-
Name
(2S-trans)-3-Amino-2-methyl-4-oxoazetidine-1-sulphonic acid
- EINECS 279-393-8
- CAS No. 80082-65-1
- Article Data15
- CAS DataBase
- Density 1.75 g/cm3
- Solubility
- Melting Point 206-211oC
- Formula C4H8N2O4S
- Boiling Point
- Molecular Weight 180.185
- Flash Point
- Transport Information
- Appearance White to white powder
- Safety
- Risk Codes
-
Molecular Structure
- Hazard Symbols
- Synonyms 1-Azetidinesulfonicacid, 3-amino-2-methyl-4-oxo-, (2S-trans)-;
- PSA 109.08000
- LogP 0.06620
Synthetic route
-
-
80082-60-6
(3S)-trans-3-<(tert-butoxycarbonyl)amino>-4-methyl-2-oxoazetidine-1-sulfonic acid tetra-n-butylammonium salt

-
-
80082-65-1
(3S-trans)-3-Amino-4-methyl-2-oxo-1-azetidinesulfonic acid

| Conditions | Yield |
|---|---|
| With formic acid for 4.5h; Ambient temperature; | 93% |
| With formic acid for 5h; | 70% |
| With formic acid for 5h; Ambient temperature; | 56% |
-
-
80082-62-8
(3S-trans)-2-Oxo-3-[[(phenylmethoxy)carbonyl]amino]-4-methyl-1-azetidinesulfonic acid,tetrabutylammonium salt

-
-
80082-65-1
(3S-trans)-3-Amino-4-methyl-2-oxo-1-azetidinesulfonic acid

| Conditions | Yield |
|---|---|
| With hydrogen; palladium on activated charcoal In ethanol | 50% |
-
-
80082-64-0
tetrabutylammonium (3S,4S)-3-phenylacetamido-4-methyl-2-oxoazetidine-1-sulfonate

-
-
80082-65-1
(3S-trans)-3-Amino-4-methyl-2-oxo-1-azetidinesulfonic acid

| Conditions | Yield |
|---|---|
| With pyridine; phosgene In dichloromethane at 0℃; for 41h; |
-
-
115887-91-7
(3S,4R)-3-benzyloxycarbonylamino-4-methyl-2-oxoazetidine-1-sulphonic acid sodium salt

-
-
80082-65-1
(3S-trans)-3-Amino-4-methyl-2-oxo-1-azetidinesulfonic acid

| Conditions | Yield |
|---|---|
| With hydrogen; palladium on activated charcoal In methanol; water for 2h; Ambient temperature; | |
| With nitrogen; palladium In methanol; water | |
| With hydrogenchloride; palladium on activated charcoal; hydrogen In methanol; water for 2h; pH=6.8 - 7; | |
| Stage #1: (3S,4R)-3-benzyloxycarbonylamino-4-methyl-2-oxoazetidine-1-sulphonic acid sodium salt With 5%-palladium/activated carbon; hydrogen In methanol at 15℃; for 4h; Large scale; Stage #2: With hydrogenchloride In methanol; water at 0℃; for 6h; Large scale; |
-
-
88144-19-8
(3S,trans)-4-methyl-3-triphenylmethylamino-2-azetidinone

-
-
80082-65-1
(3S-trans)-3-Amino-4-methyl-2-oxo-1-azetidinesulfonic acid

| Conditions | Yield |
|---|---|
| Yield given. Multistep reaction; |

-
-
80082-65-1
(3S-trans)-3-Amino-4-methyl-2-oxo-1-azetidinesulfonic acid

| Conditions | Yield |
|---|---|
| With formic acid at 35 - 40℃; for 4.5h; Inert atmosphere; | 12.7 g |

-
-
80082-65-1
(3S-trans)-3-Amino-4-methyl-2-oxo-1-azetidinesulfonic acid

| Conditions | Yield |
|---|---|
| With ammonia; lithium at -40℃; for 0.5h; | 56 g |

-
-
80082-65-1
(3S-trans)-3-Amino-4-methyl-2-oxo-1-azetidinesulfonic acid

| Conditions | Yield |
|---|---|
| Stage #1: C10H19N2O9S2(1-)*Na(1+) at 45℃; for 1.5h; Stage #2: With sulfuric acid at 30℃; for 6h; | 60 g |
-
-
2052-49-5
tetra(n-butyl)ammonium hydroxide

