GL Biochem (Shanghai) Ltd.
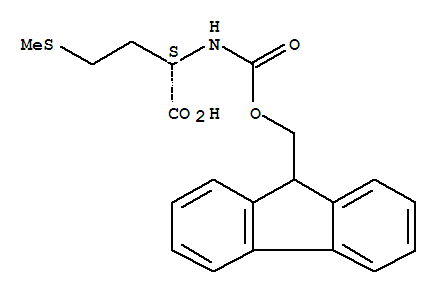
CAS#71989-28-1 Fmoc-L-Met-OH which is coded as 35601 is a catalogue compound synthesized/supplied by GL Biochem. Its specifications & structural Indications are as follows. This material is intended for the R&D and industrial production
Cas:71989-28-1
Min.Order:1 Gram
FOB Price: $6.0 / 30.0
Type:Manufacturers
inquiryDayang Chem (Hangzhou) Co.,Ltd.
DayangChem exported this product to many countries and regions at best price. If you are looking for the material's manufacturer or supplier in China, DayangChem is your best choice. Pls contact with us freely for getting detailed product spe
Cas:71989-28-1
Min.Order:1 Kilogram
FOB Price: $3.0
Type:Lab/Research institutions
inquirySimagchem Corporation
Welcome to Simagchem, your partner in China as a premier supply of bulk specialty chemicals for industry and life science. We introduce experienced quality product and exceptional JIT service with instant market intelligence in China to benefit our
Cas:71989-28-1
Min.Order:1 Kilogram
Negotiable
Type:Manufacturers
inquiryWuhan Fortuna Chemical Co.,Ltd
Unique advantages Guaranteed purity High quality & competitive price Quality control Fast feedback Prompt shipment Appearance:White powder Storage:cool dry place Package:1kg/foil bag;25kg/drum Application:pharmaceutical intermediate Transpo
Cas:71989-28-1
Min.Order:1 Kilogram
Negotiable
Type:Trading Company
inquiryHebei yanxi chemical co.,LTD.
Hebei yanxi chemical co. LTD. has expanded a compositive entity from initially only as a small manufacturer. The company dedicated to the development, production and marketing of chemicals. After many years of efforts, we have established stable
Cas:71989-28-1
Min.Order:1 Metric Ton
FOB Price: $1.0 / 2.0
Type:Manufacturers
inquiryHenan Allgreen Chemical Co.,Ltd
high purity Appearance:White to white powder Storage:cold storage Package:meet the demand of customer Application:Biomedical reagents Transportation:by sea or by air Port:shanghai
SICHUAN TONGSHENG AMINOACID CO.LTD
Sichuan Tongsheng is the most strongest manufacturer and exporter of amino acids and their derivatives in China, we guarantee high quality, competive price and reliable service. We fully compliance with ISO9001:2008, production and quality manage
Chemwill Asia Co., Ltd.
Our main production base is located in Xuzhou industry park. We are certified both to the ISO 9001 and ISO 14001 Standards, have a safety management system in place.Our R&D team masters core technology for process-design of target building block
Cas:71989-28-1
Min.Order:5 Kiloliter
FOB Price: $1.2 / 5.0
Type:Manufacturers
inquiryHubei DiBo chemical co., LTD
Name:FMOC-L-Methionine CAS NO:71989-28-1 Grade:Medical scientific research and export Molecular formula:C20H21NO4S Molecular weight:371.45 Product Quality 12 years of chemical raw materials Mature operation of the industry System stability
Cas:71989-28-1
Min.Order:25 Kilogram
FOB Price: $2.0 / 3.0
Type:Other
inquiryLeader Biochemical Group
About Product Technical Details
Cas:71989-28-1
Min.Order:1 Kilogram
FOB Price: $1.0 / 3.0
Type:Lab/Research institutions
inquiryHebei Nengqian Chemical Import and Export Co., LTD
With our good experience, we offer detailed technical support and advice to assist customers. We communicate closely with customers to establish their quality requirements. Consistent Quality Our plant has strict quality control in each manufacturin
Cas:71989-28-1
Min.Order:1 Kilogram
FOB Price: $3.0 / 10.0
Type:Trading Company
inquiryZhuozhou Wenxi import and Export Co., Ltd
WITH US,YOUR MONEY IN SAFE,YOUR BUSINESS IN SAFE 1)Quick Response Within 12 hours; 2)Quality Guarantee: All products are strictly tested by our QC, confirmed by QA and approved by third party lab in China, USA, Canada, Germany, UK, Italy, France et
Cas:71989-28-1
Min.Order:10 Gram
FOB Price: $146.0 / 176.0
Type:Trading Company
inquiryHenan Sinotech Import&Export Corporation
Product Name Fmoc-Met-OH CAS No. 71989-28-1 Synonyms FMOC-L-Methionine Purity 98.0% min Molecular Formula C20H21NO4S Molecular Weight 371.5 Appearance: white powder Stor
Hubei Langyou International Trading Co., Ltd
1. Guaranteed purity; 2. Large quantity in stock; 3. Largest manufacturer; 4. Best service after shipment with email; 5. High quality & competitive price; Appearance:White to off-white crystalline powder Storage:Store in seale
Cas:71989-28-1
Min.Order:100 Gram
Negotiable
Type:Other
inquiryShanghai Upbio Tech Co.,Ltd
1.No Less 8 years exporting experience. Clients can 100% received goods 2.Lower Price with higher quality 3,Free sample 4,We are sincerely responsible for the "product quality" and "After Service" Upbio is Specialized
Cas:71989-28-1
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryBaoji Guokang Healthchem co.,ltd
