 °C View Scheme | |
| Multi-step reaction with 3 steps 1.1: methanesulfonyl chloride; N-ethyl-N,N-diisopropylamine / tetrahydrofuran / Cooling with acetone-dry ice 1.2: 20 °C 2.1: pyridine; trifluoroacetic anhydride / 0.5 h / Cooling with ice 2.2: 7.55 h / Cooling 3.1: dichloromethane / methanol / 20 °C / Inert atmosphere View Scheme |

-
-
709031-78-7
Saxagliptin hydrochloride

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1.1: benzotriazol-1-ol; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride; triethylamine / dichloromethane / 12 h / 5 - 35 °C 2.1: pyridine; trifluoroacetic anhydride / tetrahydrofuran / 3.5 h / 0 - 10 °C 2.2: 3.5 h / 25 °C / pH 10 - 11 3.1: water; hydrogenchloride / methanol / 3 h / 25 °C View Scheme |
-
-
361440-67-7
[1S-(1α,3β,5α)]-3-(aminocarbonyl)-2-azabicyclo[3.1.0]hexane-2-carboxylic acid 1,1-dimethylethyl ester

-
-
709031-78-7
Saxagliptin hydrochloride

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1.1: isopropyl alcohol / 5 h / 60 - 70 °C 2.1: dichloromethane / 0.25 h / 25 - 35 °C 2.2: 13.25 h / 5 - 35 °C 3.1: pyridine; trifluoroacetic anhydride / tetrahydrofuran / 3.5 h / 0 - 10 °C 3.2: 3.5 h / 25 °C / pH 10 - 11 4.1: water; hydrogenchloride / methanol / 3 h / 25 °C View Scheme | |
| Multi-step reaction with 4 steps 1.1: isopropyl alcohol / 5 h / 60 - 70 °C 2.1: benzotriazol-1-ol; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride; triethylamine / dichloromethane / 12 h / 5 - 35 °C 3.1: pyridine; trifluoroacetic anhydride / tetrahydrofuran / 3.5 h / 0 - 10 °C 3.2: 3.5 h / 25 °C / pH 10 - 11 4.1: water; hydrogenchloride / methanol / 3 h / 25 °C View Scheme | |
| Multi-step reaction with 4 steps 1.1: hydrogenchloride / water; tetrahydrofuran; ethyl acetate / 18 h / 20 °C 2.1: methanesulfonyl chloride; N-ethyl-N,N-diisopropylamine / tetrahydrofuran / Cooling with acetone-dry ice 2.2: 20 °C 3.1: pyridine; trifluoroacetic anhydride / 0.5 h / Cooling with ice 3.2: 7.55 h / Cooling 4.1: dichloromethane / methanol / 20 °C / Inert atmosphere View Scheme |
-
-
709031-45-8
(1S,3S,5S)-2-azabicyclo[3.1.0]hexane-3-carboxamide methane sulphonic acid

-
-
709031-78-7
Saxagliptin hydrochloride

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1.1: benzotriazol-1-ol; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride; triethylamine / dichloromethane / 12 h / 5 - 35 °C 2.1: pyridine; trifluoroacetic anhydride / tetrahydrofuran / 3.5 h / 0 - 10 °C 2.2: 3.5 h / 25 °C / pH 10 - 11 3.1: water; hydrogenchloride / methanol / 3 h / 25 °C View Scheme | |
| Multi-step reaction with 3 steps 1.1: dichloromethane / 0.25 h / 25 - 35 °C 1.2: 13.25 h / 5 - 35 °C 2.1: pyridine; trifluoroacetic anhydride / tetrahydrofuran / 3.5 h / 0 - 10 °C 2.2: 3.5 h / 25 °C / pH 10 - 11 3.1: water; hydrogenchloride / methanol / 3 h / 25 °C View Scheme |
-
-
709031-38-9
(5S)-5-aminocarbonyl-4,5-dihydro-1H-pyrrole-1-carboxylic acid 1-(1,1-dimethylethyl) ester

-
-
709031-78-7
Saxagliptin hydrochloride

| Conditions | Yield |
|---|---|
| Multi-step reaction with 5 steps 1.1: diethylzinc / dichloromethane; toluene; 1,2-dimethoxyethane / 0.75 h / -30 - -20 °C / Large scale 1.2: -25 - 24 °C / Large scale 2.1: hydrogenchloride / water; tetrahydrofuran; ethyl acetate / 18 h / 20 °C 3.1: methanesulfonyl chloride; N-ethyl-N,N-diisopropylamine / tetrahydrofuran / Cooling with acetone-dry ice 3.2: 20 °C 4.1: pyridine; trifluoroacetic anhydride / 0.5 h / Cooling with ice 4.2: 7.55 h / Cooling 5.1: dichloromethane / methanol / 20 °C / Inert atmosphere View Scheme |

-
-
709031-78-7
Saxagliptin hydrochloride

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1.1: methanesulfonyl chloride; N-ethyl-N,N-diisopropylamine / tetrahydrofuran / Cooling with acetone-dry ice 1.2: 20 °C 2.1: pyridine; trifluoroacetic anhydride / 0.5 h / Cooling with ice 2.2: 7.55 h / Cooling 3.1: dichloromethane / methanol / 20 °C / Inert atmosphere View Scheme |
-
-
141-82-2
malonic acid

-
-
709031-78-7
Saxagliptin hydrochloride

| Conditions | Yield |
|---|---|
| In methanol at 20 - 25℃; for 0.75h; | 100% |

-
-
709031-78-7
Saxagliptin hydrochloride

| Conditions | Yield |
|---|---|
| In isopropyl alcohol for 0.75h; Solvent; | 100% |

| Conditions | Yield |
|---|---|
| In ethyl acetate at 65℃; | 93% |
-
-
79-14-1
glycolic Acid

-
-
709031-78-7
Saxagliptin hydrochloride

| Conditions | Yield |
|---|---|
| In 5,5-dimethyl-1,3-cyclohexadiene at 20 - 25℃; | 83% |
-
-
709031-78-7
Saxagliptin hydrochloride

| Conditions | Yield |
|---|---|
| In dichloromethane; water | 81% |
| With sodium carbonate In dichloromethane; water at 25 - 30℃; for 0.5h; pH=9 - 10; | 74% |
| With sodium carbonate In dichloromethane; water at 25 - 30℃; for 0.5h; pH=9 - 10; | 74% |
Related products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View