-
Name
3-Chloropropyltriethoxysilane
- EINECS 225-805-6
- CAS No. 5089-70-3
- Article Data28
- CAS DataBase
- Density 0.989 g/cm3
- Solubility 113-1000000mg/L at 20℃
- Melting Point
- Formula C9H21ClO3Si
- Boiling Point 230.8 °C at 760 mmHg
- Molecular Weight 240.802
- Flash Point 78.8 °C
- Transport Information UN 1993 3/PG 3
- Appearance colorless transparent liquid
- Safety 16-26-36
- Risk Codes 10-36/37/38
-
Molecular Structure
-
Hazard Symbols
 Xi
Xi
- Synonyms (3-Chloropropyl)triethoxysilane;(g-Chloropropyl)triethoxysilane;3-(Triethoxysilyl)-1-chloropropane;3-(Triethoxysilyl)propyl chloride;KH 230;NSC 252156;Si 223;Si 230;Triethoxy(g-chloropropyl)silane;g-Chloropropylltriethoxysilane;3-chloropropyl-triethoxy-silane;
- PSA 27.69000
- LogP 2.66370
Synthetic route

| Conditions | Yield |
|---|---|
| With Ru-B/γ-Al2O3 at 115℃; for 1h; Temperature; Inert atmosphere; | 99.44% |
| With dihydrogen hexachloroplatinate; Triethoxyvinylsilane; isopropyl alcohol at 50 - 80℃; for 4h; Product distribution; Mechanism; influence unsaturated Si-comp. on rate of hydrosilylation; effect of other additives; | 22.7 % Chromat. |
| bis(1,5-cyclooctadiene)diiridium(I) dichloride at -78 - 40℃; for 2h; Product distribution / selectivity; Inert atmosphere; pressure tight tube; | 21.8 %Chromat. |
| With di-tert-butyl peroxide In methanol at 80 - 91℃; for 4h; | 65 %Chromat. |

| Conditions | Yield |
|---|---|
| at 80 - 90℃; under 600 Torr; Large scale; | 99% |
| With tin(IV) chloride; cyclohexene for 1h; Ambient temperature; | 94.5% |
| In dichloromethane for 3h; Heating; | 92% |

| Conditions | Yield |
|---|---|
| In para-xylene | 83% |

-
-
954114-17-1
(3-chloropropyl)trivinylsilane

-
-
64-17-5
ethanol

-
-
5089-70-3
(3-chloropropyl)triethoxysilane

| Conditions | Yield |
|---|---|
| With hydrogenchloride; chlorobis(cyclooctene)rhodium(I) dimer In 1,4-dioxane; chloroform at 20℃; for 2h; | 100 % Chromat. |
| hydrogenchloride; chlorobis(cyclooctene)rhodium(I) dimer In 1,4-dioxane; chloroform for 2h; Product distribution / selectivity; |
-
-
998-30-1
Triethoxysilane

-
-
107-05-1
3-chloroprop-1-ene

-
A
-
78-10-4
tetraethoxy orthosilicate

-
B
-
135795-91-4
(3-chloropropyl)-diethoxy-chlorosilane

-
C
-
4667-99-6
chlorotriethoxysilane

-
D
-
2550-02-9
propyltriethoxysilane

-
E
-
5089-70-3
(3-chloropropyl)triethoxysilane

| Conditions | Yield |
|---|---|
| Stage #1: Triethoxysilane; ruthenium trichloride at 20 - 120℃; Under N2 atmosphere; Stage #2: 3-chloroprop-1-ene at 70 - 120℃; for 1h; Product distribution / selectivity; Under N2 atmosphere; | |
| chloro(1,5-cyclooctadiene)iridium(I) dimer at 20 - 70℃; for 18h; Product distribution / selectivity; Under N2 atmosphere; | |
| ruthenium trichloride at 25℃; for 18h; Product distribution / selectivity; Under N2 atmosphere; |