EINECS No.:204-879-3 Place of Origin:Shaanxi, China (Mainland) Type:Anesthetic Agents, Anti-Allergic Agents, Antibiotic and Antimicrobial Agents, Antineoplastic Agents, Central Nervous System Agents Grade Standard:Cosmetic Grade, Feed Grade,
Cas:71989-28-1
Min.Order:1 Kilogram
FOB Price: $8.0 / 256.0
Type:Trading Company
inquiryQingdao Beluga Import and Export Co., LTD
FMOC-L-Methionine CAS:71989-28-1 Qingdao Belugas Import and Export Co., Ltd. is a scientific and technological company integrating research and development, production and trade of chemical intermediates, specializing in high quality organic interme
Cas:71989-28-1
Min.Order:1 Gram
Negotiable
Type:Lab/Research institutions
inquiryHangzhou ZeErRui Chemical Co., Ltd.
Hangzhou ZeErRui Chemical Co., Ltd. located in Lingang industrial areas, our plant covers an area of 6000 square meters.ZeErRui dedicated to the development, production and marketing of chemicals. We have earned ourselves a good reputation at home
Hangzhou Keyingchem Co.,Ltd
Hangzhou KeyingChem Co., Ltd. exported this product to many countries and regions at best price. If you are looking for the material’s manufacturer or supplier in China, KeyingChem is your best choice. Pls contact with us freely for getting det
Cas:71989-28-1
Min.Order:0 Metric Ton
Negotiable
Type:Lab/Research institutions
inquiryWuhan Han Sheng New Material Technology Co.,Ltd
Our Advantage: high quality with competitive price High quality standard: BP/USP/EP Enterprise standard All purity customized Fast and safe delivery We have reliable forwarder who can help us deliver our goods more fast and safe. We
Afine Chemicals Limited
Company Introduction 1. Established in 2005, with two independent business divisions: Fine chemicals division; Pharmaceutical division. 2. Main product: Optical brightener Textile auxiliary Dye stuff Pigments
SHANGHAI T&W PHARMACEUTICAL CO., LTD.
A substitute for perfluorooctanoic acid, mainly used as a surfactant, dispersant, additive, etc Appearance:White solid or Colorless liquid Purity:99.3 % We will ship the goods in a timely manner as required We can provide relevant documents acc
Ality Chemical Corporation
The above product is Ality Chemical's strong item with best price, good quality and fast supply. Ality Chemical has been focusing on the research and production of this field for over 14 years. At the same time, we are always committed to providi
Cas:71989-28-1
Min.Order:0 Metric Ton
Negotiable
Type:Other
inquiryHangzhou J&H Chemical Co., Ltd.
J&H CHEM R&D center can offer custom synthesis according to the contract research and development services for the fine chemicals, pharmaceutical, biotechnique and some of the other chemicals. J&H CHEM has some Manufacturing base in Jia
Zibo Hangyu Biotechnology Development Co., Ltd
Zibo Hangyu Biotechnology Development Co., Ltd is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality pharmaceuticals, intermediates, special chemicals and OLED intermediates and other fine chemi
Cas:71989-28-1
Min.Order:10 Gram
FOB Price: $100.0
Type:Lab/Research institutions
inquiryHangzhou Lingrui Chemical Co.,Ltd.
High purity Fmoc-Met-OH 71989-28-1 in stock immediately delivery good supplierAppearance:Powder Storage:Dry and ventilated Package:according to customers' requirements Application:APIs Transportation:By air(EMS or EUB or FedEx or TNT ect...) or by s
Cas:71989-28-1
Min.Order:1 Gram
Negotiable
Type:Other
inquiryHANGZHOU YUNUO CHEMICAL CO.,LTD
Superior quality, moderate price & quick delivery. Appearance:white powder Storage:Stored in cool, dry and ventilation place; Away from fire and heat Package:25kg/drum, or as per your request. Application:Used for Pesticide intermediates T
Henan Tianfu Chemical Co., Ltd.
FMOC-L-Methionine 71989-28-1 FMOC-L-Methionine Basic information Product Name: FMOC-L-Methionine Synonyms: N-ALPHA-FMOC-L-METHIONINE;N-(9-FLUORENYLMETHOXYCARBONYL)-L-METHIONINE;N-[(9H-
Cas:71989-28-1
Min.Order:1 Metric Ton
Negotiable
Type:Lab/Research institutions
inquirySiwei Development Group Ltd.
Product name: FMOC-L-Met-OH CAS No.:71989-28-1 Molecule Formula:C20H20NO4S Molecule Weight:370.44 Purity: 99.0% Package: 25kg/drum Description:White to off-white powder Manufacture Standards:Enterprise Standard TES
Cas:71989-28-1
Min.Order:1 Kilogram
Negotiable
Type:Trading Company
inquiryXiamen Jenny Chemical Technology Co., Ltd.
GMP standard, high purity, competitive price, in stock 1. Quick Response: within 6 hours after receiving your email. 2. Quality Guarantee: All products are strictly tested by our QC, confirmed by QA, and approved by a third-party lab in China, USA,
Synthetic route
-
-
500872-34-4
2-(9H-fluoren-9-ylmethoxycarbonylamino)-4-methylsulfanyl-butyric acid methyl ester

