-
Name
3-Azetidinecarboxylic acid
- EINECS 674-976-7
- CAS No. 36476-78-5
- Article Data15
- CAS DataBase
- Density 1.275 g/cm3
- Solubility Soluble in water.
- Melting Point 286 °C (dec.)(lit.)
- Formula C4H7NO2
- Boiling Point 242 °C at 760 mmHg
- Molecular Weight 101.105
- Flash Point 100.1 °C
- Transport Information
- Appearance light yellow powder
- Safety 24/25-22-36-26
- Risk Codes 36/37/38
-
Molecular Structure
-
Hazard Symbols
 Xi
Xi
- Synonyms 3-Carboxyazetidine;Azetidine-3-carboxylic acid;
- PSA 49.33000
- LogP -0.38080
Synthetic route
-
-
94985-27-0
N-benzyl azetidine-3-carboxylic acid

-
-
36476-78-5
3-azetidinecarboxylic acid

| Conditions | Yield |
|---|---|
| palladium In methanol | 90% |
| With hydrogen; palladium dihydroxide In methanol; water at 60℃; under 2068.59 Torr; for 16h; | 806.2 g |
| palladium In methanol |
-
-
97628-91-6
1-acetylazetidine-3-carboxylic acid

-
-
36476-78-5
3-azetidinecarboxylic acid

| Conditions | Yield |
|---|---|
| With palladium 10% on activated carbon; hydrogen In ethanol at 40℃; under 15001.5 Torr; for 24h; | 27.9% |

-
-
36476-78-5
3-azetidinecarboxylic acid

| Conditions | Yield |
|---|---|
| With formic acid In water |
-
-
26096-30-0
N-benzyl-3,3-bis-(hydroxymethyl)-azetidine

-
-
87-69-4
tartaric acid

-
-
94985-27-0
N-benzyl azetidine-3-carboxylic acid

-
-
36476-78-5
3-azetidinecarboxylic acid

| Conditions | Yield |
|---|---|
| With potassium hydroxide; phosphoric acid; ammonium formate; palladium on charcoal In methanol; water |

-
-
142253-55-2
1-(t-butyloxycarbonyl)-azetidine-3-carboxylic acid

-
-
36476-78-5
3-azetidinecarboxylic acid

| Conditions | Yield |
|---|---|
| With palladium 10% on activated carbon; hydrogen In ethanol at 50℃; under 18751.9 Torr; for 30h; | 13.8 g |
-
-
36476-78-5
3-azetidinecarboxylic acid

-
-
501-53-1
benzyl chloroformate

-
-
97628-92-7
1-benzyloxycarbonyl-3-azetidinecarboxylic acid

| Conditions | Yield |
|---|---|
| Stage #1: 3-azetidinecarboxylic acid; benzyl chloroformate With sodium hydroxide In water at 0℃; for 16h; Stage #2: With hydrogenchloride In water | 100% |
| With potassium carbonate In water at 0 - 20℃; | 91% |
| Stage #1: 3-azetidinecarboxylic acid; benzyl chloroformate With potassium carbonate In 1,4-dioxane; water at 20℃; for 6h; Stage #2: With piperazine In 1,4-dioxane; water for 0.5h; | 83% |
-
-
24424-99-5
di-tert-butyl dicarbonate

-
-
36476-78-5
3-azetidinecarboxylic acid

-
-
142253-55-2
1-(t-butyloxycarbonyl)-azetidine-3-carboxylic acid

| Conditions | Yield |
|---|---|
| With triethylamine In methanol at 20℃; for 18h; | 100% |
| With triethylamine In methanol at 20℃; for 18h; | 100% |
| With sodium hydroxide In tetrahydrofuran; water at 20℃; for 24h; Product distribution / selectivity; | 95% |
-
-
919354-88-4
ethyl 6-chloro-5-cyano-2-(difluoromethyl)nicotinate

-
-
36476-78-5
3-azetidinecarboxylic acid

-
-
919354-92-0
1-[3-cyano-6-(difluoromethyl)-5-(ethoxycarbonyl)pyridin-2-yl]azetidine-3-carboxylic acid

| Conditions | Yield |
|---|---|
| With triethylamine In ethanol; water at 120℃; for 0.166667h; Microwave irradiation; | 100% |
-
-
36476-78-5
3-azetidinecarboxylic acid

-
-
407-25-0
trifluoroacetic anhydride

-
-
1001026-41-0
1-(trifluoroacetyl)azetidine-3-carboxylic acid

| Conditions | Yield |
|---|---|
| at 0℃; for 3.33333h; | 100% |
| at 0℃; for 2.83333h; | 100% |
-
-
67-56-1
methanol