-
-
40372-72-3
bis(3-triethoxysilylpropyl) tetrasulfide

-
-
111-64-8
n-octanoic acid chloride

-
A
-
220727-26-4
3-(octanoylsulfanyl)-1-propyltriethoxysilane

-
B
-
106-32-1
octanoic acid ethyl ester

-
C
-
5089-70-3
(3-chloropropyl)triethoxysilane

-
D
-
14814-09-6
3-Mercaptopropyltriethoxysilane

-
E
-
60764-86-5
bis-[γ-(triethoxysilyl)propyl]sulfide

-
F
-
56706-10-6
4,4,13,13-tetraethoxy-3,14-dioxa-8,9-dithia-4,13-disilahexadecane

-
G
-
56706-11-7
4,4,14,14-tetraethoxy-3,15-dioxa-8,9,10-trithia-4,14-disilaheptadecane

| Conditions | Yield |
|---|---|
| Stage #1: bis(3-triethoxysilylpropyl) tetrasulfide With sodium In Solvent 140 at 110℃; for 0.75h; Stage #2: n-octanoic acid chloride In Solvent 140 at 45 - 104℃; for 1h; Stage #3: With water In toluene at 45℃; Product distribution / selectivity; |

-
-
14814-09-6
3-Mercaptopropyltriethoxysilane

-
-
111-64-8
n-octanoic acid chloride

-
A
-
220727-26-4
3-(octanoylsulfanyl)-1-propyltriethoxysilane

-
B
-
106-32-1
octanoic acid ethyl ester

-
C
-
5089-70-3
(3-chloropropyl)triethoxysilane

-
D
-
60764-86-5
bis-[γ-(triethoxysilyl)propyl]sulfide

| Conditions | Yield |
|---|---|
| Stage #1: 3-Mercaptopropyltriethoxysilane With sodium In toluene at 105℃; for 0.5 - 1.36667h; Stage #2: n-octanoic acid chloride In toluene at 20 - 62℃; for 1 - 1.58333h; Stage #3: With water In toluene at 32 - 50℃; Product distribution / selectivity; |

-
-
56706-10-6
4,4,13,13-tetraethoxy-3,14-dioxa-8,9-dithia-4,13-disilahexadecane

-
-
111-64-8
n-octanoic acid chloride

-
A
-
220727-26-4
3-(octanoylsulfanyl)-1-propyltriethoxysilane

-
B
-
106-32-1
octanoic acid ethyl ester

-
C
-
5089-70-3
(3-chloropropyl)triethoxysilane

-
D
-
14814-09-6
3-Mercaptopropyltriethoxysilane

-
E
-
60764-86-5
bis-[γ-(triethoxysilyl)propyl]sulfide

-
F
-
56706-11-7
4,4,14,14-tetraethoxy-3,15-dioxa-8,9,10-trithia-4,14-disilaheptadecane

| Conditions | Yield |
|---|---|
| Stage #1: 4,4,13,13-tetraethoxy-3,14-dioxa-8,9-dithia-4,13-disilahexadecane With sodium In toluene at 110℃; for 0.75 - 1.15h; Stage #2: n-octanoic acid chloride In toluene at 45 - 60℃; for 1 - 2h; Stage #3: With water In toluene at 45℃; Product distribution / selectivity; |

-
-
998-30-1
Triethoxysilane

-
-
107-05-1
3-chloroprop-1-ene

-
A
-
4667-99-6
chlorotriethoxysilane

-
B
-
2550-02-9
propyltriethoxysilane

-
C
-
5089-70-3
(3-chloropropyl)triethoxysilane

| Conditions | Yield |
|---|---|
| bis(1,5-cyclooctadiene)diiridium(I) dichloride at -78 - 60℃; for 1h; Product distribution / selectivity; Inert atmosphere; pressure tight tube; | A 33.2 %Chromat. B 5.7 %Chromat. C 34.8 %Chromat. |

-
-
998-30-1
Triethoxysilane

-
-
107-05-1
3-chloroprop-1-ene

-
A
-
4667-99-6
chlorotriethoxysilane

-
B
-
5089-70-3
(3-chloropropyl)triethoxysilane

| Conditions | Yield |
|---|---|
| bis(1,5-cyclooctadiene)diiridium(I) dichloride at -78 - 30℃; for 2h; Product distribution / selectivity; Inert atmosphere; pressure tight tube; | A 16.2 %Chromat. B 48.8 %Chromat. |
| With platinum(0)-1,3-divinyl-1,1,3,3-tetramethyldisiloxane complex In toluene at 40 - 60℃; for 16h; Overall yield = 32 %Spectr.; |


| Conditions | Yield |
|---|---|
| In tetrahydrofuran at 30 - 180℃; |
-
-
998-30-1
Triethoxysilane