-
-
71989-28-1
N-[(9H-fluoren-9-ylmethoxy)carbonyl]-L-methionine

| Conditions | Yield |
|---|---|
| With aluminium trichloride; N,N-dimethyl-aniline In dichloromethane | 98% |
-
-
63-68-3
L-methionine

-
-
88744-04-1
(9-fluorenyl)methyl pentafluorophenyl carbonate

-
-
71989-28-1
N-[(9H-fluoren-9-ylmethoxy)carbonyl]-L-methionine

| Conditions | Yield |
|---|---|
| With sodium hydrogencarbonate In water; acetone Ambient temperature; | 84% |
-
-
63-68-3
L-methionine

-
-
1131148-55-4
1-[(9-fluorenylmethyloxycarbonyl)]benzotriazole

-
-
71989-28-1
N-[(9H-fluoren-9-ylmethoxy)carbonyl]-L-methionine

| Conditions | Yield |
|---|---|
| With triethylamine In water; acetonitrile at 20℃; for 2h; | 84% |
-
-
63-68-3
L-methionine

-
-
71989-28-1
N-[(9H-fluoren-9-ylmethoxy)carbonyl]-L-methionine

| Conditions | Yield |
|---|---|
| With triethylamine In water Ambient temperature; | 80% |
-
-
63-68-3
L-methionine

-
-
28920-43-6
(fluorenylmethoxy)carbonyl chloride

-
-
71989-28-1
N-[(9H-fluoren-9-ylmethoxy)carbonyl]-L-methionine

| Conditions | Yield |
|---|---|
| Stage #1: L-methionine With chloro-trimethyl-silane In dichloromethane for 1h; Inert atmosphere; Reflux; Stage #2: (fluorenylmethoxy)carbonyl chloride With N-ethyl-N,N-diisopropylamine In dichloromethane at 5 - 20℃; for 1.5h; | 80% |
| In acetonitrile at 20℃; for 0.25h; pH=9.2; aq. borate buffer; | |
| Stage #1: L-methionine With sodium carbonate In 1,4-dioxane; water Cooling with ice; Stage #2: (fluorenylmethoxy)carbonyl chloride In 1,4-dioxane; water at 20℃; for 5h; |
-
-
63-68-3
L-methionine

-
-
82911-69-1
N-(9H-fluoren-2-ylmethoxycarbonyloxy)succinimide

-
-
71989-28-1
N-[(9H-fluoren-9-ylmethoxy)carbonyl]-L-methionine

| Conditions | Yield |
|---|---|
| With sodium hydrogencarbonate In N,N-dimethyl-formamide at 50℃; | 78% |
| Stage #1: L-methionine With N-cyclohexyl-cyclohexanamine In acetone at 20℃; Stage #2: N-(9H-fluoren-2-ylmethoxycarbonyloxy)succinimide With sodium carbonate In water; acetonitrile at 0 - 20℃; pH=8; Stage #3: With potassium hydrogensulfate In water; acetonitrile pH=2 - 3; | 61% |
| With sodium hydrogencarbonate In water; N,N-dimethyl-formamide at 0 - 20℃; for 2h; |

-
-
71989-28-1
N-[(9H-fluoren-9-ylmethoxy)carbonyl]-L-methionine

| Conditions | Yield |
|---|---|
| With methanol In dichloromethane for 0.5h; Ambient temperature; Yield given; |

-
-
71989-28-1
N-[(9H-fluoren-9-ylmethoxy)carbonyl]-L-methionine

| Conditions | Yield |
|---|---|
| With magnesium; acetic acid In methanol; N,N-dimethyl-formamide at 20℃; for 75h; |
-
-
63-68-3
L-methionine

-
-
71989-28-1
N-[(9H-fluoren-9-ylmethoxy)carbonyl]-L-methionine

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: CH2Cl2 / 1 h / Heating 2: CH2Cl2 / 72 h / Ambient temperature 3: MeOH / CH2Cl2 / 0.5 h / Ambient temperature View Scheme |
-
-
5269-64-7
O-(Trimethylsilyl)methionine