-
-
36476-78-5
3-azetidinecarboxylic acid

-
-
100202-39-9
methyl 3-azetidinecarboxylate hydrochloride

| Conditions | Yield |
|---|---|
| With thionyl chloride In acetonitrile at 0 - 20℃; for 2.33333h; | 100% |
| With thionyl chloride at 0 - 25℃; for 2.25h; | 100% |
| With thionyl chloride at 5 - 65℃; | 100% |

| Conditions | Yield |
|---|---|
| With thionyl chloride at 0 - 20℃; for 10h; | 100% |
| With chloro-trimethyl-silane Heating / reflux; | |
| With chloro-trimethyl-silane Heating / reflux; |

-
-
36476-78-5
3-azetidinecarboxylic acid

| Conditions | Yield |
|---|---|
| Stage #1: 2-fluoro-4-[5-(4-isobutylphenyl)-1,2,4-oxadiazol-3-yl]-benzaldehyde; 3-azetidinecarboxylic acid With acetic acid In tetrahydrofuran at 20℃; for 2h; Stage #2: With methanol; sodium cyanoborohydride In tetrahydrofuran at 15 - 20℃; for 17h; Temperature; Concentration; Solvent; | 92.6% |
| Stage #1: 2-fluoro-4-[5-(4-isobutylphenyl)-1,2,4-oxadiazol-3-yl]-benzaldehyde; 3-azetidinecarboxylic acid With acetic acid In methanol at 20℃; for 2h; Large scale; Stage #2: With sodium cyanoborohydride In methanol at 20℃; for 17h; Large scale; | 79% |
| Stage #1: 2-fluoro-4-[5-(4-isobutylphenyl)-1,2,4-oxadiazol-3-yl]-benzaldehyde; 3-azetidinecarboxylic acid With acetic acid In tetrahydrofuran; methanol at 20℃; for 2h; Stage #2: With methanol; sodium cyanoborohydride In tetrahydrofuran at 20℃; for 16h; | 2 g |
-
-
1230487-01-0
4-[(1E)-1-([[4-cyclohexyl-3-(trifluoromethyl)phenyl]methoxy]imino)ethyl]-2-ethylbenzaldehyde

-
-
36476-78-5
3-azetidinecarboxylic acid

-
-
1230487-00-9
Siponimod

| Conditions | Yield |
|---|---|
| Stage #1: 4-[(1E)-1-([[4-cyclohexyl-3-(trifluoromethyl)phenyl]methoxy]imino)ethyl]-2-ethylbenzaldehyde; 3-azetidinecarboxylic acid In methanol at 20 - 25℃; for 0.5h; Stage #2: With methanol; sodium tris(acetoxy)borohydride | 89% |
| Stage #1: 4-[(1E)-1-([[4-cyclohexyl-3-(trifluoromethyl)phenyl]methoxy]imino)ethyl]-2-ethylbenzaldehyde; 3-azetidinecarboxylic acid In methanol at 20 - 25℃; for 0.5h; Stage #2: With sodium tris(acetoxy)borohydride In methanol Reagent/catalyst; Temperature; | 89% |
| Stage #1: 4-[(1E)-1-([[4-cyclohexyl-3-(trifluoromethyl)phenyl]methoxy]imino)ethyl]-2-ethylbenzaldehyde; 3-azetidinecarboxylic acid With acetic acid In methanol at 20℃; for 0.333333h; Stage #2: With sodium cyanoborohydride In methanol at 20℃; | 20% |
-
-
1446358-48-0
3-(3-(4-chloro-5-fluoropyrimidin-2-yl)-1-(2-fluorobenzyl)-1H-pyrazol-5-yl)isoxazole

-
-
36476-78-5
3-azetidinecarboxylic acid

| Conditions | Yield |
|---|---|
| With triethylamine In tetrahydrofuran at 75℃; for 18h; | 88% |

-
-
36476-78-5
3-azetidinecarboxylic acid

| Conditions | Yield |
|---|---|
| With N-ethyl-N,N-diisopropylamine; HATU In dichloromethane at 30℃; | 88% |
-
-
1220708-88-2
N-butyl-2-fluoro-N-(5-(4-formylphenyl)-1,3,4-thiadiazol-2-yl)benzamide

-
-
36476-78-5
3-azetidinecarboxylic acid

-
-
1220709-59-0
1-(4-(5-(N-butyl-2-fluorobenzamido)-1,3,4-thiadiazol-2-yl)benzyl)azetidine-3-carboxylic acid

| Conditions | Yield |
|---|---|
| Stage #1: N-butyl-2-fluoro-N-(5-(4-formylphenyl)-1,3,4-thiadiazol-2-yl)benzamide; 3-azetidinecarboxylic acid With acetic acid In methanol at 20℃; for 1h; Stage #2: With methanol; sodium cyanoborohydride at 20℃; Stage #3: With acetic acid In methanol pH=3 - 4; | 87% |
| Stage #1: N-butyl-2-fluoro-N-(5-(4-formylphenyl)-1,3,4-thiadiazol-2-yl)benzamide; 3-azetidinecarboxylic acid With acetic acid In methanol at 20℃; for 1h; Stage #2: With sodium cyanoborohydride In methanol at 20℃; | 87% |

