-
Name
1-Tetradecanol
- EINECS 204-000-3
- CAS No. 112-72-1
- Article Data102
- CAS DataBase
- Density 0.833 g/cm3
- Solubility insoluble in water
- Melting Point 35-39 °C(lit.)
- Formula C14H30O
- Boiling Point 263.2 °C at 760 mmHg
- Molecular Weight 214.392
- Flash Point 125.9 °C
- Transport Information
- Appearance white low melting solid or flakes
- Safety 24/25
- Risk Codes 38
-
Molecular Structure
-
Hazard Symbols
 Xi
Xi
- Synonyms Tetradecyl alcohol;n-Tetradecan-1-ol;n-Tetradecanol;n-Tetradecyl alcohol;Tetradecanol;Tetradecanol(7CI);1-Hydroxytetradecane;1-Tetradecyl alcohol;Adol 18;Alfol 14;Conol1495;Hinol 14SS;Kalcohl 40;Kalcohl 4098;Kalcol 4098;Lanette 14;Lanette K;Lanette Wax KS;Lorol C 14;Loxanol V;Myristic alcohol;Myristyl alcohol;NSC8549;Nacol 14-95;
- PSA 20.23000
- LogP 4.67980
Synthetic route

| Conditions | Yield |
|---|---|
| With C32H36ClNO2P2Ru; potassium tert-butylate; hydrogen In neat (no solvent) at 120℃; under 38002.6 Torr; for 20h; Autoclave; Green chemistry; | 99% |
| With [RuCl2(2-(diphenylphosphino)-N-((6-((diphenylphosphino)methyl)pyridin-2-yl)methyl)ethan-1-amine)]; potassium tert-butylate; hydrogen In tetrahydrofuran at 80℃; under 38002.6 Torr; for 5h; Catalytic behavior; Autoclave; | 97% |
| Stage #1: methyl myristoate With phenylsilane; fac-[Mn-(xantphos)(CO)3Br] at 120℃; for 12h; Inert atmosphere; Stage #2: With water; sodium hydroxide In methanol at 20℃; Inert atmosphere; | 96% |

-
-
112-72-1
1-Tetradecanol

| Conditions | Yield |
|---|---|
| With 18-crown-6 ether; tert-butylamine; tert-butyl alcohol; potassium In tetrahydrofuran at 20℃; for 2h; | 98% |
-
-
144708-82-7
1-Methoxymethoxy-tetradecane

-
-
112-72-1
1-Tetradecanol

| Conditions | Yield |
|---|---|
| With methanol; DOWEX-50W-X2 for 30h; Heating; | 96% |
-
-
108-88-3
toluene

-
A
-
112-72-1
1-Tetradecanol

-
B
-
620-47-3
1-methyl-3-(phenylmethyl)-benzene

-
C
-
620-83-7
1-methyl-4-(phenylmethyl)benzene

-
D
-
713-36-0
2-benzyltoluene

| Conditions | Yield |
|---|---|
| With boron trifluoride diethyl etherate at 120℃; for 2h; | A 96% B n/a C n/a D n/a |

-
-
77774-34-6
tetradecyl tert-butyldimethylsilyl ether

-
-
112-72-1
1-Tetradecanol

| Conditions | Yield |
|---|---|
| With toluene-4-sulfonic acid In methanol; dichloromethane at 20 - 25℃; for 0.333333h; Flow reactor; | 96% |

| Conditions | Yield |
|---|---|
| With zirconium(IV) borohydride In tetrahydrofuran at 25℃; for 1h; Reduction; | 95% |
| With sodium tetrahydroborate; benzene-1,2-diol; trifluoroacetic acid In tetrahydrofuran at 25℃; for 4h; | 91% |
| With hydrogen In neat (no solvent) at 160℃; under 37503.8 Torr; for 24h; | 88% |

| Conditions | Yield |
|---|---|
| With C30H34Cl2N2P2Ru; potassium methanolate; hydrogen In tetrahydrofuran at 100℃; under 38002.6 - 76005.1 Torr; for 15h; Glovebox; Autoclave; | 94% |
| With kieselguhr; copper at 350℃; under 73550.8 - 147102 Torr; Hydrogenation; | |
| With lithium aluminium tetrahydride |
-
-
146404-39-9
C21H38N2O2S