-
-
75315-63-8
N-(benzyloxycarbonyl)succinimide

-
-
80082-65-1
(3S-trans)-3-Amino-4-methyl-2-oxo-1-azetidinesulfonic acid

-
-
80082-62-8
(3S-trans)-2-Oxo-3-[[(phenylmethoxy)carbonyl]amino]-4-methyl-1-azetidinesulfonic acid,tetrabutylammonium salt

| Conditions | Yield |
|---|---|
| Stage #1: N-(benzyloxycarbonyl)succinimide; (3S-trans)-3-Amino-4-methyl-2-oxo-1-azetidinesulfonic acid With triethylamine In ethanol; water at 25℃; for 16h; Stage #2: tetra(n-butyl)ammonium hydroxide In water | 94% |

-
-
13139-17-8
N-(Benzyloxycarbonyloxy)succinimide

-
-
2052-49-5
tetra(n-butyl)ammonium hydroxide

-
-
80082-65-1
(3S-trans)-3-Amino-4-methyl-2-oxo-1-azetidinesulfonic acid

-
-
80082-62-8
(3S-trans)-2-Oxo-3-[[(phenylmethoxy)carbonyl]amino]-4-methyl-1-azetidinesulfonic acid,tetrabutylammonium salt

| Conditions | Yield |
|---|---|
| Stage #1: N-(Benzyloxycarbonyloxy)succinimide; (3S-trans)-3-Amino-4-methyl-2-oxo-1-azetidinesulfonic acid With triethylamine In ethanol; water at 20℃; for 16h; Stage #2: tetra(n-butyl)ammonium hydroxide In water | 94% |

-
-
80082-65-1
(3S-trans)-3-Amino-4-methyl-2-oxo-1-azetidinesulfonic acid

-
-
147742-76-5
(E)-α-(3-Chloroisoxazol-5-yl)-α-methoxyiminoacetic acid

| Conditions | Yield |
|---|---|
| With ion exchange column (Na form); benzotriazol-1-ol; triethylamine; dicyclohexyl-carbodiimide 1.) DMF, 3 d, 2.) H2O, CH3OH; | 91% |
-
-
57612-86-9
(isoxazol-3-yl)acetic acid

-
-
80082-65-1
(3S-trans)-3-Amino-4-methyl-2-oxo-1-azetidinesulfonic acid

| Conditions | Yield |
|---|---|
| With ion exchange column (Na form); benzotriazol-1-ol; triethylamine; dicyclohexyl-carbodiimide 1.) DMF, 3 d, 2.) H2O, CH3OH; | 87% |
-
-
80082-65-1
(3S-trans)-3-Amino-4-methyl-2-oxo-1-azetidinesulfonic acid

-
-
104164-45-6
3-chloro-5-isoxazolecarboxylic acid chloride

| Conditions | Yield |
|---|---|
| With ion exchange column (Na form); triethylamine; dicyclohexyl-carbodiimide 1.) DMF, 0 to 5 deg C, 1 h, 2.) H2O; | 87% |
-
-
80082-65-1
(3S-trans)-3-Amino-4-methyl-2-oxo-1-azetidinesulfonic acid

-
-
89604-92-2
(Z)-2-<<<1-(2-amino-4-thiazolyl)-2-<(2-benzothiazolyl)thio>-2-oxoethylidene>amino>oxy>-2-methyl propanoic acid 1,1-dimethylethyl ester

-
-
78110-38-0
azthreonam

| Conditions | Yield |
|---|---|
| Stage #1: (3S-trans)-3-Amino-4-methyl-2-oxo-1-azetidinesulfonic acid; (Z)-2-<<<1-(2-amino-4-thiazolyl)-2-<(2-benzothiazolyl)thio>-2-oxoethylidene>amino>oxy>-2-methyl propanoic acid 1,1-dimethylethyl ester With pyridine In acetonitrile at -5℃; for 4h; Stage #2: With nitric acid at 35℃; for 2h; pH=1; Solvent; Reagent/catalyst; Temperature; | 86.9% |
-
-
80082-65-1
(3S-trans)-3-Amino-4-methyl-2-oxo-1-azetidinesulfonic acid

-
-
114875-61-5
(Z)-2-(((1,5-bis(benzhydryloxy)-4-oxo-1,4-dihydropyridin-2-yl)methoxy)imino)-2-(2-(tritylamino)thiazol-4-yl)acetic acid

