-
Name
1,4,5,8-TETRAHYDROXYANTHRAQUINONE
- EINECS
- CAS No. 81-60-7
- Article Data17
- CAS DataBase
- Density 1.781g/cm3
- Solubility
- Melting Point >274.85°C
- Formula C14H8 O6
- Boiling Point 554.8°Cat760mmHg
- Molecular Weight 272.214
- Flash Point 303.4°C
- Transport Information
- Appearance
- Safety Moderately toxic by intraperitoneal route. A skin and eye irritant. When heated to decomposition it emits acrid smoke and irritating fumes.
- Risk Codes
-
Molecular Structure
- Hazard Symbols
- Synonyms Anthraquinone,1,4,5,8-tetrahydroxy- (6CI,7CI,8CI); 1,4,5,8-Tetrahydroxy-9,10-anthracenedione;1,4,5,8-Tetrahydroxy-9,10-anthraquinone; 1,4,5,8-Tetrahydroxyanthraquinone; NSC401144
- PSA 115.06000
- LogP 1.28440
Synthetic route

| Conditions | Yield |
|---|---|
| With water; trifluoroacetic acid at 65℃; for 1h; | 100% |
-
-
145-49-3
4,8-diamino-1,5-dihydroxyanthraquinone

-
A
-
81-60-7
1,4,5,8-tetrahydroxy-anthraquinone

-
B
-
6374-78-3
1-amino-4,5,8-trihydroxyanthraquinone

-
C
-
69837-14-5
leuco-1,4,5,8-tetrahydroxyanthraquinone

| Conditions | Yield |
|---|---|
| With sodium dithionite In water at 100℃; for 0.666667h; | A 2.5% B 1.3% C 92% D 6.4% |

-
-
23478-60-6
leuco-1,4,5,8-tetrahydroxyanthraquinone

-
-
81-60-7
1,4,5,8-tetrahydroxy-anthraquinone

| Conditions | Yield |
|---|---|
| With nitrobenzene for 0.25h; Heating; | 92% |

-
A
-
81-60-7
1,4,5,8-tetrahydroxy-anthraquinone

-
B
-
6374-78-3
1-amino-4,5,8-trihydroxyanthraquinone

-
C
-
69837-14-5
leuco-1,4,5,8-tetrahydroxyanthraquinone

| Conditions | Yield |
|---|---|
| With sodium dithionite In water at 100℃; for 0.666667h; | A 3.7% B 1.4% C 90% D 7.1% |
| With hydrogenchloride at 85 - 90℃; for 2h; Product distribution; | A 6.8% B 22.2% C 46% D 16.5% |

-
-
6374-78-3
1-amino-4,5,8-trihydroxyanthraquinone

-
A
-
81-60-7
1,4,5,8-tetrahydroxy-anthraquinone

-
B
-
69837-14-5
leuco-1,4,5,8-tetrahydroxyanthraquinone

| Conditions | Yield |
|---|---|
| With sodium dithionite In water at 100℃; for 0.666667h; | A 2.1% B 88% C 4.44% |


-
A
-
81-60-7
1,4,5,8-tetrahydroxy-anthraquinone

-
B
-
6374-78-3
1-amino-4,5,8-trihydroxyanthraquinone

-
C
-
69837-14-5
leuco-1,4,5,8-tetrahydroxyanthraquinone

| Conditions | Yield |
|---|---|
| With sodium dithionite In water at 100℃; for 0.666667h; | A 2.7% B 0.2% C 87% D 5.1% |
| With sodium hydroxide at 90 - 100℃; for 0.666667h; Product distribution; NaOH conc.; | A 17.4% B 12.7% C 8.47% D 24.9% |


| Conditions | Yield |
|---|---|
| With sodium hydroxide; sodium dithionite at 100℃; for 1h; | 80% |
| With hydrogenchloride at 180℃; | |
| With sodium dithionite; nitrobenzene 1.) 100 deg C; Multistep reaction; |
-
-
16517-70-7
1,4-diamino-5,8-dihydroxy-9,10-anthracenedione

-
-
81-60-7
1,4,5,8-tetrahydroxy-anthraquinone

| Conditions | Yield |
|---|---|
| With sodium hydroxide In water for 3h; Reflux; | 75% |

| Conditions | Yield |
|---|---|
| Stage #1: 1,5-diaminoanthraquinone With sodium hydroxide; water for 3h; Heating / reflux; Stage #2: With sodium hydrogensulfite In water for 3h; Heating / reflux; Stage #3: With hydrogenchloride In water pH=3; | 73% |
-
-
14597-12-7
3,6-dimethoxyphthalic anhydride