-
-
107-05-1
3-chloroprop-1-ene

-
A
-
78-10-4
tetraethoxy orthosilicate

-
B
-
5089-70-3
(3-chloropropyl)triethoxysilane

| Conditions | Yield |
|---|---|
| With dihydrogen hexachloroplatinate; Triethoxyvinylsilane at 55 - 65℃; |


| Conditions | Yield |
|---|---|
| With potassium hydroxide at 240℃; for 3h; Autoclave; Molecular sieve; |
-
-
5089-70-3
(3-chloropropyl)triethoxysilane

| Conditions | Yield |
|---|---|
| In N,N-dimethyl acetamide at 100℃; for 2h; | 100% |
-
-
5089-70-3
(3-chloropropyl)triethoxysilane

| Conditions | Yield |
|---|---|
| at 120℃; for 24h; Inert atmosphere; | 100% |
-
-
590-28-3
potassium cyanate

-
-
5089-70-3
(3-chloropropyl)triethoxysilane

-
-
24801-88-5
3-(triethoxypropyl) isocyanate

| Conditions | Yield |
|---|---|
| at 100℃; for 12h; Temperature; Inert atmosphere; Ionic liquid; | 99.5% |
-
-
279238-29-8
tert-butyl (3-(1H-imidazol-1-yl)propyl)carbamate

-
-
5089-70-3
(3-chloropropyl)triethoxysilane

-
-
1005216-91-0
1-[(N-tert-butoxycarbonyl)aminopropyl]-3-(3-triethoxysilylpropyl)imidazolinium chloride

| Conditions | Yield |
|---|---|
| at 90℃; for 60h; | 99% |
-
-
5089-70-3
(3-chloropropyl)triethoxysilane

| Conditions | Yield |
|---|---|
| With hydrogenchloride In ethanol; water for 5h; Heating / reflux; | 99% |
-
-
280-57-9
1,4-diaza-bicyclo[2.2.2]octane

-
-
5089-70-3
(3-chloropropyl)triethoxysilane

-
-
1403993-57-6
DABCO-SIL

| Conditions | Yield |
|---|---|
| In ethanol; acetone for 24h; Reflux; | 99% |
| In toluene for 48h; Reflux; Inert atmosphere; | 96% |
| With sodium hydride In tetrahydrofuran at 60℃; for 16h; Sonication; |

| Conditions | Yield |
|---|---|
| Stage #1: 2-chloroethanamine hydrochloride With potassium hydroxide for 1h; Cooling with ice; Stage #2: (3-chloropropyl)triethoxysilane In N,N-dimethyl-formamide at 110℃; for 24h; Inert atmosphere; | 99% |

| Conditions | Yield |
|---|---|
| Stage #1: 3-chloropropylamine hydrochloride With potassium hydroxide for 1h; Cooling with ice; Stage #2: (3-chloropropyl)triethoxysilane In N,N-dimethyl-formamide at 110℃; for 24h; Inert atmosphere; | 99% |

| Conditions | Yield |
|---|---|
| With sodium iodide In dimethyl sulfoxide at 90℃; for 2h; | 98.6% |

| Conditions | Yield |
|---|---|
| Stage #1: (3-chloropropyl)triethoxysilane With sodium hydride In tetrahydrofuran at 50℃; for 0.5h; Inert atmosphere; Stage #2: 1,1,3-trimethylthiourea In tetrahydrofuran at 66℃; for 4h; Inert atmosphere; | 98.5% |

| Conditions | Yield |
|---|---|
| With potassium hydroxide at 90℃; under 100 - 350 Torr; for 4h; | 98.2% |

| Conditions | Yield |
|---|---|
| at 78℃; for 24h; Inert atmosphere; | 98% |
| at 80℃; for 30h; Inert atmosphere; | 96% |
| at 245℃; for 0.0111111h; microwave irradiation; | 95% |

-
-
5089-70-3
(3-chloropropyl)triethoxysilane

| Conditions | Yield |
|---|---|
| In DMF (N,N-dimethyl-formamide) at 100℃; for 8h; | 98% |
-
-
5089-70-3
(3-chloropropyl)triethoxysilane

