Refine
 This product is a nationally controlled contraband, and the Lookchem platform doesn't provide relevant sales information.
This product is a nationally controlled contraband, and the Lookchem platform doesn't provide relevant sales information.
Synthetic route
Conditions

| Conditions | Yield |
|---|---|
| In water byproducts: PH3; addn. of 600 ml H2O to 120 g CaO, addn. of 20 g crushed, yellow P at boiling heat, boiling for 8-10 hours with replacement of vaporized H2O; introduction of CO2 into filtrate at 70-80°C; filtration after short boiling; vaporization till crystn.;; recrystn. from 3-4 parts H2O; yield 18-22 g pure Ca(PH2O2)2;; | |
| In water byproducts: PH3; addn. of 600 ml H2O to 120 g CaO, addn. of 20 g crushed, yellow P at boiling heat, boiling for 8-10 hours with replacement of vaporized H2O; introduction of CO2 into filtrate at 70-80°C; filtration after short boiling; vaporization till crystn.;; recrystn. from 3-4 parts H2O; yield 18-22 g pure Ca(PH2O2)2;; |
...Expand

Conditions

| Conditions | Yield |
|---|---|
| With Ca2P2O7 In water decompn. of phosphorus lime with boiling water;; treatment of filtrate with CO2, crystn., drying at 100°C;; | |
| With Ca2P2O7 In water decompn. of phosphorus lime with boiling water;; treatment of filtrate with CO2, crystn., drying at 100°C;; | |
| In water |
Conditions

| Conditions | Yield |
|---|---|
| In chloroform; water byproducts: PH3; stirring on slight warming (ground temp. 44°C, surface temp. 37°C) for ca 80 hours; after storage for 24 hours decantation of clear layer, saturating with CO2, filtration;; vaporization in vac.;; | |
| In calcium hydroxide byproducts: PH3; addn. of small amt. of yellow P to boiling lime milk, till there is no formation of PH3;; removal of excessive Ca(OH)2 by introduction of CO2 at elevated temp. into filtrate; vaporization on air or over H2SO4 in vac.;; | |
| In water byproducts: PH3; aq. Ca(OH)2 suspension; in closed vessel;; |
Conditions

| Conditions | Yield |
|---|---|
| With calcium hydroxide In neat (no solvent) byproducts: H2; on boiling water bath; |
...Expand

Conditions

| Conditions | Yield |
|---|---|
| With calcium hydroxide In not given byproducts: H2; by heating; |
Conditions

| Conditions | Yield |
|---|---|
| Stage #1: calcium bis(hypophosphite); calcium(II) nitrate In water Stage #2: at 200 - 252℃; under 33098.3 Torr; for 0.733333h; |
Conditions

| Conditions | Yield |
|---|---|
| With water without warming; |
Conditions

| Conditions | Yield |
|---|---|
| With Na acetate In not given pH=5.0-5.5 (NaOH) , 92-93°C; |
Conditions

| Conditions | Yield |
|---|---|
| filtration, recrystn. from water or alc.; |
Conditions

| Conditions | Yield |
|---|---|
| In water Cu(II) : Ca(H2PO2)2 : (NH2)2CO = 1:1:2; evapn. of the soln. for several d at room temp.; elem. anal.; |
...Expand

Conditions

| Conditions | Yield |
|---|---|
| In water |
Conditions

| Conditions | Yield |
|---|---|
| In hydrogenchloride byproducts: CaCl2; introduction of a hydrochloric solution of Ca(H2PO4)2 in boiling aq. MgCl2;; |
...Expand

Conditions

| Conditions | Yield |
|---|---|
| filtration and evapn. of filtrate; product contaminated with Ca; | |
| filtration and evapn. of filtrate; product contaminated with Ca; |
Conditions

| Conditions | Yield |
|---|---|
| In nitric acid introduction of a nitric solution of Ca(H2PO4)2 in boiling aq. Mg(NO3)2;; | |
| In nitric acid aq. HNO3; introduction of a nitric solution of Ca(H2PO4)2 in boiling aq. Mg(NO3)2;; |
Conditions

| Conditions | Yield |
|---|---|
| In water byproducts: Ca-oxalate; addn. of a soln. of oxalic acid to a soln. of Ca(H2PO2)2; pptn. in coldness;; removal of Ca-oxalate impurities by heating at 100 °C, standing for 5 days and by repeated filtration;; | |
| In water byproducts: H3PO4, H3PO3; addn. of a hot soln. of 2 mol H2C2O4 in 100 ml H2O to a hot soln. of 2 mol Ca(H2PO2)2 in 2500 ml H2O (stirring); cooling, filtration, concentrating to a volume of 384 ml in a flow of N2 at 45 °C under reduced pressure;; content: 52 % H3PO2, less than 0.1 % H3PO3 and H3PO4;; |
-
-
7789-79-9
calcium bis(hypophosphite)

