COLORCOM LTD.
Colorcom is a global leader in industrial chemical manufacturing and is continuously innovating and transforming to exceed client expectations and industry standards. Colorcom prides itself on superior customer and technical focus, while focusing on
Cas:709031-38-9
Min.Order:1 Metric Ton
Negotiable
Type:Manufacturers
inquiryDayang Chem (Hangzhou) Co.,Ltd.
As a leading manufacturer and supplier of chemicals in China, DayangChem not only supply popular chemicals, but also DayangChem’s R&D center offer custom synthesis according to the contract research and development services for the fine chemicals, ph
Cas:709031-38-9
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryXiamen Hisunny Chemical Co.,Ltd
Best quality & Attractive price & Professional service; Trial & Pilot & Commercial Hisunny Chemical is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality intermediates, specia
Cas:709031-38-9
Min.Order:0
Negotiable
Type:Manufacturers
inquiryShanghai Hope Chem Co., Ltd
1. Product advantages: exquisite appearance and unique functions. 2. Product Advantages: Our products are the best,fast speed ,in large stock 3. High-quality products and thoughtful service are your greatest satisfaction. 4. The product
Cas:709031-38-9
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryTaiChem Taizhou Limited
Established in May 2015, TaiChem Ltd. is initially invested by a British research and development company and started by PhDs back from aboard. The company is registered in China Medical City (CMC), Taizhou, Jiangsu Province, and the production site
Cas:709031-38-9
Min.Order:10 Gram
Negotiable
Type:Lab/Research institutions
inquiryXiamen Jenny Chemical Technology Co., Ltd.
GMP standard, high purity, competitive price, in stock 1. Quick Response: within 6 hours after receiving your email. 2. Quality Guarantee: All products are strictly tested by our QC, confirmed by QA, and approved by a third-party lab in China, USA,
Cas:709031-38-9
Min.Order:1 Milligram
Negotiable
Type:Trading Company
inquiryTAIZHOU ZHENYU BIOTECHNOLOGY CO., LTD
Zhenyu biotech exported this product to many countries and regions at best price. if you are looking for the material's manufacturer or supplier in china, zhenyu biotech is your best choice. pls contact with us freely for getting detailed
Cas:709031-38-9
Min.Order:1 Kilogram
FOB Price: $2.0
Type:Lab/Research institutions
inquiryBluecrystal chem-union
We are a Union of chemistry in China, consists of chemists,engineers, laboratories,factories in China. We organize surplus capacity of R&D and production as well as custom synthesis for chemical products and chemical business project. We are supp
Cas:709031-38-9
Min.Order:1 Metric Ton
FOB Price: $1.0
Type:Trading Company
inquiryHangzhou Dingyan Chem Co., Ltd
Enterprise standard Application:Saxagliptin Intermediate
Cas:709031-38-9
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryAntimex Chemical Limied
Our own factory produces direct sales with absolute price advantage Application:Pharmaceutical industry Transportation:By sea Port:Shanghai/tianjin
Cas:709031-38-9
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryHangzhou Fandachem Co.,Ltd
(S)-Boc-2-carbamoyl-2,3-dihydro-1H-pyrroleAppearance:white crystalline powder Storage:Store in dry, dark and ventilated place Package:25KG drum Application:pharmaceutical intermediate Transportation:by air, by sea, by express
Cas:709031-38-9
Min.Order:0
Negotiable
Type:Other
inquiryHunan Longxianng Runhui Trading Co.,Ltd
R & D enterprises have their own stock in stockAppearance:To be subject to the object Package:Customized Application:pharmaceutical intermediates Transportation:Air Port:Shanghai;Guangzhou
Cas:709031-38-9
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryZHEJIANG JIUZHOU CHEM CO.,LTD
factory?direct?saleAppearance:White powder Storage:Sealed and preserved Package:200/Kilograms Application:healing drugs Transportation:By sea Port:Shanghai/tianjin
Cas:709031-38-9
Min.Order:0
Negotiable
Type:Trading Company
inquiryAmadis Chemical Co., Ltd.
1.Professional synthesis laboratory and production base. 2.Strong synthesis team and service team. 3.Professional data management system. 4.We provide the professional test date and product information ,ex. HNMR ,CNMR,FNMR, HPLC/G
Cas:709031-38-9
Min.Order:10 Milligram
Negotiable
Type:Lab/Research institutions
inquiryShanghai Hanhong Scientific Co.,Ltd.
factory,reasonable price Appearance:detailed see specifications Storage:Store in dry, dark and ventilated place. Package:according to the clients requirement Application:pharmaceutical intermediates Transportation:by courier,air or sea Port:S
Cas:709031-38-9
Min.Order:0
Negotiable
Type:Trading Company
inquiryNanjing Norris Pharm Technology Co., Ltd.
R & D enterprises have their own stock in stockAppearance:To be subject to the object Package:Customized Application:pharmaceutical intermediates Transportation:Air Port:Shanghai;Guangzhou
Cas:709031-38-9
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryHangzhou Haiqiang Chemical Co.,Ltd.
Enterprise standard Application:Saxagliptin Intermediate
Cas:709031-38-9
Min.Order:0 Metric Ton
Negotiable
Type:Other
inquiryENAO Chemical Co, Limited
kodgyq8oAppearance:white powders Storage:Store at 2-8°C. Package:As per buyer requirement Application:Pharmaceuticals, Skincare, Medical Ingedients and others Transportation:By courier or By sea Port:As per buyer requirement
Cas:709031-38-9
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquirySkyrun Industrial Co.,Ltd
Adequate stocks available,prompt shipment,Strictly control on quality,Timely after-sales Application:Intermediate
Cas:709031-38-9
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryTopbatt Chemical Co., Ltd.
Topbatt Chemical Co., Ltd., Established in 2019, located in Shenzhen, Guangdong Province, is a Manufacturer and Trading company which specialized in fine chemicals like Pharmaceutical Reference Standards and Stable Isotopes. Our Stable Isotopes produ
Cas:709031-38-9
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquirycoolpharm Ltd
Known for its best quality and competitve price, this chemicals we offered is widely appreciated by our customers.Appearance:yellow to white solid Storage:keep sealed and keep from direct light Package:According client's requirements Application:Phar
Cas:709031-38-9
Min.Order:0
Negotiable
Type:Other
inquiryShenzhen Sumshine Biotech Co., Ltd.
1. Our staff are all biomedical related majors with rich experience in the pharmaceutical industry, and can provide you with more professional services.2. Our supplier has a good quality management system, and the quality of products is reliable and
Beyond Pharmaceutical Co., Ltd
Cas:709031-38-9
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryNovaChemistry
High purity Application:Pharmaceutical intermediates, Material synthesis and analyzing the expectant ingredients
Synthetic route
-
-
178172-26-4
(5S)-4,5-dihydro-1H-pyrrol-1,5-dicarboxylic acid [1-(1,1-dimethylethyl)] 5-ethyl ester