-
-
14814-09-6
3-Mercaptopropyltriethoxysilane

-
-
60764-86-5
bis-[γ-(triethoxysilyl)propyl]sulfide

| Conditions | Yield |
|---|---|
| Stage #1: (3-chloropropyl)triethoxysilane; 3-Mercaptopropyltriethoxysilane at 60℃; for 2h; Stage #2: With sodium ethanolate In ethanol at 65℃; for 2h; Temperature; | 98% |

| Conditions | Yield |
|---|---|
| at 100℃; for 0.0833333h; Inert atmosphere; | 98% |

| Conditions | Yield |
|---|---|
| Stage #1: (3-chloropropyl)triethoxysilane With sodium hydride In toluene at 50℃; for 0.5h; Inert atmosphere; Stage #2: N,N',N''-triphenylguanidine In tetrahydrofuran at 110℃; for 5h; | 97.9% |
-
-
5089-70-3
(3-chloropropyl)triethoxysilane

-
-
40372-72-3
bis(3-triethoxysilylpropyl) tetrasulfide

| Conditions | Yield |
|---|---|
| With sodium polysulfide In ethanol at 78℃; Product distribution / selectivity; | 97.8% |
| With sodium sulfide; sodium hydrogencarbonate; sulfur In ethanol; water | |
| With sodium sulfide; sulfur In ethanol; toluene for 4h; Reflux; Green chemistry; | |
| With sodium sulfide; sodium borate; dodecyltrimethylammonium bromide; sulfur; potassium iodide In water at 40 - 55℃; for 1h; Temperature; Reagent/catalyst; |

| Conditions | Yield |
|---|---|
| Stage #1: (3-chloropropyl)triethoxysilane With sodium hydroxide In toluene at 50℃; for 0.5h; Inert atmosphere; Stage #2: N,N'-diethylthiourea In toluene at 110℃; for 5h; Inert atmosphere; | 97.8% |

| Conditions | Yield |
|---|---|
| Stage #1: (3-chloropropyl)triethoxysilane With sodium hydride In tetrahydrofuran at 50℃; for 0.5h; Inert atmosphere; Stage #2: 1,3-Di(2-tolyl)guanidine In tetrahydrofuran at 65℃; for 4h; | 97.5% |

| Conditions | Yield |
|---|---|
| Stage #1: (3-chloropropyl)triethoxysilane With sodium hydride In tetrahydrofuran at 50℃; for 0.5h; Inert atmosphere; Stage #2: 1-methyl-1H-benzimidazole-2-thiol In tetrahydrofuran at 65 - 67℃; for 5h; | 97.1% |

| Conditions | Yield |
|---|---|
| With tetra-(n-butyl)ammonium iodide; potassium iodide In acetonitrile at 90℃; for 24h; Inert atmosphere; | 97% |
-
-
5089-70-3
(3-chloropropyl)triethoxysilane

| Conditions | Yield |
|---|---|
| In neat (no solvent) at 120℃; for 120h; Inert atmosphere; | 97% |

| Conditions | Yield |
|---|---|
| Stage #1: (3-chloropropyl)triethoxysilane With sodium hydroxide In tetrahydrofuran at 50℃; for 0.5h; Inert atmosphere; Stage #2: 2-methylsulfanylbenzimidazole In tetrahydrofuran at 65℃; for 5h; | 96.8% |

| Conditions | Yield |
|---|---|
| With dmap; potassium iodide; sodium hydroxide In 1,4-dioxane; Petroleum ether at 60℃; for 2h; Reagent/catalyst; Solvent; Temperature; | 96.7% |
-
-
5089-70-3
(3-chloropropyl)triethoxysilane

| Conditions | Yield |
|---|---|
| at 50℃; for 12h; | 96.5% |
-
-
5089-70-3
(3-chloropropyl)triethoxysilane

| Conditions | Yield |
|---|---|
| at 50℃; for 12h; | 96.5% |
-
-
5089-70-3
(3-chloropropyl)triethoxysilane

| Conditions | Yield |
|---|---|
| With sodium azide In butanone for 72h; Inert atmosphere; Reflux; | 96% |
| With sodium azide In dimethyl sulfoxide at 60℃; | 95% |
| With sodium azide; tetrabutylammomium bromide In acetonitrile for 20h; Reflux; Inert atmosphere; | 95% |
-
-
5089-70-3
(3-chloropropyl)triethoxysilane