-
-
497-19-8
sodium carbonate

-
-
10039-56-2
sodium hypophosphite monohydrate
Conditions

| Conditions | Yield |
|---|---|
| In ethanol Ca(H2PO2)2 decompd. with Na2CO3 and alcoholic soln. evapd.;; | |
| In ethanol Ca(H2PO2)2 decompd. with Na2CO3 and alcoholic soln. evapd.;; |
Conditions

| Conditions | Yield |
|---|---|
| In water byproducts: cadmium calcium phosphinite;; boiling an excess of Cd-oxalate with Ca-phosphinite in water; formation of crystals of cadmium calcium phosphinite by evaporation of the filtrate;; cadmium calcium phosphinite was not isolated; decomposition on heating under evolution of PH3;; | |
| In water byproducts: cadmium calcium phosphinite;; boiling an excess of Cd-oxalate with Ca-phosphinite in water; formation of crystals of cadmium calcium phosphinite by evaporation of the filtrate;; cadmium calcium phosphinite was not isolated; decomposition on heating under evolution of PH3;; |
-
-
7789-79-9
calcium bis(hypophosphite)

-
-
10039-56-2
sodium hypophosphite monohydrate
Conditions

| Conditions | Yield |
|---|---|
| In water byproducts: CaCO3; addn. of 50g Ca(H2PO2)2 in 150ml H2O to a boiling soln. of 42g Na2CO3*10H2O in 100ml H2O, pptn. of CaCO3, filtration, evapn. of filtrate;; | |
| In water byproducts: CaCO3; addn. of 50g Ca(H2PO2)2 in 150ml H2O to a boiling soln. of 42g Na2CO3*10H2O in 100ml H2O, pptn. of CaCO3, filtration, evapn. of filtrate;; |
Conditions

| Conditions | Yield |
|---|---|
| In neat (no solvent) byproducts: P, calcium phosphate; calcination in retort; first small amt. of H2O and PH3, later sublimation of small amt. of P with formation of calcium phosphate;; | |
| In neat (no solvent) byproducts: P, calcium phosphate; calcination in retort; first small amt. of H2O and PH3, later sublimation of small amt. of P with formation of calcium phosphate;; |
Conditions

| Conditions | Yield |
|---|---|
| With sulfur dioxide In not given in acidic soln.; | |
| With SO2 In not given in acidic soln.; |
Conditions

| Conditions | Yield |
|---|---|
| With hydrogen In neat (no solvent) heating in H2;; | |
| With H2 In neat (no solvent) heating in H2;; |
...Expand

Conditions

| Conditions | Yield |
|---|---|
| In neat (no solvent) decompn. by heating over 300°C;; | |
| In neat (no solvent) decompn. by heating over 300°C;; |
-
-
7789-79-9
calcium bis(hypophosphite)

-
-
752190-89-9
calcium sulfite
Conditions

| Conditions | Yield |
|---|---|
| With sulfur dioxide In not given in neutral soln.; | |
| With SO2 In not given in neutral soln.; |
-
-
7789-79-9
calcium bis(hypophosphite)

-
-
547-66-0, 879683-44-0
magnesium(II) oxalate
Conditions

| Conditions | Yield |
|---|---|
| In not given boiling a solution of Ca(H2PO2)2 with an excess of a solution of Mg-oxalate; evaporation of the filtrate;; | |
| In not given boiling a solution of Ca(H2PO2)2 with an excess of a solution of Mg-oxalate; evaporation of the filtrate;; |
-
-
7789-79-9
calcium bis(hypophosphite)

-
-
553-91-3
lithium oxalate

-
-
14832-79-2
lithium dihydrogenphosphate
Conditions

| Conditions | Yield |
|---|---|
| In water reaction of aq. soln. of Li2C2O4 and Ca(H2PO2)2 at 283-293 K; |
Conditions

| Conditions | Yield |
|---|---|
| In water slow evapn. aq. soln. of Co(II) hypophosphite, prepared by adding Ca(H2PO2)2 to CoSO4 (H2O);; mixt. filtered; crystals grown at 293 K in air; |
Related products
Raw Materials
Downstream Products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View

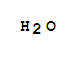

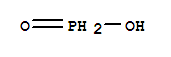


 F
F