-
-
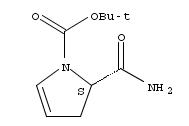
709031-38-9
(5S)-5-aminocarbonyl-4,5-dihydro-1H-pyrrole-1-carboxylic acid 1-(1,1-dimethylethyl) ester

| Conditions | Yield |
|---|---|
| With sodium isopropylate; formamide In diethyl ether at 20℃; for 2.16667h; Solvent; Temperature; Cooling with ice; | 93% |
| With Candida antarctica lipase B; ammonium carbamate; Ascarite; calcium chloride at 50℃; for 72h; Product distribution; Kinetics; Further Variations:; Reagents; Temperatures; | 81% |
| Multi-step reaction with 2 steps 1: water; lithium hydroxide / ethanol 2: ammonia; N-ethyl-N,N-diisopropylamine; methanesulfonyl chloride / tetrahydrofuran View Scheme |

-
-
709031-38-9
(5S)-5-aminocarbonyl-4,5-dihydro-1H-pyrrole-1-carboxylic acid 1-(1,1-dimethylethyl) ester

| Conditions | Yield |
|---|---|
| Stage #1: N-BOC dehydroproline DIPEA With methanesulfonyl chloride; N-ethyl-N,N-diisopropylamine In tetrahydrofuran at -25 - -20℃; for 2h; Stage #2: With ammonia In tetrahydrofuran at 0 - 20℃; for 3h; | 90% |
-
-
709031-37-8
(S)-1-(tert-butoxycarbonyl)-2,3-dihydro-1H-pyrrole-2-carboxylic acid diisopropylethylamine salt

-
-
709031-38-9
(5S)-5-aminocarbonyl-4,5-dihydro-1H-pyrrole-1-carboxylic acid 1-(1,1-dimethylethyl) ester

| Conditions | Yield |
|---|---|
| Stage #1: (S)-1-(tert-butoxycarbonyl)-2,3-dihydro-1H-pyrrole-2-carboxylic acid diisopropylethylamine salt With methanesulfonyl chloride; N-ethyl-N,N-diisopropylamine In tetrahydrofuran at -20 - 0℃; for 3h; Stage #2: With ammonium carbonate In tetrahydrofuran at 0 - 20℃; | 90% |
| With ammonia; methanesulfonyl chloride; N-ethyl-N,N-diisopropylamine In tetrahydrofuran | 1.5 g |
-
-
90104-21-5
1-(tert-butoxycarbonyl)-2,3-dihydro-1H-pyrrole-2-carboxylic acid

-
-
709031-38-9
(5S)-5-aminocarbonyl-4,5-dihydro-1H-pyrrole-1-carboxylic acid 1-(1,1-dimethylethyl) ester

| Conditions | Yield |
|---|---|
| Stage #1: 1-(tert-butoxycarbonyl)-2,3-dihydro-1H-pyrrole-2-carboxylic acid With methanesulfonyl chloride; N-ethyl-N,N-diisopropylamine In tetrahydrofuran at -25 - -20℃; for 2h; Large scale; Stage #2: With ammonia In tetrahydrofuran at 0 - 20℃; for 3h; Large scale; | 90% |
| With 4-methyl-morpholine; ammonia; methanesulfonyl chloride at -15 - -8℃; | 359.7 g |