-
-
404824-78-8
(3S)-trans-3-[[2-(2-tritylamino)thiazol-4-yl]-(Z)-2-{(1,5-dibenzhydryloxy-4-pyridon-2-ylmethoxy)imino}acetamido]-4-methyl-2-oxoazetidine-1-sulfonic acid

| Conditions | Yield |
|---|---|
| Stage #1: (3S-trans)-3-Amino-4-methyl-2-oxo-1-azetidinesulfonic acid; (Z)-2-(((1,5-bis(benzhydryloxy)-4-oxo-1,4-dihydropyridin-2-yl)methoxy)imino)-2-(2-(tritylamino)thiazol-4-yl)acetic acid With benzotriazol-1-ol; dicyclohexyl-carbodiimide In DMF (N,N-dimethyl-formamide) at 20℃; for 0.5h; Stage #2: With sodium hydrogencarbonate In DMF (N,N-dimethyl-formamide) at 20℃; | 85.9% |

| Conditions | Yield |
|---|---|
| In tetrahydrofuran; water at 20℃; for 3h; Acylation; | 84% |

-
-
80082-65-1
(3S-trans)-3-Amino-4-methyl-2-oxo-1-azetidinesulfonic acid

| Conditions | Yield |
|---|---|
| With ion exchange column (Na form); benzotriazol-1-ol; triethylamine; dicyclohexyl-carbodiimide 1.) DMF, 3 d, 2.) H2O, CH3OH; | 82% |
-
-
80082-65-1
(3S-trans)-3-Amino-4-methyl-2-oxo-1-azetidinesulfonic acid

| Conditions | Yield |
|---|---|
| With ion exchange column (Na form); benzotriazol-1-ol; triethylamine; dicyclohexyl-carbodiimide 1.) DMF, 3 d, 2.) H2O, CH3OH; | 81% |
-
-
139714-28-6
(Z)-2-[[[1-(2-amino-4-thiazolyl)-2-(3-oxido-1H-benzotriazol-1-yl)-2-oxoethylidene]amino]oxy]-2-methylpropanoic acid methanesulfonate (1:1) salt

-
-
80082-65-1
(3S-trans)-3-Amino-4-methyl-2-oxo-1-azetidinesulfonic acid

-
-
78110-38-0
azthreonam

| Conditions | Yield |
|---|---|
| Stage #1: (Z)-2-[[[1-(2-amino-4-thiazolyl)-2-(3-oxido-1H-benzotriazol-1-yl)-2-oxoethylidene]amino]oxy]-2-methylpropanoic acid methanesulfonate (1:1) salt; (3S-trans)-3-Amino-4-methyl-2-oxo-1-azetidinesulfonic acid With triethylamine In ethanol; water at -6 - -3℃; for 3h; Stage #2: With hydrogenchloride In ethanol; water at -3 - 0℃; | 81% |

-
-
80082-65-1
(3S-trans)-3-Amino-4-methyl-2-oxo-1-azetidinesulfonic acid

| Conditions | Yield |
|---|---|
| Stage #1: C35H38N4O6S2 With benzotriazol-1-ol; diisopropyl-carbodiimide In N,N-dimethyl-formamide at 20℃; for 1h; Stage #2: (3S-trans)-3-Amino-4-methyl-2-oxo-1-azetidinesulfonic acid With triethylamine In N,N-dimethyl-formamide at 20℃; | 78% |
-
-
80082-65-1
(3S-trans)-3-Amino-4-methyl-2-oxo-1-azetidinesulfonic acid

-
-
54489-25-7
(3-Chloroisoxazol-5-yl)acetic acid

| Conditions | Yield |
|---|---|
| With ion exchange column (Na form); triethylamine; dicyclohexyl-carbodiimide 1.) DMF, 24 h, 2.) H2O, CH3OH; | 75% |
-
-
80082-65-1
(3S-trans)-3-Amino-4-methyl-2-oxo-1-azetidinesulfonic acid

-
-
93276-75-6
(4-Cyano-5-methylisoxazol-3-yl)acetic acid

| Conditions | Yield |
|---|---|
| With ion exchange column (Na form); triethylamine; dicyclohexyl-carbodiimide 1.) DMF, 24 h, 2.) H2O, CH3OH; | 73% |
-
-
80082-65-1
(3S-trans)-3-Amino-4-methyl-2-oxo-1-azetidinesulfonic acid