-
-
36476-78-5
3-azetidinecarboxylic acid

-
-
847585-32-4
1-{[6-(2,3-dihydro-1H-inden-2-ylmethoxy)-1-methyl-3,4-dihydro-2-naphthalenyl]methyl}-3-azetidinecarboxylic acid hydrochloride

| Conditions | Yield |
|---|---|
| Stage #1: 6-(2,3-dihydro-1H-inden-2-ylmethoxy)-1-methyl-3,4-dihydro-2-naphthalenecarbaldehyde; 3-azetidinecarboxylic acid With sodium hydroxide; trimethyl orthoformate In tetrahydrofuran; methanol at 0℃; for 4h; Stage #2: With sodium tetrahydroborate In tetrahydrofuran; methanol at 0℃; for 0.5h; Stage #3: With hydrogenchloride In tetrahydrofuran; water at 20℃; for 0.5h; | 87% |
-
-
36476-78-5
3-azetidinecarboxylic acid

-
-
17341-93-4
2,2,2-Trichloroethyl chloroformate

| Conditions | Yield |
|---|---|
| Stage #1: 3-azetidinecarboxylic acid With sodium hydroxide In water at -10℃; Stage #2: 2,2,2-Trichloroethyl chloroformate In water at -10 - 24℃; for 3h; | 87% |
-
-
36476-78-5
3-azetidinecarboxylic acid

-
-
123-11-5
4-methoxy-benzaldehyde

-
-
1127402-49-6
1-(4-methoxy-benzyl)-azetidine-3-carboxylic acid

| Conditions | Yield |
|---|---|
| With palladium 10% on activated carbon; hydrogen In methanol at 20℃; under 1034.32 Torr; | 85% |
| With palladium 10% on activated carbon; hydrogen In methanol at 25℃; under 760.051 Torr; for 1h; | 82% |
-
-
36476-78-5
3-azetidinecarboxylic acid

-
-
67-64-1
acetone

-
-
959238-28-9
1-isopropyl-azetidine-3-carboxylic acid

| Conditions | Yield |
|---|---|
| With palladium 10% on activated carbon; hydrogen In methanol at 20℃; under 1034.32 Torr; | 85% |

-
-
36476-78-5
3-azetidinecarboxylic acid

| Conditions | Yield |
|---|---|
| Stage #1: 4-(5-(4-(2-fluoroethoxy)phenyl)-1,2,4-oxadiazol-3-yl)benzaldehyde; 3-azetidinecarboxylic acid With acetic acid In methanol for 0.5h; Stage #2: With sodium cyanoborohydride In methanol | 85% |
-
-
175277-73-3
ethyl 6-chloro-5-cyano-2-(trifluoromethyl)nicotinate

-
-
36476-78-5
3-azetidinecarboxylic acid

-
-
919354-83-9
1-[3-cyano-5-(ethoxycarbonyl)-6-(trifluoromethyl)pyridin-2-yl]azetidine-3-carboxylic acid

| Conditions | Yield |
|---|---|
| With triethylamine In ethanol for 0.333333h; Microwave irradiation; | 84% |
| With triethylamine In ethanol for 0.333333h; Microwave irradiation; | 84% |
-
-
635701-88-1
4-[5-(4-isobutylphenyl)-1,2,4-oxadiazol-3-yl]-benzaldehyde

-
-
36476-78-5
3-azetidinecarboxylic acid

| Conditions | Yield |
|---|---|
| Stage #1: 4-[5-(4-isobutylphenyl)-1,2,4-oxadiazol-3-yl]-benzaldehyde; 3-azetidinecarboxylic acid With acetic acid In tetrahydrofuran; methanol at 20℃; for 2h; Stage #2: With sodium cyanoborohydride In tetrahydrofuran; methanol at 20℃; for 16h; | 82% |
| Stage #1: 4-[5-(4-isobutylphenyl)-1,2,4-oxadiazol-3-yl]-benzaldehyde; 3-azetidinecarboxylic acid With acetic acid In methanol at 20℃; for 0.333333h; Stage #2: With sodium cyanoborohydride In methanol for 1h; | 74% |
| Stage #1: 4-[5-(4-isobutylphenyl)-1,2,4-oxadiazol-3-yl]-benzaldehyde; 3-azetidinecarboxylic acid With acetic acid In methanol at 20℃; for 0.333333h; Stage #2: With sodium cyanoborohydride In methanol for 1h; | 74% |
| Stage #1: 4-[5-(4-isobutylphenyl)-1,2,4-oxadiazol-3-yl]-benzaldehyde; 3-azetidinecarboxylic acid With acetic acid In methanol at 20℃; for 0.333333h; Stage #2: With sodium cyanoborohydride In methanol for 1h; | 74% |
-
-
1396776-39-8
N-((4,6-dimethyl-2-oxo-1,2-dihydropyridin-3-yl)methyl)-1-isopropyl-6-(1,2,3,6-tetrahydropyridin-4-yl)-1H-pyrazolo[3,4-b]pyridine-4-carboxamide

