-
Name
1-(2-Furoyl)piperazine
- EINECS 254-823-7
- CAS No. 40172-95-0
- Article Data30
- CAS DataBase
- Density 1.175 g/cm3
- Solubility
- Melting Point 67-70 °C(lit.)
- Formula C9H12N2O2
- Boiling Point 320.3 °C at 760 mmHg
- Molecular Weight 180.206
- Flash Point 147.5 °C
- Transport Information
- Appearance white to light yellow solid
- Safety 26-36-24/25
- Risk Codes 36/37/38
-
Molecular Structure
-
Hazard Symbols
 Xi
Xi
- Synonyms Piperazine,1-(2-furanylcarbonyl)- (9CI);(Furan-2-yl)(piperazin-1-yl)methanone;1-(2-Furanylcarbonyl)piperazine;1-(2-Furylcarbonyl)piperazine;1-(Furan-2-carbonyl)piperazine;4-(Furan-2-ylcarbonyl)piperazine;N-(2-Furancarbonyl)piperazine;N-(2-Furoyl)piperazine;N-(Furan-2-oyl)piperazine;Methanone,2-furanyl-1-piperazinyl-;
- PSA 45.48000
- LogP 0.59170
Synthetic route

| Conditions | Yield |
|---|---|
| With 4-methyl-morpholine; 2-chloro-4,6-dimethoxy-1 ,3,5-triazine In dichloromethane at 20℃; for 0.166667h; | 92% |
| Stage #1: 2-furanoic acid With N-Bromosuccinimide; triphenylphosphine In dichloromethane at 0 - 5℃; for 0.25h; Stage #2: piperazine With pyridine In dichloromethane at 20℃; for 0.583333h; | 86% |
-
-
110-85-0
piperazine

-
-
614-99-3
Ethyl 2-furoate

-
A
-
31350-27-3
N,N'-bis(furan-2-carbonyl)piperazine

-
B
-
40172-95-0
N-(furan-2-carbonyl)piperazine

| Conditions | Yield |
|---|---|
| at 110℃; for 3h; | A 6% B 89% |

-
-
110-85-0
piperazine

-
-
88-14-2
2-furanoic acid

-
A
-
31350-27-3
N,N'-bis(furan-2-carbonyl)piperazine

-
B
-
40172-95-0
N-(furan-2-carbonyl)piperazine

| Conditions | Yield |
|---|---|
| With 1,1,1,3,3,3-hexamethyl-disilazane at 110℃; for 8h; Yields of byproduct given; | A n/a B 83% |

-
-
110-85-0
piperazine

-
-
88-14-2
2-furanoic acid

-
-
999-97-3
1,1,1,3,3,3-hexamethyl-disilazane

-
-
40172-95-0
N-(furan-2-carbonyl)piperazine

| Conditions | Yield |
|---|---|
| In dichloromethane | 83% |


| Conditions | Yield |
|---|---|
| 64% |
-
-
527-69-5
2-furancarbonyl chloride

-
-
57260-71-6
1-t-Butoxycarbonylpiperazine

-
-
40172-95-0
N-(furan-2-carbonyl)piperazine

| Conditions | Yield |
|---|---|
| With hydrogenchloride; triethylamine 1.) THF, r.t., 2 h, 2.) THF, 20 deg C, 2 h; Yield given. Multistep reaction; |

| Conditions | Yield |
|---|---|
| With sodium hydroxide; hydrogen bromide; potassium carbonate 1.) aq. monoglyme, 3 h; Yield given. Multistep reaction; |
-
-
527-69-5
2-furancarbonyl chloride

-
-
142-63-2
piperazine hexahydrate

-
-
40172-95-0
N-(furan-2-carbonyl)piperazine

| Conditions | Yield |
|---|---|
| With sodium hydroxide In water | 108.2 g product (60%) |
| With sodium hydroxide In water | 108.2 g product (60%) |
| With sodium hydroxide In water | 108.2 g product (60%) |
| With sodium hydroxide In water | 108.2 g product (60%) |
| With sodium hydroxide | 108.2 g. product (60%) |

| Conditions | Yield |
|---|---|
| With hydrogenchloride; sodium hydroxide In diethyl ether; ethanol; acetone |