-
-
71989-28-1
N-[(9H-fluoren-9-ylmethoxy)carbonyl]-L-methionine

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: CH2Cl2 / 72 h / Ambient temperature 2: MeOH / CH2Cl2 / 0.5 h / Ambient temperature View Scheme |
-
-
28920-43-6
(fluorenylmethoxy)carbonyl chloride

-
-
71989-28-1
N-[(9H-fluoren-9-ylmethoxy)carbonyl]-L-methionine

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: triethylamine / dichloromethane / 2 h / 10 °C 2: triethylamine / water; acetonitrile / 2 h / 20 °C View Scheme |
-
-
1361400-97-6
picolyl N-Fmoc-L-methioninate

-
-
71989-28-1
N-[(9H-fluoren-9-ylmethoxy)carbonyl]-L-methionine

| Conditions | Yield |
|---|---|
| With magnesium In methanol at 20℃; for 4h; |
-
-
28920-43-6
(fluorenylmethoxy)carbonyl chloride

-
A
-
71989-28-1
N-[(9H-fluoren-9-ylmethoxy)carbonyl]-L-methionine

-
B
-
71989-28-1, 144701-23-5, 112883-40-6
Nα-fluorenyl-9-methoxycarbonyl-D-Met

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1.1: sodium carbonate / water; 1,4-dioxane / Cooling with ice 1.2: 5 h / 20 °C 2.1: trifluoroacetic acid / isopropyl alcohol; hexane / 25 °C / Resolution of racemate View Scheme |
-
-
144701-23-5
N-Fmoc-methionine

-
A
-
71989-28-1
N-[(9H-fluoren-9-ylmethoxy)carbonyl]-L-methionine

-
B
-
71989-28-1, 144701-23-5, 112883-40-6
Nα-fluorenyl-9-methoxycarbonyl-D-Met

| Conditions | Yield |
|---|---|
| With trifluoroacetic acid In hexane; isopropyl alcohol at 25℃; Reagent/catalyst; Resolution of racemate; | |
| With chiral column based on 3,5-dimethylphenylcarbamoylated β-cyclodextrin combining cinchona alkaloid immobilized on silica gel In methanol; formic acid; triethylamine; acetonitrile at 25℃; Reagent/catalyst; Resolution of racemate; enantioselective reaction; |
-
-
59-51-8
DL-methionine

-
A
-
71989-28-1
N-[(9H-fluoren-9-ylmethoxy)carbonyl]-L-methionine

-
B
-
71989-28-1, 144701-23-5, 112883-40-6
Nα-fluorenyl-9-methoxycarbonyl-D-Met

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1.1: sodium carbonate / water; 1,4-dioxane / Cooling with ice 1.2: 5 h / 20 °C 2.1: trifluoroacetic acid / isopropyl alcohol; hexane / 25 °C / Resolution of racemate View Scheme |

-
-
71989-28-1
N-[(9H-fluoren-9-ylmethoxy)carbonyl]-L-methionine

| Conditions | Yield |
|---|---|
| Stage #1: mono-oxidized L-methionine With chloro-trimethyl-silane In N,N-dimethyl-formamide at 20℃; Pummerer Sulfoxide Rearrangement; Stage #2: With 1,3-dihydro-imidazole-2-thione In N,N-dimethyl-formamide at 20℃; Solvent; Reagent/catalyst; Pummerer Sulfoxide Rearrangement; | 90 %Chromat. |
-
-
872-35-5
1,3-dihydro-imidazole-2-thione

-
C
-
71989-28-1
N-[(9H-fluoren-9-ylmethoxy)carbonyl]-L-methionine

| Conditions | Yield |
|---|---|
| Stage #1: mono-oxidized L-methionine With chloro-trimethyl-silane In ethyl acetate at 20℃; Pummerer Sulfoxide Rearrangement; Stage #2: 1,3-dihydro-imidazole-2-thione In ethyl acetate at 20℃; Solvent; Reagent/catalyst; Pummerer Sulfoxide Rearrangement; | A n/a B n/a C 8 %Chromat. |

-
-
71989-28-1
N-[(9H-fluoren-9-ylmethoxy)carbonyl]-L-methionine

-
-
314732-61-1
(9H-fluoren-9-yl)methyl (S)-(1-amino-4-(methylthio)-1-oxobutan-2-yl)carbamate

| Conditions | Yield |
|---|---|
| Stage #1: N-[(9H-fluoren-9-ylmethoxy)carbonyl]-L-methionine With chloroformic acid ethyl ester; triethylamine In tetrahydrofuran at 0℃; for 0.5h; Stage #2: With ammonium chloride In tetrahydrofuran; water at 0℃; for 0.5h; | 100% |
| With pyridine; di-tert-butyl dicarbonate; ammonium bicarbonate In 1,4-dioxane; N,N-dimethyl-formamide at 20℃; for 48h; Inert atmosphere; | 91% |
| With ammonium bicarbonate; N-ethoxycarbonyl-2-ethoxy-1,2-dihydroquinoline In acetonitrile at 20℃; for 15h; | 86% |
-
-
35661-60-0
Fmoc-Leu-OH