-
-
123-31-9
hydroquinone

-
-
81-60-7
1,4,5,8-tetrahydroxy-anthraquinone

| Conditions | Yield |
|---|---|
| With aluminium trichloride; sodium chloride |

| Conditions | Yield |
|---|---|
| With hydrogenchloride at 180℃; | |
| With sodium hydroxide; sodium disulfite 1.) n-BuOH, 15 min, 2.) n-BuOH, 60 deg C, 30 min; Yield given. Multistep reaction; |

| Conditions | Yield |
|---|---|
| With manganese(IV) oxide; sulfuric acid |

-
-
81-60-7
1,4,5,8-tetrahydroxy-anthraquinone

| Conditions | Yield |
|---|---|
| With nitrobenzene |

| Conditions | Yield |
|---|---|
| With cis-nitrous acid |
-
-
69837-14-5
leuco-1,4,5,8-tetrahydroxyanthraquinone

-
-
81-60-7
1,4,5,8-tetrahydroxy-anthraquinone

| Conditions | Yield |
|---|---|
| In nitrobenzene Heating; |

| Conditions | Yield |
|---|---|
| With sodium hydroxide at 85 - 90℃; for 2h; Product distribution; |

-
-
81-60-7
1,4,5,8-tetrahydroxy-anthraquinone

| Conditions | Yield |
|---|---|
| With sulfuric acid; boric acid |

-
-
81-60-7
1,4,5,8-tetrahydroxy-anthraquinone

| Conditions | Yield |
|---|---|
| With calcium hydroxide; water; copper at 250℃; |

-
-
81-60-7
1,4,5,8-tetrahydroxy-anthraquinone

-
-
81-60-7
1,4,5,8-tetrahydroxy-anthraquinone

| Conditions | Yield |
|---|---|
| With water; nitric acid |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: 76 percent / boric acid, nitric acid, oleum / 25 °C 2: 100 percent / sulfuric acid, iron / 0.5 h / 90 °C 3: 1.) 10percent aq. NaOH, 2.) sodium hydrosulfite / 1.) n-BuOH, 15 min, 2.) n-BuOH, 60 deg C, 30 min View Scheme |


| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 100 percent / sulfuric acid, iron / 0.5 h / 90 °C 2: 1.) 10percent aq. NaOH, 2.) sodium hydrosulfite / 1.) n-BuOH, 15 min, 2.) n-BuOH, 60 deg C, 30 min View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 88 percent / sodium dithionite / H2O / 0.67 h / 100 °C 2: nitrobenzene / Heating View Scheme |

-
-
81-60-7
1,4,5,8-tetrahydroxy-anthraquinone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 12.7 percent / NaOH, NaS2O8 / 0.67 h / 90 - 100 °C / NaOH conc. 2: 2.1 percent / sodium dithionite / H2O / 0.67 h / 100 °C View Scheme | |
| Multi-step reaction with 3 steps 1: 12.7 percent / NaOH, NaS2O8 / 0.67 h / 90 - 100 °C / NaOH conc. 2: 88 percent / sodium dithionite / H2O / 0.67 h / 100 °C 3: nitrobenzene / Heating View Scheme | |
| Multi-step reaction with 2 steps 1: 8.47 percent / NaOH, NaS2O8 / 0.67 h / 90 - 100 °C / NaOH conc. 2: nitrobenzene / Heating View Scheme | |
| Multi-step reaction with 2 steps 1: 87 percent / sodium dithionite, sodium hydroxyde / H2O / 0.67 h / 90 °C 2: nitrobenzene / Heating View Scheme |
-
-
10262-79-0
1,5-diamino-4,8-dinitro-anthraquinone

-
-
81-60-7
1,4,5,8-tetrahydroxy-anthraquinone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: sodium sulfide 2: nitrous acid View Scheme |
-
-
852177-97-0
1,8-diamino-4,5-dinitro-anthraquinone

-
-
81-60-7
1,4,5,8-tetrahydroxy-anthraquinone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: zinc dust; diluted sulfuric acid 2: nitrous acid View Scheme |
-
-
42013-63-8
1,8-diazido-anthraquinone

-
-
81-60-7
1,4,5,8-tetrahydroxy-anthraquinone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: sulfuric acid / 50 °C 2: hydrochloric acid / 180 °C View Scheme |
-
-
85192-94-5
1,5-diazido-anthraquinone