-
-
112-72-1
1-Tetradecanol

| Conditions | Yield |
|---|---|
| With potassium hydroxide; ethanol; oxygen Heating; | 91% |
-
-
14980-92-8
ethyl 2-bromomyristate

-
-
112-72-1
1-Tetradecanol

| Conditions | Yield |
|---|---|
| With sodium tetrahydroborate at 65℃; for 24h; | 90% |

-
-
112-72-1
1-Tetradecanol

| Conditions | Yield |
|---|---|
| With water; silica gel In methanol at 60℃; Inert atmosphere; | 89% |

| Conditions | Yield |
|---|---|
| With samarium diiodide; water; triethylamine; diisopropylamine In tetrahydrofuran at 20℃; for 2h; Inert atmosphere; | A 86% B 13% |

| Conditions | Yield |
|---|---|
| With 5 wt% Re/TiO2; hydrogen In neat (no solvent) at 230℃; under 37503.8 Torr; for 24h; Autoclave; | 86% |

| Conditions | Yield |
|---|---|
| Stage #1: Tetradecanoic acid 1-methylethyl ester With triethyl borane; sodium methylate In tetrahydrofuran; diethyl ether at 20℃; for 6h; Inert atmosphere; Stage #2: With sodium hydroxide In tetrahydrofuran; methanol; diethyl ether at 20℃; for 2h; Reagent/catalyst; Solvent; Time; | 84% |
| With diphenylsilane; tris(triphenylphosphine)rhodium(I) chloride In tetrahydrofuran at 50℃; for 24h; | 90 % Spectr. |

| Conditions | Yield |
|---|---|
| With potassium superoxide; tetraethylammonium bromide In N,N-dimethyl-formamide at 5 - 10℃; for 3h; | A 2% B 80% |
-
-
123-75-1
pyrrolidine

-
-
629-63-0
myristonitrile

-
A
-
74673-29-3
1-tetradecylpyrrolidine

-
B
-
2016-42-4
tetradecylamine

-
C
-
112-72-1
1-Tetradecanol

| Conditions | Yield |
|---|---|
| With samarium diiodide; water; triethylamine In tetrahydrofuran at 20℃; for 0.0833333h; Inert atmosphere; | A 24% B 71% C 4.4% |

-
-
130340-56-6
2-(3-carboxypropyl)-5-(12-hydroxydodecyl)thiophene

-
A
-
112-70-9
tridecan-1-ol

-
B
-
112-72-1
1-Tetradecanol

-
C
-
36653-82-4
1-Hexadecanol

-
D
-
62643-46-3
20-hydroxyeicosanoic acid

| Conditions | Yield |
|---|---|
| With Raney nickel W-2 In ethanol for 2h; Heating; Further byproducts given; | A 3.5% B 6% C 12.6% D 65% |

-
-
130340-56-6
2-(3-carboxypropyl)-5-(12-hydroxydodecyl)thiophene

-
A
-
112-70-9
tridecan-1-ol

-
B
-
112-72-1
1-Tetradecanol

-
C
-
117197-09-8
tridecyl hexanoate

-
D
-
62643-46-3
20-hydroxyeicosanoic acid

| Conditions | Yield |
|---|---|
| With Raney nickel W-2 In ethanol for 2h; Heating; Further byproducts given; | A 3.5% B 6% C 0.5% D 65% |

-
-
130340-56-6
2-(3-carboxypropyl)-5-(12-hydroxydodecyl)thiophene

-
A
-
112-70-9
tridecan-1-ol

-
B
-
112-72-1
1-Tetradecanol

-
C
-
71801-23-5
tetradecyl hexanoate

-
D
-
62643-46-3
20-hydroxyeicosanoic acid

| Conditions | Yield |
|---|---|
| With Raney nickel W-2 In ethanol for 2h; Heating; Further byproducts given; | A 3.5% B 6% C 0.6% D 65% |

-
-
130340-56-6
2-(3-carboxypropyl)-5-(12-hydroxydodecyl)thiophene

-
A
-
112-70-9
tridecan-1-ol

-
B
-
112-72-1
1-Tetradecanol

-
C
-
62643-46-3
20-hydroxyeicosanoic acid

-
D
-
130340-57-7
n-hexanoic acid 1-hexadecyl

| Conditions | Yield |
|---|---|
| With Raney nickel W-2 In ethanol for 2h; Heating; Further byproducts given; | A 3.5% B 6% C 65% D 0.1% |