-
-
71989-31-6
Fmoc-Pro-OH

-
-
71989-28-1
N-[(9H-fluoren-9-ylmethoxy)carbonyl]-L-methionine

-
-
76-05-1
trifluoroacetic acid


| Conditions | Yield |
|---|---|
| Multistep reaction.; | 100% |


-
-
1161-13-3
N-Cbz-L-Phe

-
-
35661-39-3
N-[(9H-fluoren-9-ylmethoxy)carbonyl]-L-alanine

-
-
122889-11-6, 83792-48-7
O-benzyl-N-Fmoc-L-serine

-
-
71989-31-6
Fmoc-Pro-OH

-
-
71989-33-8
Fmoc-Ser(tBu)-OH

-
-
71989-28-1
N-[(9H-fluoren-9-ylmethoxy)carbonyl]-L-methionine

-
-
139090-50-9, 83792-47-6
Nα-FMOC-Nω-L-tosylarginine

-
-
146346-82-9
N-(((9H-fluoren-9-yl)methoxy)carbonyl)-O-(tert-butyldimethylsilyl)-L-threonine

-
-
146982-24-3
N-alpha-(9-fluorenylmethyloxycarbonyl)-L-aspartic acid beta-allyl ester

-
-
1240360-55-7
Z-Phe-Ala-Thr(TBS)-Met-Arg(Tos)-Tyr(Pen)-Pro-Ser(tBu)-Asp(OAll)-Ser(Bzl)-Asp(OtBu)-OH

| Conditions | Yield |
|---|---|
| Stage #1: C42H37ClNO6Pol With morpholine Automated synthesizer; solid phase reaction; Stage #2: O-benzyl-N-Fmoc-L-serine With O‑(6‑chlorobezotriazol‑1‑yl)‑N,N,N,N‑tetramethyluronium hexafluorophosphate; N-ethyl-N,N-diisopropylamine; 6-chloro-1-hydroxybenzotriazole In 1-methyl-pyrrolidin-2-one Automated synthesizer; solid phase reaction; Stage #3: C29H31NO5; N-Cbz-L-Phe; N-[(9H-fluoren-9-ylmethoxy)carbonyl]-L-alanine; Fmoc-Pro-OH; Fmoc-Ser(tBu)-OH; N-[(9H-fluoren-9-ylmethoxy)carbonyl]-L-methionine; Nα-FMOC-Nω-L-tosylarginine; N-(((9H-fluoren-9-yl)methoxy)carbonyl)-O-(tert-butyldimethylsilyl)-L-threonine; N-alpha-(9-fluorenylmethyloxycarbonyl)-L-aspartic acid beta-allyl ester Further stages; | 100% |

-
-
29022-11-5
N-(fluoren-9-ylmethoxycarbonyl)glycine

-
-
35661-40-6
N-Fmoc L-Phe

-
-
71989-18-9
Fmoc-Glu(OtBu)-OH

-
-
71989-28-1
N-[(9H-fluoren-9-ylmethoxy)carbonyl]-L-methionine

| Conditions | Yield |
|---|---|
| Stage #1: N-[(9H-fluoren-9-ylmethoxy)carbonyl]-L-methionine With N-ethyl-N,N-diisopropylamine In methanol; dichloromethane; N,N-dimethyl-formamide for 1.33333h; Inert atmosphere; Stage #2: With piperidine In N,N-dimethyl-formamide for 0.333333h; Inert atmosphere; Stage #3: N-(fluoren-9-ylmethoxycarbonyl)glycine; N-Fmoc L-Phe; Fmoc-Glu(OtBu)-OH; N-[(9H-fluoren-9-ylmethoxy)carbonyl]-L-methionine Further stages; | 99% |

-
-
35661-40-6
N-Fmoc L-Phe

-
-
71989-28-1
N-[(9H-fluoren-9-ylmethoxy)carbonyl]-L-methionine

| Conditions | Yield |
|---|---|
| Stage #1: N-[(9H-fluoren-9-ylmethoxy)carbonyl]-L-methionine With N-ethyl-N,N-diisopropylamine In methanol; dichloromethane; N,N-dimethyl-formamide for 1.33333h; Inert atmosphere; Stage #2: With piperidine In N,N-dimethyl-formamide for 0.333333h; Inert atmosphere; Stage #3: N-Fmoc L-Phe; N-[(9H-fluoren-9-ylmethoxy)carbonyl]-L-methionine Further stages; | 99% |
| Stage #1: N-[(9H-fluoren-9-ylmethoxy)carbonyl]-L-methionine With N-ethyl-N,N-diisopropylamine In dichloromethane for 1.08333h; Inert atmosphere; Stage #2: With piperidine In N,N-dimethyl-formamide for 0.166667h; Inert atmosphere; Stage #3: N-Fmoc L-Phe; N-[(9H-fluoren-9-ylmethoxy)carbonyl]-L-methionine Further stages; | 99% |
-
-
29022-11-5
N-(fluoren-9-ylmethoxycarbonyl)glycine