-
-
3945-69-5
4-(4,6-dimethoxy-1,3,5-triazin-2-yl)-4-methylmorpholinium chloride

-
A
-
709031-38-9
(5S)-5-aminocarbonyl-4,5-dihydro-1H-pyrrole-1-carboxylic acid 1-(1,1-dimethylethyl) ester

| Conditions | Yield |
|---|---|
| With sodium hydroxide; sodium dihydrogenphosphate; ammonium chloride at 20℃; for 4h; pH=6.20; Conversion of starting material; | A 87% B 12% |

-
-
178172-26-4
(5S)-4,5-dihydro-1H-pyrrol-1,5-dicarboxylic acid [1-(1,1-dimethylethyl)] 5-ethyl ester

-
A
-
709031-38-9
(5S)-5-aminocarbonyl-4,5-dihydro-1H-pyrrole-1-carboxylic acid 1-(1,1-dimethylethyl) ester

| Conditions | Yield |
|---|---|
| With sodium methylate; formamide In methanol at 20℃; for 4.5h; |
-
-
24424-99-5
di-tert-butyl dicarbonate

-
-
709031-38-9
(5S)-5-aminocarbonyl-4,5-dihydro-1H-pyrrole-1-carboxylic acid 1-(1,1-dimethylethyl) ester

| Conditions | Yield |
|---|---|
| Multi-step reaction with 5 steps 1: dmap 2: lithium triethylborohydride / toluene / -70 - -60 °C 3: dmap; trifluoroacetic anhydride; N-ethyl-N,N-diisopropylamine 4: water; lithium hydroxide / ethanol 5: ammonia; N-ethyl-N,N-diisopropylamine; methanesulfonyl chloride / tetrahydrofuran View Scheme | |
| Multi-step reaction with 4 steps 1.1: dmap / toluene / 3 h / 25 °C / Large scale 2.1: lithium triethylborohydride / toluene; tetrahydrofuran / 0.5 h / -45 °C / Large scale 2.2: 3 h / 25 °C / Large scale 3.1: lithium hydroxide monohydrate / water; ethanol / 2 h / 25 °C / Large scale 4.1: methanesulfonyl chloride; N-ethyl-N,N-diisopropylamine / tetrahydrofuran / 2 h / -25 - -20 °C / Large scale 4.2: 3 h / 0 - 20 °C / Large scale View Scheme |
-
-
194594-23-5
5-hydroxy-pyrrolidine-1,2-dicarboxylic acid 1-tert-butyl ester 2-ethyl ester

-
-
709031-38-9
(5S)-5-aminocarbonyl-4,5-dihydro-1H-pyrrole-1-carboxylic acid 1-(1,1-dimethylethyl) ester

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: dmap; trifluoroacetic anhydride; N-ethyl-N,N-diisopropylamine 2: water; lithium hydroxide / ethanol 3: ammonia; N-ethyl-N,N-diisopropylamine; methanesulfonyl chloride / tetrahydrofuran View Scheme |
-
-
144978-35-8, 144978-12-1
(S)-ethyl N-tert-butoxycarbonylpyroglutamate

-
-
709031-38-9
(5S)-5-aminocarbonyl-4,5-dihydro-1H-pyrrole-1-carboxylic acid 1-(1,1-dimethylethyl) ester

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: lithium triethylborohydride / toluene / -70 - -60 °C 2: dmap; trifluoroacetic anhydride; N-ethyl-N,N-diisopropylamine 3: water; lithium hydroxide / ethanol 4: ammonia; N-ethyl-N,N-diisopropylamine; methanesulfonyl chloride / tetrahydrofuran View Scheme | |
| Multi-step reaction with 3 steps 1.1: lithium triethylborohydride / toluene; tetrahydrofuran / 0.5 h / -45 °C / Large scale 1.2: 3 h / 25 °C / Large scale 2.1: lithium hydroxide monohydrate / water; ethanol / 2 h / 25 °C / Large scale 3.1: methanesulfonyl chloride; N-ethyl-N,N-diisopropylamine / tetrahydrofuran / 2 h / -25 - -20 °C / Large scale 3.2: 3 h / 0 - 20 °C / Large scale View Scheme |
-
-
7149-65-7
ethyl (S)-pyroglutamate

-
-
709031-38-9
(5S)-5-aminocarbonyl-4,5-dihydro-1H-pyrrole-1-carboxylic acid 1-(1,1-dimethylethyl) ester

| Conditions | Yield |
|---|---|
| Multi-step reaction with 5 steps 1: dmap 2: lithium triethylborohydride / toluene / -70 - -60 °C 3: dmap; trifluoroacetic anhydride; N-ethyl-N,N-diisopropylamine 4: water; lithium hydroxide / ethanol 5: ammonia; N-ethyl-N,N-diisopropylamine; methanesulfonyl chloride / tetrahydrofuran View Scheme | |
| Multi-step reaction with 4 steps 1.1: dmap / toluene / 3 h / 25 °C / Large scale 2.1: lithium triethylborohydride / toluene; tetrahydrofuran / 0.5 h / -45 °C / Large scale 2.2: 3 h / 25 °C / Large scale 3.1: lithium hydroxide monohydrate / water; ethanol / 2 h / 25 °C / Large scale 4.1: methanesulfonyl chloride; N-ethyl-N,N-diisopropylamine / tetrahydrofuran / 2 h / -25 - -20 °C / Large scale 4.2: 3 h / 0 - 20 °C / Large scale View Scheme |
-
-
709031-41-4
4,5-duhydro-1H-pyrrole-1,5-dicarboxylic acid, 1-(1,1-dimethylethyl) dicyclohexylamine salt

