-
Name
2-Naphthoxyacetic acid
- EINECS 204-380-0
- CAS No. 120-23-0
- Article Data47
- CAS DataBase
- Density 1.274 g/cm3
- Solubility Soluble in diethyl ether, hot water, ethanol, acetic acid. Slightly soluble in colde water
- Melting Point 151-154 °C(lit.)
- Formula C12H10O3
- Boiling Point 388.729 °C at 760 mmHg
- Molecular Weight 202.21
- Flash Point 155.784 °C
- Transport Information
- Appearance gray crystals
- Safety 26-36
- Risk Codes 22-36/37/38
-
Molecular Structure
-
Hazard Symbols
 Xn,
Xn, Xi
Xi
- Synonyms Aceticacid, (2-naphthalenyloxy)- (9CI);Acetic acid, (2-naphthyloxy)- (8CI);Aceticacid, 2-naphthoxy- (3CI);(2-Naphthalenyloxy)acetic acid;(2-Naphthyloxy)aceticacid;(β-Naphthyloxy)acetic acid;2-NOXA;Betapal;Betokson;Betokson Super;
- PSA 46.53000
- LogP 2.30320
Synthetic route

| Conditions | Yield |
|---|---|
| With water; triethylamine; lithium bromide In acetonitrile at 20℃; for 0.25h; | 99% |
| With lithium hydroxide In tetrahydrofuran; water at 20℃; |

| Conditions | Yield |
|---|---|
| With sodium hydroxide Irradiation; | 95% |
| With sodium hydroxide In water at 95 - 100℃; for 0.166667h; Microwave irradiation; Sonication; Green chemistry; | 91% |
| With sodium hydroxide In water at 60℃; for 7h; | 90% |

| Conditions | Yield |
|---|---|
| With potassium hydroxide In methanol; water at 20℃; for 1h; | 90% |
| With potassium hydroxide | |
| Alkaline conditions; |
-
-
3926-62-3
sodium monochloroacetic acid

-
-
875-83-2
sodium 2-naphtholate

-
-
120-23-0
(2-naphthoyl)oxyacetic acid

| Conditions | Yield |
|---|---|
| 78% | |
| With Amberlite IRA 400 1.) water, 2.) water, 100 deg C, 1 h; Yield given. Multistep reaction; |

| Conditions | Yield |
|---|---|
| Stage #1: β-naphthol With sodium carbonate In butanone at 50℃; for 0.5h; Inert atmosphere; Stage #2: bromoacetic acid In butanone for 4h; Heating; Stage #3: With hydrogenchloride In water; ethyl acetate | 72% |
-
-
120-23-0
(2-naphthoyl)oxyacetic acid

-
-
40926-77-0
(2-naphthyloxy)acetyl chloride

| Conditions | Yield |
|---|---|
| With oxalyl dichloride; N,N-dimethyl-formamide In dichloromethane at 20℃; for 1h; Inert atmosphere; | 100% |
| With thionyl chloride for 16h; Ambient temperature; | 93% |
| With thionyl chloride |
-
-
120-23-0
(2-naphthoyl)oxyacetic acid

-
-
265324-03-6
N-(4-acetylaminophenyl)-N'-(4-aminophenyl)-1,4-phenylenediamine

| Conditions | Yield |
|---|---|
| With 1,1'-carbonyldiimidazole In tetrahydrofuran at 20℃; for 12h; | 100% |
-
-
120-23-0
(2-naphthoyl)oxyacetic acid

-
-
100-46-9
benzylamine

-
-
173946-17-3
N-benzyl-2-(naphthalen-2-yloxy)acetamide

| Conditions | Yield |
|---|---|
| Stage #1: (2-naphthoyl)oxyacetic acid With titanium(IV) isopropylate In tetrahydrofuran at 40 - 70℃; Molecular sieve; Inert atmosphere; Stage #2: benzylamine In tetrahydrofuran at 70℃; Molecular sieve; Inert atmosphere; | 99% |

| Conditions | Yield |
|---|---|
| With dmap; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride In dichloromethane at 20℃; for 48h; | 99% |
-
-
120-23-0
(2-naphthoyl)oxyacetic acid

-
-
116027-17-9
2-amino-5-benzyloxybenzoic acid methyl ester

| Conditions | Yield |
|---|---|
| With dmap; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride In dichloromethane at 20℃; for 16h; | 99% |
-
-
120-23-0
(2-naphthoyl)oxyacetic acid