-
-
36476-78-5
3-azetidinecarboxylic acid

-
-
1396772-06-7
6-(1-(azetidine-3-carbonyl)-1,2,3,6-tetrahydropyridin-4-yl)-N-((4,6-dimethyl-2-oxo-1,2-dihydropyridin-3-yl)methyl)-1-isopropyl-1H-pyrazolo[3,4-b]pyridine-4-carboxamide

| Conditions | Yield |
|---|---|
| Stage #1: N-((4,6-dimethyl-2-oxo-1,2-dihydropyridin-3-yl)methyl)-1-isopropyl-6-(1,2,3,6-tetrahydropyridin-4-yl)-1H-pyrazolo[3,4-b]pyridine-4-carboxamide; 3-azetidinecarboxylic acid With triethylamine In dimethyl sulfoxide at 20℃; for 0.25h; Stage #2: With benzotriazol-1-yloxyl-tris-(pyrrolidino)-phosphonium hexafluorophosphate In dimethyl sulfoxide | 81.9% |
3-Azetidinecarboxylic acid Chemical Properties
IUPAC Name: azetidine-3-carboxylic acid
Empirical Formula: C4H7NO2
Molecular Weight: 101.1039g/mol
Structure of 3-Azetidinecarboxylic acid (CAS NO.36476-78-5):
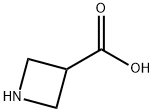
Index of Refraction: 1.499
Molar Refractivity: 23.29 cm3
Molar Volume: 79.2 cm3
Polarizability: 9.23×10-24cm3
Surface Tension: 48.2 dyne/cm
Density: 1.275 g/cm3
Flash Point: 100.1 °C
Enthalpy of Vaporization: 52.75 kJ/mol
Melting Point: 286 °C (dec.)(lit.)
Boiling Point: 242 °C at 760 mmHg
Vapour Pressure: 0.0116 mmHg at 25°C
Product Categories: Pharmaceutical Raw Materials;Unusual amino acids;Heterocycles series;Miscellaneous;Carboxylic Acids;Amino Acids 13C, 2H, 15N;Azetidines;Simple 4-Membered Ring Compounds;Peptide;Amino Acids & Derivatives;Carboxylic Acids;Ring Systems;Alicyclic Amino Acids;Peptide Synthesis;Unnatural Amino Acid Derivatives;B-Amino
Canonical SMILES: C1C(CN1)C(=O)O
InChI: InChI=1S/C4H7NO2/c6-4(7)3-1-5-2-3/h3,5H,1-2H2,(H,6,7)
InChIKey: GFZWHAAOIVMHOI-UHFFFAOYSA-N
3-Azetidinecarboxylic acid Uses
A useful intermediate in the synthesis of polypeptides.
3-Azetidinecarboxylic acid Toxicity Data With Reference
| 1. | orl-rat LD50:>5 g/kg | FMCHA2 Farm Chemicals Handbook .(Meister Publishing,Willoughy, OH.: )1991,C65. | ||
| 2. | skn-rbt LD50:>1 g/kg | FMCHA2 Farm Chemicals Handbook .(Meister Publishing,Willoughy, OH.: )1991,C65. |
3-Azetidinecarboxylic acid Safety Profile
Low toxicity by ingestion and skin contact. When heated to decomposition it emits toxic vapors of NOx.
Hazard Codes:  Xi
Xi
The Risk Statements information of 3-AZETIDINECARBOXYLIC ACID:
36/37/38: Irritating to eyes, respiratory system and skin
The Safety Statements information of 3-AZETIDINECARBOXYLIC ACID :
22: Do not breathe dust
26: In case of contact with eyes, rinse immediately with plenty of water and seek medical advice
36: Wear suitable protective clothing
24/25: Avoid contact with skin and eyes
WGK Germany:3
Hazard Note:Irritant
3-Azetidinecarboxylic acid Specification
3-Azetidinecarboxylic acid , its cas register number is 36476-78-5. It also can be called 3-Carboxyazetidine ; 5-22-01-00023 (Beilstein Handbook Reference) ; Azetidine-3-carboxylic acid ; CHA-811 ; Caswell No. 063C ; EPA Pesticide Chemical Code 128830 . 3-Azetidinecarboxylic acid (CAS NO.36476-78-5) is a light yellow powder.
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View