-
-
709031-38-9
(5S)-5-aminocarbonyl-4,5-dihydro-1H-pyrrole-1-carboxylic acid 1-(1,1-dimethylethyl) ester

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: sodium hydroxide / water 2: ammonium hydroxide / Inert atmosphere View Scheme |

-
-
709031-38-9
(5S)-5-aminocarbonyl-4,5-dihydro-1H-pyrrole-1-carboxylic acid 1-(1,1-dimethylethyl) ester

| Conditions | Yield |
|---|---|
| With ammonium hydroxide Inert atmosphere; |

-
-
709031-38-9
(5S)-5-aminocarbonyl-4,5-dihydro-1H-pyrrole-1-carboxylic acid 1-(1,1-dimethylethyl) ester

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1.1: ethyl acetate / 0.25 h / 8 °C / Inert atmosphere 1.2: 2 h / 8 - 12 °C / Inert atmosphere 2.1: ammonium hydroxide / 0.5 h / 0 - 20 °C View Scheme |

-
-
709031-38-9
(5S)-5-aminocarbonyl-4,5-dihydro-1H-pyrrole-1-carboxylic acid 1-(1,1-dimethylethyl) ester

| Conditions | Yield |
|---|---|
| With ammonium hydroxide at 0 - 20℃; for 0.5h; | 1.75 g |
-
-
108963-96-8
(S)-1-tert-butoxycarbonyl-5-methoxycarbonyl-pyrrolidin-2-one

-
-
709031-38-9
(5S)-5-aminocarbonyl-4,5-dihydro-1H-pyrrole-1-carboxylic acid 1-(1,1-dimethylethyl) ester

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1.1: lithium triethylborohydride / tetrahydrofuran / 5 - 10 °C 1.2: 3 h / 120 °C 2.1: lithium hydroxide monohydrate / methanol; water / -5 - 30 °C 3.1: 4-methyl-morpholine; methanesulfonyl chloride; ammonia / -15 - -8 °C View Scheme | |
| Multi-step reaction with 3 steps 1.1: lithium triethylborohydride / toluene; tetrahydrofuran / 3 h / -70 - -60 °C / Inert atmosphere 1.2: 3.33 h / -60 - 20 °C 2.1: water; lithium hydroxide / methanol / 2 h / 0 - 20 °C 3.1: methanesulfonyl chloride; N-ethyl-N,N-diisopropylamine / tetrahydrofuran / 3 h / -20 - 0 °C 3.2: 0 - 20 °C View Scheme |
-
-
98-79-3
L-Pyroglutamic acid

-
-
709031-38-9
(5S)-5-aminocarbonyl-4,5-dihydro-1H-pyrrole-1-carboxylic acid 1-(1,1-dimethylethyl) ester

| Conditions | Yield |
|---|---|
| Multi-step reaction with 5 steps 1.1: thionyl chloride / -5 - 30 °C 2.1: triethylamine / toluene / -5 - 0 °C 2.2: -5 - 30 °C 3.1: lithium triethylborohydride / tetrahydrofuran / 5 - 10 °C 3.2: 3 h / 120 °C 4.1: lithium hydroxide monohydrate / methanol; water / -5 - 30 °C 5.1: 4-methyl-morpholine; methanesulfonyl chloride; ammonia / -15 - -8 °C View Scheme | |
| Multi-step reaction with 5 steps 1.1: thionyl chloride / 5 h / -5 - 0 °C / Large scale 2.1: dmap / toluene / 3 h / 25 °C / Large scale 3.1: lithium triethylborohydride / toluene; tetrahydrofuran / 0.5 h / -45 °C / Large scale 3.2: 3 h / 25 °C / Large scale 4.1: lithium hydroxide monohydrate / water; ethanol / 2 h / 25 °C / Large scale 5.1: methanesulfonyl chloride; N-ethyl-N,N-diisopropylamine / tetrahydrofuran / 2 h / -25 - -20 °C / Large scale 5.2: 3 h / 0 - 20 °C / Large scale View Scheme |
-
-
4931-66-2
methyl (S)-pyroglutamate

-
-
709031-38-9
(5S)-5-aminocarbonyl-4,5-dihydro-1H-pyrrole-1-carboxylic acid 1-(1,1-dimethylethyl) ester

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1.1: triethylamine / toluene / -5 - 0 °C 1.2: -5 - 30 °C 2.1: lithium triethylborohydride / tetrahydrofuran / 5 - 10 °C 2.2: 3 h / 120 °C 3.1: lithium hydroxide monohydrate / methanol; water / -5 - 30 °C 4.1: 4-methyl-morpholine; methanesulfonyl chloride; ammonia / -15 - -8 °C View Scheme |
-
-
83548-46-3
(S)-1-tert-butyl 2-methyl 2,3-dihydro-1H-pyrrole-1,2-dicarboxylate