-
-
15485-31-1, 97221-16-4
(4-nitrobenzylidene)(4-ethoxyphenyl)amine

| Conditions | Yield |
|---|---|
| With Vilsmeier reagent; triethylamine In dichloromethane at 20℃; | 95% |
| With Vilsmeier reagent; triethylamine In dichloromethane at 0 - 20℃; | 95% |
| With (methoxymethylidene)dimethylammonium methyl sulfate; triethylamine In dichloromethane at 20℃; | 88% |
| With triethylamine; p-toluenesulfonyl chloride In dichloromethane at 20℃; | 84% |
-
-
120-23-0
(2-naphthoyl)oxyacetic acid

-
-
15484-92-1
(4-chlorobenzylidene)(4-ethoxyphenyl)amine

| Conditions | Yield |
|---|---|
| With Vilsmeier reagent; triethylamine In dichloromethane at 0 - 20℃; | 95% |
| With 1,3,5-trichloro-2,4,6-triazine; triethylamine; N,N-dimethyl-formamide In dichloromethane at 20℃; | 91% |
| With Benzoyloxymethylene-dimethyl-ammonium; chloride; triethylamine In dichloromethane at 20℃; Staudinger reaction; | 91% |
| With Vilsmeier reagent; triethylamine In dichloromethane at 20℃; | 90% |
| With acetic anhydride; dimethyl sulfoxide; triethylamine In dichloromethane at 20℃; | 88% |

| Conditions | Yield |
|---|---|
| With 1-methyl-2-fluoropyridinium p-toluenesulfonate; triethylamine In dichloromethane at 20℃; stereoselective reaction; | 95% |
| With Benzoyloxymethylene-dimethyl-ammonium; chloride; triethylamine In dichloromethane at 20℃; Staudinger reaction; | 88% |
-
-
120-23-0
(2-naphthoyl)oxyacetic acid

| Conditions | Yield |
|---|---|
| With 2,6-di-tert-butyl-pyridine; N-fluorobis(benzenesulfon)imide In acetone for 3h; Inert atmosphere; Irradiation; | 95% |
| With (5,10,15,20-tetramesitylporphyrinato)manganese(III) chloride; iodosylbenzene; triethylamine tris(hydrogen fluoride); benzoic acid In 1,2-dichloro-ethane at 45℃; Inert atmosphere; Schlenk technique; | 46% |
-
-
120-23-0
(2-naphthoyl)oxyacetic acid

-
-
22706-11-2
3-phenyl-4-amino-5-mercapto-1,2,4-triazole

| Conditions | Yield |
|---|---|
| With dmap; tetrabutylammomium bromide; trichlorophosphate Microwave irradiation; Heating; | 95% |
-
-
120-23-0
(2-naphthoyl)oxyacetic acid

-
-
36209-49-1
4-amino-5-(4-methoxyphenyl)-4H-1,2,4-triazole-3-thiol

| Conditions | Yield |
|---|---|
| With dmap; tetrabutylammomium bromide; trichlorophosphate Microwave irradiation; Heating; | 95% |
-
-
120-23-0
(2-naphthoyl)oxyacetic acid

| Conditions | Yield |
|---|---|
| With dmap; tetrabutylammomium bromide; trichlorophosphate Microwave irradiation; Heating; | 95% |
-
-
120-23-0
(2-naphthoyl)oxyacetic acid

-
-
7524-50-7
methyl (2S)-2-amino-3-phenylpropanoate hydrochloride

-
-
107491-95-2
(S)-2-[2-(naphthalen-2-yloxy)-acetylamino]-3-phenyl-propionic acid methyl ester

| Conditions | Yield |
|---|---|
| With O-(benzotriazol-1-yl)-N,N,N',N'-tetramethyluronium tetrafluoroborate; triethylamine In dichloromethane at 20℃; | 94% |
-
-
120-23-0
(2-naphthoyl)oxyacetic acid

-
-
36304-47-9
1-(2-naphthyloxyacetyl)hydrazine

| Conditions | Yield |
|---|---|
| With hydrazine hydrate; Vilsmeier reagent; triethylamine In dichloromethane at 20℃; for 7h; | 94% |
| Multi-step reaction with 2 steps 1: H2SO4 / 3 h / Heating 2: hydrazine hydrate / ethanol / 14 h / Heating View Scheme | |
| Multi-step reaction with 2 steps 1: H2SO4 / 3 h 2: hydrazine hydrate / ethanol / 14 h / Heating View Scheme |
-
-
120-23-0
(2-naphthoyl)oxyacetic acid

-
-
15475-06-6
p-anisal-p-phenetidine

| Conditions | Yield |
|---|---|
| With (methoxymethylidene)dimethylammonium methyl sulfate; triethylamine In dichloromethane at 20℃; | 94% |
| With trifluoro-[1,3,5]triazine; triethylamine In dichloromethane at 20℃; | 90% |
| With 1-methyl-2-fluoropyridinium p-toluenesulfonate; triethylamine In dichloromethane at 20℃; stereoselective reaction; | 90% |
| With diethyl chlorophosphate; triethylamine at 20℃; |

| Conditions | Yield |
|---|---|
| With dmap; dacarbazine In dichloromethane at 20℃; for 24h; | 93% |
-
-
120-23-0
(2-naphthoyl)oxyacetic acid