-
-
147742-85-6
(Z)-α-(3-Chloroisoxazol-5-yl)-α-methoxyiminoacetic acid

| Conditions | Yield |
|---|---|
| With ion exchange column (Na form); benzotriazol-1-ol; triethylamine; dicyclohexyl-carbodiimide 1.) DMF, 3 d, 2.) H2O, CH3OH; | 62% |
-
-
80082-65-1
(3S-trans)-3-Amino-4-methyl-2-oxo-1-azetidinesulfonic acid

-
-
121-44-8
triethylamine

| Conditions | Yield |
|---|---|
| In tetrahydrofuran; water at 20℃; for 3h; Acylation; | 59% |

-
-
80082-65-1
(3S-trans)-3-Amino-4-methyl-2-oxo-1-azetidinesulfonic acid

-
-
147742-81-2
α-tert-Butyloxycarbonylamino-α-(3-chloroisoxazol-5-yl)acetic Acid

| Conditions | Yield |
|---|---|
| With ion exchange column (Na form); benzotriazol-1-ol; triethylamine; dicyclohexyl-carbodiimide 1.) DMF, 3 d, 2.) H2O, CH3OH; | 57% |

| Conditions | Yield |
|---|---|
| In N,N-dimethyl-formamide at 20℃; Acylation; | 53% |

-
-
63564-50-1
α-methyl-4-chlorobenzylideneaminoxy propionic acid

-
-
80082-65-1
(3S-trans)-3-Amino-4-methyl-2-oxo-1-azetidinesulfonic acid

-
-
134468-86-3
Sodium; (2S,3S)-3-{3-[1-(4-chloro-phenyl)-eth-(E)-ylideneaminooxy]-propionylamino}-2-methyl-4-oxo-azetidine-1-sulfonate

| Conditions | Yield |
|---|---|
| With benzotriazol-1-ol; triethylamine; dicyclohexyl-carbodiimide 1) DMF, 30 min, r.t 2) DMF, 18h, r.t.; Yield given. Multistep reaction; |
-
-
103586-55-6
3-(isopropylidenamino)oxy-propanoic acid

-
-
80082-65-1
(3S-trans)-3-Amino-4-methyl-2-oxo-1-azetidinesulfonic acid

-
-
134468-81-8
Sodium; (2S,3S)-3-(3-isopropylideneaminooxy-propionylamino)-2-methyl-4-oxo-azetidine-1-sulfonate

| Conditions | Yield |
|---|---|
| With benzotriazol-1-ol; triethylamine; dicyclohexyl-carbodiimide 1) DMF, 30 min, r.t 2) DMF, 18h, r.t.; Yield given. Multistep reaction; |
-
-
103586-56-7
3-(1-Propyl-butylideneaminooxy)-propionic acid

-
-
80082-65-1
(3S-trans)-3-Amino-4-methyl-2-oxo-1-azetidinesulfonic acid

-
-
134468-83-0
Sodium; (2S,3S)-2-methyl-4-oxo-3-[3-(1-propyl-butylideneaminooxy)-propionylamino]-azetidine-1-sulfonate

| Conditions | Yield |
|---|---|
| With benzotriazol-1-ol; triethylamine; dicyclohexyl-carbodiimide 1) DMF, 30 min, r.t 2) DMF, 18h, r.t.; Yield given. Multistep reaction; |
-
-
103586-53-4
3-Cyclopentylideneaminooxy-propionic acid

-
-
80082-65-1
(3S-trans)-3-Amino-4-methyl-2-oxo-1-azetidinesulfonic acid

-
-
134468-82-9
Sodium; (2S,3S)-3-(3-cyclopentylideneaminooxy-propionylamino)-2-methyl-4-oxo-azetidine-1-sulfonate

| Conditions | Yield |
|---|---|
| With benzotriazol-1-ol; triethylamine; dicyclohexyl-carbodiimide 1) DMF, 30 min, r.t 2) DMF, 18h, r.t.; Yield given. Multistep reaction; |
-
-
103586-52-3
3-[1-Phenyl-eth-(E)-ylideneaminooxy]-propionic acid