-
-
709031-38-9
(5S)-5-aminocarbonyl-4,5-dihydro-1H-pyrrole-1-carboxylic acid 1-(1,1-dimethylethyl) ester

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: lithium hydroxide monohydrate / methanol; water / -5 - 30 °C 2: 4-methyl-morpholine; methanesulfonyl chloride; ammonia / -15 - -8 °C View Scheme | |
| Multi-step reaction with 2 steps 1.1: water; lithium hydroxide / methanol / 2 h / 0 - 20 °C 2.1: methanesulfonyl chloride; N-ethyl-N,N-diisopropylamine / tetrahydrofuran / 3 h / -20 - 0 °C 2.2: 0 - 20 °C View Scheme |
-
-
709031-38-9
(5S)-5-aminocarbonyl-4,5-dihydro-1H-pyrrole-1-carboxylic acid 1-(1,1-dimethylethyl) ester

-
-
25628-09-5
tetramethylammonium trifluoromethanesulphonate

-
-
361440-67-7
[1S-(1α,3β,5α)]-3-(aminocarbonyl)-2-azabicyclo[3.1.0]hexane-2-carboxylic acid 1,1-dimethylethyl ester

| Conditions | Yield |
|---|---|
| With n-butyllithium; C40H54NiP2 In tetrahydrofuran; hexane at 0 - 20℃; for 16h; Reagent/catalyst; Solvent; Inert atmosphere; | 93% |
-
-
709031-38-9
(5S)-5-aminocarbonyl-4,5-dihydro-1H-pyrrole-1-carboxylic acid 1-(1,1-dimethylethyl) ester

-
-
75-11-6
diiodomethane

-
-
361440-67-7
[1S-(1α,3β,5α)]-3-(aminocarbonyl)-2-azabicyclo[3.1.0]hexane-2-carboxylic acid 1,1-dimethylethyl ester

| Conditions | Yield |
|---|---|
| Stage #1: (5S)-5-aminocarbonyl-4,5-dihydro-1H-pyrrole-1-carboxylic acid 1-(1,1-dimethylethyl) ester; diiodomethane With diethylzinc In water; ethyl acetate; toluene at -10 - 0℃; for 105h; Inert atmosphere; Stage #2: With trifluoroacetic acid In water; ethyl acetate; toluene at -10 - 16℃; for 1.66667h; Reagent/catalyst; Inert atmosphere; | 70% |
| Stage #1: diiodomethane With copper(I) bromide; zinc In tert-butyl methyl ether at 20℃; for 0.5h; Simmons-Smith Cyclopropanation; Inert atmosphere; Stage #2: (5S)-5-aminocarbonyl-4,5-dihydro-1H-pyrrole-1-carboxylic acid 1-(1,1-dimethylethyl) ester In tert-butyl methyl ether at 20℃; for 4.5h; Simmons-Smith Cyclopropanation; Inert atmosphere; | 68% |
| Stage #1: diiodomethane With diethylzinc In 1,2-dimethoxyethane; dichloromethane; toluene at -30 - -25℃; for 0.75h; Simmons-Smith Reaction; Stage #2: (5S)-5-aminocarbonyl-4,5-dihydro-1H-pyrrole-1-carboxylic acid 1-(1,1-dimethylethyl) ester In 1,2-dimethoxyethane; dichloromethane; toluene at 22 - 24℃; Stage #3: With sodium hydrogencarbonate In 1,2-dimethoxyethane; dichloromethane; water; toluene at 15℃; for 1h; | 55% |
| With diethylzinc In 1,2-dimethoxyethane; dichloromethane at -30 - 20℃; Simmons-Smith Cyclopropanation; stereoselective reaction; | 45% |
| Stage #1: diiodomethane With diethylzinc In 1,2-dimethoxyethane; dichloromethane; toluene at -30 - -20℃; for 0.75h; Large scale; Stage #2: (5S)-5-aminocarbonyl-4,5-dihydro-1H-pyrrole-1-carboxylic acid 1-(1,1-dimethylethyl) ester In 1,2-dimethoxyethane; dichloromethane; toluene at -25 - 24℃; Large scale; |
-
-
709031-38-9
(5S)-5-aminocarbonyl-4,5-dihydro-1H-pyrrole-1-carboxylic acid 1-(1,1-dimethylethyl) ester

| Conditions | Yield |
|---|---|
| Stage #1: diiodomethane With diethylzinc In 1,2-dimethoxyethane; dichloromethane; toluene at -30 - 25℃; for 0.75h; Simmons-Smith Reaction; Stage #2: (5S)-5-aminocarbonyl-4,5-dihydro-1H-pyrrole-1-carboxylic acid 1-(1,1-dimethylethyl) ester In 1,2-dimethoxyethane; dichloromethane; toluene at 22 - 24℃; | 55% |
-
-
709031-38-9
(5S)-5-aminocarbonyl-4,5-dihydro-1H-pyrrole-1-carboxylic acid 1-(1,1-dimethylethyl) ester