-
-
40172-95-0
N-(furan-2-carbonyl)piperazine

| Conditions | Yield |
|---|---|
| Acidic conditions; | |
| With hydrogenchloride; water In dichloromethane at 20℃; | |
| With hydrogenchloride In methanol; diethyl ether |
-
-
527-69-5
2-furancarbonyl chloride

-
-
40172-95-0
N-(furan-2-carbonyl)piperazine

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: triethylamine / dichloromethane / 20 °C 2: hydrogenchloride; water / dichloromethane / 20 °C View Scheme |
-
-
88-14-2
2-furanoic acid

-
-
40172-95-0
N-(furan-2-carbonyl)piperazine

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: benzotriazol-1-ol; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride; N-ethyl-N,N-diisopropylamine / dichloromethane / 18 h / 20 °C 2: trifluoroacetic acid / dichloromethane / 25 °C View Scheme | |
| Multi-step reaction with 2 steps 1: O-(1H-benzotriazol-1-yl)-N,N,N',N'-tetramethyluronium hexafluorophosphate / dichloromethane / 20 °C 2: trifluoroacetic acid / dichloromethane / 1 h / 20 °C View Scheme |
-
-
108-22-5
Isopropenyl acetate

-
-
40172-95-0
N-(furan-2-carbonyl)piperazine

-
-
768292-07-5
1-(4-(furan-2-carbonyl)piperazin-1-yl)ethanone

| Conditions | Yield |
|---|---|
| In neat (no solvent) at 20℃; for 1h; Green chemistry; | 100% |

| Conditions | Yield |
|---|---|
| Stage #1: 2-Chloroethanesulfonyl chloride; 4-(4-((p-methylbenzylamino)methyl)-3-methoxyphenoxy)butyryl AM resin With N-ethyl-N,N-diisopropylamine In dichloromethane at 20℃; for 2h; solid phase reaction; Stage #2: N-(furan-2-carbonyl)piperazine With lithium perchlorate In dichloromethane; isopropyl alcohol at 75℃; for 24h; Michael addition; Stage #3: With trifluoroacetic acid In water for 1h; | 99% |

-
-
83178-89-6
6-ethoxycarbonyl-7-chlorothieno<3,2-b>pyridin-5(4H)-one

-
-
40172-95-0
N-(furan-2-carbonyl)piperazine

| Conditions | Yield |
|---|---|
| With 1,4-diaza-bicyclo[2.2.2]octane In ISOPROPYLAMIDE at 120℃; for 2 - 4h; | 99% |
-
-
111218-89-4
6-bromo-2-chloroquinazolin-4-amine

-
-
40172-95-0
N-(furan-2-carbonyl)piperazine

-
-
111218-73-6
[4-(4-Amino-6-bromo-quinazolin-2-yl)-piperazin-1-yl]-furan-2-yl-methanone; hydrochloride

| Conditions | Yield |
|---|---|
| In N,N-dimethyl-formamide at 95℃; for 14h; | 96.4% |
-
-
75-15-0
carbon disulfide

-
-
40172-95-0
N-(furan-2-carbonyl)piperazine

| Conditions | Yield |
|---|---|
| With sodium hydroxide In methanol at 0℃; for 3h; | 95% |
| With sodium hydroxide In water; ethyl acetate at 0 - 5℃; for 0.5h; | |
| With sodium hydroxide In ethanol at 20℃; for 1h; Cooling with ice; | |
| With sodium hydroxide In ethanol for 2h; Cooling with ice; |
-
-
814-68-6
acryloyl chloride