-
-
80082-65-1
(3S-trans)-3-Amino-4-methyl-2-oxo-1-azetidinesulfonic acid

-
-
134468-85-2
Sodium; (2S,3S)-2-methyl-4-oxo-3-{3-[1-phenyl-eth-(E)-ylideneaminooxy]-propionylamino}-azetidine-1-sulfonate

| Conditions | Yield |
|---|---|
| With benzotriazol-1-ol; triethylamine; dicyclohexyl-carbodiimide 1) DMF, 30 min, r.t 2) DMF, 18h, r.t.; Yield given. Multistep reaction; |
-
-
110596-23-1
(E)-3-<(benzylidene)iminoxy>-(E)-2-(methoxyimino)propionic acid

-
-
80082-65-1
(3S-trans)-3-Amino-4-methyl-2-oxo-1-azetidinesulfonic acid

-
-
134468-87-4
Sodium; (2S,3S)-3-{2-[(E)-methoxyimino]-3-[1-phenyl-meth-(E)-ylideneaminooxy]-propionylamino}-2-methyl-4-oxo-azetidine-1-sulfonate

| Conditions | Yield |
|---|---|
| With benzotriazol-1-ol; triethylamine; dicyclohexyl-carbodiimide 1) DMF, 30 min, r.t 2) DMF, 18h, r.t.; Yield given. Multistep reaction; |
-
-
110596-24-2
(E)-3-<<(benzylidene)methyl>iminoxy>-(E)-2-(methoxyimino)propionic acid

-
-
80082-65-1
(3S-trans)-3-Amino-4-methyl-2-oxo-1-azetidinesulfonic acid

-
-
134468-91-0
Sodium; (2S,3S)-3-{2-[(E)-methoxyimino]-3-[1-phenyl-eth-(E)-ylideneaminooxy]-propionylamino}-2-methyl-4-oxo-azetidine-1-sulfonate

| Conditions | Yield |
|---|---|
| With benzotriazol-1-ol; triethylamine; dicyclohexyl-carbodiimide 1) DMF, 30 min, r.t 2) DMF, 18h, r.t.; Yield given. Multistep reaction; |
(2S-trans)-3-Amino-2-methyl-4-oxoazetidine-1-sulphonic acid Chemical Properties
IUPAC name: (2S,3S)-3-Amino-2-methyl-4-oxoazetidine-1-sulfonic acid
Molecular Formula: C4H8N2O4S
Molecular Weight: 180.18 g/mol
The molecular structure of (2S-trans)-3-Amino-2-methyl-4-oxoazetidine-1-sulphonic acid (CAS NO.80082-65-1): 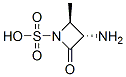
Density: 1.75 g/cm3
EINECS: 279-393-8
Index of Refraction: 1.627
Surface Tension of (2S-trans)-3-Amino-2-methyl-4-oxoazetidine-1-sulphonic acid (CAS NO.80082-65-1): 87.4 dyne/cm
Appearance of (2S-trans)-3-Amino-2-methyl-4-oxoazetidine-1-sulphonic acid (CAS NO.80082-65-1): White to white powder
Canonical SMILES: CC1C(C(=O)N1S(=O)(=O)O)N
Isomeric SMILES: C[C@H]1[C@@H](C(=O)N1S(=O)(=O)O)N
InChI: InChI=1S/C4H8N2O4S/c1-2-3(5)4(7)6(2)11(8,9)10/h2-3H,5H2,1H3,(H,8,9,10)/t2-,3-/m0/s1
InChIKey: ISUIVWNWEDIHJD-HRFVKAFMSA-N
(2S-trans)-3-Amino-2-methyl-4-oxoazetidine-1-sulphonic acid Uses
(2S-trans)-3-Amino-2-methyl-4-oxoazetidine-1-sulphonic acid (CAS NO.80082-65-1) is a useful intermediate in the preparation of Lactams.
(2S-trans)-3-Amino-2-methyl-4-oxoazetidine-1-sulphonic acid Specification
(2S-trans)-3-Amino-2-methyl-4-oxoazetidine-1-sulphonic acid , its cas register number is 80082-65-1. The chemical synonyms of (2S-Trans)-3-Amino-2-methyl-4-oxoazetidine-1-sulphonic acid (CAS NO.80082-65-1) are (2s,3s)-3-Amino-2-methyl-4-oxo-1-azetidinesulfonic acid ; (2s-trans)-3-Methyl-4-oxo-1-azetidinesulfonic acid(foraztreonam) ; 3s-trans-3-amino-4-methyl-2-oxo-1-azetidine sulfonic acid ; and 3-Amino-4-methylmonobactamic acid .
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View