-
-
80039-92-5
C19H14N4O2

| Conditions | Yield |
|---|---|
| With 1,3,5-trichloro-2,4,6-triazine; triethylamine; N,N-dimethyl-formamide In dichloromethane at 20℃; | 93% |
| With (methoxymethylidene)dimethylammonium methyl sulfate; triethylamine In dichloromethane at 20℃; | 92% |
2-Naphthoxyacetic acid Specification
The 2-Naphthoxyacetic acid, with the CAS registry number 120-23-0, is also known as (β-Naphthyloxy)acetic acid. It belongs to the product categories of Pharmaceutical Raw Materials; Phenoxyacetic Acids and Alcohols (substituted); Acetics acid and esters; Auxins; Biochemistry; Plant Growth Regulators; Building Blocks; C11 to C12; Carbonyl Compounds; Carboxylic Acids; Chemical Synthesis; Organic Building Blocks. Its EINECS number is 204-380-0. This chemical's molecular formula is C12H10O3 and molecular weight is 202.21. What's more, its systematic name is (2-Naphthyloxy)acetic acid. Its classification codes are: (1)Agricultural Chemical; (2)Growth regulator / Fertilizer. This chemical should be sealed and stored in a cool and dry place. It should be ensured that the workshop is well ventilated or equipped with exhaust devices. Moreover, it should be protected from oxides and fire. It is used as plant hormone to promote the growth of roots on clippings, to prevent fruit from falling prematurely.
Physical properties of 2-Naphthoxyacetic acid are: (1)ACD/LogP: 2.077; (2)# of Rule of 5 Violations: 0; (3)ACD/LogD (pH 5.5): -0.23; (4)ACD/LogD (pH 7.4): -1.55; (5)ACD/BCF (pH 5.5): 1.00; (6)ACD/BCF (pH 7.4): 1.00; (7)ACD/KOC (pH 5.5): 1.59; (8)ACD/KOC (pH 7.4): 1.00; (9)#H bond acceptors: 3; (10)#H bond donors: 1; (11)#Freely Rotating Bonds: 3; (12)Polar Surface Area: 46.53 Å2; (13)Index of Refraction: 1.637; (14)Molar Refractivity: 56.971 cm3; (15)Molar Volume: 158.684 cm3; (16)Polarizability: 22.585×10-24cm3; (17)Surface Tension: 53.6 dyne/cm; (18)Density: 1.274 g/cm3; (19)Flash Point: 155.784 °C; (20)Enthalpy of Vaporization: 67.289 kJ/mol; (21)Boiling Point: 388.729 °C at 760 mmHg; (22)Vapour Pressure: 0 mmHg at 25°C.
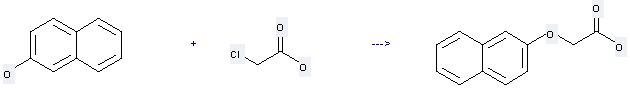
Preparation: this chemical can be prepared by chloroacetic acid and naphthalen-2-ol. This reaction will need reagent NaOH and solvent H2O with the reaction time of 2 hours.
Uses of 2-Naphthoxyacetic acid: it can be used to produce by naphthalen-2-yloxy-acetyl chloride at the ambient temperature. It will need reagent thionyl chloride with the reaction time of 16 hours. The yield is about 93%.

When you are using this chemical, please be cautious about it as the following:
This chemical is harmful if swallowed. It is irritating to eyes, respiratory system and skin. In case of contact with eyes, you should rinse immediately with plenty of water and seek medical advice. When using it, you need wear suitable protective clothing.
You can still convert the following datas into molecular structure:
(1)SMILES: O=C(O)COc2ccc1c(cccc1)c2
(2)Std. InChI: InChI=1S/C12H10O3/c13-12(14)8-15-11-6-5-9-3-1-2-4-10(9)7-11/h1-7H,8H2,(H,13,14)
(3)Std. InChIKey: RZCJYMOBWVJQGV-UHFFFAOYSA-N
The toxicity data is as follows:
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| rat | LD50 | oral | 600mg/kg (600mg/kg) | Pesticide Manual. Vol. 9, Pg. 609, 1991. |
Related Products
- 2-Naphthoxyacetic acid
- 1202-34-2
- 12023-54-0
- 120237-76-5
- 12023-91-5
- 1202-39-7
- 12024-10-1
- 1202-41-1
- 120241-31-8
- 12024-21-4
- 12024-22-5
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View