-
-
35661-40-6
N-Fmoc L-Phe

-
-
71989-28-1
N-[(9H-fluoren-9-ylmethoxy)carbonyl]-L-methionine

-
-
144120-54-7
Nα-(9-fluorenylmethoxycarbonyl)-L-glutamic acid α-allyl ester

| Conditions | Yield |
|---|---|
| Stage #1: Nα-(9-fluorenylmethoxycarbonyl)-L-glutamic acid α-allyl ester With N-ethyl-N,N-diisopropylamine In dichloromethane for 0.916667h; Stage #2: With piperidine In N,N-dimethyl-formamide for 0.333333h; Stage #3: N-(fluoren-9-ylmethoxycarbonyl)glycine; N-Fmoc L-Phe; N-[(9H-fluoren-9-ylmethoxy)carbonyl]-L-methionine Further stages; | 99% |

-
-
71989-28-1
N-[(9H-fluoren-9-ylmethoxy)carbonyl]-L-methionine

-
-
147221-33-8
1-methyl-1-phenylethyl 2,2,2-trichloroacetimidoate

| Conditions | Yield |
|---|---|
| In dichloromethane | 98% |
| In dichloromethane at 20℃; for 16h; | 95% |
-
-
103647-81-0
N-(2,2-diethoxyethyl)-3-methylbutan-1-amine

-
-
71989-28-1
N-[(9H-fluoren-9-ylmethoxy)carbonyl]-L-methionine

| Conditions | Yield |
|---|---|
| With N-ethyl-N,N-diisopropylamine; HATU In dichloromethane at 20℃; Cooling with ice; | 98% |
-
-
71989-28-1
N-[(9H-fluoren-9-ylmethoxy)carbonyl]-L-methionine

-
-
28230-32-2
3-hydroxy-3,4-dihydrobenzotriazine-4-one

-
-
119767-83-8
Fmoc-Met-ODhbt

| Conditions | Yield |
|---|---|
| With dicyclohexyl-carbodiimide In tetrahydrofuran at -15 - 4℃; | 95% |
| With thionyl chloride In dichloromethane Heating; | 92% |
-
-
98946-18-0
tert-Butyl 2,2,2-trichloroacetimidate

-
-
71989-28-1
N-[(9H-fluoren-9-ylmethoxy)carbonyl]-L-methionine

| Conditions | Yield |
|---|---|
| In dichloromethane Heating; | 95% |
-
-
913987-81-2
(4-iodomethylphenoxy)acetic acid 2-oxo-2-phenylethyl ester

-
-
71989-28-1
N-[(9H-fluoren-9-ylmethoxy)carbonyl]-L-methionine

-
-
913987-86-7
4-(Fmoc-methionyloxymethyl)phenoxyacetic acid 2-phenacyl ester

| Conditions | Yield |
|---|---|
| With N-ethyl-N,N-diisopropylamine at 0℃; | 95% |
-
-
71989-28-1
N-[(9H-fluoren-9-ylmethoxy)carbonyl]-L-methionine

-
-
63-68-3
L-methionine

| Conditions | Yield |
|---|---|
| With sodium azide In N,N-dimethyl-formamide at 50℃; for 16h; | 95% |
-
-
50-00-0
formaldehyd

-
-
71989-28-1
N-[(9H-fluoren-9-ylmethoxy)carbonyl]-L-methionine

-
-
84000-02-2
(4S)-3-(9-fluorenylmethyloxycarbonyl)-4-<2-(methylthio)ethyl>-5-oxooxazolidine

| Conditions | Yield |
|---|---|
| With toluene-4-sulfonic acid In toluene for 2h; Reflux; | 94% |
| With toluene-4-sulfonic acid In toluene for 0.5h; Heating; | 88% |
-
-
71989-28-1
N-[(9H-fluoren-9-ylmethoxy)carbonyl]-L-methionine

-
-
70-11-1
α-bromoacetophenone

| Conditions | Yield |
|---|---|
| With triethylamine In ethyl acetate; N,N-dimethyl-formamide at 0℃; for 18h; | 94% |

-
-
71989-28-1
N-[(9H-fluoren-9-ylmethoxy)carbonyl]-L-methionine

| Conditions | Yield |
|---|---|
| Stage #1: N-[(9H-fluoren-9-ylmethoxy)carbonyl]-L-methionine With 1-hydroxy-pyrrolidine-2,5-dione; dicyclohexyl-carbodiimide In dichloromethane at 0℃; for 1h; Inert atmosphere; Stage #2: (E)-1α,25-dihydroxy-2-(1′-amino-3′,6′,9′-trioxadodecan-12′-ylidene)-19-norvitamin D3 In dichloromethane Inert atmosphere; | 94% |
-
-
771-61-9
2,3,4,5,6-pentafluorophenol