-
-
40172-95-0
N-(furan-2-carbonyl)piperazine

| Conditions | Yield |
|---|---|
| With triethylamine In dichloromethane at 0 - 20℃; for 24.3333h; Inert atmosphere; | 95% |
| With triethylamine In dichloromethane at 0 - 20℃; for 24.3333h; Inert atmosphere; | 95% |
-
-
864265-14-5
4-(chloromethyl)-N-cyclohexylbenzamide

-
-
40172-95-0
N-(furan-2-carbonyl)piperazine

| Conditions | Yield |
|---|---|
| Stage #1: N-(furan-2-carbonyl)piperazine With potassium carbonate In acetonitrile for 0.5h; Reflux; Stage #2: 4-(chloromethyl)-N-cyclohexylbenzamide In acetonitrile | 94% |
-
-
1146300-32-4
7-chloro-1-(2,4-difluorophenyl)-1,4-dihydro-8-methyl-6-nitro-4-oxoquinolone-3-carboxylic acid

-
-
40172-95-0
N-(furan-2-carbonyl)piperazine

-
-
1146300-52-8
1-(2,4-difluorophenyl)-1,4-dihydro-7-(4-(2-furoyl)piperazin-1-yl)-8-methyl-6-nitro-4-oxoquinoline-3-carboxylic acid

| Conditions | Yield |
|---|---|
| With potassium carbonate In dimethyl sulfoxide Microwave irradiation; | 93% |
-
-
328-26-7
2-chloro-N-(2-chloro-5-(trifluoromethyl)phenyl)acetamide

-
-
40172-95-0
N-(furan-2-carbonyl)piperazine

-
-
899713-86-1
N-(2-chloro-5-(trifluoromethyl)phenyl)-2-(4-(furan-2-carbonyl)piperazin-1-yl)acetamide

| Conditions | Yield |
|---|---|
| With potassium carbonate at 60℃; for 12h; Inert atmosphere; | 93% |
-
-
119072-55-8, 7188-38-7
tert-butylisonitrile

-
-
1356545-79-3
1-formyl-3-ethoxycarbonyl-β-carboline

-
-
40172-95-0
N-(furan-2-carbonyl)piperazine

| Conditions | Yield |
|---|---|
| Stage #1: tert-butylisonitrile; 1-formyl-3-ethoxycarbonyl-β-carboline; N-(furan-2-carbonyl)piperazine In methanol at 20℃; for 0.166667h; Ugi Condensation; Stage #2: With sodium azide In methanol at 20℃; | 92% |


-
-
40172-95-0
N-(furan-2-carbonyl)piperazine

| Conditions | Yield |
|---|---|
| With sodium tris(acetoxy)borohydride In dichloromethane at 20℃; Inert atmosphere; | 92% |
-
-
40172-95-0
N-(furan-2-carbonyl)piperazine

-
-
43091-89-0
2-isothiocyanato-4,5-dimethoxybenzonitrile

-
-
67817-56-5
3,4-Dimethoxy-6-<4-(2-furoyl)piperazin-1-ylthiocarbamido>benzonitrile

| Conditions | Yield |
|---|---|
| In ethyl acetate at 0℃; for 20h; | 90% |

-
-
40172-95-0
N-(furan-2-carbonyl)piperazine

| Conditions | Yield |
|---|---|
| Stage #1: N-(furan-2-carbonyl)piperazine With potassium carbonate In acetonitrile for 0.5h; Reflux; Stage #2: C10H9Br4NO2 In acetonitrile Reflux; | 90% |
-
-
96783-89-0
O-phenyl N-(2-chloroethyl)-carbamate

-
-
40172-95-0
N-(furan-2-carbonyl)piperazine

| Conditions | Yield |
|---|---|
| Stage #1: N-(furan-2-carbonyl)piperazine With potassium carbonate In acetonitrile for 0.5h; Reflux; Stage #2: O-phenyl N-(2-chloroethyl)-carbamate In acetonitrile Reflux; | 90% |
-
-
40172-95-0
N-(furan-2-carbonyl)piperazine

| Conditions | Yield |
|---|---|
| Stage #1: N-(furan-2-carbonyl)piperazine With potassium carbonate In acetonitrile for 0.5h; Reflux; Stage #2: N-benzyl-4-(chloromethyl)benzamide In acetonitrile | 90% |
-
-
52759-43-0
4-amino-2-chloro-7,8-dihydro-1H-[1,4]dioxino[2,3-g]quinazoline