-
-
130340-56-6
2-(3-carboxypropyl)-5-(12-hydroxydodecyl)thiophene

-
A
-
112-70-9
tridecan-1-ol

-
B
-
112-72-1
1-Tetradecanol

-
C
-
62643-46-3
20-hydroxyeicosanoic acid

-
D
-
14331-11-4
hexadecyl hexanoate

| Conditions | Yield |
|---|---|
| With Raney nickel W-2 In ethanol for 2h; Heating; Further byproducts given; | A 3.5% B 6% C 65% D 0.25% |

-
-
130340-56-6
2-(3-carboxypropyl)-5-(12-hydroxydodecyl)thiophene

-
A
-
112-70-9
tridecan-1-ol

-
B
-
112-72-1
1-Tetradecanol

-
C
-
62643-46-3
20-hydroxyeicosanoic acid

-
D
-
29710-33-6
tetradecyl heptanoate

| Conditions | Yield |
|---|---|
| With Raney nickel W-2 In ethanol for 2h; Heating; Further byproducts given; | A 3.5% B 6% C 65% D 0.4% |


| Conditions | Yield |
|---|---|
| With Au0998Ag0002; hydrogen; triethylamine at 90℃; under 6080.41 Torr; for 24h; chemoselective reaction; | 63% |
| With acetic acid; zinc | |
| With ethanol; sodium |

| Conditions | Yield |
|---|---|
| With potassium fluoride; benzylnixantphos; iron(II) acetate In tetrahydrofuran at 0 - 40℃; for 44h; Negishi Coupling; Inert atmosphere; | 60% |

| Conditions | Yield |
|---|---|
| With samarium diiodide; water; triethylamine In tetrahydrofuran at 20℃; for 2h; Inert atmosphere; | 55% |

| Conditions | Yield |
|---|---|
| With potassium fluoride; benzylnixantphos; iron(II) acetate In tetrahydrofuran at 0 - 40℃; for 44h; Negishi Coupling; Inert atmosphere; | 38% |

| Conditions | Yield |
|---|---|
| With triethyl borane; tri-n-butyl-tin hydride In hexane; benzene at 20℃; for 0.333333h; | A 18% B 30% |

| Conditions | Yield |
|---|---|
| With tert.-butylhydroperoxide; zirconocene dichloride; sodium bis(2-methoxyethoxy)aluminium dihydride 1.) THF, 72 h, 40 deg C, 2.) ClCH2CH2Cl, 1 h, room temp.; Yield given. Multistep reaction; | A 10% B n/a |

| Conditions | Yield |
|---|---|
| With 3,6,9-trioxaundecane; diborane und anschliessend mit wss.Wasserstoffperoxid und wss.Natronlauge; |
-
-
62936-14-5
(E)-5-tetradecenol

-
-
112-72-1
1-Tetradecanol

| Conditions | Yield |
|---|---|
| Hydrogenation; |

| Conditions | Yield |
|---|---|
| With sodium; butan-1-ol |
1-TETRADECANOL Chemical Properties
Synonyms: Dytol R-52;dytolr-52;Dehydag wax 14;Emery 3334;Epal 14 ;Alfol 14;alfol14;Cachalot M-43 NF;
The Molecular formula of 1-TETRADECANOL(112-72-1): C14H30O
The Molecular Weight of 1-TETRADECANOL(112-72-1): 214.39
Molecular Structure :
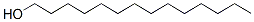
EINECS: 204-000-3
Melting point: 35-39 °C(lit.)
Boiling point: 289 °C(lit.)
Flash point: >230 °F
density: 0.823 g/mL at 25 °C(lit.)
vapor density: 7.4 (vs air)
storage temp.:-20°C
Water Solubility: insoluble
1-TETRADECANOL Uses
1-TETRADECANOL Toxicity Data With Reference
| 1. | skn-hmn 75 mg/3D-I MOD | 85DKA8 Cutaneous Toxicity Drill, V.A. andP. Lazar, eds.,New York, NY.: Academic Press,1977,127. |
1-TETRADECANOL Consensus Reports
1-TETRADECANOL Safety Profile
The Hazard Codes of 1-TETRADECANOL(112-72-1):
 Xi
Xi The Risk Statements information of 1-TETRADECANOL(112-72-1):
38: Irritating to the skin
The Safety Statements information of 1-TETRADECANOL(112-72-1):
24/25: Avoid contact with skin and eyes
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View