-
-
71989-28-1
N-[(9H-fluoren-9-ylmethoxy)carbonyl]-L-methionine

-
-
86060-94-8
N-α-Fmoc-L-methionine pentafluorophenyl ester

| Conditions | Yield |
|---|---|
| With dicyclohexyl-carbodiimide In ethyl acetate 0 deg C, 1 h; r.t., 1 h; | 93% |
| With dicyclohexyl-carbodiimide In ethyl acetate at 0℃; for 2h; Yield given; |
-
-
71989-28-1
N-[(9H-fluoren-9-ylmethoxy)carbonyl]-L-methionine

-
-
244633-31-6
pentafluorophenyl 4-nitrobenzenesulfonate

-
-
86060-94-8
N-α-Fmoc-L-methionine pentafluorophenyl ester

| Conditions | Yield |
|---|---|
| With benzotriazol-1-ol; N-ethyl-N,N-diisopropylamine In N,N-dimethyl-formamide at 20℃; Esterification; | 93% |
-
-
15028-41-8
methyl 2-aminoisobutyrate hydrochloride

-
-
71989-28-1
N-[(9H-fluoren-9-ylmethoxy)carbonyl]-L-methionine

-
-
1124322-46-8
Fmoc-L-Met-Aib-OMe

| Conditions | Yield |
|---|---|
| With 4-methyl-morpholine; 1-hydroxy-7-aza-benzotriazole; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride In dichloromethane at 20℃; for 24h; | 93% |
-
-
1313025-22-7
2-docosyloxy-4-methoxybenzyl alcohol

-
-
71989-28-1
N-[(9H-fluoren-9-ylmethoxy)carbonyl]-L-methionine

-
-
1313025-44-3
C50H73NO6S

| Conditions | Yield |
|---|---|
| With dmap; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride In chloroform at 20℃; for 1h; Cooling with ice; | 93% |
-
-
1738-77-8
L-leucine benzyl ester p-toluenesulfonate

-
-
71989-28-1
N-[(9H-fluoren-9-ylmethoxy)carbonyl]-L-methionine

| Conditions | Yield |
|---|---|
| Stage #1: L-leucine benzyl ester p-toluenesulfonate With triethylamine In dichloromethane at 20℃; for 0.5h; Stage #2: N-[(9H-fluoren-9-ylmethoxy)carbonyl]-L-methionine With benzotriazol-1-ol; dicyclohexyl-carbodiimide In dichloromethane at 20℃; | 93% |
-
-
71989-28-1
N-[(9H-fluoren-9-ylmethoxy)carbonyl]-L-methionine

-
-
130859-02-8
(9H-fluoren-9-yl)methyl (S)-(1-fluoro-4-(methylthio)-1-oxobutan-2-yl)carbamate

| Conditions | Yield |
|---|---|
| With tetramethylammonium trifluoromethanethiolate In dichloromethane at 20℃; for 0.0833333h; | 92% |
| With pyridine; trifluoro-[1,3,5]triazine In dichloromethane for 4h; Ambient temperature; | 80% |
| With pyridine; trifluoro-[1,3,5]triazine In dichloromethane at 20℃; | 74% |
| With pyridine; trifluoro-[1,3,5]triazine In dichloromethane for 2h; | 72% |
-
-
2462-35-3
L-leucine benzyl ester hydrochloride

-
-
71989-28-1
N-[(9H-fluoren-9-ylmethoxy)carbonyl]-L-methionine

| Conditions | Yield |
|---|---|
| With C36H24B4N2O3 In toluene at 80℃; for 48h; Inert atmosphere; Molecular sieve; | 92% |
-
-
71989-28-1
N-[(9H-fluoren-9-ylmethoxy)carbonyl]-L-methionine

-
-
119706-37-5
(S)-2-((S)-2-Amino-4-methyl-pentanoylamino)-3-phenyl-propionic acid (benzhydryl-carbamoyl)-methyl ester; compound with trifluoro-acetic acid

-
-
119706-25-1
Fmoc-Met-Leu-Phe-OBg

| Conditions | Yield |
|---|---|
| With 4-methyl-morpholine; (benzotriazo-1-yloxy)tris(dimethylamino)phosphonium hexafluorophosphate In N,N-dimethyl-formamide for 0.5h; Ambient temperature; | 91% |
-
-
71989-28-1
N-[(9H-fluoren-9-ylmethoxy)carbonyl]-L-methionine

| Conditions | Yield |
|---|---|
| With diethyl cyanophosphonate; N-ethyl-N,N-diisopropylamine In dichloromethane at 20℃; Addition; Peptide-bond formation; | 91% |
-
-
95-14-7
1,2,3-Benzotriazole