-
-
131242-36-9
2-sulfanylpyrimidine

| Conditions | Yield |
|---|---|
| With triethylamine In N,N-dimethyl-formamide at 115℃; for 48h; | 96% |
3-Chloropropyltriethoxysilane Specification
The 3-Chloropropyltriethoxysilane, with the CAS registry number 5089-70-3, is also known as Triethoxy(gamma-chloropropyl)silane. It belongs to the product categories of Industrial/Fine Chemicals; Chloro; Chloroalkylsilanes, etc. (Silane Coupling Agents); Functional Materials; Si (Classes of Silicon Compounds); Silane Coupling Agents; Si-O Compounds; Trialkoxysilanes; Alkoxy Silanes; Silane series; Organometallic Reagents; Organosilicon. Its EINECS registry number is 225-805-6. Its IUPAC name is called 3-chloropropyl(triethoxy)silane. The product which is colorless transparent liquid should be sealed and stored in dark, cool and dry place. What's more, it can be used for making silicone and silane coupling agent. Meanwhile, its own can also be used as a coupling agent of epoxy composite materials.
Physical properties of 3-Chloropropyltriethoxysilane: (1)ACD/LogP: 2.72; (2)# of Rule of 5 Violations: 0; (3)ACD/LogD (pH 5.5): 2.72; (4)ACD/LogD (pH 7.4): 2.72; (5)ACD/BCF (pH 5.5): 68.43; (6)ACD/BCF (pH 7.4): 68.43; (7)ACD/KOC (pH 5.5): 716.63; (8)ACD/KOC (pH 7.4): 716.63; (9)#H bond acceptors: 3; (10)#H bond donors: 0; (11)#Freely Rotating Bonds: 9; (12)Index of Refraction: 1.427; (13)Molar Refractivity: 62.54 cm3; (14)Molar Volume: 243.3 cm3; (15)Surface Tension: 25.5 dyne/cm; (16)Density: 0.989 g/cm3; (17)Flash Point: 78.8 °C; (18)Enthalpy of Vaporization: 44.85 kJ/mol; (19)Boiling Point: 230.8 °C at 760 mmHg; (20)Vapour Pressure: 0.0978 mmHg at 25°C.
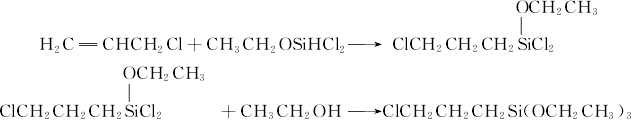
Preparation: this chemical can be prepared by allyl chloride and dichloro-silane which will conduct the addition reaction, and they will conduct alcoholysis reaction with ethanol. At last, you can obtaine γ-chloropropyl triethoxysilane.
Uses of 3-Chloropropyltriethoxysilane: it can be used to produce Triethoxy-(3-iodopropyl)-silan by heating. This reaction is will need reagent NaI and solvent acetone with reaction time of 24 hours. The yield is about 58%.
.png)
When you are using this chemical, please be cautious about it as the following:
This chemical may cause inflammation to the skin or other mucous membranes. It is flammable. In addition, it is irritating to eyes, respiratory system and skin. You should keep away from sources of ignition - No smoking. In case of contact with eyes, you should rinse immediately with plenty of water and seek medical advice. Whenever you will contact it, please wear suitable protective clothing.
You can still convert the following datas into molecular structure:
(1)Canonical SMILES: CCO[Si](CCCCl)(OCC)OCC
(2)InChI: InChI=1S/C9H21ClO3Si/c1-4-11-14(12-5-2,13-6-3)9-7-8-10/h4-9H2,1-3H3
(3)InChIKey: KSCAZPYHLGGNPZ-UHFFFAOYSA-N
Related Products
- 3-Chloropropyltriethoxysilane
- 5089-72-5
- 50899-10-0
- 50901-18-3
- 50903-76-9
- 50904-38-6
- 50906-05-3
- 50906-56-4
- 50907-20-5
- 50907-23-8
- 50907-31-8
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View