-
-
1217038-24-8
(13)C(2)H2I2

-
-
1572922-54-3
(1S,3S,5S)-tert-butyl 3-carbamoyl-6-[13CD2]-2-azabicyclo[3.1.0]hexane-2-carboxylate

| Conditions | Yield |
|---|---|
| Stage #1: (5S)-5-aminocarbonyl-4,5-dihydro-1H-pyrrole-1-carboxylic acid 1-(1,1-dimethylethyl) ester With diethylzinc In dichloromethane; toluene at 0℃; for 0.5h; Inert atmosphere; Stage #2: (13)C(2)H2I2 In dichloromethane; toluene at 0 - 20℃; for 17h; Simmons-Smith Cyclopropanation; Inert atmosphere; | 40% |
-
-
709031-38-9
(5S)-5-aminocarbonyl-4,5-dihydro-1H-pyrrole-1-carboxylic acid 1-(1,1-dimethylethyl) ester

-
-
58401-84-6
<14C>methylene iodide

| Conditions | Yield |
|---|---|
| With water; diethylzinc In dichloromethane; toluene; pentane at -10 - 20℃; for 21h; Simmons-Smith Cyclopropanation; Inert atmosphere; | 10.2% |
-
-
709031-38-9
(5S)-5-aminocarbonyl-4,5-dihydro-1H-pyrrole-1-carboxylic acid 1-(1,1-dimethylethyl) ester

-
-
709031-43-6
3-cyano-(αS)-α-(3-hydroxytricyclo[3.3.1.13,7]dec-1-yl)-β-oxo-(1S,3S,5S)-2-azabicyclo[3.1.0]hexane-2-ethanecarbamic acid 1,1-dimethylethyl ester

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1.1: diethylzinc / 1,2-dimethoxyethane; dichloromethane / -30 - 20 °C 2.1: isopropyl alcohol / 60 °C 3.1: benzotriazol-1-ol; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride; N-ethyl-N,N-diisopropylamine / ethyl acetate; acetonitrile / 3 h 4.1: trifluoroacetic anhydride; pyridine / tetrahydrofuran / 0.5 h / 0 °C 4.2: 18 h / 20 °C View Scheme | |
| Multi-step reaction with 4 steps 1.1: diethylzinc / dichloromethane; toluene; 1,2-dimethoxyethane / 0.75 h / -30 - -20 °C / Large scale 1.2: -25 - 24 °C / Large scale 2.1: hydrogenchloride / water; tetrahydrofuran; ethyl acetate / 18 h / 20 °C 3.1: methanesulfonyl chloride; N-ethyl-N,N-diisopropylamine / tetrahydrofuran / Cooling with acetone-dry ice 3.2: 20 °C 4.1: pyridine; trifluoroacetic anhydride / 0.5 h / Cooling with ice 4.2: 7.55 h / Cooling View Scheme |
-
-
709031-38-9
(5S)-5-aminocarbonyl-4,5-dihydro-1H-pyrrole-1-carboxylic acid 1-(1,1-dimethylethyl) ester

-
-
1564266-92-7
(R)-N-Boc-3-hydroxyadamantylglycine-L-cis-4,5-methanoprolinenitrile

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1.1: diethylzinc / 1,2-dimethoxyethane; dichloromethane / -30 - 20 °C 2.1: isopropyl alcohol / 60 °C 3.1: benzotriazol-1-ol; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride; N-ethyl-N,N-diisopropylamine / ethyl acetate; acetonitrile / 3 h 4.1: trifluoroacetic anhydride; pyridine / tetrahydrofuran / 0.5 h / 0 °C 4.2: 18 h / 20 °C View Scheme |
-
-
709031-38-9
(5S)-5-aminocarbonyl-4,5-dihydro-1H-pyrrole-1-carboxylic acid 1-(1,1-dimethylethyl) ester

| Conditions | Yield |
|---|---|
| Multi-step reaction with 5 steps 1.1: diethylzinc / 1,2-dimethoxyethane; dichloromethane / -30 - 20 °C 2.1: isopropyl alcohol / 60 °C 3.1: benzotriazol-1-ol; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride; N-ethyl-N,N-diisopropylamine / ethyl acetate; acetonitrile / 3 h 4.1: trifluoroacetic anhydride; pyridine / tetrahydrofuran / 0.5 h / 0 °C 4.2: 18 h / 20 °C 5.1: hydrogenchloride / isopropyl alcohol; water / 1.5 h / 65 °C View Scheme | |
| Multi-step reaction with 6 steps 1.1: diethylzinc / dichloromethane; toluene; 1,2-dimethoxyethane / 0.75 h / -30 - -20 °C / Large scale 1.2: -25 - 24 °C / Large scale 2.1: hydrogenchloride / water; tetrahydrofuran; ethyl acetate / 18 h / 20 °C 3.1: methanesulfonyl chloride; N-ethyl-N,N-diisopropylamine / tetrahydrofuran / Cooling with acetone-dry ice 3.2: 20 °C 4.1: pyridine; trifluoroacetic anhydride / 0.5 h / Cooling with ice 4.2: 7.55 h / Cooling 5.1: dichloromethane / methanol / 20 °C / Inert atmosphere 6.1: water; dichloromethane View Scheme |
-
-
709031-38-9
(5S)-5-aminocarbonyl-4,5-dihydro-1H-pyrrole-1-carboxylic acid 1-(1,1-dimethylethyl) ester