-
-
81-60-7
1,4,5,8-tetrahydroxy-anthraquinone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: sulfuric acid 2: hydrochloric acid / 180 °C View Scheme |
-
-
81-60-7
1,4,5,8-tetrahydroxy-anthraquinone

-
-
62-53-3
aniline

-
-
66181-84-8
1,4,5,8-tetraphenylaminoanthraquinone

| Conditions | Yield |
|---|---|
| With C24H24Br2ClN3Rh; 1,8-diazabicyclo[5.4.0]undec-7-ene at 120℃; for 10h; | 93% |
-
-
81-60-7
1,4,5,8-tetrahydroxy-anthraquinone

-
-
107-11-9
1-amino-2-propene

-
-
100495-33-8
1,4-bis(2,3-propyleneamino)-5,8-dihydroxy-9,10-anthracenedione

| Conditions | Yield |
|---|---|
| In ethanol 1.) 60 deg C, 4 h, 2.) 25 deg C, 15 h; | 90% |

-
-
81-60-7
1,4,5,8-tetrahydroxy-anthraquinone

-
-
135649-20-6
(NH2)2(CS)2(C6H2)2(CO)2(OH)3ONiCl(H2O)

| Conditions | Yield |
|---|---|
| In ethanol byproducts: HCl, (H2NCS)2; refluxed 3-5 h; concd., filtered, washed (ethanol), dried in vac. over P2O5; elem. anal.; | 83% |
-
-
81-60-7
1,4,5,8-tetrahydroxy-anthraquinone

-
-
143-15-7
1-dodecylbromide

-
-
146519-48-4
1,4,5,8-tetrakis(dodecyloxy)anthraquinone

| Conditions | Yield |
|---|---|
| With caesium carbonate In N,N-dimethyl-formamide at 100℃; for 15h; | 81% |
-
-
81-60-7
1,4,5,8-tetrahydroxy-anthraquinone

-
-
77-78-1
dimethyl sulfate

-
-
63229-37-8
1,4,5,8-Tetramethoxy-9,10-anthrachinon

| Conditions | Yield |
|---|---|
| With sodium hydride In tetrahydrofuran for 408h; Ambient temperature; | 76% |
| With potassium carbonate In acetone |

-
-
81-60-7
1,4,5,8-tetrahydroxy-anthraquinone

-
-
135649-22-8
(NH2)2(CS)2(C6H2)2(CO)2(OH)3OCoCl(H2O)

| Conditions | Yield |
|---|---|
| In ethanol byproducts: HCl, (NH2CS)2; refluxed 3-5 h; concd., filtered, washed (ethanol), dried in vac. over P2O5; elem. anal.; | 76% |
-
-
77339-03-8
((NH2)2(CS)2)2CdCl2

-
-
81-60-7
1,4,5,8-tetrahydroxy-anthraquinone

| Conditions | Yield |
|---|---|
| In ethanol byproducts: HCl; refluxed 3-5 h; concd., filtered, washed (ethanol), dried in vac. over P2O5; elem. anal.; | 73% |

-
-
81-60-7
1,4,5,8-tetrahydroxy-anthraquinone

-
-
135649-16-0
(NH2)2(CS)2(C6H2)2(CO)2(OH)3OCuCl(H2O)

| Conditions | Yield |
|---|---|
| In ethanol byproducts: HCl; refluxed 3-5 h, concd.; filtered, washed (ethanol), dried in vac. over P2O5; elem. anal.; | 73% |
-
-
81-60-7
1,4,5,8-tetrahydroxy-anthraquinone

-
-
108-00-9
N,N-dimethylethylenediamine

-
-
70476-63-0
1,4-bis{[2-(dimethylamino)ethyl]amino}-5,8-dihydroxyanthracene-9,10-dione

| Conditions | Yield |
|---|---|
| In ethanol; water for 25h; Reflux; Inert atmosphere; | 40% |
-
-
857637-03-7
[1-(2-aminoethyl)-3-piperidin-2-yl]methanol

-
-
81-60-7
1,4,5,8-tetrahydroxy-anthraquinone

-
-
919487-89-1
1,4-bis-{[2-(3-hydroxymethylpiperidine-1-yl)ethyl]amino}-5,8-dihydroxyanthracene-9,10-dione

| Conditions | Yield |
|---|---|
| In ethanol at 20℃; for 23h; Heating / reflux; | 21% |
-
-
81-60-7
1,4,5,8-tetrahydroxy-anthraquinone

| Conditions | Yield |
|---|---|
| With palladium on activated charcoal; acetic acid Hydrogenation; |
-
-
81-60-7
1,4,5,8-tetrahydroxy-anthraquinone