-
-
71989-28-1
N-[(9H-fluoren-9-ylmethoxy)carbonyl]-L-methionine

-
-
850232-62-1
9H-fluoren-9-ylmethyl N-[(1S)-1-(1H-1,2,3-benzotriazol-1-ylcarbonyl)-3-(methylsulfanyl)propyl]carbamate

| Conditions | Yield |
|---|---|
| Stage #1: 1,2,3-Benzotriazole With thionyl chloride In tetrahydrofuran at 25℃; for 0.5h; Inert atmosphere; Stage #2: N-[(9H-fluoren-9-ylmethoxy)carbonyl]-L-methionine In tetrahydrofuran at 20℃; for 2h; Inert atmosphere; | 91% |
| Stage #1: 1,2,3-Benzotriazole With thionyl chloride In tetrahydrofuran at 35 - 40℃; for 0.333333h; Stage #2: N-[(9H-fluoren-9-ylmethoxy)carbonyl]-L-methionine In tetrahydrofuran at 25℃; for 2h; | 87% |
| Stage #1: 1,2,3-Benzotriazole With thionyl chloride In tetrahydrofuran at 40 - 50℃; for 0.333333h; Stage #2: N-[(9H-fluoren-9-ylmethoxy)carbonyl]-L-methionine In tetrahydrofuran at 20℃; for 2h; | 87% |
| With thionyl chloride In tetrahydrofuran at 20℃; for 2h; |
-
-
147556-16-9
curcumin

-
-
71989-28-1
N-[(9H-fluoren-9-ylmethoxy)carbonyl]-L-methionine

| Conditions | Yield |
|---|---|
| With dmap; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride In dichloromethane at -5 - 0℃; | 91% |
-
-
71989-28-1
N-[(9H-fluoren-9-ylmethoxy)carbonyl]-L-methionine

-
-
103321-54-6
Fmoc-methioninechloride

| Conditions | Yield |
|---|---|
| With thionyl chloride; N,N-dimethyl-formamide In dichloromethane for 1h; Ambient temperature; | 90.3% |
| With thionyl chloride In dichloromethane for 24h; Ambient temperature; | 90% |
| With pyridine; thionyl chloride In dichloromethane Heating; |
-
-
6638-79-5
N,O-dimethylhydroxylamine*hydrochloride

-
-
71989-28-1
N-[(9H-fluoren-9-ylmethoxy)carbonyl]-L-methionine

| Conditions | Yield |
|---|---|
| Stage #1: N-[(9H-fluoren-9-ylmethoxy)carbonyl]-L-methionine With 2,4,6-tripropyl-1,3,5,2,4,6-trioxatriphosphinane-2,4,6-trioxide; 1,8-diazabicyclo[5.4.0]undec-7-ene In ethyl acetate; acetonitrile at 0℃; for 0.166667h; Stage #2: N,O-dimethylhydroxylamine*hydrochloride With triethylamine In ethyl acetate; acetonitrile at 0℃; for 0.5h; | 90% |
| Stage #1: N-[(9H-fluoren-9-ylmethoxy)carbonyl]-L-methionine With trichloroacetonitrile; triphenylphosphine In tetrahydrofuran at 20℃; for 1h; Inert atmosphere; Stage #2: N,O-dimethylhydroxylamine*hydrochloride With triethylamine In tetrahydrofuran at 20℃; for 2h; Inert atmosphere; | 78% |
| With O-(1H-benzotriazol-1-yl)-N,N,N',N'-tetramethyluronium hexafluorophosphate; N-ethyl-N,N-diisopropylamine | |
| Stage #1: N,O-dimethylhydroxylamine*hydrochloride; N-[(9H-fluoren-9-ylmethoxy)carbonyl]-L-methionine With O-(benzotriazol-1-yl)-N,N,N',N'-tetramethyluronium tetrafluoroborate In acetonitrile at 0℃; Inert atmosphere; Stage #2: With N-ethyl-N,N-diisopropylamine In acetonitrile at 0 - 20℃; Inert atmosphere; |
-
-
35661-60-0
Fmoc-Leu-OH

-
-
35661-40-6
N-Fmoc L-Phe

-
-
71989-28-1
N-[(9H-fluoren-9-ylmethoxy)carbonyl]-L-methionine

| Conditions | Yield |
|---|---|
| Stage #1: N-Fmoc L-Phe With N-ethyl-N,N-diisopropylamine In N,N-dimethyl-formamide at 20℃; for 0.666667h; Stage #2: With N-ethyl-N,N-diisopropylamine In methanol; dichloromethane at 20℃; for 0.166667h; Stage #3: Fmoc-Leu-OH; N-[(9H-fluoren-9-ylmethoxy)carbonyl]-L-methionine; Nα-(9-fluorenylmethyloxycarbonyl)-Nγ-2,2,4,6,7-pentamethyldihydrobenzofuran-5-sulfonyl-L-arginine Further stages; | 90% |

Related products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View
 Xi
Xi