| Conditions | Yield |
|---|---|
| Multi-step reaction with 5 steps 1.1: diethylzinc / 1,2-dimethoxyethane; dichloromethane / -30 - 20 °C 2.1: isopropyl alcohol / 60 °C 3.1: benzotriazol-1-ol; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride; N-ethyl-N,N-diisopropylamine / ethyl acetate; acetonitrile / 3 h 4.1: trifluoroacetic anhydride; pyridine / tetrahydrofuran / 0.5 h / 0 °C 4.2: 18 h / 20 °C 5.1: hydrogenchloride / isopropyl alcohol; water / 1.5 h / 65 °C View Scheme |
-
-
709031-38-9
(5S)-5-aminocarbonyl-4,5-dihydro-1H-pyrrole-1-carboxylic acid 1-(1,1-dimethylethyl) ester

-
-
709031-45-8
(1S,3S,5S)-2-azabicyclo[3.1.0]hexane-3-carboxamide methane sulphonic acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: diethylzinc / 1,2-dimethoxyethane; dichloromethane / -30 - 20 °C 2: isopropyl alcohol / 60 °C View Scheme |
-
-
709031-38-9
(5S)-5-aminocarbonyl-4,5-dihydro-1H-pyrrole-1-carboxylic acid 1-(1,1-dimethylethyl) ester

-
-
361442-01-5
tert-butyl [(1S)-2-[(1S,3S,5S)-3-carbamoyl-2-azabicyclo[3.1.0]hex-2-yl]-1-(3-hydroxytricyclo[3.3.1.13'7]dec-1-yl)-2-oxoethyl]carbamate

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: diethylzinc / 1,2-dimethoxyethane; dichloromethane / -30 - 20 °C 2: isopropyl alcohol / 60 °C 3: benzotriazol-1-ol; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride; N-ethyl-N,N-diisopropylamine / ethyl acetate; acetonitrile / 3 h View Scheme | |
| Multi-step reaction with 3 steps 1.1: diethylzinc / dichloromethane; toluene; 1,2-dimethoxyethane / 0.75 h / -30 - -20 °C / Large scale 1.2: -25 - 24 °C / Large scale 2.1: hydrogenchloride / water; tetrahydrofuran; ethyl acetate / 18 h / 20 °C 3.1: methanesulfonyl chloride; N-ethyl-N,N-diisopropylamine / tetrahydrofuran / Cooling with acetone-dry ice 3.2: 20 °C View Scheme |
-
-
709031-38-9
(5S)-5-aminocarbonyl-4,5-dihydro-1H-pyrrole-1-carboxylic acid 1-(1,1-dimethylethyl) ester

-
-
1528611-63-3
(R)-N-Boc-3-hydroxyadamantylglycine-L-cis-4,5-methanoprolinamide

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: diethylzinc / 1,2-dimethoxyethane; dichloromethane / -30 - 20 °C 2: isopropyl alcohol / 60 °C 3: benzotriazol-1-ol; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride; N-ethyl-N,N-diisopropylamine / ethyl acetate; acetonitrile / 3 h View Scheme |
-
-
709031-38-9
(5S)-5-aminocarbonyl-4,5-dihydro-1H-pyrrole-1-carboxylic acid 1-(1,1-dimethylethyl) ester

-
-
1572922-58-7
tert-butyl((S)-2-((1S,3S,5S)-3-(6-[13CD2])-cyano-2-azabicyclo[3.1.0]hexan-2-yl)-1-(3,5-dihydroxyadamantan-1-yl)-2-oxoethyl)carbamate

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1.1: diethylzinc / dichloromethane; toluene / 0.5 h / 0 °C / Inert atmosphere 1.2: 17 h / 0 - 20 °C / Inert atmosphere 2.1: 1,3,5-trichloro-2,4,6-triazine / N,N-dimethyl-formamide / 1.2 h / 0 °C 3.1: hydrogenchloride / 1,4-dioxane / 1.2 h / 47 °C / Inert atmosphere 4.1: N-[(dimethylamino)-3-oxo-1H-1,2,3-triazolo[4,5-b]pyridin-1-yl-methylene]-N-methylmethanaminium hexafluorophosphate; N-ethyl-N,N-diisopropylamine / N,N-dimethyl-formamide View Scheme |
-
-
709031-38-9
(5S)-5-aminocarbonyl-4,5-dihydro-1H-pyrrole-1-carboxylic acid 1-(1,1-dimethylethyl) ester