-
-
75313-28-9
2-butyl-1,4,5,8-tetrahydroxy-anthraquinone

| Conditions | Yield |
|---|---|
| With morpholine; sodium dithionite anschliessend mit Butyraldehyd und Oxidation mit Luft; |
-
-
81-60-7
1,4,5,8-tetrahydroxy-anthraquinone

-
-
69837-14-5
leuco-1,4,5,8-tetrahydroxyanthraquinone

| Conditions | Yield |
|---|---|
| With sodium dithionite |
1,4,5,8-Tetrahydroxyanthraquinone Chemical Properties
Chemical Name: 1,4,5,8-Tetrahydroxyanthraquinone
IUPAC NAME: 1,4,5,8-Tetrahydroxyanthracene-9,10-dione
CAS No.: 81-60-7
EINECS: 201-365-0
Molecular Formula: C14H8O6
Molecular Weight: 272.21 g/mol
Density: 1.781 g/cm3
Flash Point: 303.4 °C
Boiling Point: 554.8 °C at 760 mmHg
Following is the structure of 1,4,5,8-Tetrahydroxyanthraquinone (CAS No.81-60-7):
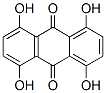
Product Categories about 1,4,5,8-Tetrahydroxyanthraquinone (CAS No.81-60-7) are Intermediates of Dyes and Pigments
The chemical synonymous of 1,4,5,8-Tetrahydroxyanthraquinone (CAS No.81-60-7) are 1,4,5,8-Tetrahydroxyanthracene-9,10-Dione ; 1,4,5,8-Tetrahydroxyanthraquinone ; Rarechem Aq Bd An29 ; 1,4,5,8-Leucotetraoxyanthraquinone ; 1,4,5,8-Ltoa ; 1,4,5,8-Tetrahydroxy-10-Anthracenedione ; 1,4,5,8-Tetrahydroxy-9,10-Anthacendione ; 1,4,5,8-Tetrahydroxyanthrachinon
1,4,5,8-Tetrahydroxyanthraquinone Toxicity Data With Reference
| 1. | skn-rbt 500 mg/24H MLD | 28ZPAK Sbornik Vysledku Toxixologickeho Vysetreni Latek A Pripravku Marhold, J.V.,Institut Pro Vychovu Vedoucicn Pracovniku Chemickeho Prumyclu Praha,Czechoslovakia.: 1972,104. | ||
| 2. | eye-rbt 500 mg/24H MLD | 28ZPAK Sbornik Vysledku Toxixologickeho Vysetreni Latek A Pripravku Marhold, J.V.,Institut Pro Vychovu Vedoucicn Pracovniku Chemickeho Prumyclu Praha,Czechoslovakia.: 1972,104. | ||
| 3. | ipr-rat LD50:2200 mg/kg | GTPZAB Gigiena Truda i Professionalnye Zabolevaniia. Labor Hygiene and Occupational Diseases. 21 (12)(1977),27. |
1,4,5,8-Tetrahydroxyanthraquinone Safety Profile
Moderately toxic by intraperitoneal route. A skin and eye irritant. When heated to decomposition it emits acrid smoke and irritating fumes.
Related Products
- 10,10'-Bis([1,1'-biphenyl]-4-yl)-9,9'-bianthracene
- 10,10'-Dibromo-9,9'-bianthryl
- 10,10-Dimethylanthrone
- 10,10-Oxybisphenoxarsine
- 10-[1,1'-Biphenyl]-4-yl-2-(1-methylethyl)-9-oxo-9H-thioxanthenium hexafluorophosphate
- 10,11-Dehydroimipramine
- 10,11-Dihydro-11-oxodibenzo[b,f][1,4]thiazepine
- 10,11-Dihydro-2-(4-methyl-1-piperazinyl)-11-(2-athiazolyl)-pyridazino(3,4-b)(1,4)benzoxazepine
- 10,11-DIHYDRO-2-(4-METHYL-1-PIPERAZINYL)-11-(3,4-XYLYL)PYRIDAZINO(3,4b)(1,4)-BENZOXAZEPINE MALEATE
- 10,11-Dihydro-5-(3-dimethylamino-2-methylpropyl)-5h-dibenz (b,f)azepine
- 81607-67-2
- 81608-27-7
- 81608-30-2
- 81613-61-8
- 81-61-8
- 816-27-3
- 81627-83-0
- 81627-84-1
- 81-62-9
- 81-63-0
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View