-
-
40172-95-0
N-(furan-2-carbonyl)piperazine

-
-
1067672-06-3
[4-(4-amino-7,8-dihydro[1,4]dioxino[2,3-g]quinazolin-2-yl)piperazin-1-yl]furan-2-yl methanone hydrochloride

| Conditions | Yield |
|---|---|
| In pentan-1-ol for 1h; Heating; | 89% |
-
-
760-93-0
methacryloyl anhydride

-
-
40172-95-0
N-(furan-2-carbonyl)piperazine

| Conditions | Yield |
|---|---|
| In chloroform at 0℃; for 0.25h; | 88% |
-
-
132814-23-4
5-bromomethyl-6-phenyl-3(2H)-pyridazinone

-
-
40172-95-0
N-(furan-2-carbonyl)piperazine

-
-
132814-15-4
6-phenyl-5-(N4-(2-furoyl)-N1-piperazinylmethyl)-2H-pyridazin-3-one

| Conditions | Yield |
|---|---|
| In methanol for 12h; Ambient temperature; | 87% |
-
-
33904-04-0
4-isothiocyanato-1,2-dimethoxybenzene

-
-
40172-95-0
N-(furan-2-carbonyl)piperazine

| Conditions | Yield |
|---|---|
| In ethyl acetate <5 deg C; | 87% |

-
-
40172-95-0
N-(furan-2-carbonyl)piperazine

| Conditions | Yield |
|---|---|
| Stage #1: N-(furan-2-carbonyl)piperazine With potassium carbonate In acetonitrile for 0.5h; Reflux; Stage #2: Phenyl-(β-brom-propyl)-carbamat In acetonitrile Reflux; | 87% |

-
-
40172-95-0
N-(furan-2-carbonyl)piperazine

| Conditions | Yield |
|---|---|
| Stage #1: N-(furan-2-carbonyl)piperazine With potassium carbonate In acetonitrile for 0.5h; Reflux; Stage #2: 3-Chloromethyl-N-cyclohexyl-benzamide In acetonitrile | 87% |
-
-
67-68-5
dimethyl sulfoxide

-
-
40172-95-0
N-(furan-2-carbonyl)piperazine

| Conditions | Yield |
|---|---|
| With potassium iodide at 20℃; for 8h; Electrolysis; | 87% |
-
-
1146300-33-5
1-tert-butyl-7-chloro-1-1,4-dihydro-8-methyl-6-nitro-4-oxoquinolone-3-carboxylic acid

-
-
40172-95-0
N-(furan-2-carbonyl)piperazine

-
-
1146300-69-7
1-tert-butyl-1,4-dihydro-7-(4-(2-furoyl)piperazin-1-yl)-8-methyl-6-nitro-4-oxoquinoline-3-carboxylic acid

| Conditions | Yield |
|---|---|
| With potassium carbonate In dimethyl sulfoxide Microwave irradiation; | 86% |

-
-
40172-95-0
N-(furan-2-carbonyl)piperazine

| Conditions | Yield |
|---|---|
| Stage #1: N-(furan-2-carbonyl)piperazine With potassium carbonate In acetonitrile for 0.5h; Reflux; Stage #2: C10H9BrN4O8 In acetonitrile Reflux; | 86% |

-
-
40172-95-0
N-(furan-2-carbonyl)piperazine

| Conditions | Yield |
|---|---|
| With triethylamine In iso-butanol at 135℃; for 12h; Sealed tube; | 85.1% |
-
-
1146300-31-3
7-chloro-1-cyclopropyl-1,4-dihydro-8-methyl-6-nitro-4-oxoquinolone-3-carboxylic acid