-
-
1572922-53-2
C17(13)CH23(2)H2N3O3*ClH

| Conditions | Yield |
|---|---|
| Multi-step reaction with 5 steps 1.1: diethylzinc / dichloromethane; toluene / 0.5 h / 0 °C / Inert atmosphere 1.2: 17 h / 0 - 20 °C / Inert atmosphere 2.1: 1,3,5-trichloro-2,4,6-triazine / N,N-dimethyl-formamide / 1.2 h / 0 °C 3.1: hydrogenchloride / 1,4-dioxane / 1.2 h / 47 °C / Inert atmosphere 4.1: N-[(dimethylamino)-3-oxo-1H-1,2,3-triazolo[4,5-b]pyridin-1-yl-methylene]-N-methylmethanaminium hexafluorophosphate; N-ethyl-N,N-diisopropylamine / N,N-dimethyl-formamide 5.1: hydrogenchloride / water; isopropyl alcohol View Scheme |
-
-
709031-38-9
(5S)-5-aminocarbonyl-4,5-dihydro-1H-pyrrole-1-carboxylic acid 1-(1,1-dimethylethyl) ester

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: diethylzinc; water / dichloromethane; pentane; toluene / 21 h / -10 - 20 °C / Inert atmosphere 2: 1,3,5-trichloro-2,4,6-triazine / N,N-dimethyl-formamide / 1.2 h / 0 °C 3: hydrogenchloride / 1,4-dioxane / 1.2 h / 47 °C / Inert atmosphere 4: N-[(dimethylamino)-3-oxo-1H-1,2,3-triazolo[4,5-b]pyridin-1-yl-methylene]-N-methylmethanaminium hexafluorophosphate; N-ethyl-N,N-diisopropylamine / N,N-dimethyl-formamide View Scheme |
-
-
709031-38-9
(5S)-5-aminocarbonyl-4,5-dihydro-1H-pyrrole-1-carboxylic acid 1-(1,1-dimethylethyl) ester

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: diethylzinc; water / dichloromethane; pentane; toluene / 21 h / -10 - 20 °C / Inert atmosphere 2: 1,3,5-trichloro-2,4,6-triazine / N,N-dimethyl-formamide / 1.2 h / 0 °C View Scheme |
-
-
709031-38-9
(5S)-5-aminocarbonyl-4,5-dihydro-1H-pyrrole-1-carboxylic acid 1-(1,1-dimethylethyl) ester

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: diethylzinc; water / dichloromethane; pentane; toluene / 21 h / -10 - 20 °C / Inert atmosphere 2: 1,3,5-trichloro-2,4,6-triazine / N,N-dimethyl-formamide / 1.2 h / 0 °C 3: hydrogenchloride / 1,4-dioxane / 1.2 h / 47 °C / Inert atmosphere View Scheme |
-
-
709031-38-9
(5S)-5-aminocarbonyl-4,5-dihydro-1H-pyrrole-1-carboxylic acid 1-(1,1-dimethylethyl) ester

| Conditions | Yield |
|---|---|
| Multi-step reaction with 5 steps 1.1: diethylzinc; water / dichloromethane; pentane; toluene / 21 h / -10 - 20 °C / Inert atmosphere 2.1: 1,3,5-trichloro-2,4,6-triazine / N,N-dimethyl-formamide / 1.2 h / 0 °C 3.1: hydrogenchloride / 1,4-dioxane / 1.2 h / 47 °C / Inert atmosphere 4.1: N-[(dimethylamino)-3-oxo-1H-1,2,3-triazolo[4,5-b]pyridin-1-yl-methylene]-N-methylmethanaminium hexafluorophosphate; N-ethyl-N,N-diisopropylamine / N,N-dimethyl-formamide 5.1: hydrogenchloride / water; isopropyl alcohol 5.2: 10 - 20 °C View Scheme |
-
-
709031-38-9
(5S)-5-aminocarbonyl-4,5-dihydro-1H-pyrrole-1-carboxylic acid 1-(1,1-dimethylethyl) ester

-
-
1572922-55-4
(1S,3S,5S)-tert-butyl 3-cyano-6-[13CD2]-2-azabicyclo[3.1.0]hexane-2-carboxylate

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1.1: diethylzinc / dichloromethane; toluene / 0.5 h / 0 °C / Inert atmosphere 1.2: 17 h / 0 - 20 °C / Inert atmosphere 2.1: 1,3,5-trichloro-2,4,6-triazine / N,N-dimethyl-formamide / 1.2 h / 0 °C View Scheme |
-
-
709031-38-9
(5S)-5-aminocarbonyl-4,5-dihydro-1H-pyrrole-1-carboxylic acid 1-(1,1-dimethylethyl) ester

-
-
1572922-56-5
(1S,3S,5S)-6-[13CD2]-2-azabicyclo[3.1.0]hexane-3-carbonitrile hydrochloride

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1.1: diethylzinc / dichloromethane; toluene / 0.5 h / 0 °C / Inert atmosphere 1.2: 17 h / 0 - 20 °C / Inert atmosphere 2.1: 1,3,5-trichloro-2,4,6-triazine / N,N-dimethyl-formamide / 1.2 h / 0 °C 3.1: hydrogenchloride / 1,4-dioxane / 1.2 h / 47 °C / Inert atmosphere View Scheme |
Related products
Downstream Products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View