-
-
40172-95-0
N-(furan-2-carbonyl)piperazine

-
-
1146300-35-7
1-cyclopropyl-1,4-dihydro-7-(4-(2-furoyl)piperazin-1-yl)-8-methyl-6-nitro-4-oxoquinoline-3-carboxylic acid

| Conditions | Yield |
|---|---|
| With potassium carbonate In dimethyl sulfoxide Microwave irradiation; | 85% |

-
-
40172-95-0
N-(furan-2-carbonyl)piperazine

| Conditions | Yield |
|---|---|
| Stage #1: N-(furan-2-carbonyl)piperazine With potassium carbonate In acetonitrile for 0.5h; Reflux; Stage #2: C9H7Br3ClNO2 In acetonitrile Reflux; | 85% |
1-(2-Furoyl)piperazine Chemical Properties
IUPAC Name: Furan-2-yl(piperazin-1-yl)methanone
Synonyms: (Furan-2-yl)(piperazin-1-yl)methanone ; Piperazine,1-(2-furanylcarbonyl)- (9CI) ; 1-(2-Furanoyl)piperazine ; 1-(2-Furanylcarbonyl)piperazine ; 1-(Furan-2-carbonyl)piperazine ; 4-(Furan-2-ylcarbonyl)piperazine ; N-(2-Furoyl)piperazine
EINECS: 254-823-7
Molecular Weight: 180.20378 [g/mol]
Molecular Formula: C9H12N2O2
XLogP3-AA: 0.1
H-Bond Donor: 1
H-Bond Acceptor: 3
Melting Point: 67-70 °C(lit.)
Index of Refraction: 1.529
Molar Refractivity: 47.3 cm3
Molar Volume: 153.3 cm3
Surface Tension: 41.5 dyne/cm
Density: 1.175 g/cm3
Flash Point: 147.5 °C
Enthalpy of Vaporization: 56.19 kJ/mol
Boiling Point: 320.3 °C at 760 mmHg
Vapour Pressure: 0.000321 mmHg at 25 °C
Sensitive: Hygroscopic
Appearance: white to light yellow solid
Product Categories: Piperidines, Piperidones, Piperazines; BUILDING BLOCKS; API intermediates
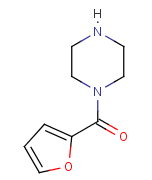
Following is the molecular structure of 1-(2-Furoyl)piperazine (CAS NO.40172-95-0) is:
1-(2-Furoyl)piperazine Safety Profile
Hazard Codes:  Xi
Xi
Risk Statements:
R36/37/38:Irritating to eyes, respiratory system and skin.
Safety Statements
S26: In case of contact with eyes, rinse immediately with plenty of water and seek medical advice.
S36:Wear suitable protective clothing.
S24/25:Avoid contact with skin and eyes.
WGK Germany: 3
F: 10-34
Hazard Note: Irritant
HazardClass: IRRITANT
1-(2-Furoyl)piperazine Specification
1-(2-Furoyl)piperazine (CAS NO.40172-95-0) is a white to light yellow solid.
First Aid Measures about it:
Eyes: Flush eyes with plenty of water for at least 15 minutes, occasionally lifting the upper and lower eyelids. Get medical aid.
Skin: Flush skin with plenty of water for at least 15 minutes while removing contaminated clothing and shoes.
Ingestion: Get medical aid. Wash mouth out with water.
Inhalation: Remove from exposure and move to fresh air immediately. If not breathing, give artificial respiration. If breathing is difficult, give oxygen.
Handling and Storage about it:
Handling: Avoid contact with eyes, skin, and clothing. Avoid ingestion and inhalation.
Storage: Store in a cool, dry place. Keep container closed when not in use.
Related Products
- 1-(2-Furoyl)piperazine
- 401-73-0
- 4017-32-7
- 40174-39-8
- 40175-06-2
- 4017-56-5
- 401-77-4
- 40178-22-1
- 401-78-5
- 401788-98-5
- 401-79-6
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View